6
The Quarantine Facility
The quarantine facility for Mars samples called for in previous chapters has several purposes. First, it will prevent back contamination of Earth’s environment by martian organisms, if the samples contain them, by sealing the samples in a biologically contained space. Second, it will prevent contamination of the samples by terrestrial organisms and chemical contaminants. Third, it will contain a laboratory and the necessary equipment for initial processing of the samples: unpacking, preliminary examination, baseline characterization, weighing, photography, splitting, repackaging, and storage. Another important operation will be the preparation of heat- and/or radiation-treated samples for distribution to the scientific community. In addition, certain life-detection studies that cannot be made on sterilized1 samples—such as testing for biohazards—will have to be carried out in the quarantine facility. There is widespread agreement that the quarantine facility should be a biological containment facility of the most rigorous possible design, technically equivalent to the laboratories in which the most dangerous pathogens known are studied at the Centers for Disease Control and Prevention (CDC, Atlanta, Georgia) and at the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID, Ft. Detrick, Maryland).
BIOLOGICAL CONTAINMENT FACILITIES
Four categories of biological containment facilities are recognized, ranging from biosafety laboratories in which minimal common-sense practices are followed (BSL-1; a high school biology laboratory would qualify) to highly engineered, tightly sealed facilities where diseases that cause high mortality, and for which no immunization or treatment are known, are studied (BSL-4). Table 6.1 summarizes the major distinctions among the four biosafety levels. The higher levels of BSL facilities employ ventilated Biological Safety cabinets (BSCs) as work stations. These, too, are numbered, but there is no correspondence between the BSL number of a facility and the BSC number of the cabinets in it. The properties of three classes of BCSs are shown in Table 6.2.
The maximum level of biological security is furnished by a BSL-4 facility, and the Mars Quarantine Facility should be designed to that level. There are two principal forms a BSL-4 laboratory can take (Figure 6.1). (1) Inside an augmented BSL-3 facility, the samples can be enclosed in Class III Biological Safety cabinets with
TABLE 6.1 A Summary of the Four Formally Defined Levels of Biological Containment
|
|
Biosafety Laboratory Level |
|||
|
BSL-1 |
BSL-2 |
BSL-3 |
BSL-4 |
|
|
Biological agents present |
Well-characterized agents not known to cause disease in healthy adult humans (high school, college biology labs are BSL-1) |
Agents of moderate potential hazard to personnel and environment, for example, measles virus, salmonellae, hepatitis B virus |
Agents capable of causing serious or lethal disease by inhalation, for example, tuberculosis, St. Louis encephalitis virus |
Dangerous and exotic agents that pose a high risk of aerosol-transmitted infections and life-threatening disease, for example, hanta, ebola, arena viruses |
|
Building |
(No restriction) |
(No restriction) |
Separate building or isolated zone; double-door entry; negative air pressure (inward air flow); room exhaust; rooms sealed |
As in BSL-3, plus independent air supply and HEPA-filtered to permit gaseous penetrations sealed decontamination |
|
Laboratory facilities |
Designed for ease of cleaning and decontamination |
As in BSL-1, plus Class II biosafety cabinets, autoclave, eyewash station available |
As in BSL-1, BSL-2 |
Class III biosafety cabinets or Class II cabinets with spacesuits |
|
Safety equipment |
Lab coats, gloves, face and eye protection if needed |
As in BSL-1, plus Class II cabinets for work with infectious agents involving aerosols and splashes, large volumes, high concentrations |
As in BSL-2, plus Class II or Class III biosafety cabinets to manipulate infectious material; respiratory protection if needed |
BSL-3 where applicable; interlocking double-door pass-through autoclave |
|
Laboratory operations |
Mechanical pipetting, hand washing, restricted access; no eating, drinking, smoking; splashes and aerosols minimized; daily decontamination of work surfaces; decontamination of waste, control of insects and rodents |
As in BSL-1, plus emphasis on gloves, mechanical pipetting; extreme precaution with contaminated needles, sharp instruments, broken glass; use of plasticware where possible; restricted entry; biosafety training and biosafety manual specific to lab; personnel immunized, baseline serum samples taken; spills and accidents reported; supervisor is a competent scientist |
As in BSL-2, plus all work in biosafety cabinets; use of bioaerosol-containing equipment; prompt decontamination of spills; personnel strictly follow guidelines, demonstrate proficiency, participate in medical surveillance |
BSL-3 where applicable, plus liquid, solid waste sterilized; exhaust air HEPA filtered; all work in Class III biosafety cabinets or Class II cabinets with spacesuits; all surfaces decontaminated before removal; no infectious organisms can leave the laboratory except in sealed containers |
TABLE 6.2 Summary of Properties of Three Classes of Biological Safety Cabinet
|
|
Class of Biological Safety Cabinet |
||
|
Class I |
Class II |
Class III |
|
|
Interior pressure |
Ambient |
Ambient |
Negative pressure (closed cabinet) |
|
Air supply |
Through opening for arms |
HEPA-filtered supply, also through arm opening |
HEPA-filtered supply only |
|
Air exhaust |
HEPA-filtered |
HEPA-filtered |
Doubly HEPA-filtered |
|
Glove ports |
Optional |
Optional |
Required |
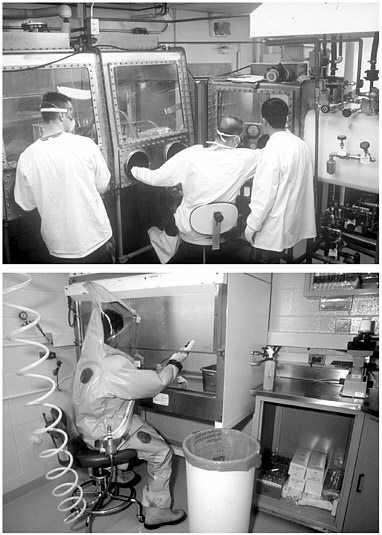
FIGURE 6.1 Different work modes in a BSL-4 laboratory at USAMRIID. At the top, hazardous samples are enclosed in a Class III Biological Safety cabinet. Below, the samples are more exposed in a Class II cabinet, but the worker is protected from them by a spacesuit. Using a glove box (Class III cabinet) is preferable for repetitive tasks that require limited lateral mobility. The spacesuit offers more flexibility, but at the cost of increased fatigue. SOURCE: Photographs courtesy of Steve Ferendo, USAMRIID.
glove ports, which are used by operators with only minimal protective garments (top of Figure 6.1). The box is typically made of stainless steel, glass, or Plexiglas, and gloves are sealed to the arm holes. (2) The samples can be manipulated in Class II cabinets that are ventilated but not tightly sealed, by operators wearing ventilated protective garments that completely isolate them from the environment they are working in (bottom of Figure 6.1). These garments are formally positive pressure personnel protection suits, but in practice they are called “spacesuits.”
In the first case, the Class III Biological Safety cabinets have internal gas pressures that are less than atmospheric, so if there is slight leakage, the gas flow is from the surrounding room into the cabinet and pathogens in the cabinet have no path out of it. The surrounding room is effectively a large Class III cabinet itself. In the second case, the spacesuits have internal gas pressures higher than that of the room being worked in, so leaking gas flows out of the suits and pathogens in the laboratory cannot enter the suits. Ideally, a BSL-4 laboratory offers both work environments.
All waste products from BSL-4 laboratories must be specially treated. Exhaust air is passed through two high-efficiency particulate air (HEPA) filters. Liquid effluents are routinely decontaminated by a heating process before discharge to a sanitary sewer. In small operations, this decontamination can be accomplished with chemical treatment. Other waste materials are decontaminated by steam sterilization in an autoclave. Large objects (e.g., equipment) may be sterilized by exposure to gaseous formaldehyde, hydrogen peroxide, ethylene oxide, or some other sterilant. Small quantities of material are sometimes gamma-irradiated.
Some Operational Details2
Flexibility and adaptability are essential in the design of a BSL-4 facility. Experience has shown that the ideal BSL-4 environment is one in which a Class III cabinet line is joined to a spacesuit laboratory. After training and acclimatization, most workers prefer the flexibility of the spacesuit environment but report that they cannot perform satisfactorily for more than about 4 hours (continuously) in that environment. To the extent that segments of the work routine can be compartmentalized, those elements that require little lateral mobility can be performed with more comfort in a cabinet. The cabinet can be held at modest negative pressure; 0.98 to 0.99 atmospheres is sufficient. The gloves need be no thicker than those on spacesuits, provided that they are checked regularly for perforations.
The responsibilities surrounding a BSL-4 facility are not to be taken lightly. Engineering controls for such a system must be monitored around the clock, and support staff must be on continuous alert to correct any problems that arise. Access to BSL-4 laboratories must be controlled; passage through control points is monitored by guards who can track movement via key cards and visually, through windows to the corridors and with television monitors. Firefighters and emergency medical care providers also must be trained for the contingency that they might be required to enter an active BSL-4 suite. Backup generators to supply power to all critical systems must be available and routinely tested. The necessary systems must incorporate redundancy so that no critical system is ever compromised, even momentarily. Battery backups for such systems are also advisable, and they also should be routinely exercised and performance tested. It is also essential to have a backup supply of breathing air.
Extensive documentation regarding design and maintenance specifications, environmental impact statements, internal and external security systems, emergency response plans, and standard operating procedures (especially with regard to decontamination) must be published.
Staffing and Training
A fully functional BSL-4 facility requires more than the elaborately engineered laboratory itself. The physical plant is useless without a highly trained and dedicated cadre of scientists and support personnel. At most BSL-4
|
2 |
The technical discussions in this subsection and in the next (“Staffing and Training”) are from the experience of COMPLEX member P. Jahrling, USAMRIID. Some of the guidelines differ at other containment facilities, such as the National Center for Infectious Diseases at the CDC. |
laboratories, technical staff and scientists who wish to work in that environment must first demonstrate that they can perform whatever manipulations are contemplated in a less hazardous (BSL-2) environment. Typically, these personnel begin their special immunization series (required to work at BSL-3) while working at BSL-2. After a period of several months they graduate to BSL-3, where they will work for at least several additional months. Experience has shown that a number of potential BSL-4 workers may drop out at this stage, for a variety of reasons, but others will elect to undergo the specialized training for BSL-4. This training involves familiarization with wearing and adjusting the spacesuit, and understanding the critical life support systems (breathing air supply and backups), communications, redundancy of all systems, and emergency procedures. The neophyte never works alone in the early stages of training; he is assigned a mentor who oversees his every move. Although most special situations are detailed in written procedures, it is only through personal supervision and daily reinforcement that these rules become ingrained and routine. Working with authentic pathogens removes the abstraction from such rules. It is unlikely that any worker who has not handled pathogenic agents will fully appreciate and respect the importance of strict adherence. A worker without this experience is more likely to cut corners, especially if he believes the possibility of encountering a true pathogen to be remote. At least 1 year of experience is required before BSL-4 workers should be permitted to work on their own, or to train others in the principles of biocontainment.
At the Mars Quarantine Facility, the goal of rigorous adherence to biosafety principles will be facilitated by providing a core staff of veterans to run the laboratory and serve as mentors for new recruits and visiting scientists. A policy of zero tolerance for shortcuts or lapses should be enforced. The core staff should include scientists with training similar to that of the visiting specialists, in addition to experts in biosafety, decontamination, tissue culture, media preparation, communication, and conceivably animal care.
In facilities used for the study of disease, the first indication that there has been a break in containment may come when a worker develops a febrile illness. In this case, circumstances may suggest that the prudent course is to place the worker in BSL-4 quarantine. This requires that there be a dedicated medical care facility at hand, and trained medical care providers. Again, experience with authentic agents is desirable, since highly trained workers who lack this experience sometimes panic when faced with the reality of a truly sick patient. This experience can come only from working at a facility where such patients are cared for on a routine basis.
These measures sound drastic, in the context of handling samples that are unlikely to contain life and still less likely to contain pathogenic life; but if maximum precautions are to be taken, they must be adhered to. COMPLEX concurs with the conclusion reached by earlier studies that initial BSL-4 containment is dictated for the Mars samples.3-7
THE QUARANTINE FACILITY FOR MARS SAMPLES
The initial processing of returned martian samples should be restricted to a BSL-4 laboratory in the quarantine facility (Chapter 6). A very modest gas-tight glove box (Class III cabinet) in a “clean room” (class 10; however, see following section) will be sufficient for this purpose. Within this cabinet, the samples should be inventoried and subdivided for various analyses. One portion of each sample should be sterilized and sent to an outside laboratory to be analyzed for total reduced carbon (organic carbon; see Chapter 2). If this analysis finds all samples to be devoid of reduced carbon, it will be reasonable to conduct almost all additional analyses under less
stringent conditions of biocontainment (BSL-3). However, if additional effort is invested in isolating replicating agents by encouraging them to grow in artificial media, or to amplify their genomes by polymerase chain reaction or related techniques, this work should be restricted to BSL-4. The small original sample sizes will preclude testing in a variety of animals; animal infectivity studies will be practical only in the unlikely event that a seed stock of a microbe is successfully replicated from the minute sample of original material. The need for extensive BSL-4 facilities, especially animal facilities, is remote.
Special Requirements
Earlier studies have recognized a special problem associated with the Mars Quarantine Facility,8-9 in that it must be not only a biological containment facility (to protect the environment), but also a clean room (to protect the samples). No existing facility combines these qualities. BSL-4 facilities are usually rather “dirty,” both chemically and biologically. Clean conditions are needed to avert false biological positives, and to make precise isotopic dating and other geochemical measurements possible.
The need for cleanliness to protect the samples from terrestrial contamination is even more important now than it was at the time of the Apollo program (Appendix B). Analytical techniques have improved to the point that low levels of contamination that would have escaped detection 30 years ago are now significant signals. For example, analyses of amino acids are commonly made on samples with a sensitivity of 10–11 g, and 10–14 g can be achieved with state-of-the-art apparatus. By the time the samples are returned, it is reasonable to expect sensitivities substantially better than these. In the area of geochemical analyses of inorganic minerals, the sensitivity for measurements of isotopic ratios has improved to the point that, for example, researchers can typically measure the ratio of 143Nd/144Nd to a precision of 1 part in 105 on a mass of 10–9 g of the element. It is only because of such high sensitivity and precision that many of the new isotopic dating schemes in use have become possible. The tiniest amount of terrestrial contamination can alter the isotopic ratio of a sample to the point that application of a dating technique gives an erroneous result.
Contamination can be less of a problem when isotopic ratios are measured by secondary ion mass spectrometry (SIMS) on polished sections of rocks, which can be freshly cut and kept relatively clean. Technological improvements in the capability of SIMS have made possible some forms of isotopic dating, but these SIMS techniques are only applicable to samples where there are large chemical fractionations and thus large isotope-ratio differences. Such large fractionations have been found in samples thought to contain interstellar grains, but they are not expected in the Mars samples. Our ability to understand the Mars samples would be severely limited if quarantine or curation activities allowed contamination of the samples to a degree that precluded all dating techniques except SIMS.
The problem of combining quarantine with cleanliness is technically challenging, because contradictory measures (i.e., pressure differentials) are normally employed to produce biological containment and a clean room environment. Biological Safety cabinets are held at negative gas pressures, so leakage (which must be considered inevitable) will produce gas flow into the cabinets, away from personnel in the room with the cabinets. Clean rooms are held at positive gas pressures, relative to ambient, so gas will flow out of them and contaminants cannot flow in. Nesting the two types of enclosures (Figure 6.2) would seem to offer a first-order solution to the problem, but that stratagem raises new difficulties. If a BSL-4 cabinet is housed in a clean room, the clean room protection is partly negated because contamination from workers in the room and the ambient atmosphere is drawn by leakage into the cabinet. A clean cabinet in a BSL-4 enclosure would protect both samples and environment, but workers would have to wear spacesuits and use glove ports in the clean cabinet, which would limit their dexterity
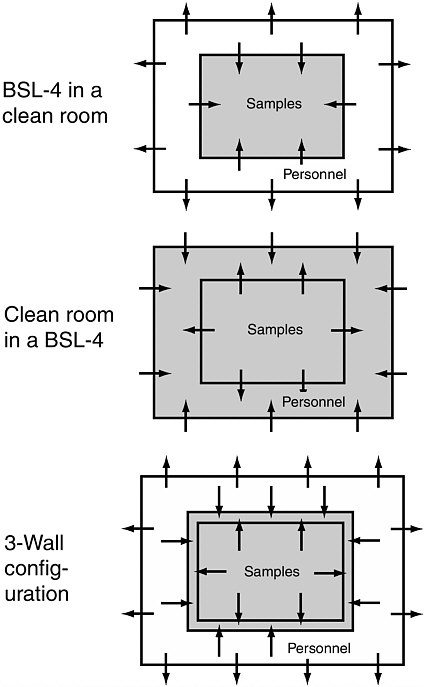
FIGURE 6.2 Top and center: simple options for the combination of a biological containment facility with a clean room. Arrows show gas flow (via leakage) caused by pressure differentials in the spaces shown. Gray areas are potentially contaminated by any organisms the Mars samples might contain. Bottom: a more complex arrangement with double walls separating workers from samples in which the sequence of gas pressures is personnel space > outside atmosphere > sample cabinet > exhaust space between double walls.
to a degree that might prevent useful work from being done. The bottom panel of Figure 6.2 suggests an approach in which double walls separate workers from the samples, with the lowest gas pressure in the space between the double walls. Leakage from both the sample cabinet and the workers’ space would be collected between the walls, sterilized, and exhausted. Glove ports would have to penetrate both walls, however, and leakage of air through pinholes in the gloves would still have the potential of contaminating the samples, but gas in the sample cabinet could be monitored for traces of contaminant gases as was done in the Lunar Receiving Laboratory (Figure B.2, Appendix B), and glove ports sealed if it is detected.
A possibility that should be explored is the use of remote or robotic manipulation of samples within the containment facility, in lieu of human hands.
The solution of this problem is in the area of implementation, and beyond the scope of a COMPLEX study. However this committee recognizes the importance of the problem, and the fact that design work on a quarantine facility cannot proceed until it is solved.
Recommendation. A major obstacle to the design of the Mars Quarantine Facility is the problem of combining biological containment with clean-room conditions. It is essential that work on the solution of this problem be started immediately, to include mockups of containment/clean-room combinations whose efficacy can be tested, so that the design of a quarantine facility can proceed.
Scale and Use of the Facility
It must be understood that imposing biological containment and clean-room conditions on any kind of scientific operation complicates it by a huge factor. Working through glove ports or spacesuit gloves is inherently clumsy: Fairly coarse, simple operations are possible but impeded; delicate operations on a fine scale may be prohibited. The use of bulky, complex, failure-prone scientific instrumentation within such an enclosure, with the potential for system contamination by expendable supplies the instrument uses, and the need to accommodate the technicians and repair personnel who keep the instrument working, would be a nightmare, and an expensive one. When thought is given to the many kinds of studies (and attendant instrumentation) the international scientific community will want to subject the Mars samples to, it is clear that the required instrumentation and personnel cannot possibly be housed within the quarantine facility. The quarantine facility should contain a minimum amount of scientific equipment, only the devices needed to carry out research that cannot be done elsewhere. Other research should be conducted on sterilized samples, in the home laboratories of investigators approved for the Mars sample research program. Chapter 4 identifies the types of studies that should be done in the quarantine facility: first-order characterization of the samples, and studies that cannot be carried out on sterilized samples because of damage that would be done to them by the sterilization procedure, especially efforts to demonstrate the presence of microbial life by methods of microbial replication. (It is possible, however, that technological advances during the time before sample return may make available compact analytical devices that could feasibly be included within the quarantine facility, which would allow searches for biological structures and/or reduced carbon within the facility.)
Recommendation
-
The Mars Quarantine Facility should be designed to the smallest and simplest possible scale consistent with its role as a biological containment and clean room facility. No scientific investigations should be carried out in the quarantine facility that can be executed on sterilized samples outside the facility.
-
Protocols should be developed that specify in detail the steps and procedures to be followed for handling Mars samples in the quarantine facility. Necessary protocols include those for inventorying and preliminary analyses of the samples, searching for evidence of biological activity, testing for biohazards, and preparing sterilized aliquots of the samples for distribution to the scientific community.
-
Because it cannot be carried out on sterilized samples, biohazard assessment should be performed in the quarantine facility prior to any release of samples from the facility. Elements of these studies might include culturing experiments; attempts to infect animals, plants, and cell cultures; and genome detection via the polymerase chain reaction or similar techniques.
Siting of the Facility
The quarantine facility could be built as an independent, free-standing laboratory, or it could be physically part of and affiliated with an institution that already contains BSL-4 laboratories (USAMRIID, in Ft. Detrick, Maryland; CDC, in Atlanta, Georgia; or the Medical Branch of the University of Texas at Galveston, where a BSL-4 facility is being constructed). COMPLEX considers that the second of these options, affiliation with an ongoing containment facility, is preferable for several reasons.
-
Institutional support. A collaborative agreement with a host institution would mean that the Mars Quarantine Facility could draw on that institution for personnel, training, experience, security, and specialized utilities (in return for appropriate augmentation of the institution’s support).
-
Economy. Sharing the resources named under 1 above should effect a large economy in operation of the Mars Quarantine Facility.
-
Environmental impact. Clearing an environmental impact statement for a BSL-4 facility can take years. Ideally, the Mars facility would operate under the environmental impact statement (EIS) of its host institution. Failing this, existence of the original EIS should pave the way for approval of a new statement applicable to the quarantine facility.
The Mars Quarantine Facility should be either a free-standing building located at an existing institution, or a segregated portion of an extant building in that institution’s complex. The BSL-4 laboratory that handles the martian samples must not be shared physically with other activities that involve BSL-4 agents, but this restriction would not prevent the Mars Quarantine Facility from sharing the manpower pool of the host institution.
The Mars Quarantine Facility should remain effectively under the control of NASA, which is able to supply the science perspective that will guide study of the samples without compromising the required standard of biological containment and planetary protection. It is important that the laboratory be located in or near an active biological science community, where those workers using the facility would be able to draw from a critical mass of supporting scientific disciplines and infrastructure, including other BSL-4 facilities. Without this support, operation of the Mars Quarantine Facility would be greatly handicapped.
Recommendation. The Mars Quarantine Facility should be affiliated with an ongoing containment facility that has BSL-4 capability and should be physically part of it or proximate to it, but control of the Mars Quarantine Facility should be under the jurisdiction of NASA.
TIME FOR CONSTRUCTION
The design of a BSL-4 facility dedicated to the processing of martian samples will be more complex than any BSL-4 laboratory constructed to date, because of the need to prevent contamination of the samples with terrestrial organisms. The design will probably entail enveloping a conventional BSL-4 facility within a class 10 clean room, and operational controls will have to be implemented to reduce the likelihood of terrestrial organisms being carried on clothing, instruments, or reagents into the BSL-4 zone (either cabinet or hood line).
Conventional BSL-4 laboratories (without the clean room envelope) are being constructed now with increasing frequency, both in the United States and abroad. The record for speed of construction, from inception to commissioning, belongs to a private laboratory built by Pasteur Merrieux Connaught in Lyon, France. This
laboratory progressed from first design to completion in exactly 2 years, although the commissioning process and shakedown have consumed an additional 18 months. This is a conventional spacesuit laboratory, with tissue culture laboratories, animal (primate) laboratories, and a surgery/necropsy suite; however, no class III cabinets are installed. A smaller spacesuit-only lab was created at the National Institutes of Health in Bethesda, Maryland, using existing BSL-3 space. Construction and commissioning took 4 years, although some of this time was expended on a divisive public-relations problem with concerned residents in the community. A more extensive BSL-4 facility has been constructed by the Canadian Departments of Health and Agriculture in Winnipeg; more than 5 years have elapsed, and it has still not been commissioned.
With the exception of the clean room envelope, a BSL-4 laboratory dedicated to processing the martian samples should be designed with simplicity and flexibility in mind. Experience has shown that the ideal BSL-4 environment is one in which a Class III cabinet line (see Table 6.2) is joined to a spacesuit laboratory. Class III cabinetry should be selected “off the shelf.” Custom-designed Class III BSC hoods that accommodate specialized equipment items are very expensive and inherently inflexible; such equipment is usually better accommodated in a spacesuit laboratory. An optimistic estimate for design and construction of this dedicated laboratory is 4 years, plus shakedown and commissioning. Cross-training of personnel in adjacent or affiliated BSL-4 facilities would reduce the start-up time in activating a trained BSL-4 cadre.10
A schedule for the planning, building, and certification of the Mars Quarantine Facility is given in Table 6.3. This schedule assumes that the facility is affiliated with an ongoing containment facility with BSL-4 capability; for a free-standing facility, more time would be required to clear an environmental impact statement, establish needed support services, and train personnel (since these activities cannot proceed in parallel with facility construction). An estimated 7 years is required from beginning to end. This estimate is not padded; it is based on experience with the Apollo Lunar Receiving Laboratory (Chapter 7) and contemporary biological containment facilities. The schedule cannot be shortened without compromising the quality and effectiveness of the facility. Thus it is imperative that planning and construction of the facility be put in motion 7 years prior to projected sample return, which is the most important recommendation that COMPLEX makes in this report.
Recommendation. It is imperative that planning and construction of the Mars Quarantine Facility be begun at least 7 years in advance of the anticipated return of Mars samples. This responsibility cannot be deferred without compromising the quarantine and study of the Mars samples.
INTERNATIONAL ACCESS TO THE MARS SAMPLES
It is possible if not probable that the Mars sample return program will be an international venture, with other nations playing an important role in flight operations. Plans for the original Mars Surveyor 2005 mission called for a substantial part of the program to be carried out by France. The role of international partners in a sample-return program should be carefully defined. The question of international access to the samples is not an issue; traditionally the United States has freely shared the fruits of its space program with other nations (e.g., the Apollo lunar samples). However, the potentially sizeable contribution of another nation to the Mars program raises questions of how the earliest access and ultimate curation of the samples will be shared. The simplest option would be a single path through quarantine and curation, with management and operation of the facilities, and continuing studies within a quarantine facility in the United States, shared by the international partners. Separate curation facilities in the United States and abroad could also be pictured.
It is beyond the scope of COMPLEX’s charge to comment on the ultimate curation of the samples, but the committee believes strongly that their preliminary examination, baseline description, cataloguing, and packaging should be carried out at a single quarantine facility in the United States. The sample canister containing Mars samples probably will be landed in the continental United States, recovered by a mobile retrieval unit, and then
TABLE 6.3 An Estimate of the Time Required to Construct, Prepare, and Certify the Mars Quarantine Facility
|
Time |
Action: Facility |
Action: Personnel |
|
0 years |
Funding of facility is authorized. Negotiate site for laboratory and collaborative relationship with in-being containment facility. Determine requirements for sample sterilization, certification of absence of biohazardous material, and sample handling and storage. (12 months) |
|
|
1 year |
Write draft protocols designed to meet requirements named above and for other critical procedures, such as those for: • Sample entry, • Sample inventory, • Preliminary analyses, • Search for evidence of biological activity, • Assessment of whether sample contains biohazardous material, • Sterilization of aliquots of sample in preparation for removal from the facility, • Removal of samples from the facility, • Storage of samples within the facility, and • Sterilization/cleaning of laboratory facility in preparation for sample entry. (6 months) |
|
|
1.5 years |
Design a facility around the requirements for Mars sample quarantine and the particular protocols that have been written. (24 monthsa) |
|
|
3.5 years |
Begin construction. (12 monthsa) |
Begin recruiting, training, and inoculation regime of staff for the facility. (18 monthsb) |
|
4.5 years |
Begin troubleshooting and certification of mechanics of facility (air, water, pumps, backup systems, etc.). (6 months) |
|
|
5 years |
Approve and officially certify building and laboratory mechanics (air, water, pumps, backup systems, etc.). Perfect all protocols that will be used in the facility (listed above). Test them with a full dress rehearsal. (12 months) |
Training of initial staff completedb |
|
6 years |
Review and, as necessary, revise specified requirements for critical procedures in light of demonstrated facility capabilities. (6 months) |
|
|
6.5 years |
Clean the facility, which is now biologically contaminated and organically dirty. (6 months) |
|
|
7 years |
Facility is certified as ready to receive Mars samples. |
|
|
aFrom Lunar Receiving Laboratory experience (Chapter 7). bFrom the section “Staffing and Training” in this chapter. |
||
transported to a fixed quarantine facility for opening and initial processing. It would be a mistake to attempt to open and divide the sample before it entered the quarantine facility, inviting contamination of the sample and environment. Once in the facility, it would be a mistake to divide the sample before baseline characterization was completed, as this would compromise optimal allocation of the samples to outside investigators for study.
Recommendation. All samples in the initial collection returned from Mars should be placed in a quarantine facility in the United States, at least until the preliminary examination of the samples has been completed. Management and operation of the Mars Quarantine Facility should be shared between the United States and major international partners that participated in the collection of martian samples.
After sample inventory, an international partner may wish to establish an independent BSL-4 facility in which to study unsterilized samples of martian material. Division of the samples in this manner is not excluded by COMPLEX’s recommendation, providing that containers and transfer procedures conform to protocols established by a panel of experts (e.g., from the CDC) in containment (Chapter 4).
It is important that formal agreements (memorandums of understanding) be completed with major partners, in the planning stages of an international program of Mars sample return, to define each partner’s degree of participation in the preliminary examination of the samples and its entitlement to a share of samples for an independent quarantine facility (and/or its degree of participation in a continuing U.S. quarantine facility).
OVERSIGHT COMMITTEE
COMPLEX endorses the 1997 National Research Council report’s recommendation that:
A panel of experts, including representatives of relevant governmental and scientific bodies, should be established as soon as possible once serious planning for a Mars sample-return mission has begun, to coordinate regulatory responsibilities and to advise NASA on the implementation of planetary protection measures for sample-return missions. The panel should be in place at least one year prior to the establishment of the sample-receiving facility (at least three years prior to launch).11
The panel described would be equivalent to the Interagency Committee on Back Contamination (ICBC) that provided oversight during the Apollo program (Appendix B). However, Appendix B also describes another committee, the Lunar Sample Analysis and Planning Team (LSAPT), which played a different and equally important role in the lunar program. The formal charge of LSAPT was to advise NASA on the following:
-
Equipment and procedures for use in the Lunar Receiving Laboratory for conduct of time critical experiments, gross preliminary examinations of samples and curatorial functions.
-
Proficiency, capability and adequacy of Lunar Receiving Laboratory and its staff to carry out functional roles of Lunar Receiving Laboratory.
-
Sequence of sample analysis and the allocation and distribution of samples after quarantine release.
-
Review findings of preliminary examination team during quarantine and advise on operational procedures and techniques.12
The responsibilities of ICBC lay mostly in the quarantine period (limited to three weeks after return of the lunar samples), and LSAPT’s responsibilities were mostly in the curation and allocation period after that, but there was overlap of the committees’ roles, and, inevitably, conflict. Put in simplest terms, the job of ICBC was to tighten the screws, while LSAPT tried to loosen them and move the samples. In this LSAPT was very effective,
and much of the timeliness of the Apollo scientific program is attributable to LSAPT’s exertions. Since the lunar samples proved to be devoid of life or its products, the efforts of ICBC are less celebrated.
The Mars Quarantine Facility would profit from the efforts of a committee modeled after LSAPT. It should differ from the planetary protection panel described above in being more of an advocacy group, concerned with getting the most science possible from the samples, and making political representations where necessary. It should differ from LSAPT in having a larger component of biological scientists, and a more deeply felt appreciation for planetary protection and the potential of the Mars samples to be repositories of life. But like LSAPT it should be a continuing committee, with the capacity and motivation to follow through on the reception of its recommendations, something that advisory groups like COMPLEX, the Space Studies Board Task Group on Issues in Sample Return,13 and MSHARP (the NASA Mars Sample Handling and Requirements Panel14 cannot do.
The Mars sample advocacy committee should consist mostly of experts from outside NASA. Qualified persons from outside NASA (e.g., from the National Academy of Sciences) should be substantially involved in nominating the committee. The committee should report to a high level in NASA, either the Office of the Associate Administrator for Space Sciences or the Office of the Director of the NASA center principally concerned with Mars sample quarantine and curation.
Recommendation. A continuing committee of senior biologists and geochemists that includes appropriate international representation should be formed and charged with reviewing every step of the planning, construction, and employment of the Mars Quarantine Facility. The committee should be formed during the earliest stages of planning of a Mars sample-return mission. Members of the committee should also participate in the design of the spacecraft and those portions of the mission profile where biological contamination is a threat.
Early formation of the committee is advocated because there is work for it to do at every stage of planning, including response to the recommendations of this report and earlier reports.15-17













