1
Introduction
BACKGROUND
In the years following World War II, the successful development and use of vaccines and antibiotics led to speculation that infectious diseases were vanquished foes (CDC, 1998; IOM, 1992, 2001; WHO, 1996, 2000c). Defying these predictions, infectious diseases continue to be a leading cause of morbidity and mortality globally (IOM, 2001; NIC, 2000; WHO, 2000c). Old scourges, such as tuberculosis, malaria, dengue, cholera, and influenza linger still. New challenges, such as human immunodeficiency virus (HIV) infection, multi-drug-resistant tuberculosis and other bacterial diseases, and various encephalitides and viral hemorrhagic fevers, have surfaced with grim frequency in recent years and are reminders that emerging and reemerging infectious diseases are threats worthy of continued concern and due vigilance.
In the 1980s, recognition of the devastating global HIV/AIDS epidemic focused the attention of the public and the public health community on the issue of emerging infectious diseases. Between 1987 and 1992, the Institute of Medicine (IOM) released three reports collectively urging improvements in the public health infrastructure in place nationally and internationally to address infectious diseases (IOM, 1987, 1988, 1992). The 1992 report, Emerging Infections: Microbial Threats to Health in the United States, was particularly instrumental in focusing attention on the threat of infectious disease emergence. These concerns spurred a number of national and international initiatives, including the development of a Centers for Disease Control and Prevention (CDC) strategic plan for address-
ing emerging infectious diseases (CDC, 1994), a World Health Organization (WHO) resolution calling for strengthened infectious disease surveillance and response capabilities (WHO, 1995), and a formative National Science and Technology Council Report (CISET, 1995).
In 1996, the Executive Office of the President issued Presidential Decision Directive NSTC-7 (NSTC is the National Science and Technology Council of the Executive Office of the President), which declared that national and international capabilities for infectious disease surveillance, prevention, and response were inadequate to protect the health of U.S. citizens from emerging infectious diseases and which called for a more robust national policy to improve these capabilities (NSTC, 1996). NSTC-7 directs U.S. federal agencies, namely, the U.S. Departments of Health and Human Services, State, Defense (DoD), Justice, Commerce, Agriculture, Interior, and Energy; the U.S. Agency for International Development; the National Aeronautics and Space Administration; the Environmental Protection Agency; the Intelligence Community; the White House Office of Science and Technology Policy; the National Security Council; the Domestic Policy Council; and the Office of Management and Budget to take action (NSTC, 1996). Responsibility for coordinating the multi-agency response to the directive rests with the National Science and Technology Council Task Force on Emerging Infections (CISET, 1997; NSTC, 1996).
NSTC-7 provides the following direction to DoD (NSTC, 1996, pp. 4–5):
The mission of the DoD will be expanded to include support of global surveillance, training, research, and response to emerging infectious disease threats. DoD will strengthen its global disease reduction efforts through: centralized coordination, improved preventive health programs and epidemiologic capabilities; and enhanced involvement with military treatment facilities and overseas laboratories.
DoD will ensure the availability of diagnostic capabilities at its three domestic and six1 overseas laboratories, using existing DoD resources. DoD will make available its overseas laboratory facilities, as appropriate, to serve as focal points for the training of foreign technicians and epidemiologists. If necessary, DoD will seek Chief of Mission concurrence to raise personnel ceilings at overseas laboratories, in accordance with NSDD [National Security Decision Directive]-38 procedures.
Since NSTC-7 was issued, U.S. agencies and international partners have taken many steps to achieve the goals of the directive and, more
generally, to address global emerging infectious disease priorities (CDC, 1998; CISET, 1997; DoS, 1999; GEIS, 1998; NIC, 2000; NIAID, 1996, 2000; USAID, 1998; WHO, 1996, 2000b). The DoD Global Emerging Infections Surveillance and Response System (GEIS) embodies the DoD response to Presidential Decision Directive NSTC-7.
THE DEPARTMENT OF DEFENSE GLOBAL EMERGING INFECTIONS SURVEILLANCE AND RESPONSE SYSTEM
GEIS was established in 1997 by the assistant secretary of defense for health affairs, in response to NSTC-7, to serve as the focal point for “initiating and coordinating the identification, reporting and responding to emerging infectious disease problems” (Bancroft and Schlagel, 1997, p. 1) within the DoD. GEIS is a new program that supplements, but that does not replace, the ongoing research mission of DoD laboratories (Bancroft and Schlagel, 1997; Mazzuchi, 1999). GEIS was first allocated start-up funds in fiscal year 1997, although GEIS is officially considered to have begun in late 1998 (GEIS, 2001a). In fiscal year 2000, GEIS funding totaled $7 million (GEIS, 2000b). In fiscal year 2001, funding for GEIS is expected to total approximately $8 million (GEIS, 2000c).2 GEIS’s annual budget is projected to increase over the next several years—to $9 million in fiscal year 2002, $10 million in 2004, and $11 million in 2005 (Kelley, 2000a).
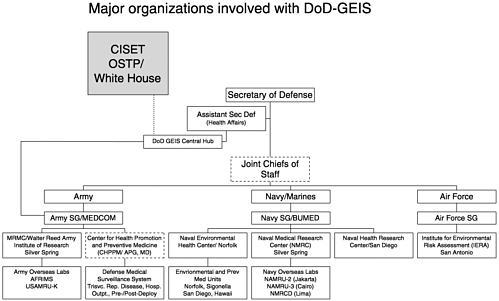
The U.S. Army is the designated executive agent for GEIS (Lister, 1997). GEIS is organizationally responsible to the Office of the Surgeon General of the U.S. Army (Martin, 1997c). GEIS is a triservice program, and its activities are implemented within all three branches (Army, Navy, and Air Force) of the armed forces, although GEIS has no direct command authority over the facilities that implement its activities. GEIS operates predominantly within the five currently operating Army and Navy overseas medical research laboratories and the infrastructure of the military health system3 (MHS) (GEIS, 2000b). Within the MHS, GEIS has designated service hubs at the Naval Health Research Center, San Diego, California, and the U.S. Air Force Epidemiology Services Branch, Brooks Air Force Base, San Antonio, Texas, that serve as focal points for GEIS MHS
activities (Gaydos, 1999). GEIS also collaborates with other components of the DoD, including the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID), Fort Detrick, Maryland; the Naval Environmental Health Center, Norfolk, Virginia; the U.S. Army Center for Health Promotion and Preventive Medicine, Aberdeen Proving Ground, Maryland; and other DoD sites within the United States and abroad. GEIS also provides support for and collaborates with non-DoD partners.
GEIS is managed by a Central Hub office that operates within and receives administrative support from the Walter Reed Army Institute of Research. Planning documents call for the Central Hub to be supported by a triservice staff (Bancroft and Schlagel, 1997), although no Air Force personnel are assigned to the Central Hub at present. The Central Hub is directed by a preventive medicine officer of the same service as the program’s executive agent (the Army). GEIS receives periodic scientific input from the Armed Forces Epidemiology Board and periodic program management direction from a flag level4 advisory board (Christopherson and Baldwin, 1997; Lister, 1997). GEIS occasionally receives non-DoD input as well, for example, through the Committee on International Science, Engineering, and Technology (CISET) (CISET, 1997) and through publicly attended GEIS symposia (GEIS, 1999b). In addition to providing GEIS program management, the Central Hub also directs surveillance, systems research, and capacity-building activities.
The MHS network of clinical and laboratory resources provides infrastructure and surveillance capabilities important for GEIS. Within the MHS, the emphasis of GEIS is on the strengthening of laboratory-based surveillance, particularly for influenza and other respiratory illnesses (GEIS, 1998), for reportable illnesses, for changes in antibiotic resistance patterns, and for other evidence of emerging infections.
Since the early 1900s, the DoD has maintained a network of medical research laboratory facilities overseas to help identify infectious diseases and to evaluate control measures (Gambel and Hibbs, 1996). These DoD overseas laboratories provide forward sites for GEIS activities. At present the DoD has five overseas laboratories, which are based in Lima, Peru; Jakarta, Indonesia; Bangkok, Thailand; Cairo, Egypt; and Nairobi, Kenya. In addition, these laboratories have field activities that operate in other nearby countries. The laboratory capacities that these facilities possess are often rare or unique within their regions of operation. GEIS was active in 38 countries in fiscal year 2000 (GEIS, 2000b). Many activities were directed through the overseas laboratories. At overseas laboratories, the focus of GEIS is primarily on the development of sentinel surveillance of indigenous and expatriate populations, as defined in its ambitious 1998
strategic plan, Addressing Emerging Infectious Disease Threats: A Strategy for the Department of Defense (GEIS, 1998).
The GEIS mission is centered around four goals that are similar to those of the CDC (CDC, 1994, 1998; GEIS, 1998). GEIS goals include the following (GEIS, 1998, p. 1):
Surveillance. Detect and monitor emerging pathogens, the diseases they cause, and the factors influencing their emergence to protect military readiness, the health of DoD beneficiary populations, and other national interests.
Systems Research, Development, and Integration. Integrate public health practices and improve capabilities in clinical medicine, military medicine, laboratory science, epidemiology, public health, and military medical research to facilitate the rapid identification and response to emerging infections
Response. Enhance the prompt implementation of all prevention and control strategies for emerging infections to include improving communication of information about emerging agents.
Training and Capacity Building. Leverage DoD and international public health infrastructures through training, networking, and other forms of assistance to support surveillance, assessment, response, and prevention of emerging infections.
These goals represent the organizing principles of GEIS activities carried out within the overseas laboratories, the infrastructure of the MHS, and the Central Hub—the constituent parts of the GEIS consortium (DoD partners in GEIS). GEIS has identified four “pillar” areas of infectious disease focus to guide surveillance efforts, particularly at overseas laboratories. Pillar areas are formally influenza, drug-resistant enteric organisms, drug-resistant malaria, and febrile illnesses, including dengue (GEIS, 1998). These target areas are intended to address a balance of disease areas that are important to the DoD, the U.S. population, and the international community, duly recognizing resource constraints.
CHARGE TO THE COMMITTEE
This committee’s task was defined as follows:
The committee will assess the extent to which the strategic plans of the five Department of Defense Global Emerging Infections System laboratories appropriately address the surveillance needs for the identification and tracking of emerging infectious disease in their respective locations. Specific questions to be addressed by the committee are:
-
How well does each plan meet the minimal infectious disease surveillance requirements as prescribed in Presidential Decision Directive NSTC-7 and the DoD Strategic Plan?
-
How well do the laboratory programs agree with the approach to emerging infection surveillance espoused in the IOM report on emerging infections and the Centers for Disease Control and Prevention (CDC) national strategic plan?
-
How well do the laboratories meet the World Health Organization and/or CDC standards for evaluating surveillance systems making reasonable allowances for (1) the need to balance the interests of the host country against those of DoD and the U.S., and (2) the varying governmental infrastructures in the different countries?
-
How well does each strategic plan respond to the particular infectious disease surveillance challenges and the cultural dictates extant in the region in which it is to be implemented?
-
How well are the laboratories working on the other goals of GEIS (response, training and capacity building, and research) in support of their surveillance activities?
The committee will also assess:
-
How well are GEIS program goals, objectives, and activities being carried out domestically, namely, within the military health system (MHS) and the GEIS Central Hub?
THE STUDY PROCESS
The IOM convened a committee, the Committee to Review the Department of Defense Global Emerging Infections Surveillance and Response System, at the request of GEIS management. The study began in April 2000 with a meeting, held in Washington, D.C., to familiarize the committee with GEIS. As part of that meeting, the committee received overview presentations and documentation regarding GEIS laboratory programs and was provided with the 5-year GEIS project plans developed by each of the overseas laboratories (the plans referred to in the committee’s statement of task).
Thereafter, committee members made site visits to each of five DoD overseas laboratories. Between June and October 2000, teams of two to three committee members each visited Naval Medical Research Center Detachment, Peru; Naval Medical Research Unit 2, Indonesia; the Armed Forces Research Institute of Medical Sciences, Thailand; Naval Medical Research Unit 3, Egypt; and the U.S. Army Medical Research Unit, Kenya. The visits lasted approximately 1 week at each laboratory. During the site
visits, committee members toured laboratory facilities, attended briefings and presentations, and met with laboratory staff and collaborators.
The scope of the study was expanded in November 2000 to include review of GEIS activities within the MHS and the Central Hub as well. As part of that review, the committee visited the Naval Health Research Center,5 San Diego, California, and USAMRIID in Washington, D.C. It was not practical to visit all MHS sites hosting GEIS-funded projects. Instead, the committee relied heavily on written documents and summary presentations from and discussions with DoD personnel and collaborators to gain an understanding of these efforts. All told, six site visits and five committee meetings (one of which included a trip to USAMRIID) were held during the course of the study.
APPROACHING THE TASK
As the committee learned more about GEIS, it became increasingly apparent that addressing several elements of the statement of task required careful consideration and interpretation. These are described in the following sections.
Focus on Written Plans
The committee reviewed considerable written background information, including the 5-year operating plans for each overseas laboratory, during the course of its study. The laboratory plans, presented to the committee in April 2000, detail planned GEIS projects at overseas laboratories for fiscal years 2000 through 2004. These plans are informative, but they do not in and of themselves provide a particularly complete, current, or in-practice picture of GEIS activities at the overseas laboratories. Site visits and presentations—contractually specified elements of the study process—provided much richer information. The committee interpreted its charge as intending consideration of all information available, including, but not limited to, information contained in laboratory plan documents.
Similarly, the committee found that its charge left room for interpretation regarding how narrowly or broadly the review should be focused. It was not feasible to conduct a technical review of GEIS-supported projects (such is the level of detail presented in the laboratory plans). The duration of the study (18 months) exceeded the annual GEIS project cycle, rendering any project-specific guidance that the committee might offer
potentially obsolete by the time of the study’s completion. Although timely, scientific evaluation of projects is certainly valuable, the IOM committee process did not seem to provide an appropriate vehicle for this exercise. As such, although the committee considered specific projects in the course of its review, the committee focused its efforts on developing conclusions and recommendations with long-term, program-level relevance rather than on evaluating specific GEIS projects.
Focus on Overseas Laboratories
As noted previously, the initial intent of the study was to provide a review of GEIS implementation at DoD overseas laboratories. The scope of the committee’s task was later expanded to incorporate evaluation of non-overseas laboratory GEIS elements as well, specifically, GEIS activities addressed within the infrastructure of the MHS and through the GEIS Central Hub. The purpose of this task expansion was, as the committee understood it, to permit a complete review of GEIS. Although a task item was added (task f), the wording “overseas laboratory” still permeates the charge. The committee interprets the elements of its task as collectively intending review of GEIS as a whole.
Tools for Review
GEIS is a composite program sustained through a diverse collection of activities carried out by a diverse and diffuse set of consortium members. WHO and CDC standard guidelines for the evaluation of surveillance systems (CDC, 1999, 2000; Klaucke et al., 1988; WHO, 1999a,b, 2001b) exist to guide formal reviews of national and state surveillance systems. They are not appropriate tools for direct application when reviewing GEIS activities at overseas laboratories and other DoD facilities or for use in evaluating GEIS as a whole. The committee interpreted its task as indicating that WHO and CDC guidelines may be used as reference tools but that strict adherence to WHO or CDC surveillance system review formats is not required.
The committee used WHO and CDC guidelines to direct the form and content of its site visits to the overseas laboratories. The type of descriptive information that the committee collected and the approach used to collect it particularly reflect CDC guidance (CDC, 1999, 2000). The overseas laboratory evaluations presented in this report are loosely modeled on WHO-specified surveillance system assessment domains6 (WHO,
1999a, 2001b), which were adapted to reflect the characteristics of GEIS, the unique circumstances of the overseas laboratories, and the requirements and limitations of this review. Furthermore, members of this committee are quite familiar with CDC and WHO surveillance system evaluation procedures and were able to bring this knowledge to bear in identifying information areas to be addressed.
Similarly, the committee interprets its charge as calling for comparison of the GEIS approach to emerging infectious disease surveillance with IOM recommendations7 and CDC strategies at the program level (rather than using GEIS activities at each laboratory as the basis of comparison). It is GEIS as a whole that has an “approach”—presented in the GEIS strategic plan (GEIS, 1998)—to emerging infectious disease surveillance. GEIS activities carried out at DoD laboratories are a part of this approach.
THE FORMAT OF THIS REPORT
Chapters 2 through 6 of this report contain descriptions, conclusions, and recommendations regarding the implementation of GEIS at each of the five DoD overseas laboratories. Conclusions and recommendations are specific to the respective chapters. Chapter 7 of this report presents the committee’s descriptions, conclusions, and recommendations regarding GEIS activities carried out within the MHS infrastructure and through the GEIS Central Hub. The committee’s evaluations of these activities were conducted differently than the overseas laboratory evaluations, and the format of Chapter 7 reflects this. The recommendations and conclusions presented in Chapter 7 reflect summary committee assessments of MHS and Central Hub activities and are specific to that chapter.
Chapter 8 of this report presents summary conclusions and recommendations regarding GEIS as a whole. These are based on what the committee learned during its assessments of the respective parts of GEIS. The conclusions and recommendations presented in Chapter 8 are derived from information presented in Chapters 2 through 7 but are not repetitions of the text in those chapters.