Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
11 Macronutrients and Healthful Diets SUMMARY Acceptable Macronutrient Distribution Ranges (AMDRs) for indi- viduals have been set for carbohydrate, fat, n-6 and n-3 poly- unsaturated fatty acids, and protein based on evidence from interventional trials, with support of epidemiological evidence that suggests a role in the prevention or increased risk of chronic dis- eases, and based on ensuring sufficient intakes of essential nutrients. The AMDR for fat and carbohydrate is estimated to be 20 to 35 and 45 to 65 percent of energy for adults, respectively. These AMDRs are estimated based on evidence indicating a risk for coro- nary heart disease (CHD) at low intakes of fat and high intakes of carbohydrate and on evidence for increased risk for obesity and its complications (including CHD) at high intakes of fat. Because the evidence is less clear on whether low or high fat intakes during childhood can lead to increased risk of chronic diseases later in life, the estimated AMDRs for fat for children are primarily based on a transition from the high fat intakes that occur during infancy to the lower adult AMDR. The AMDR for fat is 30 to 40 percent of energy for children 1 to 3 years of age and 25 to 35 percent of energy for children 4 to 18 years of age. The AMDR for carbo- hydrate for children is the same as that for adultsâ45 to 65 percent of energy. The AMDR for protein is 10 to 35 percent of energy for adults and 5 to 20 percent and 10 to 30 percent for children 1 to 3 years of age and 4 to 18 years of age, respectively. 769
770 DIETARY REFERENCE INTAKES Based on usual median intakes of energy, it is estimated that a lower boundary level of 5 percent of energy will meet the Adequate Intake (AI) for linoleic acid (Chapter 8). An upper boundary for linoleic acid is set at 10 percent of energy for three reasons: (1) individual dietary intakes in the North American population rarely exceed 10 percent of energy, (2) epidemiological evidence for the safety of intakes greater than 10 percent of energy are generally lacking, and (3) high intakes of linoleic acid create a pro-oxidant state that may predispose to several chronic diseases, such as CHD and cancer. Therefore, an AMDR of 5 to 10 percent of energy is estimated for n-6 polyunsaturated fatty acids (linoleic acid). An AMDR for α-linolenic acid is estimated to be 0.6 to 1.2 percent of energy. The lower boundary of the range meets the AI for α-linolenic acid (Chapter 8). The upper boundary corresponds to the highest α-linolenic acid intakes from foods consumed by indi- viduals in the United States and Canada. A growing body of litera- ture suggests that higher intakes of α-linolenic acid, eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA) may afford some degree of protection against CHD. Because the physiological potency of EPA and DHA is much greater than that for α-linolenic acid, it is not possible to estimate one AMDR for all n-3 fatty acids. Approximately 10 percent of the AMDR can be consumed as EPA and/or DHA. No more than 25 percent of energy should be consumed as added sugars. This maximal intake level is based on ensuring sufficient intakes of certain essential micronutrients that are not present in foods and beverages that contain added sugars. A daily intake of added sugars that individuals should aim for to achieve a healthy diet was not set. A Tolerable Upper Intake Level (UL) was not set for saturated fatty acids, trans fatty acids, or cholesterol (see Chapters 8 and 9). This chapter provides some guidance in ways of minimizing the intakes of these three nutrients while consuming a nutritionally adequate diet. INTRODUCTION Unlike micronutrients, macronutrients (fat, carbohydrate, and pro- tein) are sources of body fuel that can be used somewhat interchangeably. Thus, for a certain level of energy intake, increasing the proportion of one macronutrient necessitates decreasing the proportion of one or both of the other macronutrients. The majority of energy is consumed as carbo-
771 M ACRONUTRIENTS AND HEALTHFUL DIETS hydrate (approximately 35 to 70 percent, primarily as starch and sugars), and fat (approximately 20 to 45 percent), while the contribution of protein to energy intake is smaller and less varied (10 to 23 percent) (Appendix Tables E-3, E-6, and E-17). Therefore, a high fat diet (high percent of energy from fat) is usually low in carbohydrate and vice versa. In addition to these macronutrients, alcohol can provide on average up to 3 percent of energy of the adult diet (Appendix Table E-18). A small amount of carbohydrate and as n-6 (linoleic acid) and n-3 (α-linolenic acid) polyunsaturated fatty acids and a number of amino acids that are essential for metabolic and physiological processes, are needed by the brain. The amounts needed, however, each constitute only a small percentage of total energy requirements. Food sources vary in their con- tent of particular macro- and micronutrients. While some nutrients are present in both animal- and plant-derived foods, others are only present or are more abundant in either animal or plant foods. For example, animal-derived foods contain significant amounts of protein, saturated fatty acids, long-chain n-3 polyunsaturated fatty acids, and the micronutrients iron, zinc, and vitamin B12, while plant-derived foods provide greater amounts of carbohydrate, Dietary Fiber, linoleic and α-linolenic acids, and micronutrients such as vitamin C and the B vitamins. It may be difficult to achieve sufficient intakes of certain micronutrients when consuming foods that contain very low amounts of a particular macronutrient. Alternatively, if intake of certain macronutrients from nutrient-poor sources is too high, it may also be difficult to consume sufficient micronutrients and still remain in energy balance. Therefore, a diet containing a variety of foods is considered the best approach to ensure sufficient intakes of all nutrients. This concept is not new and has been part of nutrition education pro- grams since the early 1900s. For example, the first U.S. food guide was developed by the U.S. Department of Agriculture in 1916 and suggested consumption of a combination of five different food groups (Guthrie and Derby, 1998). This food guide has evolved to become known as the Food Guide Pyramid (USDA, 1996). Similarly, Canada has developed Canadaâs Food Guide to Healthy Eating (Health Canada, 1997). A growing body of evidence indicates that an imbalance in macro- nutrients (e.g., low or high percent of energy), particularly with certain fatty acids and relative amounts of fat and carbohydrate, can increase risk of several chronic diseases. Much of this evidence is based on epidemio- logical studies of clinical endpoints such as coronary heart disease (CHD), diabetes, cancer, and obesity. However, these studies demonstrate associa- tions; they do not necessarily infer causality, such as would be derived from controlled clinical trials. Robust clinical trials with specified clinical endpoints are generally lacking for macronutrients. Of importance, fac- tors other than diet contribute to chronic disease, and multifactorial cau-
772 DIETARY REFERENCE INTAKES sality of chronic disease can confound the long-term adverse effects of a given macronutrient distribution. It is not possible to determine a defined level of intake at which chronic disease may be prevented or may develop. For example, high fat diets may predispose to obesity, but at what percent of energy intake does this occur? The answer depends on whether energy intake exceeds energy expenditure or is balanced with physical activity. This chapter reviews the scientific evidence on the role of macro- nutrients in the development of chronic disease. In addition, the nutrient limitations that can occur with the consumption of too little or too much of a particular macronutrient are discussed. In consideration of the inter- relatedness of macronutrients, their role in chronic disease, and their association with other essential nutrients in the diet, Acceptable Macro- nutrient Distribution Ranges (AMDRs) are estimated and represented as percent of energy intake. These ranges represent (1) intakes that are asso- ciated with reduced risk of chronic disease, (2) intakes at which essential dietary nutrients can be consumed at sufficient levels, and (3) intakes based on adequate energy intake and physical activity to maintain energy balance. When intakes of macronutrients fall above or below the AMDR, the risk for development of chronic disease (e.g., diabetes, CHD, cancer) appears to increase. DIETARY FAT AND CARBOHYDRATE There are a number of adverse health effects that may result from consuming a diet that is too low or high in fat or carbohydrate (starch and sugars). Furthermore, chronic consumption of a low fat, high carbohydrate or high fat, low carbohydrate diet may result in the inadequate intake of certain essential nutrients. Low Fat, High Carbohydrate Diets of Adults The chronic diseases of greatest concern with respect to relative intakes of macronutrients are CHD, diabetes, and cancer. In this section, the rela- tionship between total fat and total carbohydrate intakes are considered. Comparisons are made in terms of percentage of total energy intake. For example, a low fat diet signifies a lower percentage of fat relative to total energy. It does not imply that total energy intake is reduced because of consumption of a low amount of fat. The distinction between hypocaloric diets and isocaloric diets is important, particularly with respect to impact on body weight. Low and high fat diets can still be isocaloric. The failure to identify this distinction has led to considerable confusion in terms of the role of dietary fat in chronic disease.
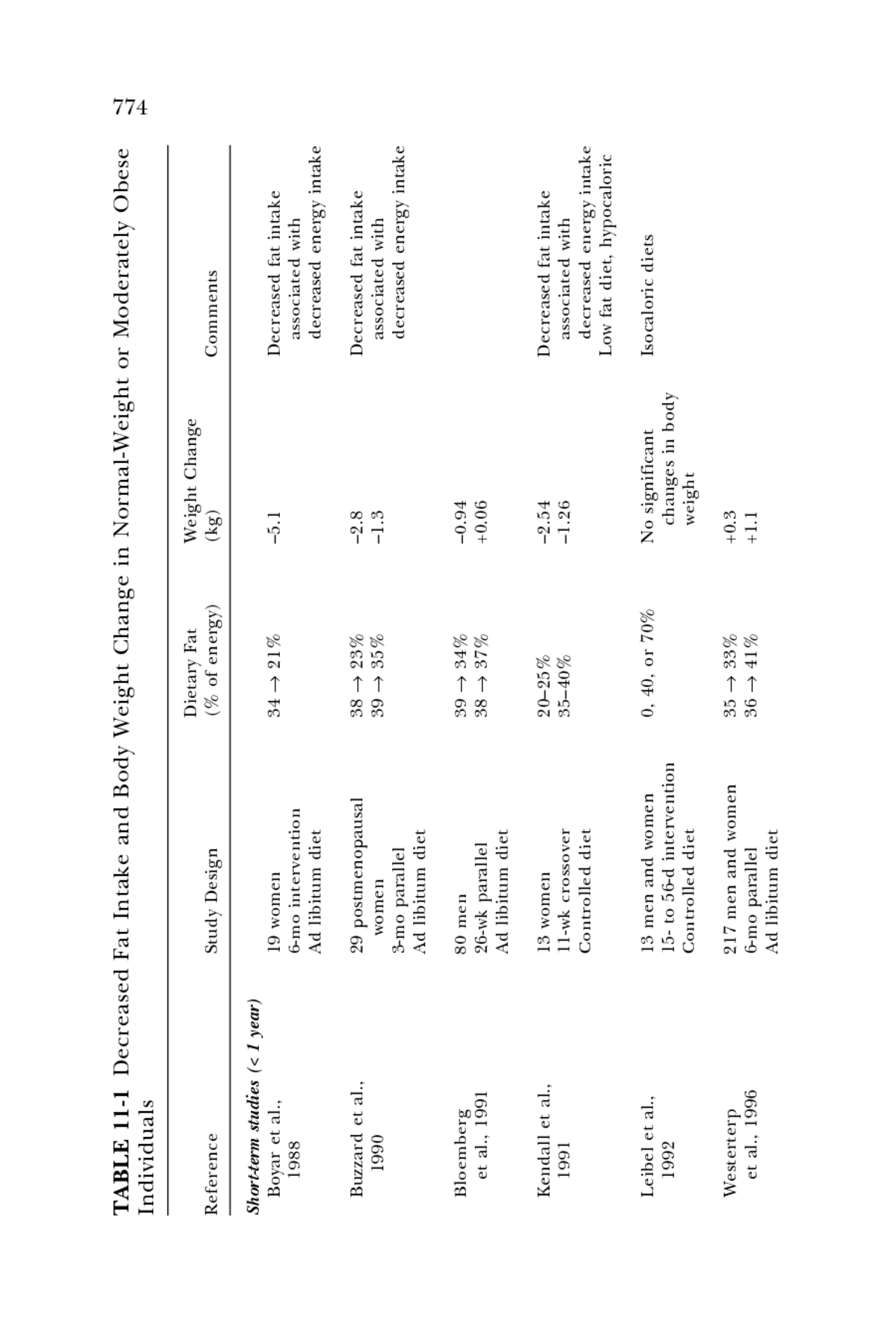
773 M ACRONUTRIENTS AND HEALTHFUL DIETS In the past few decades, the prevalence of overweight and obesity has increased at an alarming rate in many populations, particularly in the United States. Overweight and obesity contribute significantly to various chronic diseases. Consequently, there are two issues to consider for the distribution of fat and carbohydrate intakes in high-risk populations: the distributions that predispose to the development of overweight and obesity, and the distributions that worsen the metabolic consequences in popula- tions that are already overweight or obese. These issues will be considered in the following sections. Maintenance of Body Weight A first issue is whether a certain macronutrient distribution interferes with sufficient intake of total energy, that is, sufficient energy to maintain a healthy weight. Sonko and coworkers (1994) concluded that an intake of 15 percent fat was too low to maintain body weight in women, whereas an intake of 18 percent fat was shown to be adequate even with a high level of physical activity (Jéquier, 1999). Moreover, some populations, such as those in Asia, have habitual very low fat intakes (about 10 percent of total energy) and apparently maintain adequate health (Weisburger, 1988). Whether these low fat intakes and consequent low energy consumptions have con- tributed to a historically small stature in these populations is uncertain. An issue of more importance for well-nourished but sedentary popula- tions, such as that of the United States, is whether the distribution between intakes of total fat and total carbohydrate influences the risk for weight gain (i.e., for development of overweight or obesity). It has been shown that when men and women were fed isocaloric diets containing 20, 40, or 60 percent fat, there was no difference in total daily energy expenditure (Hill et al., 1991). Similar observations were reported for individuals who consumed diets containing 10, 40, or 70 percent fat, where no change in body weight was observed (Leibel et al., 1992), and for men fed diets containing 9 to 79 percent fat (Shetty et al., 1994). Horvath and colleagues (2000) reported no change in body weight after runners consumed a diet containing 16 percent fat for 4 weeks. These studies contain two important findings: fat and carbohydrate provide similar amounts of metabolic energy predicted from their true energy content, and isocaloric diets provide similar metabolic energy expenditure, regardless of their fatâcarbohydrate distribution. In other words, at isocaloric intakes, low fat diets do not produce weight loss. A number of short- and long-term intervention studies have been con- ducted on normal-weight or moderately obese individuals to ascertain the effects of altering the fat and energy density content of the diet on body weight (Table 11-1). In general, significant reductions in the percent of
TABLE 11-1 Decreased Fat Intake and Body Weight Change in Normal-Weight or Moderately Obese 774 Individuals Dietary Fat Weight Change Reference Study Design (% of energy) (kg) Comments Short-term studies (< 1 year) Boyar et al., 19 women â5.1 Decreased fat intake 34 â 21% 1988 6-mo intervention associated with Ad libitum diet decreased energy intake Buzzard et al., 29 postmenopausal â2.8 Decreased fat intake 38 â 23% 1990 women â1.3 associated with 39 â 35% 3-mo parallel decreased energy intake Ad libitum diet Bloemberg 80 men â0.94 39 â 34% et al., 1991 26-wk parallel +0.06 38 â 37% Ad libitum diet Kendall et al., 13 women 20â25% â2.54 Decreased fat intake 1991 11-wk crossover 35â40% â1.26 associated with Controlled diet decreased energy intake Low fat diet, hypocaloric Leibel et al., 13 men and women 0, 40, or 70% No significant Isocaloric diets 1992 15- to 56-d intervention changes in body Controlled diet weight Westerterp 217 men and women +0.3 35 â 33% et al., 1996 6-mo parallel +1.1 36 â 41% Ad libitum diet
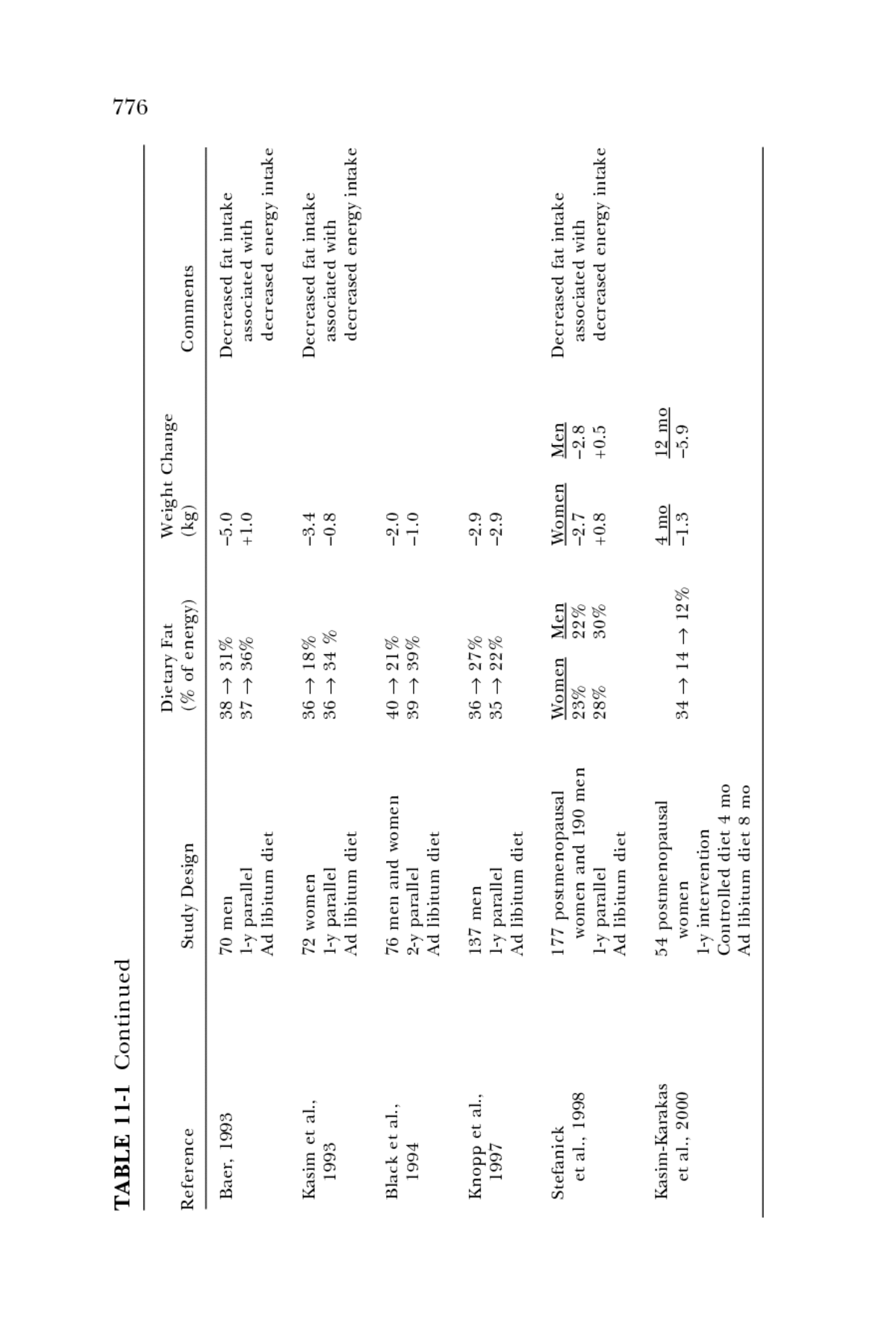
Raben et al., 11 women â0.7 Decreased fat intake 46 â 28% 1997 14-d crossover associated with Ad libitum decreased energy intake Gerhard et al., 22 women 20% â1.1 Low fat diet, hypocaloric 2000 4-wk crossover 40% â0.3 Controlled diet Saris et al., 398 men and women â0.9 Decreased fat intake 36 â 26% 2000 6-mo parallel â1.8 associated with 36 â 28% Ad libitum diet +0.8 decreased energy intake 36 â 37% Long-term studies (⥠1 year) 6 mo 12 mo Decreased fat intake Lee-Han et al., 57 women 1988 1-y parallel â1.16 â0.93 associated with 36 â 23 â 26% Ad libitum diet +0.07 +0.62 decreased energy intake 36 â 34 â 36% Boyd et al., 206 women â1.0 37 â 21% 1990 1-y parallel 0 37 â 35% Ad libitum diet Sheppard et al., 276 women 0 to 1 y Decreased fat intake 1991 1- and 2-y parallel â3.0 associated with 39 â 22% Ad libitum diet â0.4 decreased energy intake 39 â 37% 1 y to 2 y +1.1 22 â 23% continued 775
TABLE 11-1 Continued 776 Dietary Fat Weight Change Reference Study Design (% of energy) (kg) Comments Baer, 1993 70 men â5.0 Decreased fat intake 38 â 31% 1-y parallel +1.0 associated with 37 â 36% Ad libitum diet decreased energy intake Kasim et al., 72 women â3.4 Decreased fat intake 36 â 18% 1993 1-y parallel â0.8 associated with 36 â 34 % Ad libitum diet decreased energy intake Black et al., 76 men and women â2.0 40 â 21% 1994 2-y parallel â1.0 39 â 39% Ad libitum diet Knopp et al., 137 men â2.9 36 â 27% 1997 1-y parallel â2.9 35 â 22% Ad libitum diet Women Men Women Men Decreased fat intake Stefanick 177 postmenopausal et al., 1998 women and 190 men 23% 22% â2.7 â2.8 associated with 1-y parallel 28% 30% +0.8 +0.5 decreased energy intake Ad libitum diet Kasim-Karakas 54 postmenopausal 4 mo 12 mo et al., 2000 women â1.3 â5.9 34 â 14 â 12% 1-y intervention Controlled diet 4 mo Ad libitum diet 8 mo
777 M ACRONUTRIENTS AND HEALTHFUL DIETS energy consumed as fat (greater than 4 percent) resulted in small losses in body weight. The only study that provided isocaloric diets showed no dif- ferences in weight gain or loss, despite a wide range in the percent of energy from fat (Leibel et al., 1992). Four meta-analyses of long-term intervention studies associating a low fat diet with body weight concluded that lower fat diets lead to modest weight loss or prevention of weight gain (Astrup et al., 2000; Bray and Popkin, 1998; Hill et al., 2000; Yu-Poth et al., 1999). These studies thus suggest that low fat diets (low percentage of fat) tend to be slightly hypocaloric compared to higher fat diets when com- pared in outpatient intervention trials. The finding that higher fat diets are moderately hypercaloric when compared with reduced fat intakes under ad libitum conditions provides a rationale for setting an upper boundary for percentage of fat intake in a population that already has a high prevalence of overweight and obesity. However, a second issue must also be addressed: whether the distribution of fat and carbohydrate modifies the metabolic consequences of over- weight and obesity. Two of the more important consequences of obesity are dyslipidemic changes in serum lipoproteins (which predispose to CHD) and changes in glucose and insulin metabolism that accentuate an under- lying insulin resistance (which may predispose to both CHD and diabetes). These consequences are discussed in the following sections. Risk of CHD Low fat, high carbohydrate diets, compared to higher fat intakes, can induce a lipoprotein pattern called the atherogenic lipoprotein pheno- type (Krauss, 2001) or atherogenic dyslipidemia (National Cholesterol Education Program, 2001). In populations where people are routinely physically active and lean, the atherogenic lipoprotein phenotype is mini- mally expressed. In sedentary populations that tend to be overweight or obese, very low fat, high carbohydrate diets clearly promote the develop- ment of this phenotype. Whether this phenotype promotes development of coronary atherosclerosis when it is specifically induced by low fat diets is uncertain, but it is a pattern that is associated with increased risk for CHD when expressed in the general American population. The atherogenic lipoprotein phenotype is characterized by higher triacylglycerol and decreased high density lipoprotein (HDL) cholesterol concentrations and small low density lipoprotein (LDL) particles. A predominance of small LDL particles is associated with a greater risk of CHD (Austin et al., 1990), but it is not known if this association is independent of increased triacylglycerol and decreased HDL cholesterol concentrations. Table 11-2 and Figures 11-1 and 11-2 show that with decreasing fat and increasing carbohydrate intake, plasma triacylglycerol concentrations
778 DIETARY REFERENCE INTAKES TABLE 11-2 Fat and Carbohydrate Intake and Blood Lipid Concentrations in Healthy Individuals Total Fat/ Carbohydrate Intake Study Designa Reference (% of energy) Coulston et al., 11 men and women 1983 10-d crossover 21 P/S = 1.2â1.3 41 Bowman et al., 19 men 29/60 1988 10-wk parallel 33/58 P/S = 0.4 45/42 46/42 Borkman et al., 8 men and women 20/55 P/S = 0.46 1991 3-wk crossover 50/31 P/S = 0.22 Kasim et al., 72 women 18 1993 1-y parallel 34 P/S = 0.68â0.75 Leclerc et al., 7 men and women 11/64 1993 7-d crossover 30/45 40/45 Krauss and 105 men 24/60 Dreon, 1995 6-wk crossover 46/39 P/S = 0.69â0.74 OâHanesian 10 men and women 17/63 P/S = 0.25 et al., 1996 10-d crossover 28/57 P/S = 2.2 42/39 P/S = 1.7 Jeppesen et al., 10 postmenopausal 25/60 1997 women 45/40 3-wk crossover P/S = 1.0 Kasim-Karakas 14 postmenopausal 14 P/S = 1.2 et al., 1997 women 23 P/S = 1.0 4-mo intervention 31 P/S = 0.9 Yost et al., 25 men and women 25/55 1998 15-d crossover 50/30 P/S = 0.3 Straznicky 14 men 25/54 P/S = 1.3 et al., 1999 2-wk crossover 47/36 P/S = 0.1 Kasim-Karakas 54 postmenopausal 12/71 et al., 2000 women 14/69 4- to 12-mo 34/50 crossover P/S = 0.64
779 M ACRONUTRIENTS AND HEALTHFUL DIETS Postintervention Blood Lipid Concentration (mmol/L)b Triacylglycerol HDL-C LDL-C 1.51c 0.98c 1.02d 1.16d 0.91c 1.42c 2.35c 1.11c 1.22c 2.17c 0.84c 1.53c 2.59c 1.01c 1.50c 2.40c 0.82c (+49%) 0.84c (â24%) 2.88c (â20%) 0.55c 1.10d 3.60d 1.35c 1.44c (â8%) 2.79c (â10%) 1.25d 1.56d 3.09d 1.11c 1.03c 2.29c 1.29c 1.15d 2.47c 0.87d 1.32e 3.05d 1.59c 1.09c 3.26c 1.13d 1.27d 3.69d 0.8 1.1 2.4 0.8 1.2 2.5 0.8 1.3 3.0 1.97c 1.38c 2.74c 1.29d 1.49d 2.81c 2.47c 1.24c 2.61c 2.10d 1.32d 2.93d 1.85e 1.34d 2.89d 1.14c 1.22c 0.88d 1.30d 0.8c 1.05c 2.6c 0.8c 1.28d 3.5d 1.49c 1.40c 3.49c 2.00c 1.29c 3.18c 1.57c 1.53d 3.57c continued
780 DIETARY REFERENCE INTAKES TABLE 11-2 Continued Total Fat/ Carbohydrate Intake Study Designa Reference (% of energy) Marckmann 20 women 28/59 P/S = 0.7 et al., 2000 2-wk crossover 46/41 P/S = 0.4 Obarzanek 459 men and 27/58 P/S = 1.1 et al., 2001b women, 8-wk 37/52 P/S = 0.5 parallel a P/S = polyunsaturated/saturated fatty acid ratio. b H DL-C = high density lipoprotein cholesterol, LDL-C = low density lipoprotein cholesterol. FIGURE 11-1 Relationship between percent of total fat intake and change in triacylglycerol (TAG) ( ) and high density lipoprotein (HDL) cholesterol (- - - ) concentrations. Regression equations for percent change in serum TAG and HDL cholesterol predicted by percent total fat in the experimental diets of controlled- feeding studies comparing low fat, high carbohydrate diets to high fat diets. Weighted least-squares regression analyses were performed using the mixed procedure to test for differences in lipid concentrations (SAS Statistical package, version 8.00, SAS Institute, Inc., 1999). Percent of energy from total fat varied from 18.3% to 50%. All diets were low in saturated fat (less than 10% energy). Using these equa- tions, for every 5% decrease in total fat, HDL cholesterol would decrease by 2.2% and triacylglycerol would increase by 6%. DATA SOURCES: Berry et al. (1992); Curb et al. (2000); Garg et al. (1988, 1992a, 1994); Ginsberg et al. (1990); Grundy (1986); Grundy et al. (1988); Jansen et al. (1998); Kris-Etherton et al. (1999); Lefevre et al., unpublished; Lopez-Segura et al. (1996); Mensink and Katan (1987); Nelson et al. (1995); Parillo et al. (1992); Pelkman et al. (2001); Perez-Jimenez et al. (1995, 1999, 2001).
781 M ACRONUTRIENTS AND HEALTHFUL DIETS Postintervention Blood Lipid Concentration (mmol/L)b Triacylglycerol HDL-C LDL-C 0.81 c 1.34 c 2.43 c 0.70 d 1.56 d 2.71 d +0.4 â0.09 â0.29 â0.09 â0.005 â0.05 c,d,e Within each study, LDL-C, HDL-C, or Lp(a) concentrations that are significantly different between treatment groups have a different superscript. Serum HDL Cholesterol Concentration (mmol/L) mg/dL Proportion of Energy Derived from Carbohydrates (%) FIGURE 11-2 Relationship between proportion of energy from carbohydrates and serum high density lipoprotein (HDL) cholesterol concentration. ⢠= Mean values for approximately 120 boys from five countries, o = individuals values for boys from the Philippines, FI= Finland, NE = Netherlands, GH = Ghana, IT = Italy, PH = Philippines. SOURCE: Knuiman et al. (1987).
782 DIETARY REFERENCE INTAKES 25 y = 0.578x + 14.787 20 2 R = 0.3779 Percent Changes in TC:HDL-C Ratios 15 10 5 0 -5 -10 -15 -20 -25 10 15 20 25 30 35 40 45 50 55 Dietary Total Fat (% energy) FIGURE 11-3 Relationship between total fat intake and change in total cholester- ol (TC):high density lipoprotein (HDL) cholesterol ratio. Weighted least-squares regression analyses were performed using the mixed procedure to test for differ- ences in lipid concentrations (SAS Statistical package, version 8.00, SAS Institute, Inc., 1999). DATA SOURCES: Berry et al. (1992); Curb et al. (2000); Garg et al. (1988, 1992a, 1994); Ginsberg et al. (1990); Grundy (1986); Grundy et al. (1988); Jansen et al. (1998); Kris-Etherton et al. (1999); Lefevre et al., unpublished; Lopez-Segura et al. (1996); Mensink and Katan (1987); Nelson et al. (1995); Parillo et al. (1992); Pelkman et al. (2001); Perez-Jimenez et al. (1995, 1999, 2001). increase and plasma HDL cholesterol concentrations decrease. The reduc- tion in HDL cholesterol concentration with low fat intake results in a rise in the total:HDL cholesterol concentration ratio (Figure 11-3). The total:HDL cholesterol ratio has been shown to be an important risk factor for CHD (Castelli et al., 1992; Kannel, 2000). Whether diet-induced changes in the total:HDL cholesterol ratio predispose to CHD remains unclear (Brussard et al., 1982; Jeppesen et al., 1997; Krauss and Dreon, 1995; West et al., 1990; Yost et al., 1998). In support of the interventional studies, carbohydrate intake is nega- tively associated with HDL cholesterol concentrations (Table 11-3). None- theless, the association between atherogenic lipoprotein phenotype (higher
TABLE 11-3 Epidemiological Studies on Carbohydrate Intake and Blood Lipid Concentrations Low Density Lipoprotein (LDL) High Density Lipoprotein Cholesterol (HDL) Cholesterol Triacylglycerol Reference Study Design Concentration Concentration Concentration Ernst et al., 4,855 men and women Inversely related to 1980 Cross-sectional carbohydrate intake Knuiman et al., Multicountry regression Inversely related to 1987 analysis carbohydrate intake Fehily et al., 653 men No association Negative association No association 1988 Cross-sectional between carbohydrate regression analysis intake and HDL concentration West et al., 719 boys Decreased with Decreased with increased Increased with 1990 Multicountry regression increased carbohydrate intake increased analysis carbohydrate carbohydrate intake intake Tillotson et al., Prospective cohort, 1997 6-y follow-up < 29% carbohydrate 4.18 1.13 2.11 29â36% carbohydrate 4.13 1.11 2.26 36â41% carbohydrate 4.13 1.09 2.23 41â48% carbohydrate 4.11 1.07 2.25 > 48% carbohydrate 4.14 1.05 2.13 783
784 DIETARY REFERENCE INTAKES total:HDL cholesterol ratios) and CHD risk provides one rationale for establishing a lower boundary for the Acceptable Macronutrient Distribu- tion Range (AMDR) for high-risk populations. Risk of Hyperinsulinemia, Glucose Intolerance, and Type 2 Diabetes Other potential abnormalities accompanying changes in distribution of fat and carbohydrate intakes include increased postprandial responses in plasma glucose and insulin concentrations. These abnormalities are more likely to occur with low fat, high carbohydrate diets. They potentially could be related to the development of both type 2 diabetes and CHD. In particular, repeated daily elevations in postprandial glucose and insulin concentrations could âexhaustâ pancreatic β-cells of insulin supply, which could hasten the onset of type 2 diabetes. Some investigators have further suggested these repeated elevations could worsen baseline insulin sensitivity, which could cause susceptible persons to be at increased risk for type 2 diabetes. This form of diabetes, defined by an elevation of fasting serum glucose concentration, is characterized by two defects in glucose metabolism: insulin resistance, a defect in insulin-mediated uptake of glucose by cells, particularly skeletal muscle cells, and a decline in insulin secretory capacity by pancreatic β-cells (Turner and Clapham, 1998). Insulin resistance typi- cally precedes the development of type 2 diabetes by many years. It is known to be the result of obesity, physical inactivity, and genetic factors (Turner and Clapham, 1998). Before the onset of diabetic hyperglycemia, the pancreatic β-cells are able to respond to insulin resistance with an increased insulin secretion, enough to maintain normoglycemia. However, in some persons who are insulin resistant, insulin secretory capacity declines and hyperglycemia ensues (Reaven, 1988, 1995). The mechanisms for the decline in insulin secretion are not well understood, but one theory is that continuous overstimulation of insulin secretion by the presence of insulin resistance leads to âinsulin exhaustionâ and hence to decreased insulin secretory capacity (Turner and Clapham, 1998). Whether insulin exhaustion is secondary to a metabolic dysfunction of cellular production of insulin or to a loss of β-cells is uncertain. The accumulation of pancreatic islet-cell amyloidosis may be one mechanism for loss of insulin-secretory capacity (Höppener et al., 2000). High carbohydrate diets frequently causes greater insulin and plasma glucose responses than do low carbohydrate diets (Chen et al., 1988; Coulston et al., 1987). These excessive responses theoretically could pre- dispose individuals to the development of type 2 diabetes because of pro- longed overstimulation of insulin secretion (Grill and Björklund, 2001). The reasoning is similar to that for insulin resistance, namely, excessive stimulation of insulin secretion over a period of many years could result in
785 M ACRONUTRIENTS AND HEALTHFUL DIETS insulin exhaustion, and hence to hyperglycemia (Turner and Clapham, 1998). This mechanism, although plausible, remains hypothetical. None- theless, in the mind of some investigators, it deserves serious consideration. Other consequences of hyperglycemic responses to high carbohydrate diets might be considered. For example, higher postprandial glucose responses might lead to other changes such as âdesensitizationâ of β-cells for insulin secretion and production of glycated products or advanced glycation end-products, which could either promote atherogenesis or the âagingâ process (Lopes-Virella and Virella, 1996). Again, these are hypo- thetical consequences that need further examination. Epidemiological Evidence. A number of noninterventional, epidemio- logical studies have shown no relationship between carbohydrate intake and risk of diabetes (Colditz et al., 1992; Lundgren et al., 1989; Marshall et al., 1991; Meyer et al., 2000; Salmerón et al., 1997), whereas other studies have shown a positive association (Bennett et al., 1984; Feskens et al., 1991a). Interventional Evidence. Interventional studies in healthy individuals on the influence of high carbohydrate diets on biomarker precursors for type 2 diabetes are lacking and the available data are mixed (Table 11-4) (Beck- Nielsen et al., 1980; Chen et al., 1988; Dunnigan et al., 1970; Fukagawa et al., 1990; Rath et al., 1974; Reiser et al., 1979). Factors such as carbo- hydrate quality, body weight, exercise, and genetics make the interpretation of such findings difficult. Nonetheless, in overweight and sedentary groups (which carry a heavy burden of insulin resistance and are common in North America), the accentuation of postprandial glucose and insulin concentrations that accompany high carbohydrate diets are factors to con- sider when setting an upper boundary for AMDRs for dietary carbohydrate (and a lower boundary for dietary fat). Risk of Nutrient Inadequacy or Excess Diets Low in Fats. For usual diets that are low in total fat, the intake of essential fatty acids, such as n-6 polyunsaturated fatty acids, will be low (Appendix K). In general, with increasing intakes of carbohydrate and decreasing intakes of fat, the intake of n-6 polyunsaturated fatty acids decreases. Furthermore, low intakes of fat are associated with low intakes of zinc and certain B vitamins. The digestion and absorption of fat-soluble vitamins and provitamin A carotenoids are associated with fat absorption. Jayarajan and coworkers (1980) reported that the addition of 5 or 10 g of fat to a low fat (5 g) diet
786 DIETARY REFERENCE INTAKES TABLE 11-4 Intervention Studies on Carbohydrate Intake and Biochemical Indicators of Diabetes Reference Study Design Dunnigan et al., 9 men and women 1970 4-wk crossover 31% sucrose Sucrose-free Rath et al., 6 men 1974 2- to 5-wk crossover 17% sucrose 52% sucrose Reiser et al., 19 men and women 1979 6-wk crossover 30% starch 30% sucrose Beck-Nielsen 7-d intervention et al., 1980 Normal diet + 250 g glucose Normal diet + 250 g fructose Chen et al., 8 men 1988 3- to 5-d crossover 85% carbohydrate 41% carbohydrate 30% carbohydrate Lundgren et al., 1,462 women, 1989 Prospective cohort, 12-y follow-up Fukagawa et al., 6 men 1990 21- to 28-d intervention 40% carbohydrate 69% carbohydrate a,b,c Within each study, the indicators of diabetes that are significantly different be- tween treatment groups have a different superscript.
787 M ACRONUTRIENTS AND HEALTHFUL DIETS Results No diet effect on glucose tolerance and plasma insulin Serum insulin (µg/mL) Serum glucose (mg/dL) 5.4 a 87.0a 11.8b 81.1b Serum insulin (µ munits/mL) Serum glucose (mg/dL) 9.8 a 92.5a 11.9b 94.5a No significant difference in insulin concentrations The high fructose diet was accompanied by a significant reduction in insulin binding and insulin sensitivity Glucose disappearance Insulin sensitivity index (%/min) 5.6a 2.2a 6.1b 2.3b 3.9a,c 1.6a,c 5.6a 2.2a 6.1b 2.3b 3.9a,c 1.6a,c Carbohydrate intake of women who developed diabetes (212 g/d) was not significantly different than women who did not develop diabetes (228 g/d) Glucose disposal Serum insulin (pmol/L) (µmol/kg/min) 67.4a 21.2a 50.2b 27.8b
788 DIETARY REFERENCE INTAKES significantly improved serum vitamin A concentrations. However, the addi- tion of 10 g compared to 5 g did not provide any further benefit. The level of dietary fat has also been shown to improve vitamin K2 bioavailability (Uematsu et al., 1996). Doseâresponse data are limited on the amount of dietary fat needed to achieve the optimal absorption of fat-soluble vitamins, but it appears that the level is quite low. Diets High in Fiber. Most diets that are high in fiber are also high in carbohydrate . High fiber diets have the potential for reduced energy density, reduced energy intake, and poor growth. However, poor growth is unlikely in the United States where most children consume adequate energy and fiber intake is relatively low (Williams and Bollella, 1995). Miles (1992) tested the effects of daily ingestion of 64 g or 34 g of Dietary Fiber for 10 weeks in healthy adult males. The ingestion of 64 g/d of Dietary Fiber resulted in a reduction in protein utilization from 89.4 to 83.7 percent and in fat utilization from 95.5 to 92.5 percent. Total energy utilization decreased from 94.3 to 91.4 percent. Because most individuals consuming high amounts of fiber would also be consuming high amounts of energy, the slight depression in energy utilization is not significant (Miles, 1992). In other studies, ingestion of high amounts of fruit, vegetable, and cereal fiber (48.3 to 85.6 g/d) also resulted in decreases in apparent digestibilities of energy, crude protein, and fat (Göranzon et al., 1983; Wisker et al., 1988). Again, however, the Dietary Fiber intakes were very high, and because the recommendation for Total Fiber intake is related to energy intake, the high fiber consumers would also be high energy consumers. Diets High in Added Sugars. Increased consumption of added sugars can result in decreased intakes of certain micronutrients (Table 11-5). This can occur because of the abundance of added sugars in energy-dense, nutrient-poor foods, whereas naturally occurring sugars are primarily found in fruits, milk, and dairy products that also contain essential micro- nutrients. Because some micronutrients (e.g., vitamin B6, vitamin C, and folate), dietary fiber, and phytochemicals were not examined, the association between these nutrients and added sugars intakes is not known. Bowman (1999) used data from Continuing Survey of Food Intakes of Individuals (CSFII) (1994â1996) to assess the relationship between added sugars and intakes of essential nutrients in Americansâ diets. The sample (n = 14,704) was divided into three groups based on the percentage of energy consumed from added sugars: (1) less than 10 percent of total energy (n = 5,058), (2) 10 to 18 percent of total energy (n = 4,488), and (3) greater than 18 percent of total energy (n = 5,158). Group 3, with a mean of 26.7 percent of energy from added sugars, had the lowest absolute mean intakes of all
789 M ACRONUTRIENTS AND HEALTHFUL DIETS the micronutrients, especially vitamin A, vitamin C, vitamin B12, folate, calcium, phosphorus, magnesium, and iron. Compared with Groups 1 and 2, a decreased percentage of people in Group 3 met their Recommended Dietary Allowance (RDA) for many micronutrients. The individuals in Group 3 did not meet the 1989 RDA for vitamin E, vitamin B6, calcium, magnesium, and zinc. In addition, the high sugar consumers (Group 3) had lower intakes of grains, fruits, vegetables, meat, poultry, and fish com- pared with Groups 1 and 2. At the same time, Group 3 consumed more soft drinks, fruit drinks, punches, ades, cakes, cookies, grain-based pastries, milk desserts, and candies. Similar trends were also reported by Bolton- Smith and Woodward (1995) and Forshee and Storey (2001), but were not observed by Lewis and coworkers (1992). Emmett and Heaton (1995) reported an overall deterioration in the quality of the diet in heavy users of added sugars. Using 1990â1991 cross-sectional data, Guthrie (1996) found that women whose diets met their RDA for calcium consumed significantly more milk products, fruit, and grains, and less regular soft drinks than women who did not meet their calcium recommendations. Others have shown that intakes of soft drinks are negatively related to intakes of milk (Guenther, 1986; Harnack et al., 1999; Skinner et al., 1999). To further look at the association between added sugars and certain micronutrient intakes, the median intakes of various micronutrients at every 5th percentile of added sugars intake was determined using data from the Third National Health and Nutrition Examination Survey (NHANES III) (Appendix J). In addition, the prevalence of subpopulations not meeting the Estimated Average Requirement (EAR) or exceeding the Adequate Intake (AI) for these micronutrients was determined. Because not all micronutrients and other nutrients, such as fiber, were evaluated, it is not known what the association is between added sugars and these nutrients. While the trends are not consistent for all age groups, reduced intakes of calcium, vitamin A, iron, and zinc were observed with increasing intakes of added sugars, particularly at intake levels exceeding 25 percent of energy. Although this approach has limitations, it gives guidance for the planning of healthy diets. Diets High in Total Sugars. In one large dietary survey, linear reductions were observed for certain micronutrients when total sugars intakes increased (Bolton-Smith and Woodward, 1995), whereas no consistent reductions were observed in another survey (Gibney et al., 1995) (Table 11-6). Bolton- Smith (1996) reviewed the literature on the relation of sugars intake to micronutrient adequacy and concluded that, provided consumption of sugars is not excessive (defined as less than 20 percent of total energy intake), no health risks are likely to ensue due to micronutrient inadequacies.
790 DIETARY REFERENCE INTAKES TABLE 11-5 Survey Data on Added Sugars and Micronutrient Intake Study Reference Population/Survey Diet Information Nelson, 1991 143 children, 11â12 y 7-d weighed diet record Rugg-Gunn et al., 405 children, 11â14 y 3-d diet record 1991 Lewis et al., Nationwide Food 1992 Consumption Survey (1977â1978) Bolton-Smith and 11,626 men and women, Food frequency Woodward, 1995 25â64 y questionnaire Scottish Heart Health and MONICA studies Gibson, 1997 1,675 boys and girls, 4-d weighed diet 1.5â4.5 y record U.K. National Diet and Nutrition Survey of Children Bowman, 1999 Continuing Survey of Two 24-h recalls Food Intakes by Individuals (1994â1996) Forshee and Storey, Continuing Survey of 2001 Food Intakes by Individuals (1994â1996)
791 M ACRONUTRIENTS AND HEALTHFUL DIETS Added Sugars Intake (% of energy) Change in Micronutrient Intake 16 Decrease in nicotinic acid for girls 21 27 10 Decrease in vitamin D, protein 20 Percentile of intake 26thâ75th > 75th Decrease in calcium Men: 1.0â6.2, 6.3â8.9, Linear reduction in vitamin E, vitamin C, and 9.0â13.0, 13.1â15.7, vitamin A for both men and women 15.8â47.9 Women: 0.8â4.8, 4.9â6.3, 6.4â8.1, 8.2â11.6, 11.7â50.2 < 12 12â16 Decrease in zinc, calcium, riboflavin 16â20 20â25 > 25 Decrease in niacin, thiamin; large decrease in calcium, zinc, riboflavin < 10 10â18 Decrease in calcium > 18 Decrease in vitamin A, vitamin E, vitamin C, niacin, vitamin B6, folate, vitamin B12, phosphorus, magnesium, iron, zinc, copper; large decrease in calcium Negative correlation between added sugar intake and intake of vitamin A, calcium, and folate
792 DIETARY REFERENCE INTAKES TABLE 11-6 Survey Data on Total Sugar and Micronutrient Intake Study Reference Population/Survey Diet Information Gibson, 1993 2,705 children 7-d weighed food Department of Health record Survey of British School Children Bolton-Smith and 11,626 men and women, Food frequency Woodward, 1995 25â64 y questionnaire Scottish Heart Health and MONICA studies Gibney et al., 1995 8,296 men and women 3-d food record Nationwide Food Consumption Survey (1987â1988) Nicklas et al., 1996 568 children, 10 y 24-h dietary recall Bogalusa Heart Study Farris et al., 1998 568 children, 10 y 24-h dietary recall Bogalusa Heart Study The impact of total sugar intake on the intake of micronutrients does not appear to be as great as for added sugars. Furthermore, a preliminary analysis of data from NHANES III on the intake of various micronutrients at every 5th percentile of total sugar intake did not reveal any significant associations as was observed for added sugars (Appendix J). High Fat, Low Carbohydrate Diets of Adults Risk of Obesity Epidemiological Evidence. Cross-country epidemiological data of dietary fat intake and obesity have yielded mixed results (Bray and Popkin, 1998;
793 M ACRONUTRIENTS AND HEALTHFUL DIETS Total Sugar Intake (% of energy) Change in Micronutrient Intake < 20.7 Decrease in iron, nicotinic acid 20.7â25.2 Large decrease in iron, nicotinic acid > 25.2 No marked changes in micronutrient intake Men: 2.5â12.0, 12.1â14.7, Linear reduction in vitamin E, retinol, and 14.8â17.2, 17.3â20.7, vitamin A intake 20.8â51.4 Women: 1.5â11.7, 11.8â14.1, Linear reduction in vitamin E, retinol, carotene, 14.2â16.3, 16.4â19.6, and vitamin A intake 19.7â52.8 < 10 10â24 Decrease in riboflavin, thiamin, calcium, iron, zinc, vitamin A Decrease in vitamin B6, vitamin E > 24 18.0 22.1 26.4 31.2 Decrease in percent meeting the Recommended Dietary Allowance for niacin and zinc Linear reduction in vitamin B6, vitamin E, 16.1 23.5 thiamin, iron, zinc, and niacin intake with 28.2 increasing total sugar intake 35.6 Willett, 1998). In some countries, low fat, high carbohydrate diets are asso- ciated with a low prevalence of obesity, whereas in others they are not. Within-country surveys of dietary intake and body mass index (BMI) have also yielded mixed results. Many case-control and prospective studies failed to find a strong correlation between percent of energy intake from fat and body weight (Heitmann et al., 1995; Lissner et al., 2000; Ludwig et al., 1999b; Rissanen et al., 1991; Samaras et al., 1998; Willett, 1998), whereas some did find significant associations (Bray and Popkin, 1998; Dreon et al., 1988; George et al., 1990; Klesges et al., 1992; Miller et al., 1990; Romieu et al., 1988; Tucker and Kano, 1992). Colditz and coworkers (1990) observed no association between fat intake and weight gain prospectively, but did find a positive association between previous weight
794 DIETARY REFERENCE INTAKES gain and high fat intake. One statistically well-designed study that included direct measurements of body fat and considered potentially confounding factors such as exercise concluded that total dietary fat was positively cor- related with fat mass (adjusted for fat-free mass, r = 0.22, p < 0.0001) in adults (Larson et al., 1996). Most multiple regression studies found that about 3 percent of the total variance in body fatness was explained by diet, though some studies placed the estimate at 7 to 8 percent (Westerterp et al., 1996). Longitudinal studies generally supported dietary fat as a predic- tive factor in the development of obesity (Lissner and Heitmann, 1995). However, bias in subject participation, retention, and underreporting of intake may limit the power of these epidemiological studies to assess the relationship between dietary fat and obesity or weight gain (Lissner et al., 2000). Another line of evidence often cited to indicate that dietary fat is not an important contributor to obesity is that although there has been a reduction in the percent of energy from fat consumed in the United States, there has been an increase in energy intake and a marked gain in average weight (Willett, 1998). Survey data showed an increase in total energy intake over this period (McDowell et al., 1994), so that despite the decline in percent of energy from fat, the total intake of fat (g/d) remained stable. Another study that used food supply data showed that fat intake may indeed be rising in the United States (Harnack et al., 2000). Mechanisms for Obesity and Interventional Evidence. Several mechanisms have been proposed whereby high fat intakes could lead to excess body accumulation of fat. Foods containing high amounts of fat tend to be energy dense, and the fat is a major contributor to the excess energy con- sumed by persons who are overweight or obese (Prentice, 2001). The energy density of a food can be defined as the amount of metabolizable energy per unit weight or volume (Yao and Roberts, 2001); water and fat are the main determinants of dietary energy density. Energy density is an issue of interest to the extent that it influences energy intake and thus plays a role in energy regulation, weight maintenance, and the subsequent development of obesity. Three theoretical mechanisms have been identified by which dietary energy density may affect total energy intake and hence energy regulation (Yao and Roberts, 2001). Some studies suggest that, at least in the short- term, individuals tend to eat in order to maintain a constant volume of food intake because stomach distension triggers vagal signals of fullness (Duncan et al., 1983; Lissner et al., 1987; Seagle et al., 1997; Stubbs et al., 1995a). Thus, consumption of high energy-dense foods could lead to excess energy intake due to the high energy density to small food volume ratio.
795 M ACRONUTRIENTS AND HEALTHFUL DIETS A second proposed mechanism is that high energy-dense foods are often more palatable than low energy-dense foods (Drewnowski, 1999; Drewnowski and Greenwood, 1983). A survey of American adults reported that taste is the primary influence for food choice (Glanz et al., 1998). In single-meal studies, high palatability was also associated with increased food consumption (Bobroff and Kissileff, 1986; Price and Grinker, 1973; Yeomans et al., 1997). These results suggest that high energy-dense foods may be overeaten because of effects related to their high palatability. The third mechanism is that energy-dense foods reduce the rate of gastric emptying (Calbet and MacLean, 1997; Wisen et al., 1993). This reduction, however, does not occur proportionally to the increase in energy density. Although energy-dense foods reduce the rate at which food leaves the stomach, they actually increase the rate at which energy leaves the stomach. Thus, because energy-containing nutrients are digested more quickly, nutrient levels in the blood fall quicker and hunger returns (Friedman, 1995). While a subjective measure, highly palatable meals have also been shown to produce an increased glycemic response compared with less palatable meals that contain the same food items that are com- bined in different ways (Sawaya et al., 2001). This suggests a generalized link among palatability, gastric emptying, and glycemic response in the underlying mechanisms determining the effects of energy density on energy regulation. Further research on this potential link is needed. Researchers have used instruments such as visual analogue scales to measure differences in appetite sensations (e.g., hunger and satiety) between treatments in order to examine the effects of altering nutrients that play a major role in energy density, such as dietary fat, on energy regulation (Flint et al., 2000). A number of studies have been conducted in which preloads of differing energy density were given and hunger and satiety were measured either at the subsequent meal or for the remainder of the day. In the studies that administered preloads that had constant volume but different energy content (energy density was altered by chang- ing dietary fat content), there was no consistent difference in subsequent satiety or hunger between the various test meals (Durrant and Royston, 1979; Green et al., 1994; Hill et al., 1987; Himaya et al., 1997; Hulshof et al., 1993; Louis-Sylvestre et al., 1994; Porrini et al., 1995; Rolls et al., 1994). However, in those studies using isoenergetic preloads that differed in volume (energy density was altered by changing dietary fat content), there was consistently increased satiety and reduced hunger after consumption of the low energy-dense preload meals (i.e., those with higher volume) (Blundell et al., 1993; Holt et al., 1995; van Amelsvoort et al., 1989, 1990). It has been reported, however, that diets low in fat and high in carbo- hydrate may lead to more rapid return of hunger and increased snacking between meals (Ludwig et al., 1999a).
796 DIETARY REFERENCE INTAKES These data suggest that in the short-term, low energy-dense foods appear to increase satiety and decrease hunger compared to high energy- dense foods. Because individuals were blinded to the dietary content of the treatment diets, the results from these studies demonstrate the short- term effects of energy density after controlling for cognitive influences on food intake. It is important that cognitive factors are taken into account during the interpretation of results of preload studies. When individuals were aware of dietary changes, they generally (Ogden and Wardle, 1990; Shide and Rolls, 1995; Wooley, 1972), but not always (Mattes, 1990; Rolls et al., 1989), compensated for changes in energy density and thus minimized changes in energy intake. In well-controlled, short-term intervention studies lasting several days or more, high fat diets were consistently associated with higher spontaneous energy intake (Lawton et al., 1993; Proserpi et al., 1997; Thomas et al., 1992). From short- and longer-term studies, volunteers consistently con- sumed less dietary energy on low fat, low energy dense diets compared to high energy-dense diets (Glueck et al., 1982; Lissner et al., 1987; Poppitt and Swann, 1998; Poppitt et al., 1998; Stubbs et al., 1995b; Thomas et al., 1992; Tremblay et al., 1989, 1991). The extent to which energy intake was reduced on low energy-dense diets was similar for short- and long-term studies. An alternative way to study the effects of energy density on energy intake in short-term studies has been to compare energy intake between diets of similar energy density that differ in dietary fat content. Using this approach, when fat content was covertly varied between 20 and 60 percent of energy, there was no significant difference in energy intake between groups (Saltzman et al., 1997; Stubbs et al., 1996; van Stratum et al., 1978). These results suggest that energy density plays a more significant role than fat per se in the short-term regulation of food intake. During overfeeding, fat may be slightly more efficiently used than carbohydrate (Horton et al., 1995), but in one study, no difference was seen (McDevitt et al., 2000). Thus, high fat diets are not intrinsically fatten- ing, calorie for calorie, and will not lead to obesity unless excess total energy is consumed. It is apparent, however, that with the consumption of high fat diets by the free-living population, energy intake does increase, therefore predisposing to increased weight gain and obesity if activity level is not adjusted accordingly (see Table 11-1). While many of the short-term studies showed a more dramatic effect on weight reduction with reduced fat intake, the long-term studies showed weight loss as well. Conclusions. Epidemiological studies provide mixed results on the question of whether high fat (low carbohydrate) diets predispose to over-
797 M ACRONUTRIENTS AND HEALTHFUL DIETS weight and obesity and promote weight gain. However, a number of short- term studies suggest mechanisms whereby high fat intake could promote weight gain in the long-term. In addition, short- and long-term interven- tion studies provide evidence that reduced fat intake is accompanied by reduced energy intake and therefore moderate weight reduction or pre- vention of weight gain. For these reasons, it may be concluded that higher fat intakes are accompanied with increased energy intake and therefore increased risk for weight gain in populations that are already disposed to overweight and obesity, such as that of North America. Risk of CHD Epidemiological Evidence. In populations that consume very low fat diets, such as those of rural Asia and Africa, the prevalence of CHD is low (Campbell et al., 1998; Singh et al., 1995; Tao et al., 1989; Walker and Walker, 1978). This fact has led to the concept that low fat diets will pro- tect against CHD. However, this conclusion must be drawn with caution when it is applied to societies in which dietary and exercise habits differ markedly from societies in rural Asia and Africa. In the latter societies, people are highly active and lean (Singh et al., 1995; Walker and Walker, 1978). Both of these factors independently reduce risk for CHD and could offset any potentially detrimental effects of very low fat diets. For this reason, the effects of low fat diets must be viewed in the context of current societal habits in the United States and Canada and of changing habits in developing countries. Furthermore, in more recent years it has become clear that the relationship between fat intake and CHD is related more to the quality of fat than to the quantity. The relationship is clearly shown by cross-population studies. For example, some Mediterranean populations consume diets that are high in total fat and unsaturated fatty acids but low in saturated fatty acids; in these populations, rates of CHD are relatively low (Keys et al., 1980, 1984). In contrast, in northern Europe, where intakes of saturated fatty acids are high, so are rates of CHD (Keys et al., 1980, 1984). Two epidemiological studies showed no relationship between carbohydrate intake and LDL cholesterol concentration (Fehily et al., 1988; Tillotson et al., 1997). In several recent, long-term prospective studies of diet and chronic disease, rates of CHD did not substantially differ across populations that consumed approximately 25 to 45 percent of energy from fat (Ascherio et al., 1996; Hu et al., 1997). Men who developed CHD were shown to consume a slightly higher percentage of energy from fat (34.7 percent) compared with those who did not develop CHD (33.3 percent); however, this small difference in fat intake may not be significant since intake was based on a
798 DIETARY REFERENCE INTAKES 24-hour recall, and the data were not adjusted for energy intake (McGee et al., 1984). Furthermore, Hawaiians, who have a higher incidence of CHD than Japanese living in Hawaii, consumed more energy from fat (35 percent) than the Japanese (31 percent) (Bassett et al., 1969). It has been reported that those who developed CHD consumed slightly less energy from carbohydrate compared to those who did not develop CHD (Kushi et al., 1985; McGee et al., 1984) (Table 11-7). Other studies showed no significant association between risk of CHD and total carbohydrate or sugar intake (Bolton-Smith and Woodward, 1994; Liu et al., 1982, 2000). Interventional Evidence. Increasing fat intake, as a result of increased saturated fat intake, has been shown to increase LDL cholesterol concen- trations (Table 11-2), and therefore risk of CHD. Intervention studies that have investigated the effect of carbohydrate intake on LDL cholesterol concentration have shown mixed results (Table 11-3). Two intervention studies agree with the findings of West and colleagues (1990) in that LDL cholesterol concentration increased when the percent of energy from car- bohydrate was decreased from 55 to 31 percent (Borkman et al., 1991) and 59 to 41 percent (Marckmann et al., 2000). However, in other studies in which saturated fatty acids have remained constant, varying the percent- age of total fat was found to not alter the LDL cholesterol concentration (Garg et al., 1994; Grundy et al., 1988). Yu-Poth and colleagues (1999) conducted a meta-analysis on 37 inter- vention studies that evaluated the effects of the National Cholesterol Edu- cation Programâs Step I and Step II dietary interventions on various cardio- vascular disease risk factors. Reductions in plasma total cholesterol and LDL cholesterol concentrations were significantly correlated with reduc- tions in percentages of total dietary fat, but these also included a decrease in saturated fatty acids. Similarly, individuals who consumed the Dietary Approaches to Stop Hypertension diet, which contains 27 percent of energy from fat and only 7 percent of energy from saturated fat, had reduced total and LDL cholesterol concentrations (Obarzanek et al., 2001b). Singh and colleagues (1992) reported that mortality from CHD and other causes was significantly lower when patients with acute myocardial infarction were fed a reduced fat diet. The increase in LDL cholesterol concentration observed with increased fat intake is due to the strong positive association between total fat and saturated fat intake and the weak association between total fat and poly- unsaturated fat intake (Masironi, 1970; Stamler, 1979). This association is also observed in Appendix Tables K-4, K-5, K-7, and K-8. As shown in many studies, saturated fatty acids raise LDL cholesterol concentrations (see Chapter 8), whereas unsaturated fatty acids do not. In fact, n- 6 poly- unsaturated fatty acids reduce serum LDL cholesterol concentrations some-
799 M ACRONUTRIENTS AND HEALTHFUL DIETS what compared with carbohydrate (Hegsted et al., 1993; Mensink and Katan, 1992). The adverse effects of saturated fats are discussed in Chapter 8. It has been postulated that a high fat intake predisposes to a pro- thrombotic state, which contributes to venous thrombosis, coronary thrombosis, or thrombotic strokes (Barinagarrementeria et al., 1998; Kahn et al., 1997; Salomon et al., 1999). Consumption of diets high in fat (42 or 50 percent) have been shown to increase blood concentrations of the prothrombotic markers, blood coagulation factor VII (VIIc), and activated factor VII (VIIa) (Bladbjerg et al., 1994; Larsen et al., 1997). The concen- tration of factor VII is associated with increased risk of CHD (Kelleher, 1992). Furthermore, a significant and positive association was found between the level of dietary fat and factor VIIc concentration (Miller et al., 1989). Relation of Intakes of Saturated Fatty Acids and Total Fat. When fat is con- sumed in typical foods it contains a mixture of saturated, polyunsaturated, and monounsaturated fatty acids. Even when the content of saturated fatty acids in consumed fats is relatively low, the intakes of these fatty acids can be high with high fat intakes. For example, if all of the dietary fats con- sumed were low in saturated fatty acids (e.g., 20 percent of fat energy), a total fat intake of 35 percent of total energy would yield a saturated fatty acid intake of 7 percent of total energy. Consumption of a variety of dietary fats would likely result in an even higher percentage of saturated fatty acids. Thus, in practical terms, it would be difficult to avoid high intakes of saturated fatty acids for most persons if total fat intakes exceeded 35 per- cent of total energy. This fact is revealed by attempts to create a variety of heart-healthy menus (National Cholesterol Education Program, 2001). Moreover, data from CSFII show that with increased fat intake, there tends to be a greater increase in saturated fatty acid intake relative to poly- unsaturated fatty acid intake (Appendix Tables K-4, K-5, K-7, K-8; Masironi, 1970; Stamler, 1979). It should be pointed out, however, that when replac- ing saturated fatty acid intake with carbohydrate, there is no effect on the total cholesterol:HDL cholesterol ratio (Mensink and Katan, 1992). Conclusions. A few case-control studies have shown an association between total fat intake and risk for CHD. However, a detailed evaluation of these studies shows that it is not possible to separate total fat intake from saturated fatty acid intake, which is known to raise LDL cholesterol concentrations. Unsaturated fatty acids, which do not raise LDL cholesterol concentra- tions compared with carbohydrate, have not been implicated in risk for CHD through adverse effects on lipids or other risk factors. Nonetheless, practical efforts to create âheart-healthyâ menus reveal that intakes of total fat exceeding 35 percent of total energy result in unacceptably high intakes
800 TABLE 11-7 Epidemiological Studies on Total Carbohydrate and Sugar Intake and Risk of Coronary Heart Disease (CHD) Reference Study Design Results Comments Liu et al., Multi-country bivariate No significant association between sugar 1982 analysis intake and CHD McGee et al., 7,088 men Mean carbohydrate intake (% of energy) Those who developed CHD consumed 1984 Prospective cohort, Non-CHD 46.5a less energy as carbohydrates 10-y follow-up CHD 45.0b No association between sugar intake and risk of CHD Mean sugar intake (% of energy) Non-CHD 8.0 a CHD 8.1 a Kushi et al., 1,001 men Mean carbohydrate intake (% of energy) Those who died from CHD consumed 1985 Prospective cohort, No CHD death 42.7a significantly less total carbohydrate 20-y follow-up CHD death 41.2b No association between sugar intake and risk of CHD death Mean sugar intake (% of energy) No CHD death 17.3a CHD death 16.9a
Bolton- 11,626 men and women Mean sugar intake (% of energy) No association between risk of CHD and Smith and Cross-sectional survey Intrinsic sugar Men Women either intrinsic or extrinsic sugar Woodward, Control 2.06 3.31 intake 1994 CHD 1.89â2.19 3.15â3.31 Added sugar Control 11.2 8.55 CHD 10.5â11.4 8.63â9.10 Relative Liu et al., risk of CHD 75,521 women No significant association between risk 2000 Prospective cohort, Quintile Carbo- of CHD and total carbohydrate, 10-y follow-up of intake hydrate Sucrose Fructose sucrose, or fructose intake 1 1.00 1.00 1.00 2 1.02 1.03 0.91 3 1.09 1.16 0.96 4 1.03 1.02 1.11 5 1.23 1.22 1.07 a,b Within each study, the mean sugar or carbohydrate intakes that are significantly different between treatment groups have a different superscript. 801
802 DIETARY REFERENCE INTAKES of saturated fatty acids. Moreover, there is the possibility that high fat intakes may enhance a prothrombotic state, although the evidence to sup- port this mechanism for enhancing CHD risk is not strong enough alone to make solid recommendations. Risk of Hyperinsulinemia, Glucose Intolerance, the Metabolic Syndrome, and Type 2 Diabetes The metabolic syndrome (insulin-resistance syndrome) describes a clustering of metabolic abnormalities including insulin resistance (with or without glucose intolerance), an atherogenic lipid profile (high triacylglycerol concentration, low HDL cholesterol concentration, and high small, dense LDL), raised blood pressure, a prothrombotic state, and a proinflammatory state (Reaven, 2001). A prothrombotic state is charac- terized by elevations of plasminogen activator inhibitor and high fibrinogen concentrations, whereas a proinflammatory state is indicated by high c-reactive protein concentrations and other inflammatory markers. Abdomi- nal obesity (waist circumference > 102 cm in men and 88 cm in women) is highly correlated with the presence of insulin resistance (NHLBI/NIDDK, 1998) and is considered to be one of the clinical components of the meta- bolic syndrome (National Cholesterol Education Program, 2001). An excess of intra-abdominal fat has been identified as being highly associated with the lipid risk factors of the metabolic syndrome (Després, 1993), although total abdominal fat appears to be even more highly predictive of the insulin resistance component of the syndrome (Abate et al., 1996; Peiris et al., 1988). Many persons with the metabolic syndrome eventually develop type 2 diabetes. Thus, both obesity and weight gain are undisputed as major risk factors for the development of type 2 diabetes (defined as fasting plasma glucose ⥠7 mmol/L) (American Diabetes Association, 2001). The contribution of diet per se to the development of type 2 diabetes is less clear. In some laboratory animals (e.g., some species of rodents), a high percentage of fat in the diet will induce insulin resistance (Budohoski et al., 1993; Chisholm and OâDea, 1987). An important question is whether humans are similarly susceptible to this phenomenon independent of the effects of total fat intake on body fat content. Human studies do not pro- vide a clear answer to this question. Thus, if higher intakes of total fat lead to obesity, this in and of itself will reduce insulin sensitivity and predispose to the metabolic syndrome and type 2 diabetes. Recent studies have demonstrated that reduced fat intake and weight loss result in improved glucose tolerance and reduced risk of type 2 diabetes (Swinburn et al., 2001; Tuomilehto et al., 2001).
803 M ACRONUTRIENTS AND HEALTHFUL DIETS Epidemiological Evidence. In several population studies, investigators have attempted to determine the contribution of total fat intake to either insulin sensitivity or diabetes. These analyses are difficult to interpret because of the multiplicity of potential confounding variables. Nevertheless, several studies have reported an association between higher fat intakes and insulin resistance as indicated by high fasting insulin concentration, impaired glucose tolerance, or impaired insulin sensitivity (Lovejoy and DiGirolamo, 1992; Marshall et al., 1991; Mayer et al., 1993), as well as to the develop- ment of type 2 diabetes (West and Kalbfleisch, 1971). A number of studies, however, have not shown this association (Coulston et al., 1983; Liu et al., 1983; Salmerón et al., 2001). In the Insulin Resistance Atherosclerosis Study, total fat intake univariately correlated with less insulin sensitivity (Mayer-Davis et al., 1997); however, in multiple regression analyses, the presence of obesity appeared to be a confounding variable. Lovejoy and DiGirolamo (1992) likewise found intercorrelations among insulin resis- tance, total fat intake, and obesity. In contrast, Larsson and coworkers (1999) found no evidence of independent effects of diet on insulin secre- tory or sensitivity among 74 postmenopausal women. Although several studies suggest an association between total fat intake and the presence of insulin resistance (Lovejoy, 1999; Vessby, 2000), the degree to which the relationship is mediated by obesity remains uncertain. Decreased physical activity is also a significant predictor of higher postprandial insulin con- centrations and may confound some studies (Feskens et al., 1994; Parker et al., 1993). Interventional Evidence. A number of metabolic and intervention studies have examined the relationships among fat intake, fasting glucose and insulin concentrations, areas under curves for plasma glucose and insulin concentrations, insulin sensitivity, glucose effectiveness, and glucose disposal rates (Table 11-8). Several studies reported that diets containing 35 per- cent fat were accompanied by more impaired glucose tolerance than diets containing 25 percent fat or less (Fukagawa et al., 1990; Jeppesen et al., 1997; Straznicky et al., 1999; Swinburn et al., 1991). Coulston and coworkers (1983) found that a diet containing 41 percent fat led to significantly higher concentrations of insulin in response to meals compared with a diet containing 21 percent fat, but there were no alterations in fasting concentrations. In other studies, no effect on measures of glucose toler- ance were reported when diets varied in fat content from 11 to 30 (Leclerc et al., 1993) or 20 to 50 percent fat (Abbott et al., 1989; Borkman et al., 1991; Howard et al., 1991; Thomsen et al., 1999). When the diet was high in fat (50 percent of energy), the area under the curve for plasma glucose and insulin concentration was lower than when the diet had a low fat content (25 percent of energy) (Yost et al., 1998). In this study, the decreased
804 DIETARY REFERENCE INTAKES TABLE 11-8 Interventional Studies on the Effect of Dietary Fat on the Metabolic Parameters for Glucose and Insulin in Healthy Individuals Percent Fasting Fasting Reference Study Design of Fat Glucose Insulin NSCa Coulston et al., 11 men and women 41â21 NSC 1983 10-d crossover Chen et al., 8 young men 0 1988 3- to 5-d crossover 42 ND ND 55 ND ND 10 elderly men 0â37 ND ND 3- to 5-d crossover Abbott et al., 9 men and women 42â21 NSC NSC 1989 5-wk crossover Decreased b Decreasedb Fukagawa et al., 6 young men 42â14 1990 21- to 28-d intervention Decreasedb Decreasedb 6 elderly men and 38â15 women 21- to 28-d intervention Borkman et al., 8 men and women 20â50 NSC NSC 1991 3-wk crossover Howard et al., 1991 7 men and women 42â21 NSC NSC 5- to 7-wk crossover 9 men and women 42â21 NSC NSC 3- to 5-wk longitudinal Increased d NSC 12 Caucasians and 15â50 12 Pima Indians 2-wk crossover
805 M ACRONUTRIENTS AND HEALTHFUL DIETS Glucose Area Under Area Under Disposal/ the Curve the Curve Insulin Glucose Disappearance for Glucose for Insulin Sensitivity Effectiveness Rate Decreased b NDc NSC ND ND Decreased b ND ND NSC ND Increased b ND ND NSC ND Decreased b ND ND NSC ND ND ND ND ND ND Increased b ND ND ND ND ND ND ND ND NSC ND ND ND ND NSC NSC ND ND ND ND ND ND ND ND ND Increased e Increasede Decreasedd NSC ND continued
806 DIETARY REFERENCE INTAKES TABLE 11-8 Continued Percent Fasting Fasting Reference Study Design of Fat Glucose Insulin Increasedd Swinburn et al., 24 Caucasians and 15â50 NSC 1991 Pima Indians 2-wk crossover Leclerc et al., 7 men and women 11â30 NSC NSC 1993 7-d crossover Jeppesen et al., 10 women 25â45 ND ND 1997 3-wk crossover Yost et al., 1998 25 men and women 25â50 NSC NSC 15-d crossover Increasedb NSC Straznicky et al., 14 men 25â47 1999 2-wk crossover Thomsen et al., 16 men and women 28â42 NSC NSC 1999 4-wk crossover Kasim-Karakas 54 postmenopausal 15, 25, NSC NSC et al., 2000 women and 34 4- to 12-mo crossover a NSC = no significant change. b p < 0.05. c ND = no data available. d p < 0.001. e p < 0.01. fat intake was accompanied by an increased percentage of energy from carbohydrate. Garg and coworkers (1992b) reported that insulin sensitivity, indicated by insulin-mediated glucose disposal, was similar after almost a month of ingestion of either a reduced fat (25 percent of energy) or an increased fat diet (50 percent of energy). However, favorable effects of substituting a monounsaturated fat diet for a saturated fat diet on insulin sensitivity were seen at a total fat intake of up to 37 percent of energy (Vessby et al., 2001). A large, long-term intervention trial in adults showed that reducing total fat intake, in part, reduced the risk of the onset of type 2 diabetes by 58 percent (Tuomilehto et al., 2001). Similarly, the Diabetes Prevention Program Research Group reported that diet modification,
807 M ACRONUTRIENTS AND HEALTHFUL DIETS Glucose Area Under Area Under Disposal/ the Curve the Curve Insulin Glucose Disappearance for Glucose for Insulin Sensitivity Effectiveness Rate Increasede Increasede Decreasedd NSC ND NSC NSC ND ND ND Increased d NSC ND ND ND Decreasede Decreasedc ND ND ND Increasede Decreased b NSC ND ND ND NSC NSC NSC ND ND ND ND ND ND including a reduction of total fat intake from 34 to 27 percent of energy reduced the incidence of type 2 diabetes by 58 percent. Thus, there is no definitive evidence from metabolic and interventional studies that higher fat intakes impair insulin sensitivity in humans as they do in various labora- tory animals. Any suggestive links between fat intake and either insulin secretion or sensitivity may be mediated through confounding factors, such as body-fat content, making it difficult to detect any independent contri- bution of total fat intake to insulin sensitivity. Conclusions. Although high fat diets can induce insulin resistance in rodents, investigations in humans fail to confirm this effect. Moreover, an
808 DIETARY REFERENCE INTAKES association between dietary fat intake and risk for diabetes has been reported in some epidemiological studies, but this association is most likely confounded by various factors, such as obesity and glycemic index. Risk of Cancer High intakes of dietary fat have been implicated in the development of cancer, especially cancer of the lung, breast, colon, and prostate gland. Early support for this theory comes from laboratory animal and cross- cultural studies. The latter were based largely on international food dis- appearance data and migrant and time trend studies. In recent years, the theory that a diet high in fat predisposes to certain cancers has been weak- ened by additional epidemiological studies. Early cross-cultural and case- control studies reported strong associations between total fat intake and breast cancer (Howe et al., 1991; Miller et al., 1978; vanât Veer et al., 1990), yet a number of epidemiological studies, most in the last 15 years, have found little or no association between fat intake and breast cancer (Hunter et al., 1996; Jones et al., 1987; Kushi et al., 1992; van den Brandt et al., 1993; Velie et al., 2000; Willett et al., 1987, 1992). A meta-analysis of 23 studies yielded a relative risk of 1.01 and 1.21 from cohort and case- control studies, respectively (Boyd et al., 1993). Total fat intake in relation to colon cancer has strong support from animal studies (Reddy, 1992). However, evidence from epidemiological studies has been mixed (De Stefani et al., 1997b; Giovannucci et al., 1994; Willett et al., 1990). Howe and colleagues (1997) reported no association between fat intake and risk of colorectal cancer from the combined analysis of 13 case-control studies. Epidemiological studies tend to suggest that dietary fat intake is not associated with prostate cancer (Ramon et al., 2000; Veierød et al., 1997b). Giovannucci and coworkers (1993), however, reported a positive association between total fat consumption, primarily animal fat, and risk of advanced prostate cancer. Findings on the association between fat intake and lung cancer have been mixed (De Stefani et al., 1997a; Goodman et al., 1988; Veierød et al., 1997a; Wu et al., 1994). Risk of Nutrient Inadequacy or Excess Diets High in Fat. With increasing intakes of carbohydrate, and there- fore decreasing fat intakes, there is a trend towards reduced consumption of dietary fiber, folate, and vitamin C (Appendix K). With higher fat intakes, it is difficult to create practical high fat menus that do not contain unacceptably high amounts of saturated fatty acids (National Cholesterol Education Program, 2001).
809 M ACRONUTRIENTS AND HEALTHFUL DIETS Diets Low in Total Sugars. Micronutrient inadequacy can occur when sugars intake is very low (less than 4 percent of total energy) (Bolton- Smith and Woodward, 1995) because many foods that are abundant in micronutrients, such as fruits and dairy products, also contain naturally occurring sugars. A wide variety of foods from different food groups are needed to meet nutrient requirements. Because sugars are important for the palatability of foods, the complete omission of sugars from the diet could endanger overall nutrient adequacy by leading to low total energy intake, as well as low micronutrient intakes (Bolton-Smith, 1996). Although reduced nutrient intakes have been reported, adverse affects on health have not. Individuals with fructose intolerance, a condition caused by fructose-1-phosphate aldolase deficiency, strictly avoid foods containing fructose and sucrose and yet remain in good health (Burmeister et al., 1991). AMDRs for Adults When fat intakes are low and carbohydrate intakes are high, interven- tion studies, with the support of epidemiological studies, demonstrate a reduction in plasma HDL cholesterol concentration, an increase in the plasma total cholesterol:HDL cholesterol ratio, and an increase in plasma triacylglycerol concentration, which are all consistent with an increased risk of CHD. Conversely, many interventional studies show that when fat intake is high, many individuals consume additional energy, and therefore gain additional weight. Weight gain on high fat diets can be detrimental to individuals already susceptible to obesity and can worsen the metabolic consequences of obesity, particularly the risk of CHD. Moreover, high fat diets are usually accompanied by increased intakes of saturated fatty acids, which can raise plasma LDL cholesterol concentrations and further increase risk for CHD. Based on the apparent risk for CHD that may occur on low fat diets, and the risk for increased energy intake and therefore obesity with the consumption of high fat diets, the AMDR for fat and carbohydrate is estimated to be 20 to 35 and 45 to 65 percent of energy, respectively, for all adults. By consuming fat and carbohydrate within these ranges, the risk for obesity, as well as for CHD and diabetes, can be kept at a minimum. Furthermore, these ranges allow for sufficient intakes of essential nutri- ents while keeping the intake of saturated fatty acids at moderate levels. There is no lower limit of intake and no known adverse effects with the chronic consumption of Dietary Fiber or Functional Fiber (Chapter 7). Therefore, an AMDR is not set for Dietary, Functional, or Total Fiber.
810 DIETARY REFERENCE INTAKES Maximal Intake Level for Added Sugars Data from various national surveys show that increasing intakes of added sugars is associated with a decline in the consumption of certain micronutrients, thus increasing the prevalence of those consuming below the EAR or the AI. While such trends exist, it is not possible to determine a defined intake level at which inadequate micronutrient intakes occur. Fur- thermore, at very low or very high intakes, unusual eating habits most likely exist that allow for other factors to contribute to low micronutrient intakes. Based on the available data, no more than 25 energy from added sugars should be comsumed by adults. A daily intake of added sugars that individuals should aim for to achieve a healthy diet was not set. Total sugars intake can be lowered by consuming primarily sugars that are natu- rally occurring and present in micronutrient-rich foods, such as milk, dairy products, and fruits, while at the same time limiting consumption of added sugars from foods and beverages that contain minimal amounts of micro- nutrients, such as soft drinks, fruitades, and candies. Low Fat, High Carbohydrate Diets of Children Fat Oxidation Jones and colleagues (1998) reported a significantly greater fat oxidation in children (aged 5 to 10 years, n = 12) than in adults (aged 20 to 30 years, n = 6). Breath 13CO2 was measured in 12 children and 6 men following an oral bolus dose of [1-13C]palmitic acid (10 mg/kg of body weight) consumed with a test meal. Breath 13CO2 excretion was less in the men (35.1 percent of absorbed dose, P = 0.005) than in the children (57.0 percent of absorbed dose). The children exhibited greater fat oxida- tion in the postabsorptive state (2.43 g/h) and postprandial (11.89 g/6 h) states than the men (0.93 g/h postabsorptive, 9.86 g/6 h postprandial). The children also had greater fat oxidation compared with women studied previously by these investigators (0.53 g/h postabsorptive, 0.03 g/6 h post- prandial) (Murphy et al., 1995). Growth Most studies have reported no effect of the level of dietary fat on growth when energy intake is adequate (Boulton and Magarey, 1995; Fomon et al., 1976; Lagström et al., 1999; Lapinleimu et al., 1995; Niinikoski et al., 1997a, 1997b; Obarzanek et al., 1997; Shea et al., 1993). Two well-controlled trials demonstrated that a diet providing less than 30 percent energy from fat does not result in adverse effects on growth in
811 M ACRONUTRIENTS AND HEALTHFUL DIETS children up to 8 years of age (Lapinleimu et al., 1995; Niinikoski et al., 1997a, 1997b). A cohort study with a 25-month follow-up showed that there was no difference in stature or growth of children aged 3 to 4 years at baseline across quintiles (27 to 38 percent) of total fat intake (Shea et al., 1993). The Special Turku Coronary Risk Factor Intervention Project showed no difference in growth of children 7 months to 5 years of age when they consumed 21 to 38 percent fat (Lagström et al., 1999). Niinikoski and coworkers (1997a) reported that 1-year-old children who consistently con- sumed low fat diets (less than 28 percent) grew as well as children with higher fat intakes. A cohort study showed that children aged 2 years in the lower tertile of fat intake (less than 30 percent) had a height and weight similar to that of the higher fat intake groups (Boulton and Magarey, 1995). A few studies have observed impaired growth among hypercholsterolemic children who were advised to consume 30 percent or less of energy from fat. However, the energy intake was also reduced (Lifshitz and Moses, 1989) or not reported (Hansen et al., 1992). In a group of Canadian children 3 to 6 years of age, a fat intake of less than 30 percent of energy was associated with an odds ratio of 2.3 for weight-for-age below the 50th percentile at 6 years of age (Vobecky et al., 1995). A comprehensive evalu- ation of the effect of diet-related variables on the growth of children under 6 years of age from 18 Latin American countries (FAO/WHO, 1996) demon- strated that diets providing less than 22 percent energy from fat and with less than 45 percent of total fat from animal fat were related to low birth weight, underweight, and stunting (height-for-age ⤠2 standard deviations) (Uauy et al., 2000). The dietary determinants that best explained low birth weight were energy, protein, and animal fat, suggesting that high-quality animal protein and associated nutrients are important for growth and development. Risk of Nutrient Inadequacy or Excess Diets High in Carbohydrate and Low in Fats. Because the diets of young children are less diversified than that of adults, the risk of inadequate micronutrient intake is increased in these children. A cohort of 500 children aged 3 to 6 years showed that those who consumed less than 30 percent of energy from fat consumed less vitamin A, vitamin D, and vitamin E com- pared with those who consumed higher intakes of fat (30 to 40 percent) (Vobecky et al., 1995). Calcium intakes decreased by more than 100 mg/d for 4- and 6-year-old children who consumed less than 30 percent of energy from fat (Boulton and Magarey, 1995). Lagström and coworkers (1997, 1999), however, did not observe reduced intakes of micronutrients in chil- dren with low fat intakes (26 percent).
812 DIETARY REFERENCE INTAKES The Dietary Intervention Study in Children (DISC), a multi-center, randomized trial of children 8 to 10 years of age, demonstrated that reduc- ing the intake of fat to 28 percent of energy over a 3-year period increased the percentage of children not meeting the RDA for vitamin E and zinc; however, no biochemical evidence of deficiency of these nutrients was found (Obarzanek et al., 1997). Tonstad and Sivertsen (1997) observed no reduced intake of micronutrients with diets providing 25 percent of energy as fat. Nicklas and coworkers (1992) reported reduced intakes of certain micronutrients by 10-year-old children who consumed less than 30 per- cent of energy as fat; however, this level of fat intake was associated with marked increased intakes of candy. It has been suggested that children who consume a low fat diet can meet their micronutrient recommendation by appropriate selection of certain low fat foods (Peterson and Sigman- Grant, 1997). This is especially true for older children whose diets are typically more diverse. The tables in Appendix K show the intakes of nutrients at various intake levels of carbohydrate. With increasing intakes of carbohydrate, and therefore decreasing intakes of fat, the intake levels of calcium and zinc markedly decreased in children 1 to 18 years of age (Appendix Tables K-1 through K-3). Diets High in Added Sugars. Several surveys have evaluated the impact of added sugars intake on micronutrient intakes in children (Table 11-5). Gibson (1997) examined data from the U.K. National Diet and Nutrition Survey of Children Aged 1.5 to 4.5 Years (boys, n = 848; girls, n = 827) and found evidence of a nutrient dilution effect by nonmilk extrinsic sugars (NMES). Children consuming the highest concentrations of NMES (greater than 24 percent of energy) had intakes of most micronutrients that were between 6 and 20 percent below average. Gibson (1997) con- cluded that the inverse association of NMES with micronutrient intakes was of most significance for the 20 percent of children with the diets highest in NMES (24.9 percent of energy for boys and 24.5 percent of energy for girls). In a study of British adolescents, reduced intakes of calcium, phosphorus, iron, vitamin A, vitamin D, and folic acid were associated with increased sugars intakes (mean added sugars intake for the high sugars consumers was 122 g/d for boys and 119 g/d for girls) (Rugg-Gunn et al., 1991). In a smaller survey (n = 143), added sugars intakes at levels as high as 27 per- cent of energy did not have a significant impact on micronutrient intakes (Nelson, 1991). Similar to that observed for adults using data from NHANES III, increasing the added sugars intake by every 5th percentile tended to be associated with reduced intakes of certain micronutrients, including
813 M ACRONUTRIENTS AND HEALTHFUL DIETS calcium, vitamin A, iron, and zinc (Appendix Tables J-1 through J-3, J-6, and J-7). This reduction in micronutrient intake was most significant when added sugars intake levels exceeded 25 percent of energy. From 1989 to 1995, energy intakes increased for U.S. children aged 2 to 17 years primarily due to increased carbohydrate consumption. Bever- ages, particularly soft drinks, were important contributors to the increased carbohydrate consumption. During this period, micronutrient intakes (except for iron) did not increase and calcium intakes decreased. This was attributed to the fact that increased energy was largely obtained from soft drinks, which do not add nutrients and displace milk in childrenâs diets, with negative consequences for total diet quality (Morton and Guthrie, 1998). Children who were high consumers of nondiet soft drinks had lower intakes of riboflavin, folate, vitamin A, vitamin C, calcium, and phosphorus in comparison with children who were nonconsumers of soft drinks (Harnack et al., 1999). Several of these nutrients (folate, vitamin A, and calcium) have been identified in national surveys as âshortfallâ or âproblemâ nutrients among various age and gender groups (ARS, 1998). Ballew and colleagues (2000) demonstrated that in U.S. children, milk consumption was positively associated with the likelihood of achieving recommended vitamin A, vitamin B12, folate, calcium, and magnesium intakes in all age groups. Juice (100 percent fruit or vegetable juice) consumption was posi- tively associated with achieving vitamin C and folate recommended intakes in all age groups, as well as magnesium intake among children aged 6 years and older. Soft drink intake was negatively associated with achieving rec- ommended vitamin A intake in all age groups, calcium in children younger than 12 years of age, and magnesium in children 6 years of age and older. Others have shown that children who consumed milk at the noon meal had the highest daily intakes of vitamin A, vitamin E, calcium, and zinc, whereas the opposite was true for children who consumed soft drinks and tea (Johnson et al., 1998). Hence, beverages that are major contributors of the naturally occurring sugars, such as lactose and fructose, in the diet (e.g., milk and fruit juice) have been positively associated with nutrient adequacy, while beverages that are the principal source of added sugars in the diet (e.g., soft drinks) have been negatively associated with nutrient adequacy in the diets of U.S. children and adolescents (Ballew et al., 2000; Johnson et al., 1998). Diets High in Total Sugars. The findings from three surveys on the relationship between total sugars intake and micronutrient intake in children are mixed (Table 11-6). Gibson (1993) did not observe reduced micronutrient intakes when total sugars intake exceeded 25 percent of energy. Nicklas and coworkers (1996) reported that the percent of chil- dren meeting the RDA for only niacin and zinc was significantly reduced
814 DIETARY REFERENCE INTAKES when the intake of total sugars exceeded 31 percent of energy. A linear reduction in several micronutrients was observed with increasing total sugars intake (Farris et al., 1998). High Fat, Low Carbohydrate Diets of Children Risk of Obesity In the United States and Canada, there is evidence that children are becoming progressively overweight (Flegal, 1999; Gortmaker et al., 1987; Tremblay and Willms, 2000; Troiano et al., 1995). Furthermore, Serdula and coworkers (1993) reviewed a number of longitudinal studies with vary- ing cut-off levels for obesity and concluded that 26 to 41 percent of obese preschool children and 42 to 63 percent of obese school-age children became obese adults. Clinical evidence of disease associated with excess body weight, reduced physical activity, or high dietary fat intakes, however, are generally absent. The evidence for a role of dietary fat intakes in pro- moting higher energy intakes and thus promoting obesity in young chil- dren is conflicting. A positive trend in energy intake was associated with an increased percent of energy from fat for children up to 8 years of age (Boulton and Magarey, 1995). A positive correlation between fat intake and fat mass has been reported for boys 4 to 7 years of age (Nguyen et al., 1996). A lack of effect of dietary fat on BMI and adiposity, however, has been reported for children 1.5 to 4.5 years of age (Atkin and Davies, 2000; Davies, 1997). The DISC trial found no difference in BMI for children 8 to 10 years of age who consumed diets containing 29 or 33 percent fat over a 3-year period (Lauer et al., 2000). However, several studies showed a positive correlation between dietary fat intake and body fatness in children 8 to 12 years of age (Maffeis et al., 1996; Obarzanek et al., 1994; Ricketts, 1997). The average fat intake of nonobese children was measured to be 31 to 34 percent for children 9 to 11 years old, whereas the average fat intake of obese children was 39 percent of energy (Gazzaniga and Burns, 1993). A positive association between fat intake and several adiposity indices were observed, but only for up to 35 percent of energy (Maillard et al., 2000). Other factors that have been associated with increased BMI include physical activity. Risk of CHD Clinical studies have provided some evidence that serum cholesterol concentration is modified in children the same way as in adults, with serum total, LDL, and non-HDL cholesterol concentrations being increased by
815 M ACRONUTRIENTS AND HEALTHFUL DIETS consuming diets higher in total fat (Lauer et al., 2000; Niinikoski et al., 1996; Obarzanek et al., 2001a; Shannon et al., 1994; Simell et al., 2000; Vartiainen et al., 1986). However, no significant association between dietary fat and LDL cholesterol concentration was observed for boys and girls (aged 8 to 10 years) consuming fat ranging from 10 to 50 percent of energy (R = â0.04 to 0.14) (Kwiterovich et al., 1997). Furthermore, a significant positive association between fat intake and total cholesterol con- centration was observed in only two of five countries (Knuiman et al., 1983). Another potential indicator for childrenâs future risk of CHD is the presence of fatty streaks, which are found in the aortas of almost all chil- dren over 3 years of age in North America (Holman et al., 1958), and begin to appear in the coronary arteries about 5 to 10 years later than in the aorta (Berenson et al., 1992; McGill, 1968; Stary, 1989; Strong et al., 1992). The prevalence of aortic fatty streaks differs only slightly among children and adolescents of all populations studied, regardless of the fre- quency of atherosclerosis and coronary artery disease in adults of the respective population (Holman et al., 1958; McGill, 1968). The absence of a relation between aortic fatty streaks and the clinically relevant lesions of atherosclerosis in epidemiological and histological studies has thus raised questions on the clinical significance of fatty streaks in the aorta of young children (Newman et al., 1995; Olson, 2000). The Pathobiological Deter- minants of Atherosclerosis in Youth Study, however, has provided evidence that an unfavorable lipoprotein pattern (i.e., elevated non-HDL cholesterol and low HDL cholesterol concentrations), obesity, and hyperglycemia are associated with raised fatty streaks in the coronary artery and abdominal aorta in late teenage years (McGill et al., 2000a, 2000b). Similarly, the Bogalusa Heart Study observed a positive association between LDL choles- terol concentration and the percentage of surface with fatty streaks in the aorta (Berenson et al., 1992). These findings are consistent with the hypothesis of the progression of fatty streaks to fibrous plaques under the influence of the prevailing risk factors for coronary artery disease (McGill et al., 2000a, 2000b). It is still unclear, however, how reduction in serum cholesterol con- centration in childhood, if maintained, is associated with risk of CHD in adulthood. In addition, there are still pivotal issues that must be examined further, including the relationship between fatty streaks found in the arteries of young children and the later appearance of raised lesions associated with coronary vascular disease, the effects of dietary total fat modification on predictive risk factors in children, the safety of the diet with respect to total energy and micronutrients for the general population, and the long- term health benefit of establishing healthy dietary patterns early in childhood.
816 DIETARY REFERENCE INTAKES Risk of Nutrient Inadequacy or Excess Appendix Tables K-1 through K-3 and K-6 provide data from CFSII on the intake of various nutrients based on the level of carbohydrate intake. It can been seen from these tables that as the level of carbohydrate intake decreases, and therefore the level of fat increases, certain nutrients such as folate and vitamin C markedly decrease. Furthermore, with increasing levels of fat intake, the intake of saturated fat relative to linoleic acid intake markedly increases. AMDRs for Children The evidence suggests that children have a higher fat oxidation rate compared to adults, and that reduced intake of certain micronutrients can occur with the consumption of low fat diets, whereas there is potential risk of obesity with high fat intakes. High intakes of fat may promote increased risk for CHD and obesity. Dietary fat provides energy, which may be important for younger children with reduced food intakes, particularly during the transition from a diet high in milk to a mixed diet. Thus, there should be a transition from the high fat intake during infancy (55 and 40 percent of energy for the 0- to 6- and 7- to 12-months age groups, respectively) (Chapter 8) to an AMDR for adults (20 to 35 percent of energy). Therefore, it is estimated that the AMDR for fat intake is approxi- mately 30 to 40 percent of energy for children 1 to 3 years of age and 25 to 35 percent of energy for children 4 to 18 years of age. The AMDR for carbohydrate is the same as for adults (45 to 65 percent of energy). The ranges of fat intake include intakes of saturated fat that should be consumed at levels as low as possible while consuming a nutritionally adequate diet. Maximal Intake Level for Added Sugars As for adults, no more than 25 percent of energy from added sugars should be consumed by children to ensure adequate micronutrient intakes. For those children whose intake is above this level, added sugars intake can be reduced by consuming sugars that are primarily naturally occurring and present in foods such as milk, dairy products, and fruits, which also contain essential micronutrients. n-9 MONOUNSATURATED FATTY ACIDS Approximately 20 to 40 percent of fat is consumed as n-9 mono- unsaturated fatty acids, almost all of which is oleic acid (Appendix Tables E-1 and E-8). Monounsaturated fatty acids are not essential fatty acids, but they may have some benefit in the prevention of chronic disease. Although
817 M ACRONUTRIENTS AND HEALTHFUL DIETS early research pointed to this potential benefit, most attention has been given to it in the past decade. Low n-9 Monounsaturated Fatty Acid Diets Risk of CHD Epidemiological Evidence. Population data on monounsaturated fatty acid intake and risk of coronary heart disease (CHD) are limited. How- ever, in long-term follow-up studies of the Seven Countries Study, higher intakes of monounsaturated fatty acids were associated with decreased rates of CHD mortality (Keys et al., 1986). Other reports indicate that mono- unsaturated fatty acids have a neutral or beneficial effect on risk (Hu et al., 1997; Kromhout and de Lezenne Coulander, 1984; Pietinen et al., 1997). Interventional Evidence. Much work has been conducted and is ongoing to identify the ideal substitute for saturated fat in a blood cholesterol- lowering diet. The effects of a high monounsaturated fatty acid versus a low fat, high carbohydrate diet on serum lipid and lipoprotein concentrations have been a focus of considerable scientific inquiry. Eighteen well- controlled clinical studies that compared the effects of substituting mono- unsaturated fatty acids versus carbohydrate for saturated fat in a blood cholesterol-lowering diet have recently been reviewed (Kris-Etherton et al., 2000). In these studies, when on both high monounsaturated fat and low fat, high carbohydrate diets, saturated fatty acids contributed to 4 to 12 percent of energy and dietary cholesterol varied from less than 100 up to 410 mg/d. Diets high in monounsaturated fatty acids provided 17 to 33 percent of energy from monounsaturated fatty acids and contained more total fat (33 to 50 percent energy) than the low fat, high carbohy- drate diets (18 to 30 percent energy). The low fat, high carbohydrate diets provided 55 to 67 percent of energy from carbohydrate. Compared to baseline values, serum total cholesterol concentrations changed from â17 to +3 percent on the low fat, high carbohydrate diet, whereas it changed from â20 to â3 percent on the high monounsaturated fatty acid diet. The range of decrease in plasma low density lipoprotein (LDL) cholesterol concentration was similar (â22 to +1 percent) among individuals on the two diets. The change in serum triacylglycerol concentrations ranged from â23 to +37 percent for individuals consuming the low fat, high carbo- hydrate diets and from â43 to +12 percent for diets high in monounsaturated fatty acids. Changes in high density lipoprotein (HDL) cholesterol con- centrations ranged from â25 to +2 percent for individuals on the low fat, high carbohydrate diets compared to a â9 to +6 percent change for indi- viduals on diets high in monounsaturated fatty acids.
818 DIETARY REFERENCE INTAKES These data indicate that in weight-stable individuals, a high mono- unsaturated fatty acid, low saturated fatty acid diet results in a more favor- able metabolic profile with respect to total cholesterol, HDL cholesterol, and triacylglycerol concentrations. Figure 11-4 shows that with increased monounsaturated fatty acid intake, there is a favorable reduction in the total cholesterol:HDL cholesterol ratio. Furthermore, a meta-analysis of feeding studies estimated that the regression coefficients for the effects of monounsaturated fatty acids on LDL and HDL cholesterol concentrations were â0.008 and +0.006, respectively, suggesting a slight positive benefit (Clarke et al., 1997). 25 y = â0.698x + 8.3896 20 2 R = 0.4353 Percent Changes in TC:HDL-C Ratios 15 10 5 0 -5 -10 -15 -20 -25 0 5 10 15 20 25 30 35 Dietary MUFA (% energy) FIGURE 11-4 Relationship between monounsaturated fatty acid (MUFA) intake and total cholesterol (TC):high density lipoprotein cholesterol (HDL-C) concen- tration ratio. Weighted least-squares regression analyses were performed using the mixed procedure to test for differences in lipid concentrations (SAS Statistical package, version 8.00, SAS Institute, Inc., 1999). DATA SOURCES: Berry et al. (1992); Curb et al. (2000); Garg et al. (1988, 1992a, 1994); Ginsberg et al. (1990); Grundy (1986); Grundy et al. (1988); Jansen et al. (1998); Kris-Etherton et al. (1999); Lefevre et al., unpublished; Lopez-Segura et al. (1996); Mensink and Katan (1987); Nelson et al. (1995); Parillo et al. (1992); Pelkman et al. (2001); Perez-Jimenez et al. (1995, 1999, 2001).
819 M ACRONUTRIENTS AND HEALTHFUL DIETS Risk of Diabetes Epidemiological studies tend to suggest no association between mono- unsaturated fatty acid intake and risk of indicators for diabetes (Feskens et al., 1995; Marshall et al., 1997). Similarly, some intervention studies showed no effect of monounsaturated fatty acid intake on indicators for risk of diabetes (Fasching et al., 1996; Roche et al., 1998; Thomsen et al., 1999; Vessby et al., 2001). Uusitupa and coworkers (1994), however, reported a significantly lower area under the curve for plasma glucose concentration and a greater glucose disappearance rate when healthy women consumed a diet rich in monounsaturated fatty acids (19 to 20 percent) compared with a diet rich in saturated fatty acids. Risk of Cancer Bartsch and colleagues (1999) reported a protective effect of oleic acid on cancer of the breast, colon, and possibly the prostate. A few epide- miological studies have reported an inverse relationship between mono- unsaturated fatty acid intake and risk of breast cancer (Willett et al., 1992; Wolk et al., 1998), while a number of studies reported no association (Holmes et al., 1999; Hunter et al., 1996; Jones et al., 1987; Kushi et al., 1992; van den Brandt et al., 1993; vanât Veer et al., 1990). Increased consumption of olive oil was associated with significantly reduced breast cancer risk (La Vecchia et al., 1995; Martin-Moreno et al., 1994; Trichopoulou et al., 1995). A diet high in monounsaturated fatty acid-rich vegetable oils, includ- ing olive, canola, or peanut oils, has been associated with a protective effect or no risk of prostate cancer (Norrish et al., 2000; Ramon et al., 2000; Schuurman et al., 1999; Veierød et al., 1997b). Some speculate that the apparent protective effects of olive oil (and other vegetable oils) reflect constituents other than monounsaturated fatty acids including squalene (Newmark, 1999), phenolic compounds, antioxidants, and other com- pounds (Owen et al., 2000). No significant association has been reported for monounsaturated fatty acid intake and risk of colorectal cancer (Giovannucci et al., 1994; Howe et al., 1997). Risk of Nutrient Inadequacy In the United States, monounsaturated fatty acids provide 12 to 13 per- cent of energy intake. About 50 percent of these fatty acids are consumed via animal products, primarily meat fat (Jonnalagadda et al., 1995). Vegetable oils that are good sources of monounsaturated fatty acids include canola
820 DIETARY REFERENCE INTAKES oil and olive oil. Although the major sources of monounsaturated fatty acids (animal fat and vegetable oils) are not required to supply essential nutrients, very low intakes of monounsaturated fatty acids would require increased intakes of other types of fatty acids to achieve recommended fat intakes. Consequently, intakes of saturated and n-6 polyunsaturated fatty acids would probably exceed a desirable level of intake (see ân-6 Poly- unsaturated Fatty Acidsâ and Chapter 8). High n-9 Monounsaturated Fatty Acid Diets There are limited data on the adverse health effects from consuming high levels of n-9 monounsaturated fatty acids (see Chapter 8, âTolerable Upper Intake Levelsâ). Acceptable Macronutrient Distribution Range n-9 Monounsaturated fatty acids are not essential in the diet, and the evidence relating low and high intakes of monounsaturated fatty acids and chronic disease is limited. Therefore, an Acceptable Macronutrient Distri- bution Range (AMDR) for n-9 monounsaturated fatty acids is not pro- vided. Nonetheless, practical limits on intakes of monounsaturated fatty acids will be imposed by AMDRs for total fat and other types of fatty acids. n-6 POLYUNSATURATED FATTY ACIDS Low n-6 Polyunsaturated Fatty Acid Diets Risk of CHD Epidemiological Evidence. Many populations of the world, such as in Crete and Japan, have low total intakes of n-6 polyunsaturated fatty acids (e.g., < 4 percent of total energy) without obvious health consequences (Okita et al., 1995; Renaud et al., 1995). However, high intakes of n-6 polyunsaturated fats have been associated with blood lipid profiles (e.g., reduced total and low density lipoprotein [LDL] cholesterol, reduced triacylglycerol, and increased high density lipoprotein [HDL] cholesterol concentrations) that are associated with low risk of coronary heart disease (CHD) (Arntzenius et al., 1985; Becker et al., 1983; Sonnenberg et al., 1996). Prospective epidemiological evidence suggests that after controlling for other components of the diet, replacing saturated fats with unsaturated fats decreases risk of CHD (Hu et al., 1997); however, the doseâresponse
821 M ACRONUTRIENTS AND HEALTHFUL DIETS relationship between n-6 fatty acids and risk of CHD was not adequately established with certainty. An inverse association between linoleic acid intake and risk of coronary death was observed in several prospective studies (Arntzenius et al., 1985; Gartside and Glueck, 1993), while Pietinen and coworkers (1997) did not observe a relationship between linoleic acid intake and risk of CHD. A cross-sectional study showed that linoleic acid was inversely related to the prevalence of CHD, and this effect was stronger with higher intakes of linolenic acid (Djoussé et al., 2001). It is difficult to provide a direct assessment of n-6 fatty acids on risk of CHD without taking into consideration the impact of several dietary and nondietary factors, in addition to serum cholesterol concentrations, that lead to CHD and may be modified by the intake of saturated fat and n-6 fatty acids. Interventional Evidence. From the standpoint of blood lipid concentra- tion and CHD, higher n-6 polyunsaturated fatty acid intake generally alters blood lipid concentration to result in a decreased risk profile (Katan et al., 1994) (Table 11-9). Controlled trials have examined the effects of sub- stituting n-6 fatty acids in the diet to replace carbohydrate or saturated fatty acids (Mensink et al., 1992). In general, any fat that replaces carbo- hydrate in the diet raises HDL cholesterol and decreases triacylglycerol concentrations, with only small differences between individual fatty acids. n-6 Fatty acids decrease LDL cholesterol concentrations to a much greater degree than do saturated fatty acids (Mensink et al., 1992). Risk of Diabetes A number of epidemiological studies have been conducted to ascer- tain whether the quality of fat can affect the risk for diabetes. An inverse relationship was reported for vegetable fats and polyunsaturated fats and risk of diabetes (Colditz et al., 1992; Salmerón et al., 2001; Trevisan et al., 1990). One study reported a positive association between 2-hour glucose concentrations and polyunsaturated fatty acid intake (Mooy et al., 1995). A review of epidemiological studies on this relationship concluded that higher intakes of polyunsaturated fats could be beneficial in reducing the risk for diabetes (Hu et al., 2001). Risk of Nutrient Inadequacy Dietary n-6 polyunsaturated fatty acids have been reported to contrib- ute approximately 5 to 7 percent of total energy intake of adults (Allison et al., 1999; Fischer et al., 1985), and range up to no more than 10 percent of energy intake (Willett et al., 1987; Appendix Tables E-1 and E-9). The
822 DIETARY REFERENCE INTAKES TABLE 11-9 Interventional Studies on n-6 Fatty Acid Intake and Blood Lipid Concentrations Percent of Energy from Fatty Acid a Reference Study Design Becker et al., 1983 12 men 4.3 18:2 4-wk crossover 6.8 18:2 18 18:2 Mattson and 20 adults 3.9 18:2 Grundy, 1985 4-wk crossover 29 18:2 McDonald et al., 18 men 7.9 18:2 and 18.8 18:1 1989 18-d parallel 21.3 18:2 and 7.0 18:1 Zock and Katan, 56 men and women 3.8 18:2 (trans diet) 1992 3-wk crossover 3.9 18:2 (18:0 diet) 12 18:2 (18:2 diet) 7.2 â 1.7 18:2 Kris-Etherton 30 and 33 men 7.2 â 2.1 18:2 et al., 1993 26-d crossover 7.2 â 17.8 18:2 5.7 â 1.6 18:2 5.7 â 1.8 18:2 5.7 â 2.1 18:2 Howard et al., 1995 63 men and women 3.0 18:2 6-wk crossover 4.2 18:2 7.0 18:2 12.8 18:2 a 18:2 = linoleic acid, 18:1 = oleic acid. b LDL-C = low density lipoprotein cholesterol, HDL-C = high density lipoprotein choles- terol. main sources of n-6 polyunsaturated fatty acids are vegetable oils (e.g., soybean oil, safflower oil, and corn oil). Linoleic acid, the predominant n-6 polyunsaturated fatty acid, is essential in the diet, and therefore an Adequate Intake (AI) is set (see Chapter 8). Based on the estimated energy requirement for each age group, a minimum intake of 5 percent of energy from linoleic acid would be needed to meet the AI.
823 M ACRONUTRIENTS AND HEALTHFUL DIETS Postintervention Blood Lipid Concentration (mmol/L)b Total LDL-C HDL-C Cholesterol Triacylglycerol 2.11 1.03 3.44 0.81 1.83 1.12 3.28 0.84 1.68 1.17 3.17 0.79 3.70c 1.01c 5.80c 2.93c 3.10d 0.91c 4.94d 2.61c 2.52c 1.35c 3.97c 0.82c 2.03d 1.19d 3.39d 0.82c 3.07c 1.37c 4.90c 1.00c 3.00c 1.41c 4.89c 1.04d 2.83d 1.47d 4.74d 0.95c 2.92c 1.16c 4.55c 0.99c 2.66c 1.14c 4.27d 0.98c 2.15d 1.16c 3.59e 0.82d 3.23c 1.34c 4.89c 0.90c 2.79d 1.40c 4.45c 0.79c 2.82d 1.34c 4.40c 0.76c 4.14 1.16 5.92 1.43 4.14 1.16 5.89 1.41 4.11 1.14 5.87 1.37 4.03 1.16 5.79 1.34 c,d,e Within each study, the blood lipid concentrations that are significantly different between treatment groups have a different superscript. High n-6 Polyunsaturated Fatty Acid Diets Risk of LDL Oxidation When exposed to oxidant stress, n-6 fatty acids are vulnerable to attack by free radicals and oxidation into lipid peroxides (Halliwell and Chirico, 1993). An example of lipid peroxidation is LDL oxidation, which plays an important role in the development of atherosclerosis (Steinberg et al.,
824 DIETARY REFERENCE INTAKES 1989). Oxidation products of lipids and proteins are found in athero- sclerotic plaque and in macrophage foam cells. Compared with mono- unsaturated fatty acids, in vitro susceptibility of LDLs to undergo oxidative modification has been shown to increase with increased linoleic acid con- tent in LDLs as a result of increased intakes of linoleic acid (Abbey et al., 1993; Berry et al., 1991; Bonanome et al., 1992; Louheranta et al., 1996; Reaven et al., 1991, 1993, 1994). The mechanism whereby incorporation of polyunsaturated fatty acids into LDLs enhances susceptibility of LDL oxidation has been studied extensively (Chisolm and Steinberg, 2000; Jessup and Kritharides, 2000). Nonetheless, the hypothesis suggesting that a diet rich in polyunsaturated fat increases the polyunsaturated fatty acid content of LDL particles and increases their susceptibility to oxidation, which in turn leads to athero- sclerosis and CHD, still needs to be substantiated in human studies before measures of oxidation can be used as adequate indicators of chronic disease. Risk of Inflammatory Disorders There has been significant interest in the use of dietary n-6 fatty acids to modulate inflammatory response. γ-Linolenic acid (GLA, 18:3n-6) is the â6 desaturase product of linoleic acid and is elongated to dihomo-γ-linolenic acid (DGLA, 20:3n-6). The â6 desaturase enzyme is the initial step in desaturation of linoleic acid to arachidonic acid (see Figure 8-1). When given as a dietary supplement, GLA has been found to reduce symptoms of several chronic inflammatory diseases such as rheumatoid arthritis and atopic dermatitis (Andreassi et al., 1997; Leventhal et al., 1993, 1994; Lovell et al., 1981; Tate et al., 1989; Zurier et al., 1996). Even though GLA is the precursor to arachidonic acid, human neutrophils contain an elongase enzyme that converts GLA to DGLA, but they lack the â 5 desaturase needed to form arachidonic acid. As a result, GLA supplementation results in accumulation of DGLA, but not arachidonic acid, and a reduction in leukotriene B4 production in neutrophils (Chilton-Lopez et al., 1996; Johnson et al., 1997; Ziboh and Fletcher, 1992). However, plasma arachidonic acid concentrations increase after GLA supplementation (Johnson et al., 1997), and this could have adverse implications for other problems such as platelet aggregation (Rodier et al., 1993). Risk of Cancer An 8-year controlled clinical trial of 846 men demonstrated a signifi- cant increase in fatal carcinomas when the amount of n-6 polyunsaturated fatty acids fed was 15 percent of energy compared to 4 percent of energy
825 M ACRONUTRIENTS AND HEALTHFUL DIETS (Pearce and Dayton, 1971). Epidemiological studies, however, suggest that n-6 polyunsaturated fatty acids are not associated (or have an inverse relationship) with cancer. Howe and coworkers (1990) analyzed 12 case- control studies conducted prior to 1990 and determined that the relative risk of breast cancer for an increment of 45 g of polyunsaturated fat per day was only 1.25. More recent case-control and prospective studies fur- ther support the minimal effect of n-6 polyunsaturated fatty acids on breast cancer risk (Männistö et al., 1999; Toniolo et al., 1994). A similar relation- ship has been reported for linoleic acid intake and prostate cancer (Giovannucci et al., 1993; Schuurman et al., 1999). A meta-analysis of 7 cohort studies (Hunter et al., 1996) and a combined analysis of 12 case- control studies (Howe et al., 1990) consistently found no relationship between polyunsaturated fats or vegetable fats and risk of breast cancer. The range of intake of polyunsaturated fat was sufficiently large in these combined studies to comfortably conclude that the epidemiological evi- dence largely contradicts the animal studies; at least to date, no association between polyunsaturated fat, mainly n-6 fatty acids, and risk of breast cancer has been detected. Furthermore, in a review of the literature and meta-analyses of case-controlled and prospective epidemiological studies, Zock and Katan (1998) concluded that it was unlikely that high intakes of linoleic acid substantially raise the risk of breast, colorectal, or prostate cancer. Risk of Nutrient Excess High intakes of linoleic acid can inhibit the formation of long-chain n-3 polyunsaturated fatty acids from α-linolenic acid, which are precursors to the important eicosanoids (see Chapter 8). Acceptable Macronutrient Distribution Range Based on the median energy intakes for each age group (Appendix Table E-1), a minimum intake of 5 percent of energy from linoleic acid would be needed to meet the AI (see Chapter 8). An upper boundary of 10 percent of energy is estimated based on the following information: (1) the highest intake of n-6 polyunsaturated fatty acids for individuals in North America is approximately 10 percent of energy, (2) there is not a large body of epidemiological evidence that demonstrates the long-term safety of n-6 polyunsaturated fatty acid intakes exceeding 10 percent of energy from typical mixed diets, and (3) evidence from human studies demonstrates that enrichment of lipoproteins and cell membranes with n-6 polyunsaturated fatty acids contributes to a pro-oxidant state, thus
826 DIETARY REFERENCE INTAKES suggesting caution for recommending intakes that exceed 10 percent of energy. For these reasons, an Acceptable Macronutrient Distribution Range (AMDR) is estimated to be 5 to 10 percent of energy for children and adults. n-3 POLYUNSATURATED FATTY ACIDS Low n-3 Polyunsaturated Fatty Acid Diets Risk of CHD and Stroke Growing evidence suggests that dietary n-3 polyunsaturated fatty acids (eicosapentaenoic acid [EPA] and docosahexaenoic acid [DHA]) reduce the risk of coronary heart disease (CHD) and stroke. n-3 Polyunsaturated fatty acids may reduce CHD risk through a multitude of mechanisms by (1) preventing arrhythmias (Billman et al., 1999; Kang and Leaf, 1996; McLennan, 1993), (2) reducing atherosclerosis (von Schacky et al., 1999), (3) decreasing platelet aggregation by inhibiting the production of throm- boxane A2 (Harker et al., 1993), (4) decreasing plasma triacylglycerol con- centration (Harris, 1989), (5) slightly increasing high density lipoprotein (HDL) cholesterol concentration and decreasing triacylglycerol concen- tration (Harris, 1989, 1997), (6) modulating endothelial function (De Caterina et al., 2000), (7) decreasing proinflammatory eicosanoids (James et al., 2000), and (8) moderately decreasing blood pressure (Morris, 1994). Epidemiological Evidence. Many of the epidemiological studies used fish or fish oil intake as a surrogate for n-3 polyunsaturated fatty acid intake. The amounts of n-3 fatty acids vary greatly in fish, however, and unless the amounts of n-3 fatty acids are known, any conclusions are open to question. Furthermore, other components in fish may have effects that are similar to n-3 fatty acids and therefore may confound the results. Early epidemiological studies of Greenland Eskimos suggested that diets high in n-3 fatty acids, predominantly EPA and DHA, might protect against CHD (Bang et al., 1976; Dyerberg and Bang, 1979). Subsequent observational epidemiological studies have shown mixed results. In the Zutphen study, eating fish one or two times per week was associated with a significant reduction in CHD mortality (Kromhout et al., 1985). A similar result was found in Rotterdam that compared older people who ate fish with those who did not (Kromhout et al., 1995). In three cohorts from the Seven Countries Study, the con- sumption of fatty fish, but not total fish or lean fish, was associated with a 34 percent decrease in CHD mortality (Oomen et al., 2000). In the Chicago Western Electric Study, eating more than 35 g/d of fish resulted in decreased CHD mortality, mainly of the nonsudden death type (Daviglus
827 M ACRONUTRIENTS AND HEALTHFUL DIETS et al., 1997). Utilizing data from 36 countries, an inverse correlation was found between fish consumption and CHD and all-cause mortality (Zhang et al., 1999). In the Multiple Risk Factor Intervention Trial, CHD mortality and intake of n-3 fatty acids from fish were significantly and inversely cor- related (Dolecek, 1992). In the Physiciansâ Health Study, eating fish once per week decreased the relative risk of sudden cardiac death by 52 percent compared with eating fish less than once per month (Albert et al., 1998). In this study, although dietary total n-3 fatty acid intake correlated inversely with total mortality, no effect on total myocardial infarction, nonsudden cardiac death, or total cardiovascular mortality was observed. The relative risk of sudden death was only 0.58 when 0.3 to 2.6 g/mo of total n-3 fatty acids were consumed. Siscovick and colleagues (1995) reported that a mean intake of 2.9 and 5.5 g/mo of long-chain n-3 fatty acids reduced the risk of primary cardiac arrest by 30 and 50 percent, respectively. A cross- sectional study showed that α-linolenic acid was inversely related to the prevalence of CHD; this effect was stronger with increasing intakes of linoleic acid (Djoussé et al., 2001). In contrast to the above studies, the Health Professionalsâ Follow-up Study showed no significant association between fish intake and risk of CHD (Ascherio et al., 1995). In 16 cohorts from the Seven Countries Study, an inverse association between fish consumption and CHD mortality was found, but after correcting for saturated fat and flavonoid intakes and smoking, this association was not significant (Kromhout et al., 1996). Finally, in the EURAMIC study, adipose tissue biopsy from cases with first myocardial infarction and controls indicated lower α-linolenic acid intake in cases and a relative risk reduction of 58 percent comparing the highest versus lowest quintile of α-linolenic acid intake (Guallar et al., 1999). After adjustment for classical risk factors, the reduction was only 32 percent and no longer significant. In a meta-analysis of 11 prospective cohort studies of fish intake and CHD mortality, the two largest studies found no protective effect and the two smallest found an inverse relationship, with intermediate size studies showing intermediate benefits (Marckmann and Grønbaek, 1999). This analysis suggested that 40 to 60 g/d of fish provided a reduc- tion in CHD mortality in high-risk, but not low-risk, individuals. There are fewer data with regard to the effects of fish and n-3 poly- unsaturated fatty acids on stroke. In the Zutphen Study, consumption of more than 20 g/d of fish was associated with a decrease in the risk of stroke (Keli et al., 1994). In the NHANES Epidemiological Follow-up Study, for white women and for black women and men, but not white men, con- sumption of fish more than once a week was associated with decreased age-adjusted stroke incidence (Gillum et al., 1996). In the Nursesâ Health Study, higher consumption of fish and n-3 polyunsaturated fatty acids were associated with a reduced risk of total stroke and thrombotic infarction
828 DIETARY REFERENCE INTAKES but not hemorrhagic stroke (mainly among women who did not take aspirin regularly) (Iso et al., 2001). In contrast, in the Chicago Western Electric Study and the Physiciansâ Health Study, fish intake was not signifi- cantly associated with decreased stroke risk (Morris et al., 1995; Orencia et al., 1996). Nonclinical Interventional Evidence. Supplementation with fish oil, which is high in EPA and DHA, reduces triacylglycerol concentrations; low density lipoprotein (LDL) and HDL cholesterol concentrations are either increased or unchanged (à gren et al., 1996; Axelrod et al., 1994; Bhathena et al., 1991; Bønaa et al., 1992; DeLany et al., 1990; Eritsland et al., 1994a; Haglund et al., 1990; Lungershausen et al., 1994; Mori et al., 1991; Nelson et al., 1997a; Sanders and Hinds, 1992; Saynor and Gillott, 1992; Schmidt et al., 1992). Data from studies on the effects of EPA and DHA as a percent of energy on blood lipid concentrations in healthy individuals are presented in Table 11-10. In general, EPA+DHA intake is associated with small increases in LDL and HDL cholesterol concentrations and a significant decrease in triacylglycerol concentrations (Harris, 1997). The consumption of 3.65 to 6 g/d of n-3 polyunsaturated fatty acids inhibits platelet aggregation, which in turn prevents the risk of CHD (Mori et al., 1997; Tremoli et al., 1995). Some studies, however, did not show an effect on platelet aggregation after the consumption of 4.5 to 6 g/d of EPA+DHA (Nelson et al., 1997b; Turini et al., 1994). Randomized, Controlled Clinical Trials Evidence. There are four random- ized, controlled clinical trials that show a benefit of fish, fish oils, or α-linolenic acid on CHD prevention. In the Diet and Reinfarction Trial (DART), male myocardial infarction (MI) survivors were encouraged to increase their oily fish intake to 200 to 400 g/wk in order to increase EPA and DHA intake. Over a 2-year period, this resulted in a significant reduc- tion in total mortality, with the greatest benefit in a lower rate of fatal MI (Burr et al., 1989a, 1989b). In the DART trial, of the group randomized to ingest dietary fish, a subgroup chose to ingest 1.5 g/d of fish oil capsules rather than to consume fish. The capsule group had a significant reduction in CHD death and a significant reduction in all-cause mortality, suggesting that the benefits of the fish consumption were in the fish oil fraction (Burr et al., 1994). In the Indian Experiment of Infarct Survival, MI survivors were treated with either fish oil capsules (1.08 g/d of EPA) or mustard oil (2.9 g/d of α-linolenic acid) or placebo for 1 year (Singh et al., 1997). The fish oil and mustard oil groups had decreased total cardiac events, non- fatal infarctions, arrhythmias, left ventricular enlargement, and angina
829 M ACRONUTRIENTS AND HEALTHFUL DIETS pectoris. The fish oil group, but not the mustard oil group, had decreased cardiac deaths. In the Lyon Diet Heart Study, post-MI patients were randomized into a control group or into an experimental group that received dietary counseling and a special margarine containing α-linolenic acid (de Lorgeril et al., 1994, 1999). The control and experimental groups consumed approximately 0.27 and 0.81 percent of energy as α-linolenic acid, respectively. There was a significant reduction in risk for cardiac death for the experimental group after 27 months, and a reduction after a 4-year follow-up. The extent to which these reductions in risk were due to n-3 fatty acids is uncertain. In another trial, patients with recent MI were randomized to receive 300 mg of vitamin E, 850 mg of n-3 fatty acids (EPA+DHA), both, or neither (GISSI-Prevenzione Investigators, 1999). After 3.5 years, the n-3 fatty acid group experienced a 15 percent reduction in the primary endpoints of death, nonfatal myocardial infarction, and nonfatal stroke, and a 20 per- cent reduction in the other primary endpoints of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke. This group also expe- rienced a 20 percent reduction in all-cause mortality and a 45 percent reduction in sudden deaths compared with the control group. Vitamin E, in contrast to n-3 polyunsaturated fatty acids, had no beneficial effects on cardiovascular endpoints. n-3 Polyunsaturated fatty acids have also been reported to reduce blood pressure in hypertensive individuals. A meta-analysis of 31 placebo- controlled trials estimated a mean reduction in systolic and diastolic blood pressure of 3.0 and 1.5 mm Hg, respectively (Morris et al., 1993). Further- more, a statistically significant doseâresponse effect occurred with the smallest reduction observed with intakes of less than 3 g/d and the largest reduction observed with intakes at 15 g/d. When 55 individuals were randomized to receive either 5.2 g/d of n-3 fatty acids or a placebo for 12 weeks, heart rate variability (naturally occur- ring irregular heart beats) significantly increased after supplementation with n-3 fatty acids (Christensen et al., 1997). Because impaired heart rate variability is associated with increased arrhythmic events (Farrell et al., 1991), this finding supports the hypothesis that n-3 polyunsaturated fatty acids have antiarrhythmic effects in humans (Christensen et al., 1997). A more recent study by Christensen and coworkers (1999) reported a doseâ response effect on heart rate variability, suggesting antiarrhythmic effects in men but not women, given 3 g/d of EPA plus 2.9 g/d of DHA or 0.9 g/d of EPA plus 0.8 g/d of DHA for 12 weeks. However, the beneficial effect was found only in men with low initial heart rate variability.
830 TABLE 11-10 n-3 Fatty Acid (EPA and DHA)a Intake and Blood Lipid Concentrations Postintervention Blood Lipid Concentration (mmol/L)b Percent of Energy Reference Study Design from Fatty Acid LDL-C HDL-C Triacylglycerol Flaten et al., 1990 64 men Control diet (0 n -3) 1.28c 1.71c 6-wk parallel Control diet + 2.2 EPA/DHA 1.15c 1.23d Kestin et al., 1990 33 men 0.6 18:3n-3 4.44c 1.26c 1.62c 6-wk parallel 2.7 18:3n-3 4.55c 1.16c 1.85c 1.1 EPA/DHA 4.62d 1.28c 1.24d Bhathena et al., 1991 40 men 0 EPA/DHA 1.62c 10-wk crossover 2.2 EPA/DHA 1.17d Bønaa et al., 1992 144 men and women 0.28 EPA/DHA/22:5 4.65 1.32 1.95 Cross-sectional 0.30 EPA/DHA/22:5 4.71 1.31 1.49 0.52 EPA/DHA/22:5 4.43 1.36 1.32 0.72 EPA/DHA/22:5 4.47 1.36 1.34 Eritsland et al., 1994a 511 men and women Control diet 5.03c 1.08c 2.08c 9-mo parallel Control diet + 1.46 EPA/DHA 5.11c 1.16c 1.57d Eritsland et al., 1994b 57 men and women Control diet 4.84c 1.01c 1.80c 6-mo parallel Control diet + 1.4 EPA/DHA 5.03c 0.97c 1.71c
à gren et al., 1996 55 men 0 n-3 2.60c 1.42c 15-wk parallel 0.36 n -3 (fish) 2.56c 1.16d 0.60 n -3 (DHA oil) 2.42c 0.97d 0.76 n -3 (fish oil) 2.51c 0.89d Grimsgaard et al., 1997 224 men 0.19 n -3 (corn oil) 4.10c 1.40c 1.33c 7-wk parallel 0.52 n -3 (DHA oil) 4.13c 1.42d 1.02d 0.55 n -3 (EPA oil) 3.98c 1.34c 1.08d Sanders et al., 1997 26 men 0 EPA/DHA (saturated fat diet) 2.60c 1.18c 0.93c 3-wk crossover 0 EPA/DHA ( n-6 diet) 2.29d 1.19c 0.92c 1.5 EPA/DHA (n-3 diet) 2.30d 1.22c 0.68d a EPA = eicosapentaenoic acid, DHA = docosahexaenoic acid. b LDL-C = low density lipoprotein cholesterol, HDL-C = high density lipoprotein cholesterol. c,d Within each study, the blood lipid concentrations that are significantly different between treatment groups have a different superscript. 831
832 DIETARY REFERENCE INTAKES Risk of Obesity One study in laboratory mice suggested that diets containing n-3 poly- unsaturated fatty acids lead to lower levels of fat accumulation compared with diets containing other fatty acids (Hun et al., 1999). Several studies have examined whether n-3 polyunsaturated fatty acids affect growth of adipose tissue. Parrish and colleagues (1990, 1991) found that rats given a high fat diet supplemented with fish oil had less fat in perirenal and epididymal fat pads and decreased adipocyte volumes compared with rats fed lard. Adipose tissue growth restriction appeared to be the result of limiting the amount of triacylglycerol in each adipose tissue cell rather than by limiting the number of cells. Rustan and colleagues (1993) found similar results using rats fed either lard or lard supplemented with EPA and DHA. Although body weight gain and mean energy expenditure were similar for both groups, the mean respiratory quotient was significantly higher during both fasting and fed periods in rats fed the EPA+DHA supplement. The researchers concluded that the rats supplemented with n-3 fatty acids demonstrated reduced oxidation of fat and increased carbo- hydrate utilization. Little data exist with respect to the specific effects of dietary n-3 polyunsaturated fatty acids on adiposity in humans; therefore, prevention of obesity cannot be considered an indicator at this time. Risk of Diabetes Epidemiological Evidence. While several studies have reported a nega- tive relationship between polyunsaturated fatty acid intake and risk of diabetes (Colditz et al., 1992; Salmerón et al., 2001; Trevisan et al., 1990), fish intake has specifically been reported to have a negative association (Feskens et al., 1991b, 1995). A review of the epidemiological data on this association concluded that polyunsaturated fatty acids, and possibly long- chain n-3 fatty acids, could be beneficial in reducing the risk of diabetes (Hu et al., 2001). Interventional Evidence. Studies conducted in rodents have shown that administration of fish oil results in increased insulin sensitivity (Chicco et al., 1996) and corrected hyperinsulinemia (Luo et al., 1996). Substituting a proportion of the fat in a high fat diet with fish oil prevented the devel- opment of insulin resistance in rats (Storlien et al., 1987) and normalized insulin action in rats experiencing severe insulin resistance (Storlien et al., 1991). Additionally, rats prone to spontaneous diabetes mellitus that were given EPA in doses of 0.1, 0.3, and 1.0 g/kg/d for 8 months had reduced incidences of diabetes (92, 50, and 17 percent, respectively) (Nobukata et
833 M ACRONUTRIENTS AND HEALTHFUL DIETS al., 2000). Thus, animal evidence suggests that the fatty acid composition of the diet may be an important factor in the effect of dietary fat on insulin action. Whether a change of dietary fat composition will alter insulin sensitivity in humans remains an open question. Studies in humans have demon- strated a relationship between increased insulin sensitivity and the proportion of long-chain n-3 polyunsaturated fatty acids in skeletal muscle phospho- lipids (Borkman et al., 1993; Clore et al., 1998). Supplementation with EPA and DHA resulted in improved insulin sensitivity in diabetic individuals (Popp-Snijders et al., 1987) and increased the insulin-stimulated glucose disposal rate in patients with impaired glucose tolerance (Fasching et al., 1991). However, other studies in nondiabetic individuals (Toft et al., 1995) and individuals with type 2 diabetes (Annuzzi et al., 1991; Luo et al., 1998) reported no beneficial effect of n-3 fatty-acid supplementation on insulin action. Risk of Cancer Experimental evidence suggests several mechanisms in which n-3 poly- unsaturated fatty acids may protect against cancer. n-3 Polyunsaturated fatty acids, particularly DHA and EPA, have been shown to suppress neo- plastic transformation (Takahashi et al., 1992), inhibit cell growth and proliferation (Anti et al., 1992; Calviello et al., 1998; Grammatikos et al., 1994), induce apoptosis (Calviello et al., 1998; Lai et al., 1996), and inhibit angiogenesis (Rose and Connolly, 2000), which may occur by suppressing n-6 fatty acid eicosanoid production (see Chapter 8). Animal studies with n-3 fatty acid or fish-oil supplementation have shown inhibition of mammary carcinogenesis and tumor growth (Grammatikos et al., 1994; Karmali et al., 1984), colon carcinogenesis (Deschner et al., 1990; Reddy et al., 1991), and prostate tumorigenesis and tumor cell growth (Karmali et al., 1987). Across-country epidemiological studies have shown an inverse relation- ship between dietary fish intake and breast cancer incidence and mortality (Kaizer et al., 1989; Sasaki et al., 1993), but the intakes of n-3 fatty acids in these studies are not well defined. Moreover, despite these results, most case-control and prospective studies have not reported a protective effect of fish consumption on breast cancer (Willett, 1997). Ecological studies have also shown inverse relationships between fish and fish oil intake and colorectal cancer (Caygill and Hill, 1995; Caygill et al., 1996), although some were nonsignificant (Hursting et al., 1990). Results from case-control and prospective studies have been somewhat equivocal (Boutron et al., 1991). However, Willett and colleagues (1990) found that higher fish con- sumption was associated with less colon cancer in women. No significant
834 DIETARY REFERENCE INTAKES associations were reported in the few studies that have examined fish con- sumption and risk of prostate cancer (Giovannucci et al., 1993; Severson et al., 1989; Talamini et al., 1992). Risk of Nutrient Inadequacy Vegetable oils, such as soybean oil, flaxseed oil, and canola oil, contain high amounts of α-linolenic acid. Fatty fishes and fish oils provide a mix- ture of biologically active EPA and DHA. n-3 Polyunsaturated fatty acids (α-linolenic acid) are essential in the diet and Adequate Intakes (AIs) have been set (see Chapter 8). Intakes of α-linolenic acid range from approximately 0.6 to 1.2 percent of energy (Appendix Tables E-1 and E- 11). Low intakes of α-linolenic acid can result in inadequate biosynthesis of the longer-chain n-3 polyunsaturated fatty acids, resulting in an exces- sive ratio of n-6 polyunsaturated fatty acids (see Chapter 8). High n-3 Polyunsaturated Fatty Acid Diets There is evidence to suggest that high intakes of n-3 polyunsaturated fatty acids (EPA and DHA) may have adverse effects on immune function and may increase the risk of excessive bleeding and hemorrhagic stroke (see Chapter 8). High intakes of n-3 polyunsaturated fatty acids (α-linolenic acid) can also result in inadequate biosynthesis of long chain n-6 poly- unsaturated fatty acids that are important for prostaglandin and eicosanoid synthesis (see Chapter 8). Acceptable Macronutrient Distribution Range α-Linolenic acid is essential in the diet and therefore AIs have been set (see Chapter 8). Up to 10 percent of the AI can be consumed as EPA and/or DHA. The above studies suggest that α-linolenic acid, EPA, and DHA may provide beneficial health effects when consumed at moderate levels. Based on the median energy intake by the various age groups (Appendix Table E-1), it is estimated that approximately 0.6 percent of energy from α-linolenic acid is needed to meet the AI. This level is used as the lower boundary for the Acceptable Macronutrient Distribution Range (AMDR) for α-linolenic acid. The upper boundary of the AMDR for α-linolenic acid is set at 1.2 percent of energy and represents the highest levels of α-linolenic acid consumed in the form of foods by individuals in North America. Data from interventional studies to support the benefit of even higher intakes of α-linolenic acid were not considered strong enough to justify establishing an upper boundary greater than 1.2 percent of
835 M ACRONUTRIENTS AND HEALTHFUL DIETS energy. Approximately 10 percent of the AMDR for n-3 fatty acids (α-linolenic acid) can be consumed as EPA and/or DHA (0.06 to 0.12 per- cent of energy). SATURATED FATTY ACIDS, TRANS FATTY ACIDS, AND CHOLESTEROL Low Saturated Fatty Acid, Trans Fatty Acid, and Cholesterol Diets There are no known risks of chronic disease from consuming low intakes of saturated fatty acids, trans fatty acids, or cholesterol. In the United States, saturated fatty acids provided 11 to 12 percent of energy in adult diets and 12.2 to 13.9 percent of energy in the diets of children and adolescents (CDC, 1994). It is estimated that the intake of trans fatty acids is approximately 2.6 percent of energy (Allison et al., 1999). The intake of cholesterol by American adults ranges from less than 100 mg/d to just under 770 mg/d (Appendix Table E-15). It is important to recognize that lower intakes of saturated fatty acids and cholesterol are observed for vegetarians, especially vegans (Janelle and Barr, 1995; Shultz and Leklem, 1983). Because certain micronutrients, saturated fats, and cholesterol are consumed mainly through animal foods, it is possible that diets low in saturated fat and cholesterol are associated with low intakes of these micronutrients. When the micronutrient intakes of Seventh-day Adventist vegetarians and nonvegetarians were measured, there were no significant reductions in micronutrient intakes with the lower saturated fat (7.3 versus 12.6 percent of energy) and cholesterol intakes (186 versus 404 mg/d) of vegetarian compared to nonvegetarian men (12.6 percent of energy and 404 mg/d) (Shultz and Leklem, 1983). Similarly, the intakes of most micronutrients were not significantly lower for vegans, except for vitamin B12 (0.51 versus 3.79 mg/d), riboflavin (1.32 versus 1.72 mg/d), and calcium (578 versus 950 mg/d). Vegans had significantly lower intakes of saturated fat (6.9 versus 10.6 percent of energy) and cholesterol (94 versus 231 mg/d) than nonvegetarians (Janelle and Barr, 1995). Analysis of nutritionally adequate menus indicates that there is a mini- mum amount of saturated fat that can be consumed so that sufficient levels of linoleic and α-linolenic acid are consumed (as an example see Appendix Tables G-1 and G-2). Other than soy products that are high in n-6 and n-3 fatty acids, many vegetable-based fat sources are also high in saturated fatty acids, and these differences should be considered in plan- ning menus.
836 DIETARY REFERENCE INTAKES High Saturated Fatty Acid, Trans Fatty Acid, and Cholesterol Diets There is a body of evidence suggesting that saturated and trans fatty acids and cholesterol increase blood total and low density lipoprotein cholesterol concentrations, and therefore the risk of coronary heart dis- ease (CHD) (see Chapters 8 and 9). Because the intake of each of these three nutrients and risk of CHD is a positive linear trend, even very low intakes of each may increase risk. To minimize saturated fatty acid intake requires decreased intake of animal fats (e.g., meat fat and butter fat) and certain oils, such as coconut and palm kernel oil. Saturated fatty acids can be reduced by choosing lean cuts of meat, trimming away visible fat on meats, and eating smaller por- tions. The amount of butter that is added to foods can be minimized or replaced with vegetable oils or nonhydrogenated vegetable oil spreads. Vegetable oils, such as canola and safflower oil, can be used to replace more saturated oils such as coconut and palm oil. Such changes can reduce saturated fat intake without altering the intake of essential nutrients. A reduction in the frequency of intake or serving size of certain foods such as liver (375 mg/3 oz slice) and eggs (250 mg/egg) can help reduce the intake of cholesterol, as well as foods that contain eggs, such as cheese- cake (170 mg/slice) and custard pie (170 mg/slice). There are a number of meats and dairy products that contain low amounts of cholesterol (e.g., lean meats [30 mg/2 slices] and 2 percent milk [18 mg/cup]). Therefore, there are a variety of foods that are low in saturated fat and cholesterol and also abundant in essential nutrients such as iron, zinc, and calcium. Trans fatty acids are high in stick margarine and those foods containing vegetable shortenings that have been subjected to hydrogenation. Examples of foods that contain relatively high levels of trans fatty acids include cakes, pastries, doughnuts, and french fries (Litin and Sacks, 1993). Therefore, the intake of trans fatty acids can be reduced without limiting the intake of most essential nutrients by decreasing the serving size and frequency of intake of these foods, or by using unhardened oil. CONJUGATED LINOLEIC ACID Conjugated linoleic acid (CLA) has been shown to play a role in the alteration of body composition in animals (Park et al., 1997), the inhibition of tumor cell growth (Whigham et al., 2000), and the inhibition of experi- mental atherosclerosis in animals (Lee et al., 1994). The trans-10,cis-12 CLA isomer appears to be the isomer primarily responsible for the induc- tion of changes in body composition (de Deckere et al., 1999; Park et al., 1999). Several studies suggest that these changes are primarily due to a reduction in lipid uptake by adipocytes (Pariza et al., 2001), which results
837 M ACRONUTRIENTS AND HEALTHFUL DIETS from the action of CLA on the activities of stearoyl-coenzyme A desaturase (Choi et al., 2000; Lee et al., 1998) and lipoprotein lipase (Park et al., 1997, 1999). The trans-10,cis-12 CLA isomer has also been reported to inhibit proliferation and differentiation in cultured mouse adipocytes (Brodie et al., 1999) and to induce apoptosis in vivo in the adipose tissue of mice (Tsuboyama-Kasaoka et al., 2000). In addition to body fat reduction, dietary CLA may increase whole body protein accretion in animals, suggesting the enhancement of lean body mass (Ostrowska et al., 1999; Park et al., 1997; Stangl, 2000). Research on the effects of CLA on body composition in humans has provided conflicting results. Blankson and coworkers (2000) conducted a study in overweight and obese men and women given either placebo or 1.7, 3.4, 5.1, or 6.8 g/d of a CLA preparation consisting of equal parts of the cis-9,trans-11 and trans-10,cis-12 isomers. After 12 weeks, none of the groups exhibited significant reductions in body weight or body mass index. However, the groups given 1.7, 3.4, and 6.8 g/d of CLA showed significant decreases in body fat mass compared to the placebo group. No differences in lean body mass were observed. Zambell and coworkers (2000) studied the effects of CLA supplementation in healthy adult women given either placebo or 3 g/d of CLA for 64 days. They found no significant changes in fat-free mass, fat mass, body weight, or percentage of body fat with CLA supplementation. CLA has been studied for its potential anticancer benefits in numerous animal and in vitro models. CLA mixtures have been shown to exhibit anticarcinogenic properties in skin, lung, forestomach, colorectal, prostate, and mammary tissues (Cesano et al., 1998; Ha et al., 1990; Liew et al., 1995; Schønberg and Krokan, 1995; Shultz et al., 1992), although the majority of the research has been conducted with breast cancer. Ip and Scimeca (1997) conducted a study in female rats chemically induced for mammary tumors and fed a diet containing either 2 percent or 12 percent linoleic acid. The rats were also supplemented with 0, 0.5, 1, 1.5, or 2 per- cent CLA. The researchers found that increasing CLA from 0.5 to 1 per- cent resulted in a dose-dependent decrease in both tumor incidence and total number of tumors. No further protection was observed in the groups receiving 1.5 or 2 percent CLA. In addition to inhibiting tumor growth, CLA eliminated the spread of breast cancer cells to the lungs, peripheral bone, and bone marrow of mice supplemented with 1 percent CLA (Visonneau et al., 1997). Although the exact mechanisms of the anticarcinogenic effects of CLA are not fully understood, several explanations have been offered. It has been suggested that growth inhibition of cancer cells may be due to the ability of CLA to inhibit protein and nucleotide biosynthesis (Ip et al., 1999; Shultz et al., 1992) and to induce cell apoptosis (Ip et al., 1999,
838 DIETARY REFERENCE INTAKES 2000). Antioxidant activity of CLA has also been suggested (Ha et al., 1990; Ip et al., 1991); however, this theory has been contradicted by studies showing that CLA does not decrease lipid peroxide formation (Cunningham et al., 1997; van den Berg et al., 1995). Another possible mechanism of cancer cell growth inhibition by CLA includes alteration of eicosanoid metabolism. CLA may compete with linoleic acid in its conversion to arachidonic acid, thereby reducing the biosynthesis of eicosanoids (Banni et al., 1999), which have been associated with the proliferation of cultured breast cancer cells (Karmali, 1986; Noguchi et al., 1995). CLA has been shown to reduce leukotriene B4 and prostaglandin E2 levels in animals (Kavanaugh et al., 1999; Sugano et al., 1998). To date, there are insuffi- cient data in humans to recommended a level of CLA at which beneficial health effects may occur. DIETARY FIBER AND FUNCTIONAL FIBER Low Fiber Diets A low fiber diet is often attributed to the intake of a low carbohydrate diet. A number of adverse clinical effects, including impaired laxation and increased risk of cancer, obesity, heart disease, and type 2 diabetes, have been associated with the chronic consumption of low amounts of Dietary Fiber or Functional Fiber. The studies to support a beneficial role of these fibers are reviewed in Chapter 7. Certain animal studies have shown that some fibers can actually enhance mineral absorption (Demigné et al., 1989; Levrat et al., 1991a, 1991b). There are several potential mechanisms by which ingestion of Dietary Fiber may actually enhance mineral status. For example, a more acidic pH in the colon is produced with fiber fermentation, and this results in more ionized calcium, which is better absorbed (Rémésy et al., 1992). Dietary Fiber in the colon can also stimulate bacterial fermentation, which has been associated with increases in calcium, magnesium, and potassium absorption (Demigné et al., 1989; Levrat et al., 1991a ). Many fiber sources, such as karaya gum, sugar beet fiber, and coarse bran, are also excellent sources of minerals (Behall et al., 1987; Fairweather-Tait and Wright, 1990; Van Dokkum et al., 1982). Several investigators have shown that inulin and fructooligosaccharides actually enhance calcium and magnesium absorption (Coudray et al., 1997; Delzenne et al., 1995; Levrat et al., 1991b; Ohta et al., 1995). There is also indirect evidence of this same enhancement with calcium in humans (Trinidad et al., 1993, 1996). A direct effect of fiber on mineral absorption has also been reported in humans where inulin increased the apparent absorption and balance of calcium (Coudray et al., 1997).
839 M ACRONUTRIENTS AND HEALTHFUL DIETS High Fiber Diets There is limited data to suggest that chronic consumption of high fiber diets results in adverse health effects (see Chapter 7). Gastrointestinal distress can occur with the consumption of high fiber diets, but this often subsides with time. DIETARY PROTEIN Low Protein Diets Although uncommon in North America, proteinâenergy malnutrition (PEM) is one of the most common nutritional diseases in developing coun- tries (Torun and Chew, 1999). The etiology of PEM is complex as there are a number of factors that are attributed to its onset, including insuffi- cient food intake or intake of low protein-containing foods, which in turn is attributed to poverty, unsanitary conditions, and food insecurity. Because PEM is attributed to insufficient food intake, not only are protein and energy limited, but the micronutrients that are often present in protein- containing foods are also limited. Epidemiological analysis from 53 devel- oping countries indicated that 56 percent of deaths in young children were due to the potentiating effects of malnutrition in infectious diseases (Pelletier et al., 1995). The increased duration or susceptibility to infec- tious diseases such as respiratory infections and diarrhea are due, in part, to the involvement of protein in immune function. Impaired Immune Function Chandra (1972) showed that in individuals with PEM, a variety of immune responses were impaired. The major defects observed with severe PEM involve T lymphocytes and the complement system. With PEM, the number of lymphocytes is markedly reduced and delayed cutaneous hyper- sensitivity responses to both recall and new antigens are depressed (Chandra, 1991), as is the production of several components of the complement system (Keusch et al., 1984). Furthermore, antibody affinity (Chandra et al., 1984) and lysozyme concentrations (Chandra and Newberne, 1977) are decreased. Impaired Growth Low protein intake during pregnancy is correlated with a higher inci- dence of low birth weight (King, 2000). Furthermore, in children, diets low in protein and energy are most frequently associated with a deficit in
840 DIETARY REFERENCE INTAKES weight-for-height (wasting) and height-for-age (stunting) (Waterlow, 1976). These deficits can be corrected by the provision of a high protein diet (Badaloo et al., 1999) and with an adequate energy intake to permit catch-up growth. For these reasons, various anthropometric measures are used for diagnosis and monitoring the treatment of PEM. Low Birth Weight Rush and coworkers (1980) found decreases in both gestational length and birth weight and increases in very early premature births and mortal- ity with high density protein supplementation (additional 40 g/d) in poor, black pregnant women at risk of having low birth weight infants. In contrast, Adams and coworkers (1978) reported no differences from the controls in mean birth weights of infants of mothers at risk of having a low birth weight infant when these women were supplemented with 40 g/d of protein. No reports were found of protein toxicity in healthy pregnant or lactating women that were not at risk of having a low birth weight infant. Thus, at the present time, low birth weight cannot be utilized to set a Tolerable Upper Intake Level (UL) for protein for women. Risk of Nutritional Inadequacy High quality protein is typically consumed via animal products, and therefore vegetarians may consume less high quality protein than omni- vores. Because animal foods are the primary sources of certain nutrients, such as calcium, vitamin B12, and bioavailable iron and zinc, low protein intakes may result in inadequate intakes of these micronutrients. As an example, Janelle and Barr (1995) reported significantly lower intakes of riboflavin, vitamin B12, and calcium by vegans who also consumed lower amounts of protein (10 versus 15 percent of energy) compared with nonvegetarians. Vegetable protein has been shown to decrease plasma cholesterol con- centrations in experimental animals and humans (Nagata et al., 1998; Nicolosi and Wilson, 1997; Terpstra et al., 1991). When the ratio of casein:soybean protein in the diet was decreased, there was a reduction in total and non-high density lipoprotein cholesterol concentrations (Fernandez et al., 1999; Teixeira et al., 2000). In laboratory animals, it was shown that the onset of atherosclerosis was significantly reduced when animals were fed a textured vegetable protein diet compared to a beef protein diet (Kritchevsky et al., 1981).
841 M ACRONUTRIENTS AND HEALTHFUL DIETS High Protein Diets Osteoporosis There is a substantial amount of literature that documents the increase in urinary excretion of calcium with increasing protein intake (Allen et al., 1979; Heaney, 1993; Lemann, 1999). The magnitude of this effect for a doubling of the protein intake, in the absence of change in any other nutrient, is a 50 percent increase in urinary calcium (Heaney, 1993). This has two potential detrimental consequences: loss of bone calcium and increased risk of renal calcium stone formation. Loss of calcium from bone is thought to occur because of bone mineral resorption that provides the buffer for the acid produced by the oxidation of the sulfur amino acids of protein (Barzel and Massey, 1998). However, although increased resorption of bone with increased protein intake has been shown (Kerstetter et al., 1999; Whiting et al., 1997), whether this in practice leads to bone loss and osteoporosis is controversial (Barzel and Massey, 1998; Heaney, 1998). It has recently been concluded that there may be no need to restrain dietary protein intake. Poor protein status itself leads to bone loss, whereas increased protein intake may lead to increased calcium intake, and bone loss does not occur if calcium intake is adequate (Heaney, 1998). In a recent prospective study of men and women aged 55 to 92 years, consumption of animal protein was positively associated with bone mineral density in women, but not in men (Promislow et al., 2002). In contrast, Dawson-Hughes and Harris (2002) reported no association between protein intake and bone mineral density in 342 healthy men and women aged 65 years and older. However, when the individuals were given cal- cium citrate malate and vitamin D in addition to the high protein intake, there was a favorable change in bone mineral density. Kidney Stones It has been estimated that 12 percent of the population in the United States will suffer from a kidney stone at some time (Sierakowski et al., 1978). The most common form of kidney stone is composed of calcium oxalate, and its formation is promoted by high concentrations of calcium and oxalate in the urine. A high animal protein intake in healthy humans increases urinary calcium and oxalate and the overall probability of form- ing kidney stones by 250 percent (Robertson et al., 1979). Conversely, restricting protein intake improved the lithogenic profile in hypercalciuric patients (Giannini et al., 1999). Also, the incidence of calcium oxalate stones has been shown to be associated with consumption of animal pro- tein (Curhan et al., 1996; Robertson and Peacock, 1982). In contrast, the
842 DIETARY REFERENCE INTAKES only long-term prospective trial (4.5 years) of the effect of animal protein restriction on stone formation in newly diagnosed patients with calcium stones gave a negative result (Hiatt et al., 1996). The relative risk factor for recurrent stone formation was 5.6 (confidence interval 1.2â26.1), suggest- ing that the dietary advice was detrimental. In this study, 50 patients were given low animal protein (56 to 64 g/d) and high fiber, plus adequate fluid and calcium, whereas 49 control patients were only instructed to take adequate water and calcium. However, as protein intake was not the only variable, and in view of the data described above suggesting benefits from lower protein intake, further investigation is necessary. Renal Failure Restriction of dietary protein intake is known to lessen the symptoms of chronic renal insufficiency (Walser, 1992). This raises two related, but distinct questions: Do high protein diets have some role in the develop- ment of chronic renal failure? Do high protein intakes accelerate the pro- gression of chronic renal failure? The concept that protein restriction might delay the deterioration of the kidney with age was based on studies in rats in which low energy or low protein diets attenuated the develop- ment of chronic renal failure (Anderson and Brenner, 1986, 1987). Walser (1992) has argued that this mechanism is unlikely to operate in humans. In particular, the decline in kidney function in the rat is mostly due to glomerulosclerosis, whereas in humans it is due mostly to a decline in filtration by nonsclerotic nephrons. Also, when creatinine clearance was measured in men at 10- to 18-year intervals, the decline with age did not correlate with dietary protein intake (Tobin and Spector, 1986). Correla- tion of creatinine clearance with protein intake showed a linear relation- ship with a positive gradient (Lew and Bosch, 1991), suggesting that the low protein intake itself decreased renal function. These factors point to the conclusion that the protein content of the diet is not responsible for the progressive decline in kidney function with age. Coronary Artery Disease It is well documented that high dietary protein in rabbits induces hypercholesterolemia and arteriosclerosis (Czarnecki and Kritchevsky, 1993). However, this effect has not been consistently shown in either swine (Luhman and Beitz, 1993; Pfeuffer et al., 1988) or humans. In humans, analysis of data from the Nursesâ Health Study showed an inverse relation- ship between protein intake and risk of cardiovascular disease (Hu et al., 1999). The association was weak but suggests that high protein intake does not increase the risk of cardiovascular disease. Similar conclusions have
843 M ACRONUTRIENTS AND HEALTHFUL DIETS been reached in observational studies showing an inverse relationship between protein intake and blood pressure (Obarzanek et al., 1996) and that replacement of carbohydrate with protein resulted in lower very low density cholesterol, low density cholesterol, and triglycerides (Wolfe and Piché, 1999). Obesity A number of short-term studies indicate that protein intake exerts a more powerful effect on satiety than either carbohydrate or fat (Hill and Blundell, 1990; Rolls et al., 1988; Stubbs et al., 1996). However, some epi- demiological studies have shown a positive correlation between protein intake and body fatness, body mass index, and subscapular skinfold (Buemann et al., 1995; Rolland-Cachera et al., 1995). In contrast, a 6-month randomized trial demonstrated that the replacement of some dietary carbohydrate by protein improved weight loss as part of a reduced fat diet (Skov et al., 1999). Cancer The fact that the growth of tumor cells in culture is often increased by high amino acid concentrations (Breillout et al., 1990; Collins et al., 1998) raises concern that high dietary protein intake might enhance the inci- dence or the progression of cancer. Reviews of the literature on colon cancer have concluded that high meat intake may be associated with increased risk, but that high total protein intake is not (Clinton, 1993; Giovannucci and Willett, 1994; Parnaud and Corpet, 1997). A lack of cor- relation with total protein intake has been found in a case-control study (Slattery et al., 1997), but other studies have reported both increased (Slattery et al., 1994) and decreased (Kato et al., 1997) risk. For breast cancer, the geographical distribution of incidence is corre- lated with the availability of dietary protein, especially animal protein (Clinton, 1993). Furthermore, migration to an area with typically higher protein intakes is associated with increased risk of breast cancer (Buell, 1973; Buell and Dunn, 1965). In accord with this, several studies have indicated an association among breast cancer and the intakes of animal protein and fat (Hislop et al., 1986; Lubin et al., 1981, 1986). However, others showed a relationship with fat, but not protein intake (Miller et al., 1978; Phillips, 1975). More recently, a case-control study on 2,569 patients and 2,588 controls showed a slightly negative relationship between total protein and breast cancer (Decarli et al., 1997). Another case-control study on 180 breast-cancer patients and 829 controls also showed no relation-
844 DIETARY REFERENCE INTAKES ship with total protein intake, but there was an increased risk ratio for meat consumption (Toniolo et al., 1994). For other types of tumors, there also is no clear indication of greater risk with higher protein intakes. Total protein intake was not associated with increased risk of lung cancer (Lei et al., 1996), prostate cancer (Schuurman et al., 1999), endometrial cancer (Barbone et al., 1993; Shu et al., 1993), oral and pharynx cancer (Franceschi et al., 1999), esophogeal cancer (Gao et al., 1994), and non-Hodgkinâs lymphoma (Chiu et al., 1996; Ward et al., 1994), although some studies detected a positive relationship with animal protein (Chiu et al., 1996; Shu et al., 1993) or cured meat consumption (Schuurman et al., 1999). Moreover, in some of these studies, there was an inverse relationship with total protein intake (Barbone et al., 1993; Franceschi et al., 1999; Gao et al., 1994). On the other hand, higher protein intake was associated with an increased risk of cancer of the upper digestive tract (De Stefani et al., 1999) and kidney (Chow et al., 1994). Overall, despite the demonstration of a positive influence of dietary fat and total energy, as well as meat (especially red meat), on some types of tumors, no clear role for total protein has yet emerged. The current state of the literature, therefore, does not permit any recommendation of an upper limit to be made on the basis of cancer risk. Acceptable Macronutrient Distribution Range There is no evidence to suggest that the Acceptable Macronutrient Distribution Range (AMDR) for protein should be at levels below the Rec- ommended Dietary Allowance (RDA) for protein (about 10 percent of energy) for adults. There was insufficient evidence to suggest a UL for protein (see Chapter 10) and insufficient data to suggest an upper limit for an AMDR for protein. To complement the AMDRs for fat (20 to 35 per- cent energy) and carbohydrate (45 to 65 percent energy) for adults, pro- tein intakes may range from 10 to 35 percent of energy intake to ensure a nutritionally adequate diet. For young and older children, the RDA is approximately 5 and 10 percent of energy, respectively. To complement the AMDR for fat (30 to 40 percent of energy) and carbohydrate (45 to 65 percent of energy) for young children and for older children (25 to 35 percent of energy from fat and 45 to 65 percent of energy from carbo- hydrate), protein intakes may range from 5 to 20 percent for young chil- dren and 10 to 30 percent for older children.
845 M ACRONUTRIENTS AND HEALTHFUL DIETS REFERENCES Abate N, Garg A, Peshock RM, Stray-Gundersen J, Adams-Huet B, Grundy SM. 1996. Relationship of generalized and regional adiposity to insulin sensitivity in men with NIDDM. Diabetes 45:1684â1693. Abbey M, Belling GB, Noakes M, Hirata F, Nestel PJ. 1993. Oxidation of low- density lipoproteins: Intraindividual variability and the effect of dietary linoleate supplementation. Am J Clin Nutr 57:391â398. Abbott WGH, Boyce VL, Grundy SM, Howard BV. 1989. Effects of replacing satu- rated fat with complex carbohydrate in diets of subjects with NIDDM. Diabetes Care 12:102â107. Adams SO, Barr GD, Huenemann RL. 1978. Effect of nutritional supplementation in pregnancy. I. Outcome of pregnancy. J Am Diet Assoc 72:144â147. à gren JJ, Hänninen O, Julkunen A, Fogelholm L, Vidgren H, Schwab U, Pynnönen O, Uusitupa M. 1996. Fish diet, fish oil and docosahexaenoic acid rich oil lower fasting and postprandial plasma lipid levels. Eur J Clin Nutr 50:765â771. Albert CM, Hennekens CH, OâDonnell CJ, Ajani UA, Carey VJ, Willett WC, Ruskin JN, Manson JE. 1998. Fish consumption and risk of sudden cardiac death. J Am Med Assoc 279:23â28. Allen LH, Oddoye EA, Margen S. 1979. Protein-induced calciuria: A longer term study. Am J Clin Nutr 32:741â749. Allison DB, Egan K, Barraj LM, Caughman C, Infante M, Heimbach J. 1999. Esti- mated intakes of trans fatty and other fatty acids in the US population. J Am Diet Assoc 99:166â174. American Diabetes Association. 2001. Screening for diabetes. D iabetes Care 24:S21âS24. Anderson S, Brenner BM. 1986. Effects of aging on the renal glomerulus. Am J Med 80:435â442. Anderson S, Brenner BM. 1987. The aging kidney: Structure, function, mecha- nisms, and therapeutic implications. J Am Geriatr Soc 35:590â593. Andreassi M, Forleo P, Di Lorio A, Masci S, Abate G, Amerio P. 1997. Efficacy of γ- linolenic acid in the treatment of patients with atopic dermatitis. J Int Med Res 25:266â274. Annuzzi G, Rivellese A, Capaldo B, Di Marino L, Iovine C, Marotta G, Riccardi G. 1991. A controlled study on the effects of n-3 fatty acids on lipid and glucose metabolism in non-insulin-dependent diabetic patients. Atherosclerosis 87:65â73. Anti M, Marra G, Armelao F, Bartoli GM, Ficarelli R, Percesepe A, De Vitis I, Maria G, Sofo L, Rapaccini GL. 1992. Effect of omega-3 fatty acids on rectal mucosal cell proliferation in subjects at risk for colon cancer. Gastroenterology 103:883â891. Arntzenius AC, Kromhout D, Barth JD, Reiber JHC, Bruschke AVG, Buis B, van Gent CM, Kempen-Voogd N, Strikwerda S, van der Velde EA. 1985. Diet, lipo- proteins, and the progression of coronary atherosclerosis. The Leiden Inter- vention Trial. N Engl J Med 312:805â811. ARS (Agricultural Research Service). 1998. Food and Nutrient Intakes by Individuals in the United States, by Sex and Age, 1994â96. Washington, DC: U.S. Department of Agriculture. Ascherio A, Rimm EB, Stampfer MJ, Giovannucci EL, Willett WC. 1995. Dietary intake of marine n-3 fatty acids, fish intake, and the risk of coronary disease among men. N Engl J Med 332:977â982.
846 DIETARY REFERENCE INTAKES Ascherio A, Rimm EB, Giovannucci EL, Spiegelman D, Stampfer M, Willett WC. 1996. Dietary fat and risk of coronary heart disease in men: Cohort follow up study in the United States. Br Med J 313:84â90. Astrup A, Grunwald GK, Melanson EL, Saris WH, Hill JO. 2000. The role of low-fat diets in body weight control: A meta-analysis of ad libitum dietary intervention studies. Int J Obes Relat Metab Disord 24:1545â1552. Atkin L-M, Davies PSW. 2000. Diet composition and body composition in pre- school children. Am J Clin Nutr 72:15â21. Austin MA, King MC, Vranizan KM, Krauss RM. 1990. Atherogenic lipoprotein phenotype: A proposed genetic marker for coronary heart disease risk. Circu- lation 82:495â506. Axelrod L, Camuso J, Williams E, Kleinman K, Briones E, Schoenfeld D. 1994. Effects of a small quantity of Ï-3 fatty acids on cardiovascular risk factors in NIDDM. Diabetes Care 17:37â44. Badaloo A, Boyne M, Reid M, Persaud C, Forrester T, Millward DJ, Jackson AA. 1999. Dietary protein, growth and urea kinetics in severely malnourished chil- dren and during recovery. J Nutr 129:969â979. Baer JT. 1993. Improved plasma cholesterol levels in men after a nutrition educa- tion program at the worksite. J Am Diet Assoc 93:658â663. Ballew C, Kuester S, Gillespie C. 2000. Beverage choices affect adequacy of childrenâs nutrient intakes. Arch Pediatr Adolesc Med 154:1148â1152. Bang HO, Dyerberg J, Hjørne N. 1976. The composition of food consumed by Greenland Eskimos. Acta Med Scand 200:69â73. Banni S, Angioni E, Casu V, Melis MP, Carta G, Corongiu FP, Thompson H, Ip C. 1999. Decrease in linoleic acid metabolites as a potential mechanism in cancer risk reduction by conjugated linoleic acid. Carcinogenesis 20:1019â1024. Barbone F, Austin H, Partridge EE. 1993. Diet and endometrial cancer: A case- control study. Am J Epidemiol 137:393â403. Barinagarrementeria F, González-Duarte A, Cantú-Brito C. 1998. Prothrombic states and cerebral ischemia. Rev Neurol 26:85â91. Bartsch H, Nair J, Owen RW. 1999. Dietary polyunsaturated fatty acids and cancers of the breast and colorectum: Emerging evidence for their role as risk modifiers. Carcinogenesis 20:2209â2218. Barzel US, Massey LK. 1998. Excess dietary protein can adversely affect bone. J Nutr 128:1051â1053. Bassett DR, Abel M, Moellering RC, Rosenblatt G, Stokes J. 1969. Coronary heart disease in Hawaii: Dietary intake, depot fat, âstress,â smoking, and energy bal- ance in Hawaiian and Japanese men. Am J Clin Nutr 22:1483â1503. Becker N, Illingworth R, Alaupovic P, Connor WE, Sundberg EE. 1983. Effects of saturated, monounsaturated, and Ï-6 polyunsaturated fatty acids on plasma lipids, lipoproteins, and apoproteins in humans. Am J Clin Nutr 37:355â360. Beck-Nielsen H, Pedersen O, Lindskov HO. 1980. Impaired cellular insulin bind- ing and insulin sensitivity induced by high-fructose feeding in normal subjects. Am J Clin Nutr 33:273â278. Behall KM, Scholfield DJ, Lee K, Powell AS, Moser PB. 1987. Mineral balance in adult men: Effect of four refined fibers. Am J Clin Nutr 46:307â314. Bennett PH, Knowler WC, Baird HR, Butler WJ, Pettitt DJ, Reid JM. 1984. Diet and the development of noninsulin-dependent diabetes mellitus: An epidemio- logical perspective. In: Pozza G, ed. Diet, Diabetes, and Atherosclerosis. New York: Raven Press. Pp. 109â119.
847 M ACRONUTRIENTS AND HEALTHFUL DIETS Berenson GS, Wattigney WA, Tracy RE, Newman WP, Srinivasan SR, Webber LS, Dalferes ER, Strong JP. 1992. Atherosclerosis of the aorta and coronary arteries and cardiovascular risk factors in persons aged 6 to 30 years and studied at necropsy (The Bogalusa Heart Study). Am J Cardiol 70:851â858. Berry EM, Eisenberg S, Haratz D, Friedlander Y, Norman Y, Kaufmann NA, Stein Y. 1991. Effects of diets rich in monounsaturated fatty acids on plasma lipo- proteinsâThe Jerusalem Nutrition Study: High MUFAs vs high PUFAs. Am J Clin Nutr 53:899â907. Berry EM, Eisenberg S, Friedlander Y, Harats D, Kaufmann NA, Norman Y, Stein Y. 1992. Effects of diets rich in monounsaturated fatty acids on plasma lipopro- teinsâThe Jerusalem Nutrition Study. II. Monounsaturated fatty acids vs car- bohydrates. Am J Clin Nutr 56:394â403. Bhathena SJ, Berlin E, Judd JT, Kim YC, Law JS, Bhagavan HN, Ballard-Barbash R, Nair PP. 1991. Effects of Ï3 fatty acids and vitamin E on hormones involved in carbohydrate and lipid metabolism in men. Am J Clin Nutr 54:684â688. Billman GE, Kang JX, Leaf A. 1999. Prevention of sudden cardiac death by dietary pure Ï-3 polyunsaturated fatty acids in dogs. Circulation 99:2452â2457. Black HS, Herd JA, Goldberg LH, Wolf JE, Thornby JI, Rosen T, Bruce S, Tschen JA, Foreyt JP, Scott LW, Jaax S, Andrews K. 1994. Effect of a low-fat diet on the incidence of actinic keratosis. N Engl J Med 330:1272â1275. Bladbjerg EM, Marckmann P, Sandström B, Jespersen J. 1994. Non-fasting factor VII coagulant activity (FVII:C) increased by high fat diet. Thromb Haemost 71:755â758. Blankson H, Stakkestad JA, Fagertun H, Thom E, Wadstein J, Gudmundsen O. 2000. Conjugated linoleic acid reduces body fat mass in overweight and obese humans. J Nutr 130:2943â2948. Bloemberg BPM, Kromhout D, Goddijn HE, Jansen A, Obermann-de Boer GL. 1991. The impact of the Guidelines for a Healthy Diet of the Netherlands Nutrition Council on total and high density lipoprotein cholesterol in hyper- cholesterolemic free-living men. Am J Epidemiol 134:39â48. Blundell JE, Burley VJ, Cotton JR, Lawton CL. 1993. Dietary fat and the control of energy intake: Evaluating the effects of fat on meal size and postmeal satiety. Am J Clin Nutr 57:772Sâ778S. Bobroff EM, Kissileff HR. 1986. Effects of changes in palatability on food intake and the cumulative food intake curve in man. Appetite 7:85â96. Bolton-Smith C. 1996. Intake of sugars in relation to fatness and micronutrient adequacy. Int J Obes Relat Metab Disord 20:S31âS33. Bolton-Smith C, Woodward M. 1994. Coronary heart disease: Prevalence and dietary sugars in Scotland. J Epidemiol Community Health 48:119â122. Bolton-Smith C, Woodward M. 1995. Antioxidant vitamin adequacy in relation to consumption of sugars. Eur J Clin Nutr 49:124â133. Bønaa KH, Bjerve KS, Nordøy A. 1992. Habitual fish consumption, plasma phospholipid fatty acids, and serum lipids: The Tromsø Study. Am J Clin Nutr 55:1126â1134. Bonanome A, Pagnan A, Biffanti S, Opportuno A, Sorgato F, Dorella M, Maiorino M, Ursini F. 1992. Effect of dietary monounsaturated and polyunsaturated fatty acids on the susceptibility of plasma low density lipoproteins to oxidative modification. Arterioscler Thromb 12:529â533. Borkman M, Campbell LV, Chisholm DJ, Storlien LH. 1991. Comparison of the effects on insulin sensitivity of high carbohydrate and high fat diets in normal subjects. J Clin Endocrinol Metab 72:432â437.
848 DIETARY REFERENCE INTAKES Borkman M, Storlien LH, Pan DA, Jenkins AB, Chisholm DJ, Campbell LV. 1993. The relation between insulin sensitivity and the fatty-acid composition of skeletal-muscle phospholipids. N Engl J Med 328:238â244. Boulton TJC, Magarey AM. 1995. Effects of differences in dietary fat on growth, energy and nutrient intake from infancy to eight years of age. Acta Paediatr 84:146â150. Boutron MC, Wilpart M, Faivre J. 1991. Diet and colorectal cancer. Eur J Cancer Prev 1:13â20. Bowman MP, Van Doren J, Taper LJ, Thye FW, Ritchey SJ. 1988. Effect of dietary fat and cholesterol on plasma lipids and lipoprotein fractions in normolipidemic men. J Nutr 118:555â560. Bowman SA. 1999. Diets of individuals based on energy intakes from added sugars. Fam Econ Nutr Rev 12:31â38. Boyar AP, Rose DP, Loughridge JR, Engle A, Palgi A, Laakso K, Kinne D, Wynder EL. 1988. Response to a diet low in total fat in women with postmenopausal breast cancer: A pilot study. Nutr Cancer 11:93â99. Boyd NF, Cousins M, Beaton M, Kriukov V, Lockwood G, Tritchler D. 1990. Quan- titative changes in dietary fat intake and serum cholesterol in women: Results from a randomized, controlled trial. Am J Clin Nutr 52:470â476. Boyd NF, Martin LJ, Noffel M, Lockwood GA, Tritchler DL. 1993. A meta-analysis of studies of dietary fat and breast cancer risk. Br J Cancer 68:627â636. Bray GA, Popkin BM. 1998. Dietary fat intake does affect obesity! Am J Clin Nutr 68:1157â1173. Breillout F, Antoine E, Poupon MF. 1990. Methionine dependency of malignant tumors: A possible approach for therapy. J Natl Cancer Inst 82:1628â1632. Brodie AE, Manning VA, Ferguson KR, Jewell DE, Hu CY. 1999. Conjugated linoleic acid inhibits differentiation of pre- and post-confluent 3T3-L1 preadipocytes but inhibits cell proliferation only in preconfluent cells. J Nutr 129:602â606. Brussaard JH, Katan MB, Groot PHE, Havekes LM, Hautvast JGAJ. 1982. Serum lipoproteins of healthy persons fed a low-fat diet or a polyunsaturated fat diet for three months. A comparison of two cholesterol-lowering diets. Atherosclerosis 42:205â219. Budohoski L, Panczenko-Kresowska B, Langfort J, Zoernicka E, Dubaniewicz A, Ziemlan´ski S´, Challiss RAJ, Newsholme WA. 1993. Effects of saturated and polyunsaturated fat enriched diet on the skeletal muscle insulin sensitivity in young rats. J Physiol Pharmacol 44:391â398. Buell P. 1973. Changing incidence of breast cancer in Japanese-American women. J Natl Cancer Inst 51:1479â1483. Buell P, Dunn JE. 1965. Cancer mortality among Japanese Issei and Nisei of Cali- fornia. Cancer 18:656â664. Buemann B, Tremblay A, Bouchard C. 1995. Social class interacts with the associa- tion between macronutrient intake and subcutaneous fat. Int J Obes Relat Metab Disord 19:770â775. Burmeister LA, Valdivia T, Nuttal FQ. 1991. Adult hereditary fructose intolerance. Arch Intern Med 151:773â776. Burr ML, Fehily AM, Gilbert JF, Rogers S, Holliday RM, Sweetnam PM, Elwood PC, Deadman NM. 1989a. Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: Diet and Reinfarction Trial (DART). Lancet 2:757â761.
849 M ACRONUTRIENTS AND HEALTHFUL DIETS Burr ML, Fehily AM, Rogers S, Welsby E, King S, Sandham S. 1989b. Diet and Reinfarction Trial (DART): Design, recruitment, and compliance. Eur Heart J 10:558â567. Burr ML, Sweetnam PM, Fehily AM. 1994. Diet and reinfarction. Eur Heart J 15:1152â1153. Buzzard IM, Asp EH, Chlebowski RT, Boyar AP, Jeffery RW, Nixon DW, Blackburn GL, Jochimsen PR, Scanlon EF, Insull W, Elashoff RM, Butram R, Wynder EL. 1990. Diet intervention methods to reduce fat intake: Nutrient and food group composition of self-selected low-fat diets. J Am Diet Assoc 90:42â50, 53. Calbet JA, MacLean DA. 1997. Role of caloric content on gastric emptying in humans. J Physiol 498:553â559. Calviello G, Palozza P, Piccioni E, Maggiano N, Frattucci A, Franceschelli P, Baroli GM. 1998. Dietary supplementation with eicosapentaenoic and docosahexaenoic acid inhibits growth of Morris hepatocarcinoma 3924A in rats: Effects on proliferation and apoptosis. Int J Cancer 75:699â705. Campbell TC, Parpia B, Chen J. 1998. Diet, lifestyle, and the etiology of coronary artery disease: The Cornell China Study. Am J Cardiol 82:18Tâ21T. Castelli WP, Anderson K, Wilson PWF, Levy D. 1992. Lipids and risk of coronary heart disease. The Framingham Study. Ann Epidemiol 2:23â28. Caygill CPJ, Hill MJ. 1995. Fish, n-3 fatty acids and human colorectal and breast cancer mortality. Eur J Cancer Prev 4:329â332. Caygill CPJ, Charlett A, Hill MJ. 1996. Fat, fish, fish oil and cancer. Br J Cancer 74:159â164. CDC (Centers for Disease Control and Prevention). 1994. Daily dietary fat and total food-energy intakesâThird National Health and Nutrition Examination Survey, Phase 1, 1988â91. Morb Mortal Wkly Rep 43:116â117, 123â125. Cesano A, Visonneau S, Scimeca JA, Kritchevsky D, Santoli D. 1998. Opposite effects of linoleic acid and conjugated linoleic acid on human prostatic cancer in SCID mice. Anticancer Res 18:833â838. Chandra RK. 1972. Immunocompetence in undernutrition. J Pediatr 81:1194â1200. Chandra RK. 1991. 1990 McCollum Award lecture. Nutrition and immunity: Lessons from the past and new insights into the future. A m J Clin Nutr 53:1087â1101. Chandra RK, Newberne PM. 1977. Nutrition, Immunity, and Infection: Mechanisms of Interactions. New York: Plenum Press. Chandra RK, Chandra S, Gupta S. 1984. Antibody affinity and immune complexes after immunization with tetanus toxoid in protein-energy malnutrition. Am J Clin Nutr 40:131â134. Chen M, Bergman RN, Porte D. 1988. Insulin resistance and β-cell dysfunction in aging: The importance of dietary carbohydrate. J C lin Endocrinol Metab 67:951â957. Chicco A, DâAlessandro ME, Karabatas L, Gutman R, Lombardo YB. 1996. Effect of moderate levels of dietary fish oil on insulin secretion and sensitivity, and pancreas insulin content in normal rats. Ann Nutr Metab 40:61â70. Chilton-Lopez T, Surette ME, Swan DD, Fonteh AN, Johnson MM, Chilton FH. 1996. Metabolism of gammalinolenic acid in human neutrophils. J Immunol 156:2941â2947. Chisholm KW, OâDea K. 1987. Effect of short-term consumption of a high fat diet on glucose tolerance and insulin sensitivity in the rat. J Nutr Sci Vitaminol 3:377â390.
850 DIETARY REFERENCE INTAKES Chisolm GM, Steinberg D. 2000. The oxidative modification hypothesis of athero- genesis: An overview. Free Radic Biol Med 28:1815â1826. Chiu BC, Cerhan JR, Folsom AR, Sellers TA, Kushi LH, Wallace RB, Zheng W, Potter JD. 1996. Diet and risk of non-Hodgkin lymphoma in older women. J Am Med Assoc 275:1315â1321. Choi Y, Kim Y-C, Han Y-B, Park Y, Pariza M, Ntambi JM. 2000. The trans-10,cis-12 isomer of conjugated linoleic acid downregulates stearoyl-CoA desaturase 1 gene expression in 3T3-L1 adipocytes. J Nutr 130:1920â1924. Chow WH, Gridley G, McLaughlin JK, Mandel JS, Wacholder S, Blot WJ, Niwa S, Fraumeni JF. 1994. Protein intake and risk of renal cell cancer. J Natl Cancer Inst 86:1131â1139. Christensen JH, Gustenhoff P, Korup E, Aarøe J, Møller JM, Rasmussen K, Dyerberg J, Schmidt EB. 1997. n-3 Polyunsaturated fatty acids, heart rate variability and ventricular arrhythmias in patients with previous myocardial infarcts. Ugeskr Laeger 159:5525â5529. Christensen JH, Christensen MS, Dyerberg J, Schmidt EB. 1999. Heart rate vari- ability and fatty acid content of blood cell membranes: A dose-response study with n-3 fatty acids. Am J Clin Nutr 70:331â337. Clarke R, Frost C, Collins R, Appleby P, Peto R. 1997. Dietary lipids and blood cholesterol: Quantitative meta-analysis of metabolic ward studies. Br Med J 314:112â117. Clinton SK. 1993. Dietary protein and the origins of human cancer. In: Liepa GU, Beitz DC, Beynen AC, Gorman MA, eds. Dietary Proteins: How They Alleviate Disease and Promote Better Health. Champaign, IL: American Oil Chemistsâ Society. Pp. 84â122. Clore JN, Li J, Gill R, Gupta S, Spencer R, Azzam A, Zuelzer W, Rizzo WB, Blackard WG. 1998. Skeletal muscle phosphatidylcholine fatty acids and insulin sensitivity in normal humans. Am J Physiol 275:E665âE670. Colditz GA, Willett WC, Stampfer MJ, London SJ, Segal MR, Speizer FE. 1990. Patterns of weight change and their relation to diet in a cohort of healthy women. Am J Clin Nutr 51:1100â1105. Colditz GA, Manson JE, Stampfer MJ, Rosner B, Willett WC, Speizer FE. 1992. Diet and risk of clinical diabetes in women. Am J Clin Nutr 55:1018â1023. Collins CL, Wasa M, Souba WW, Abcouwer SF. 1998. Determinants of glutamine dependence and utilization by normal and tumor-derived breast cell lines. J Cell Physiol 176:166â178. Coudray C, Bellanger J, Castiglia-Delavaud C, Rémésy C, Vermorel M, Rayssignuier Y. 1997. Effect of soluble or partly soluble dietary fibres supplementation on absorption and balance of calcium, magnesium, iron and zinc in healthy young men. Eur J Clin Nutr 51:375â380. Coulston AM, Liu GC, Reaven GM. 1983. Plasma glucose, insulin and lipid responses to high-carbohydrate low-fat diets in normal humans. Metabolism 32:52â56. Coulston AM, Hollenbeck CB, Swislocki AL, Chen YD, Reaven GM. 1987. Deleterious metabolic effects of high-carbohydrate, sucrose-containing diets in patients with non-insulin-dependent diabetes mellitus. Am J Med 82:213â220. Cunningham DC, Harrison LY, Shultz TD. 1997. Proliferative responses of normal human mammary and MCF-7 breast cancer cells to linoleic acid, conjugated linoleic acid and eicosanoid synthesis inhibitors in culture. Anticancer Res 17:197â204.
851 M ACRONUTRIENTS AND HEALTHFUL DIETS Curb JD, Wergowske G, Dobbs JC, Abbott RD, Huang B. 2000. Serum lipid effects of a high-monounsaturated fat diet based on macadamia nuts. Arch Intern Med 160:1154â1158. Curhan GC, Willet WC, Rimm EB, Stampfer MJ. 1996. A prospective study of dietary calcium and other nutrients and the risk of kidney stones in men: 8 Year follow-up. In: Pak CY, Resnick MI, Preminger GM, eds. Urolithiasis. Dallas, TX: Millet. Pp. 164â166. Czarnecki SK, Kritchevsky D. 1993. Dietary protein and atherosclerosis. In: Liepa GU, Beitz DC, Beynen AC, Gorman MA, eds. Dietary Proteins: How They Alleviate Disease and Promote Better Health. Champaign, IL: American Oil Chemistsâ Society. Pp. 42â56. Davies PS. 1997. Diet composition and body mass index in pre-school children. Eur J Clin Nutr 51:443â448. Daviglus ML, Stamler J, Orencia AJ, Dyer AR, Liu K, Greenland P, Walsh MK, Morris D, Shekelle RB. 1997. Fish consumption and the 30-year risk of fatal myocardial infarction. N Engl J Med 336:1046â1053. Dawson-Hughes B, Harris SS. 2002. Calcium intake influences the association of protein intake with rates of bones loss in elderly men and women. Am J Clin Nutr 75:773â779. Decarli A, Favero A, La Vecchia C, Russo A, Ferraroni M, Negri E, Franceschi S. 1997. Macronutrients, energy intake, and breast cancer risk: Implications from different models. Epidemiology 8:425â428. De Caterina R, Liao JK, Libby P. 2000. Fatty acid modulation of endothelial activa- tion. Am J Clin Nutr 71:213â223. de Deckere EAM, van Amelsvoort JMM, McNeill GP, Jones P. 1999. Effects of conjugated linoleic acid (CLA) isomers on lipid levels and peroxisome proliferation in the hamster. Br J Nutr 82:309â317. DeLany JP, Vivian VM, Snook JT, Anderson PA. 1990. Effects of fish oil on serum lipids in men during a controlled feeding trial. Am J Clin Nutr 52:477â485. de Lorgeril M, Renaud S, Mamelle N, Salen P, Martin J-L, Monjaud I, Guidollet J, Touboul P, Delaye J. 1994. Mediterranean alpha-linolenic acid-rich diet in secondary prevention of coronary heart disease. Lancet 343:1454â1459. de Lorgeril M, Salen P, Martin J-L, Monjaud I, Delaye J, Mamelle N. 1999. Mediter- ranean diet, traditional risk factors, and the rate of cardiovascular complica- tions after myocardial infarction. Final report of the Lyon Diet Heart Study. Circulation 99:779â785. Delzenne N, Aertssens J, Verplaetse H, Roccaro M, Roberfroid M. 1995. Effect of fermentable fructo-oligosaccharides on mineral, nitrogen and energy diges- tive balance in the rat. Life Sci 57:1579â1587. Demigné C, Levrat M-A, Rémésy C. 1989. Effects of feeding fermentable carbo- hydrates on the cecal concentrations of minerals and their fluxes between the cecum and blood plasma in the rat. J Nutr 119:1625â1630. Deschner EE, Lytle JS, Wong G, Ruperto JF, Newmark HL. 1990. The effect of dietary omega-3 fatty acids (fish oil) on azoxymethanol-induced focal areas of dysplasia and colon tumor incidence. Cancer 66:2350â2356. Després J-P. 1993. Abdominal obesity as important component of insulin-resistance syndrome. Nutrition 9:452â459. De Stefani E, Deneo-Pellegrini H, Mendilaharsu M, Carzoglio JC, Ronco A. 1997a. Dietary fat and lung cancer: A case-control study in Uruguay. Cancer Causes Control 8:913â921.
852 DIETARY REFERENCE INTAKES De Stefani E, Mendilaharsu M, Deneo-Pellegrini H, Ronco A. 1997b. Influence of dietary levels of fat, cholesterol, and calcium on colorectal cancer. Nutr Cancer 29:83â89. De Stefani E, Ronco A, Mendilaharsu M, Deneo-Pellegrini H. 1999. Diet and risk of cancer of the upper aerodigestive tract. II. Nutrients. Oral Oncol 35:22â26. Djoussé L, Pankow JS, Eckfeldt JH, Folsom AR, Hopkins PN, Province MA, Hong Y, Ellison RC. 2001. Relation between dietary linolenic acid and coronary artery disease in the National Heart, Lung, and Blood Institute Family Heart Study. Am J Clin Nutr 74:612â619. Dolecek TA. 1992. Epidemiological evidence of relationships between dietary poly- unsaturated fatty acids and mortality in the Multiple Risk Factor Intervention Trial. Proc Soc Exp Med Biol 200:177â182. Dreon DM, Frey-Hewitt B, Ellsworth N, Williams PT, Terry RB, Wood PD. 1988. Dietary fat:carbohydrate ratio and obesity in middle-aged men. Am J Clin Nutr 47:995â1000. Drewnowski A. 1999. Intense sweeteners and energy density of foods: Implications for weight control. Eur J Clin Nutr 53:757â763. Drewnowski A, Greenwood MR. 1983. Cream and sugar: Human preferences for high-fat foods. Physiol Behav 30:629â633. Duncan KH, Bacon JA, Weinsier RL. 1983. The effects of high and low energy density diets on satiety, energy intake, and eating time of obese and nonobese subjects. Am J Clin Nutr 37:763â767. Dunnigan MG, Fyfe T, McKiddie MT, Crosbie SM. 1970. The effects of isocaloric exchange of dietary starch and sucrose on glucose tolerance, plasma insulin and serum lipids in man. Clin Sci 38:1â9. Durrant M, Royston P. 1979. Short-term effects of energy density on salivation, hunger and appetite in obese subjects. Int J Obes 3:335â347. Dyerberg J, Bang HO. 1979. Haemostatic function and platelet polyunsaturated fatty acids in Eskimos. Lancet 2:433â435. Emmett PM, Heaton KW. 1995. Is extrinsic sugar a vehicle for dietary fat? Lancet 345:1537â1540. Eritsland J, Arnesen H, Seljeflot I, Høstmark AT. 1994a. Long-term metabolic effects of n-3 polyunsaturated fatty acids in patients with coronary artery dis- ease. Am J Clin Nutr 61:831â836. Eritsland J, Seljeflot I, Abdelnoor M, Arnesen H, Torjesen PA. 1994b. Long-term effects of n-3 fatty acids on serum lipids and glycaemic control. Scand J Clin Lab Invest 54:273â280. Ernst N, Fisher M, Smith W, Gordon T, Rifkind BM, Little JA, Mishkel MA, Williams OD. 1980. The association of plasma high-density lipoprotein cholesterol with dietary intake and alcohol consumption. The Lipid Research Clinics Program Prevalence Study. Circulation 62:IV41âIV52. Fairweather-Tait SM, Wright AJA. 1990. The effects of sugar-beet fibre and wheat bran on iron and zinc absorption in rats. Br J Nutr 64:547â552. FAO/WHO (Food and Agricultural Organization/World Health Organization). 1996. Sixth World Food and Nutrition Survey. Rome: FAO. Farrell TG, Bashir Y, Cripps T, Malik M, Poloniecki J, Bennett ED, Ward DE, Camm AJ. 1991. Risk stratification for arrhythmic events in postinfarction patients based on heart rate variability, ambulatory electrocardiographic vari- ables and the signal-averaged electrocardiogram. J Am Coll Cardiol 18:687â697.
853 M ACRONUTRIENTS AND HEALTHFUL DIETS Farris RP, Nicklas TA, Myers L, Berenson GS. 1998. Nutrient intake and food group consumption of 10-year-olds by sugar intake level: The Bogalusa Heart Study. J Am Coll Nutr 17:579â585. Fasching P, Ratheiser K, Waldhäusl W, Rohac M, Osterrode W, Nowotny P, Vierhapper H. 1991. Metabolic effects of fish-oil supplementation in patients with impaired glucose tolerance. Diabetes 40:583â589. Fasching P, Ratheiser K, Schneeweiss B, Rohac M, Nowotny P, Waldhausl W. 1996. No effect of short-term dietary supplementation of saturated and poly- and monounsaturated fatty acids on insulin secretion and sensitivity in healthy men. Ann Nutr Metab 40:116â122. Fehily AM, Yarnell JWG, Bolton CH, Butland BK. 1988. Dietary determinants of plasma lipids and lipoproteins: The Caerphilly Study. Eur J Clin Nutr 42:405â413. Fernandez ML, Wilson TA, Conde K, Vergara-Jimenez M, Nicolosi RJ. 1999. Hamsters and guinea pigs differ in their plasma lipoprotein cholesterol distri- bution when fed diets varying in animal protein, soluble fiber, or cholesterol content. J Nutr 129:1323â1332. Feskens EJM, Bowles CH, Kromhout D. 1991a. Carbohydrate intake and body mass index in relation to the risk of glucose tolerance in an elderly population. Am J Clin Nutr 54:136â140. Feskens EJ, Bowles CH, Kromhout D. 1991b. Inverse association between fish intake and risk of glucose intolerance in normoglycemic elderly men and women. Diabetes Care 14:935â941. Feskens EJM, Loeber JG, Kromhout D. 1994. Diet and physical activity as determinants of hyperinsulinemia: The Zutphen Elderly Study. Am J Epidemiol 140:350â360. Feskens EJM, Virtanen SM, Räsänen L, Tuomilehto J, Stengard J, Pekkanen J, Nissinen A, Kromhout D. 1995. Dietary factors determining diabetes and impaired glucose tolerance: A 20-year follow-up of the Finnish and Dutch cohorts of the Seven Countries Study. Diabetes Care 18:1104â1112. Fischer DR, Morgan KJ, Zabik ME. 1985. Cholesterol, saturated fatty acids, poly- unsaturated fatty acids, sodium, and potassium intakes of the United States population. J Am Coll Nutr 4:207â224. Flaten H, Høstmark AT, Kierulf P, Lystad E, Trygg K, Bjerkedal T, Osland A. 1990. Fish-oil concentrate: Effects on variables related to cardiovascular disease. Am J Clin Nutr 52:300â306. Flegal KM. 1999. The obesity epidemic in children and adults: Current evidence and research issues. Med Sci Sports Exerc 31:S509âS514. Flint A, Raben A, Blundell JE, Astrup A. 2000. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int J Obes Relat Metab Disord 24:3â48. Fomon SJ, Thomas LN, Filer LJ, Anderson TA, Nelson SE. 1976. Influence of fat and carbohydrate content of diet on food intake and growth of male infants. Acta Paediatr Scand 65:136â144. Forshee RA, Storey ML. 2001. The role of added sugars in the diet quality of children and adolescents. J Am Coll Nutr 20:32â43. Franceschi S, Levi F, Conti E, Talamini R, Negri E, Dal Maso L, Boyle P, Decarli A, La Vecchia C. 1999. Energy intake and dietary pattern in cancer of the oral cavity and pharynx. Cancer Causes Control 10:439â444. Friedman MI. 1995. Control of energy intake by energy metabolism. Am J Clin Nutr 62:1096Sâ1100S.
854 DIETARY REFERENCE INTAKES Fukagawa NK, Anderson JW, Hageman G, Young VR, Minaker KL. 1990. High- carbohydrate, high-fiber diets increase peripheral insulin sensitivity in healthy young and old adults. Am J Clin Nutr 52:524â528. Gao YT, McLaughlin JK, Gridley G, Blot WJ, Ji BT, Dai Q, Fraumeni JF. 1994. Risk factors for esophageal cancer in Shanghai, China. II. Role of diet and nutri- ents. Int J Cancer 58:197â202. Garg A, Bonanome A, Grundy SM, Zhang Z-J, Unger RH. 1988. Comparison of a high-carbohydrate diet with a high-monounsaturated-fat diet in patients with non-insulin-dependent diabetes mellitus. N Engl J Med 319:829â834. Garg A, Grundy SM, Koffler M. 1992a. Effect of high carbohydrate intake on hyper- glycemia, islet function, and plasma lipoproteins in NIDDM. Diabetes Care 15:1572â1580. Garg A, Grundy SM, Unger RH. 1992b. Comparison of effects of high and low carbohydrate diets on plasma lipoproteins and insulin sensitivity in patients with mild NIDDM. Diabetes 41:1278â1285. Garg A, Bantle JP, Henry RR, Coulston AM, Griver KA, Raatz SK, Brinkley L, Chen Y-DI, Grundy SM, Huet BA, Reaven GM. 1994. Effects of varying carbohydrate content of diet in patients with non-insulin-dependent diabetes mellitus. J Am Med Assoc 271:1421â1428. Gartside PS, Glueck CJ. 1993. Relationship of dietary intake to hospital admission for coronary heart and vascular disease: The NHANES II National Probability Study. J Am Coll Nutr 6:676â684. Gazzaniga JM, Burns TL. 1993. Relationship between diet composition and body fatness, with adjustment for resting energy expenditure and physical activity, in preadolescent children. Am J Clin Nutr 58:21â28. George V, Tremblay A, Després JP, Leblanc C, Bouchard C. 1990. Effect of dietary fat content on total and regional adiposity in men and women. Int J Obes 14:1085â1094. Gerhard GT, Connor SL, Wander RC, Connor WE. 2000. Plasma lipid and lipo- protein responsiveness to dietary fat and cholesterol in premenopausal African American and white women. Am J Clin Nutr 72:56â63. Giannini S, Nobile M, Sartori L, Dalle Carbonare L, Ciuffreda M, Corro P, DâAngelo A, Calo L, Crepaldi G. 1999. Acute effects of moderate dietary protein restriction in patients with idiopathic hypercalciuria and calcium nephrolithiasis. Am J Clin Nutr 69:267â271. Gibney M, Sigman-Grant M, Stanton JL, Keast DR. 1995. Consumption of sugars. Am J Clin Nutr 62:178Sâ194S. Gibson SA. 1993. Consumption and sources of sugars in the diets of British school- children: Are high-sugar diets nutritionally inferior? J Hum Nutr Diet 6:355â371. Gibson SA. 1997. Non-milk extrinsic sugars in the diets of pre-school children: Association with intakes of micronutrients, energy, fat and NSP. Br J Nutr 78:367â378. Gillum RF, Mussolino ME, Madans JH. 1996. The relationship between fish con- sumption and stroke incidence. The NHANES I epidemiologic follow-up study. Arch Intern Med 156:537â542. Ginsberg HN, Barr SL, Gilbert A, Karmally W, Deckelbaum R, Kaplan K, Ramakrishnan R, Holleran S, Dell RB. 1990. Reduction of plasma cholesterol levels in normal men on an American Heart Association Step 1 diet or a Step 1 diet with added monounsaturated fat. N Engl J Med 322:574â579. Giovannucci E, Willet WC. 1994. Dietary factors and risk of colon cancer. Ann Med 26:443â452.
855 M ACRONUTRIENTS AND HEALTHFUL DIETS Giovannucci E, Rimm EB, Colditz GA, Stampfer MJ, Ascherio A, Chute CC, Willett WC. 1993. A prospective study of dietary fat and risk of prostate cancer. J Natl Cancer Inst 85:1571â1579. Giovannucci E, Rimm EB, Stampfer MJ, Colditz GA, Ascherio A, Willett WC. 1994. Intake of fat, meat, and fiber in relation to risk of colon cancer in men. Cancer Res 54:2390â2397. GISSI-Prevenzione Investigators. 1999. Dietary supplementation with n - 3 poly- unsaturated fatty acids and vitamin E after myocardial infarction: Results of the GISSI-Prevenzione trial. Lancet 354:447â455. Glanz K, Basil M, Maibach E, Goldberg J, Snyder D. 1998. Why Americans eat what they do: Taste, nutrition, cost, convenience, and weight control concerns as influences on food consumption. J Am Diet Assoc 98:1118â1126. Glueck CJ, Hastings MM, Allen C, Hogg E, Baehler L, Gartside PS, Phillips D, Jones M, Hollenbach EJ, Braun B, Anastasia JV. 1982. Sucrose polyester and covert caloric dilution. Am J Clin Nutr 35:1352â1359. Goodman MT, Kolonel LN, Yoshizawa CN, Hankin JH. 1988. The effect of dietary cholesterol and fat on the risk of lung cancer in Hawaii. Am J Epidemiol 128:1241â1255. Göranzon H, Forsum E, Thilén M. 1983. Calculation and determination of metabolizable energy in mixed diets to humans. Am J Clin Nutr 38:954â963. Gortmaker SL, Dietz WH, Sobol AM, Wehler CA. 1987. Increasing pediatric obesity in the United States. Am J Dis Child 141:535â540. Grammatikos SI, Subbaiah PV, Victor TA, Miller WM. 1994. n-3 and n-6 Fatty acid processing and growth effects in neoplastic and non-cancerous human mam- mary epithelial cell lines. Br J Cancer 70:219â227. Green SM, Burley VJ, Blundell JE. 1994. Effect of fat- and sucrose-containing foods on the size of eating episodes and energy intake in lean males: Potential for causing overconsumption. Eur J Clin Nutr 48:547â555. Grill V, Björklund A. 2001. Overstimulation and beta-cell function. D iabetes 50:S122âS124. Grimsgaard S, Bønaa KH, Hansen J-B, Nordøy A. 1997. Highly purified eicosapentaenoic acid and docosahexaenoic acid in humans have similar triacylglycerol-lowering effects but divergent effects on serum fatty acids. Am J Clin Nutr 66:649â659. Grundy SM. 1986. Comparison of monounsaturated fatty acids and carbohydrates for lowering plasma cholesterol. N Engl J Med 314:745â748. Grundy SM, Florentin L, Nix D, Whelan MF. 1988. Comparison of monounsaturated fatty acids and carbohydrates for reducing raised levels of plasma cholesterol in man. Am J Clin Nutr 47:965â969. Guallar E, Aro A, Jiménez FJ, MartÃn-Moreno JM, Salminen I, vanât Veer P, Kardinaal AFM, Gómez-Aracena J, Martin BC, Kohlmeier L, Kark JD, Mazaev VP, Ringstad J, Guillén J, Riemersma RA, Huttunen JK, Thamm M, Kok FJ. 1999. Omega-3 fatty acids in adipose tissue and risk of myocardial infarction. The EURAMIC Study. Arterioscler Thromb Vasc Biol 19:1111â1118. Guenther PM. 1986. Beverages in the diets of American teenagers. J Am Diet Assoc 86:493â499. Guthrie JF. 1996. Dietary patterns and personal characteristics of women consum- ing recommended amounts of calcium. Fam Econ Nutr Rev 9:33â49. Guthrie JF, Derby B. 1998. Changes in consumersâ knowledge of food guide rec- ommendations, 1990â91 versus 1994â95. Fam Econ Nutr Rev 11:42â48.
856 DIETARY REFERENCE INTAKES Ha YL, Storkson J, Pariza MW. 1990. Inhibition of benzo(a)pyrene-induced mouse forestomach neoplasia by conjugated dienoic derivatives of linoleic acid. Cancer Res 50:1097â1101. Haglund O, Wallin R, Luostarinen R, Saldeen T. 1990. Effects of a new fluid fish oil concentrate, ESKIMO-3, on triglycerides, cholesterol, fibrinogen and blood pressure. J Intern Med 227:347â353. Halliwell B, Chirico S. 1993. Lipid peroxidation: Its mechanism, measurement, and significance. Am J Clin Nutr 57:715Sâ725S. Hansen D, Michaelsen KF, Skovby F. 1992. Growth during treatment of familial hypercholesterolemia. Acta Paediatr 81:1023â1025. Harker LA, Kelly AB, Hanson SR, Krupski W, Bass A, Osterud B, Fitzgerald GA, Goodnight SH, Connor WE. 1993. Interruption of vascular thrombus forma- tion and vascular lesion formation by dietary n-3 fatty acids in fish oil in non- human primates. Circulation 87:1017â1029. Harnack L, Stang J, Story M. 1999. Soft drink consumption among US children and adolescents: Nutritional consequences. J Am Diet Assoc 99:436â441. Harnack LJ, Jeffery RW, Boutelle KN. 2000. Temporal trends in energy intake in the United States: An ecologic perspective. Am J Clin Nutr 71:1478â1484. Harris WS. 1989. Fish oils and plasma lipid and lipoprotein metabolism in humans: A critical review. J Lipid Res 30:785â807. Harris WS. 1997. n-3 Fatty acids and serum lipoproteins: Human studies. Am J Clin Nutr 65:1645Sâ1654S. Health Canada. 1997. Canadaâs Food Guide to Healthy Eating. Ottawa: Minister of Public Works and Government Services Canada. Heaney RP. 1993. Protein intake and the calcium economy. J A m Diet Assoc 93:1259â1260. Heaney RP. 1998. Excess dietary protein may not adversely affect bone. J Nutr 128:1054â1057. Hegsted DM, Ausman LM, Johnson JA, Dallal GE. 1993. Dietary fat and serum lipids: An evaluation of the experimental data. Am J Clin Nutr 57:875â883. Heitmann BL, Lissner L, Sørensen TIA, Bengtsson C. 1995. Dietary fat intake and weight gain in women genetically predisposed for obesity. Am J Clin Nutr 61:1213â1217. Hiatt RA, Ettinger B, Caan B, Quesenberry CP, Duncan D, Citron JT. 1996. Random- ized controlled trial of a low animal protein, high fiber diet in the prevention of recurrent calcium oxalate kidney stones. Am J Epidemiol 144:25â33. Hill AJ, Blundell JE. 1990. Sensitivity of the appetite control system in obese sub- jects to nutritional and serotoninergic challenges. Int J Obes 14:219â233. Hill AJ, Leathwood PD, Blundell JE. 1987. Some evidence for short-term caloric compensation in normal weight human subjects: The effects of high- and low- energy meals on hunger, food preference and food intake. Hum Nutr Appl Nutr 41:244â257. Hill JO, Peters JC, Reed GW, Schlundt DG, Sharp T, Greene HL. 1991. Nutrient balance in humans: Effects of diet composition. Am J Clin Nutr 54:10â17. Hill JO, Melanson EL, Wyatt HT. 2000. Dietary fat intake and regulation of energy balance: Implications for obesity. J Nutr 130:284Sâ288S. Himaya A, Fantino M, Antoine JM, Bronel L, Louis-Sylvestre J. 1997. Satiety power of dietary fat: A new appraisal. Am J Clin Nutr 65:1410â1418. Hislop TG, Coldman AJ, Elwood JM, Brauer G, Kan L. 1986. Childhood and recent eating patterns and risk of breast cancer. Cancer Detect Prev 9:47â58.
857 M ACRONUTRIENTS AND HEALTHFUL DIETS Holman RL, McGill HC, Strong JP, Greer JC. 1958. The natural history of athero- sclerosis. The early aortic lesions as seen in New Orleans in the middle of the 20th century. Am J Pathol 34:209â235. Holmes MD, Hunter DJ, Colditz GA, Stampfer MJ, Hankinson SE, Speizer FE, Rosner B, Willett WC. 1999. Association of dietary intake of fat and fatty acids with risk of breast cancer. J Am Med Assoc 281:914â920. Holt SH, Miller JC, Petocz P, Farmakalidid E. 1995. A satiety index of common foods. Eur J Clin Nutr 49:675â690. Höppener JWM, Ahrén B, Lips CJM. 2000. Islet amyloid and type 2 diabetes mellitus. N Engl J Med 343:411â419. Horton TJ, Drougas H, Brachey A, Reed GW, Peters JC, Hill JO. 1995. Fat and carbohydrate overfeeding in humans: Different effects on energy storage. Am J Clin Nutr 62:19â29. Horvath PJ, Eagen CK, Fisher NM, Leddy JJ, Pendergast DR. 2000. The effects of varying dietary fat on performance and metabolism in trained male and female runners. J Am Coll Nutr 19:52â60. Howard BV, Abbott WGH, Swinburn BA. 1991. Evaluation of metabolic effects of substitution of complex carbohydrates for saturated fat in individuals with obesity and NIDDM. Diabetes Care 14:786â795. Howard BV, Hannah JS, Heiser CC, Jablonski KA, Paidi MC, Alarif L, Robbins DC, Howard WJ. 1995. Polyunsaturated fatty acids result in greater cholesterol lowering and less triacylglycerol elevation than do monounsaturated fatty acids in a doseâresponse comparison in a multiracial study group. Am J Clin Nutr 62:392â402. Howe GR, Hirohata T, Hislop TG, Iscovich JM, Yuan J-M, Katsouyanni K, Lubin F, Marubini E, Modan B, Rohan T, Toniolo P, Shunzhang Y. 1990. Dietary factors and risk of breast cancer: Combined analysis of 12 case-control studies. J Natl Cancer Inst 82:561â569. Howe GR, Friedenreich CM, Jain M, Miller AB. 1991. A cohort study of fat intake and risk of breast cancer. J Natl Cancer Inst 83:336â340. Howe GR, Aronson KJ, Benito E, Castelleto R, Cornée J, Duffy S, Gallagher RP, Iscovich JM, Deng-ao J, Kaaks R, Kune GA, Kune S, Lee HP, Lee M, Miller AB, Peters RK, Potter JD, Riboli E, Slattery ML, Trichopoulos D, Tuyns A, Tzonou A, Watson LF, Whittemore AS, Wu-Willimas AH, Shu Z. 1997. The relationship between dietary fat intake and risk of colorectal cancer: Evidence from the combined analysis of 13 case-control studies. Cancer Causes Control 8:215â228. Hu FB, Stampfer MJ, Manson JE, Rimm E, Colditz GA, Rosner BA, Hennekens CH, Willett WC. 1997. Dietary fat intake and the risk of coronary heart disease in women. N Engl J Med 337:1491â1499. Hu FB, Stampfer MJ, Manson JE, Rimm E, Colditz GA, Speizer FE, Hennekens CH, Willett WC. 1999. Dietary protein and risk of ischemic heart disease in women. Am J Clin Nutr 70:221â227. Hu FB, van Dam RM, Liu S. 2001. Diet and risk of type II diabetes: The role of types of fat and carbohydrate. Diabetologia 44:805â817. Hulshof T, De Graaf C, Weststrate JA. 1993. The effects of preloads varying in physical state and fat content on satiety and energy intake. Appetite 21:273â286. Hun CS, Hasegawa K, Kawabata T, Kato M, Shimokawa T, Kagawa Y. 1999. Increased uncoupling protein2 mRNA in white adipose tissue, and decrease in leptin, visceral fat, blood glucose, and cholesterol in KK-Ay mice fed with eicosapentaenoic and docosahexaenoic acids in addition to linolenic acid. Biochem Biophys Res Comm 259:85â90.
858 DIETARY REFERENCE INTAKES Hunter DJ, Spiegelman D, Adami H-O, Beeson L, van den Brandt PA, Folsom AR, Fraser GE, Goldbohn A, Graham S, Howe GR, Kushi LH, Marshall JR, McDermott A, Miller AB, Speizer FE, Wolk A, Yaun S-S, Willett W. 1996. Cohort studies of fat intake and the risk of breast cancerâA pooled analysis. N Engl J Med 334:356â361. Hursting SD, Thornquist M, Henderson MM. 1990. Types of dietary fat and the incidence of cancer at five sites. Prev Med 19:242â253. Ip C, Scimeca JA. 1997. Conjugated linoleic acid and linoleic acid are distinctive modulators of mammary carcinogenesis. Nutr Cancer 27:131â135. Ip C, Chin SF, Scimeca JA, Pariza MW. 1991. Mammary cancer prevention by conjugated dienoic derivative of linoleic acid. Cancer Res 51:6118â6124. Ip C, Ip MM, Loftus T, Shoemaker S, Shea-Eaton W. 2000. Induction of apoptosis by conjugated linoleic acid in cultured mammary tumor cells and premalignant lesions of the rat mammary gland. Cancer Epidemiol Biomarkers Prev 9:689â696. Ip MM, Masso-Welch PA, Shoemaker SF, Shea-Eaton WK, Ip C. 1999. Conjugated linoleic acid inhibits proliferation and induces apoptosis of normal rat mam- mary epithelial cells in primary culture. Exp Cell Res 250:22â34. Iso H, Rexrode KM, Stampfer MJ, Manson JE, Colditz GA, Speizer FE, Hennekens CH, Willett WC. 2001. Intake of fish and omega-3 fatty acids and risk of stroke in women. J Am Med Assoc 285:304â312. James MJ, Gibson RA, Cleland LG. 2000. Dietary polyunsaturated fatty acids and inflammatory mediator production. Am J Clin Nutr 71:343Sâ348S. Janelle KC, Barr SI. 1995. Nutrient intakes and eating behavior scores of vegetarian and nonvegetarian women. J Am Diet Assoc 95:180â196. Jansen S, Lopez-Miranda J, Salas J, Castro P, Paniagua JA, Lopez-Segura F, Ordovas JM, Jimenez-Pereperez JA, Blanco A, Perez-Jimenez F. 1998. Plasma lipid response to hypolipidemic diets in young healthy non-obese men varies with body mass index. J Nutr 128:1144â1149. Jayarajan P, Reddy V, Mohanram M. 1980. Effect of dietary fat on absorption of β carotene from green leafy vegetables in children. Indian J Med Res 71:53â56. Jeppesen J, Schaaf P, Jones C, Zhou M-Y, Chen Y-DI, Reaven GM. 1997. Effects of low-fat, high-carbohydrate diets on risk factors for ischemic heart disease in postmenopausal women. Am J Clin Nutr 65:1027â1033. Jéquier E. 1999. Response to and range of acceptable fat intake in adults. Eur J Clin Nutr 53:S84âS93. Jessup W, Kritharides L. 2000. Metabolism of oxidized LDL by macrophages. Curr Opin Lipidol 11:473â481. Johnson MM, Swan DD, Surette ME, Stegner J, Chilton T, Fonteh AN, Chilton FH. 1997. Dietary supplementation with γ-linolenic acid alters fatty acid content and eicosanoid production in healthy humans. J Nutr 127:1435â1444. Johnson RK, Panely C, Wang MQ. 1998. The association between noon beverage consumption and the diet quality of school-age children. J Child Nutr Manage 22:95â100. Jones AE, Murphy JL, Stolinski M, Wootton SA. 1998. The effect of age and gender on the metabolic disposal of [1-13C]palmitic acid. Eur J Clin Nutr 52:22â28. Jones DY, Schatzkin A, Green SB, Block G, Brinton LA, Ziegler RG, Hoover R, Taylor PR. 1987. Dietary fat and breast cancer in the National Health and Nutrition Examination Survey. I. Epidemiologic follow-up study. J Natl Cancer Inst 79:465â471.
859 M ACRONUTRIENTS AND HEALTHFUL DIETS Jonnalagadda SS, Egan SK, Heimbach JT, Harris SS, Kris-Etherton PM. 1995. Fatty acid consumption pattern of Americans: 1987â1988 USDA Nationwide Food Consumption Survey. Nutr Res 15:1767â1781. Kahn SR, Solymoss S, Flegel KM. 1997. Nonvalvular atrial fibrillation: Evidence for a prothrombic state. Can Med Assoc J 157:673â681. Kaizer L, Boyd NF, Kriukov V, Tritchler D. 1989. Fish consumption and breast cancer risk: An ecologic study. Nutr Cancer 12:61â68. Kang JX, Leaf A. 1996. Antiarrhythmic effects of polyunsaturated fatty acids: Recent studies. Circulation 94:1774â1780. Kannel WB. 2000. The Framingham Study: Its 50-year legacy and future promise. J Atheroscler Thromb 6:60â66. Karmali RA. 1986. Eicosanoids and cancer. Prog Clin Biol Res 222:687â697. Karmali RA, Marsh J, Fuchs C. 1984. Effect of omega-3 fatty acids on growth of a rat mammary tumor. J Natl Cancer Inst 73:457â461. Karmali RA, Reichel P, Cohen LA, Terano T, Hirai A, Tamura Y, Yoshida S. 1987. The effects of dietary omega-3 fatty acids on the DU-145 transplantable human prostatic tumor. Anticancer Res 7:1173â1179. Kasim SE, Martino S, Kim P-N, Khilnani S, Boomer A, Depper J, Reading BA, Heilbrun LK. 1993. Dietary and anthropometric determinants of plasma lipo- proteins during a long-term low-fat diet in healthy women. Am J Clin Nutr 57:146â153. Kasim-Karakas SE, Lane E, Almario R, Mueller W, Walzem R. 1997. Effects of dietary fat restriction on particle size of plasma lipoproteins in postmenopausal women. Metabolism 46:431â436. Kasim-Karakas SE, Almario RU, Mueller WM, Peerson J. 2000. Changes in plasma lipoproteins during low-fat, high-carbohydrate diets: Effects of energy intake. Am J Clin Nutr 71:1439â1447. Katan MB, Zock PL, Mensink RP. 1994. Effects of fats and fatty acids on blood lipids in humans: An overview. Am J Clin Nutr 60:1017Sâ1022S. Kato I, Akhmedkhanov A, Koenig K, Toniolo PG, Shore RE, Riboli E. 1997. Prospective study of diet and female colorectal cancer: The New York Univer- sity Womenâs Health Study. Nutr Cancer 28:276â281. Kavanaugh CJ, Liu K-L, Belury MA. 1999. Effect of dietary conjugated linoleic acid on phorbol ester-induced PGE2 production and hyperplasia in mouse epidermis. Nutr Cancer 33:132â138. Keli SO, Feskens EJ, Kromhout D. 1994. Fish consumption and risk of stroke. The Zutphen Study. Stroke 25:328â332. Kelleher CC. 1992. Plasma fibrinogen and factor VII as risk factors for cardiovascular disease. Eur J Epidemiol 8:79â82. Kendall A, Levitsky DA, Strupp BJ, Lissner L. 1991. Weight loss on a low-fat diet: Consequence of the imprecision of the control of food intake in humans. Am J Clin Nutr 53:1124â1129. Kerstetter JE, Mitnick ME, Gundberg CM, Caseria DM, Ellison AF, Carpenter TO, Insogna KL. 1999. Changes in bone turnover in young women consuming different levels of dietary protein. J Clin Endocrinol Metab 84:1052â1055. Kestin M, Clifton P, Belling GB, Nestel PJ. 1990. n-3 Fatty acids of marine origin lower systolic blood pressure and triglycerides but raise LDL cholesterol com- pared with n-3 and n-6 fatty acids from plants. Am J Clin Nutr 51:1028â1034. Keusch GT, Torun B, Johnson RB, Urrutia JJ. 1984. Impairment of hemolytic complement activation by both classical and alternative pathways in serum from patients with kwashiorkor. J Pediatr 105:434â436.
860 DIETARY REFERENCE INTAKES Keys A, Aravanis C, Blackburn H, Buzina R, Djordevic BS, Dontas AS, Fidanza F, ´ Karvonen MJ, Kimura N, Menotti A, Mohacek I, Nedeljkovic S, Puddu V, ´ s Punsar S, Taylor HL, van Buchem FSP. 1980. Seven Countries. A Multivariate Analysis of Death and Coronary Heart Disease. Cambridge, MA: Harvard University Press. Keys A, Menotti A, Aravanis C, Blackburn H, Djordevic BS, Buzinz R, Dontas AS, ´ Fidanza F, Karvonen MJ, Kimura N, Mohacek I, Nedeljkovic S, Puddu V, ´ s Punsar S, Taylor HL, Conti S, Kromhout D, Toshima H. 1984. The Seven Countries Study: 2,289 deaths in 15 years. Prev Med 13:141â154. Keys A, Menotti A, Karvonen MJ, Aravanis C, Blackburn H, Buzina R, Djordjevic ´ BS, Dontas AS, Fidanza F, Keys MH. 1986. The diet and 15-year death rate in the Seven Countries Study. Am J Epidemiol 124:903â915. King JC. 2000. Physiology of pregnancy and nutrient metabolism. Am J Clin Nutr 71:1218Sâ1225S. Klesges RC, Klesges LM, Haddock CK, Eck LH. 1992. A longitudinal analysis of the impact of dietary intake and physical activity on weight change in adults. Am J Clin Nutr 55:818â822. Knopp RH, Walden CE, Retzlaff BM, McCann BS, Dowdy AA, Albers JJ, Gey GO, Cooper MN. 1997. Long-term cholesterol-lowering effects of 4 fat-restricted diets in hypercholesterolemic and combined hyperlipidemic men. J Am Med Assoc 278:1509â1515. Knuiman JT, Westenbrink S, van der Heyden L, West CE, Burema J, De Boer J, Hautvast JGAJ, Räsänen L, Virkkunen L, Viikari J, Lokko P, Pobee JOM, Ferro- Luzzi A, Ferrini AM, Scaccini C, Sette S, Villavieja GM, Bulatao-Jayme J. 1983. Determinants of total and high density lipoprotein cholesterol in boys from Finland, the Netherlands, Italy, the Philippines and Ghana with special refer- ence to diet. Hum Nutr Clin Nutr 37:237â254. Knuiman JT, West CE, Katan MB, Hautvast JGAJ. 1987. Total cholesterol and high density lipoprotein cholesterol levels in populations differing in fat and carbo- hydrate intake. Arteriosclerosis 7:612â619. Krauss RM. 2001. Atherogenic lipoprotein phenotype and diet-gene interactions. J Nutr 131:340Sâ343S. Krauss RM, Dreon DM. 1995. Low-density-lipoprotein subclasses and response to a low-fat diet in healthy men. Am J Clin Nutr 62:478Sâ487S. Kris-Etherton PM (for the DELTA Investigators). 1996. Effects of replacing satu- rated fat (SFA) with monounsaturated fat (MUFA) or carbohydrate (CHO) on plasma lipids and lipoproteins in individuals with markers for insulin resis- tance. FASEB J 10:2666. Kris-Etherton PM, Derr J, Mitchell DC, Mustad VA, Russell ME, McDonnell ET, Salabsky D, Pearson TA. 1993. The role of fatty acid saturation on plasma lipids, lipoproteins, and apolipoproteins: I. Effects of whole food diets high in cocoa butter, olive oil, soybean oil, dairy butter, and milk chocolate on the plasma lipids of young men. Metabolism 42:121â129. Kris-Etherton PM, Pearson TA, Wan Y, Hargrove RL, Moriarty K, Fishell V, Etherton TD. 1999. High-monounsaturated fatty acid diets lower both plasma choles- terol and triacylglycerol concentrations. Am J Clin Nutr 70:1009â1015. Kris-Etherton PM, Zhao G, Pelkman CL, Fishell VK, Coval SM. 2000. Beneficial effects of a diet high in monounsaturated fatty acids on risk factors for cardio- vascular disease. Nutr Clin Care 3:153â162.
861 M ACRONUTRIENTS AND HEALTHFUL DIETS Kritchevsky D, Tepper SA, Czarnecki SK, Klurfeld DM, Story JA. 1981. Experimental atherosclerosis in rabbits fed cholesterol-free diets. Part 9. Beef protein and textured vegetable protein. Atherosclerosis 39:169â175. Kromhout D, de Lezenne Coulander C. 1984. Diet, prevalence and 10-year mortality from coronary heart disease in 871 middle-aged men. Am J Epidemiol 119:733â741. Kromhout D, Bosschieter EB, de Lezenne Coulander C. 1985. The inverse relation between fish consumption and 20-year mortality from coronary heart disease. N Engl J Med 312:1205â1209. Kromhout D, Feskens EJM, Bowles CH. 1995. The protective effect of a small amount of fish on coronary heart disease mortality in an elderly population. Int J Epidemiol 24:340â345. Kromhout D, Bloemberg BPM, Feskens EJM, Hertog MGL, Menotti A, Blackburn H. 1996. Alcohol, fish, fibre and antioxidant vitamins intake do not explain population differences in coronary heart disease mortality. Int J Epidemiol 25:753â759. Kushi LH, Lew RA, Stare FJ, Ellison CR, el Lozy M, Bourke G, Daly L, Graham I, Hickey N, Mulcahy R, Kevaney J. 1985. Diet and 20-year mortality from coronary heart disease. The IrelandâBoston DietâHeart Study. N Engl J Med 312:811â888. Kushi LH, Sellers TA, Potter JD, Nelson CL, Munger RG, Kaye SA, Folsom AR. 1992. Dietary fat and postmenopausal breast cancer. J Natl Cancer Inst 84:1092â1099. Kwiterovich PO, Barton BA, McMahon RP, Obarzanek E, Hunsberger S, Simons- Morton D, Kimm SYS, Friedman LA, Lasser N, Robson A, Lauer R, Stevens V, Van Horn L, Gidding S, Snetselaar L, Hartmuller VW, Greenlick M, Franklin F. 1997. Effects of diet and sexual maturation on low-density lipoprotein cholesterol during puberty. The Dietary Intervention Study in Children (DISC). Circulation 96:2526â2533. Lagström H, Jokinen E, Seppänen R, Rönnemaa T, Viikari J, Välimäki I, Venetoklis J, Myyrinmaa A, Niinikoski H, Lapinleimu H, Simell O. 1997. Nutrient intakes by young children in a prospective randomized trial of a low-saturated fat, low- cholesterol diet. The STRIP Baby Project. Arch Pediatr Adolesc Med 151:181â188. Lagström H, Seppänen R, Jokinen E, Niinikoski H, Rönnemaa T, Viikari J, Simell O. 1999. Influence of dietary fat on the nutrient intake and growth of children from 1 to 5 y of age: The Special Turku Coronary Risk Factor Intervention Project. Am J Clin Nutr 69:516â523. Lai PBS, Ross JA, Fearson KCH, Anderson JD, Carter DC. 1996. Cell cycle arrest and induction of apoptosis in pancreatic cancer cells exposed to eicosapentaenoic acid in vitro. Br J Cancer 74:1375â1383. Lapinleimu H, Viikari J, Jokinen E, Salo P, Routi T, Leino A, Rönnemaa R, Seppänen R, Välimäki I, Simell O. 1995. Prospective randomised trial in 1062 infants of diet low in saturated fat and cholesterol. Lancet 345:471â476. Larsen LF, Bladbjerg E-M, Jespersen J, Marckmann P. 1997. Effects of dietary fat quality and quantity on postprandial activation of blood coagulation factor VII. Arterioscler Thromb Vasc Biol 17:2904â2909. Larson DE, Hunter GR, Williams MJ, Kekes-Szabo T, Nyikos I, Goran MI. 1996. Dietary fat in relation to body fat and intraabdominal adipose tissue: A cross- sectional analysis. Am J Clin Nutr 64:677â684. Larsson H, ElmstÃ¥hl S, Berglund G, Ahrén B. 1999. Habitual dietary intake versus glucose tolerance, insulin sensitivity and insulin secretion in postmenopausal women. J Intern Med 245:581â591.
862 DIETARY REFERENCE INTAKES Lauer RM, Obarzanek E, Hunsberger SA, Van Horn L, Hartmuller VW, Barton BA, Stevens VJ, Kwiterovich PO, Franklin FA, Kimm SYS, Lasser NL, Simons- Morton DG. 2000. Efficacy and safety of lowering dietary intake of total fat, saturated fat, and cholesterol in children with elevated LDL cholesterol: The Dietary Intervention Study in Children. Am J Clin Nutr 72:1332Sâ1342S. La Vecchia C, Negri E, Franceschi S, Decarli A, Giacosa A, Lipworth L. 1995. Olive oil, other dietary fats, and the risk of breast cancer (Italy). Cancer Causes Control 6:545â550. Lawton CL, Burley VJ, Wales JK, Blundell JE. 1993. Dietary fat and appetite control in obese subjects: Weak effects on satiation and satiety. Int J Obes Relat Metab Disord 17:409â416. Leclerc I, Davignon I, Lopez D, Garrel DR. 1993. No change in glucose tolerance and substrate oxidation after a high-carbohydrate, low-fat diet. Metabolism 42:365â370. Lee KN, Kritchevsky D, Pariza MW. 1994. Conjugated linoleic acid and athero- sclerosis in rabbits. Atherosclerosis 108:19â25. Lee KN, Pariza MW, Ntambi JM. 1998. Conjugated linoleic acid decreases hepatic stearoyl-CoA desaturase mRNA expression. B iochem Biophys Res Comm 2 48:817â821. Lee-Han H, Cousins M, Beaton M, McGuire V, Kriukov V, Chipman M, Boyd N. 1988. Compliance in a randomized clinical trial of dietary fat reduction in patients with breast dysplasia. Am J Clin Nutr 48:575â586. Lei YX, Cai WC, Chen YZ, Du YX. 1996. Some lifestyle factors in human lung cancer: A case control study of 792 lung cancer cases. Lung Cancer 14:S121âS136. Leibel RL, Hirsch J, Appel BE, Checani GC. 1992. Energy intake required to main- tain body weight is not affected by wide variation in diet composition. Am J Clin Nutr 55:350â355. Lemann J. 1999. Relationship between urinary calcium and net acid excretion as determined by dietary protein and potassium: A review. Nephron 81:18â25. Leventhal LJ, Boyce EG, Zurier RB. 1993. Treatment of rheumatoid arthritis with gammalinolenic acid. Ann Intern Med 119:867â873. Leventhal LJ, Boyce EG, Zurier RB. 1994. Treatment of rheumatoid arthritis with blackcurrant seed oil. Br J Rheumatol 33:847â852. Levrat M-A, Behr SR, Rémésy C, Demigné C. 1991a. Effects of soybean fiber on cecal digestion in rats previously adapted to a fiber-free diet. J Nutr 121:672â678. Levrat M-A, Rémésy C, Demigné C. 1991b. High propionic acid fermentations and mineral accumulation in the cecum of rats adapted to different levels of inulin. J Nutr 121:1730â1737. Lew SQ, Bosch JP. 1991 Effect of diet on creatinine clearance and excretion in young and elderly healthy subjects and in patients with renal disease. J Am Soc Nephrol 2:856â865. Lewis CL, Park YK, Dexter PB, Yetley EA. 1992. Nutrient intakes and body weights of persons consuming high and moderate levels of added sugars. J Am Diet Assoc 92:708â713. Liew C, Schut HAJ, Chin SF, Pariza MW, Dashwood RH. 1995. Protection of conju- gated linoleic acids against 2-amino-3-methylimidazo[4,5-f]quinoline-induced colon carcinogenesis in the F344 rat: A study of inhibitory mechanisms. Carcinogenesis 16:3037â3043. Lifshitz F, Moses N. 1989. Growth failure. A complication of dietary treatment of hypercholesterolemia. Am J Dis Child 143:537â542.
863 M ACRONUTRIENTS AND HEALTHFUL DIETS Lissner L, Heitmann BL. 1995. Dietary fat and obesity: Evidence from epidemiology. Eur J Clin Nutr 49:79â90. Lissner L, Levitsky DA, Strupp BJ, Kalkwarf HJ, Roe DA. 1987. Dietary fat and the regulation of energy intake in human subjects. Am J Clin Nutr 46:886â892. Lissner L, Heitmann BL, Bengtsson C. 2000. Population studies of diet and obesity. Br J Nutr 83:S21âS24. Litin L, Sacks F. 1993. Trans-fatty-acid content of common foods. N Engl J Med 329:1969â1970. Liu GC, Coulston AM, Reaven GM. 1983. Effect of high-carbohydrate-low-fat diets on plasma glucose, insulin and lipid responses in hypertriglyceridemic humans. Metabolism 32:750â753. Liu K, Stamler J, Trevisan M, Moss D. 1982. Dietary lipids, sugar, fiber, and mortality from coronary heart disease. Bivariate analysis of international data. Arterio- sclerosis 2:221â227. Liu S, Willett WC, Stampfer MJ, Hu FB, Franz M, Sampson L, Hennekens CH, Manson JE. 2000. A prospective study of dietary glycemic load, carbohydrate intake, and risk of coronary heart disease in US women. A m J Clin Nutr 71:1455â1461. Lopes-Virella MF, Virella G. 1996. Modified lipoproteins, cytokines and macrovascular disease in non-insulin-dependent diabetes mellitus. Ann Med 28:347â354. Lopez-Segura F, Velasco F, Lopez-Miranda J, Castro P, Lopez-Pedrera R, Blanco A, Jimenez-Pereperez J, Torres A, Trujillo J, Ordovas JM, Perez-Jiminez F. 1996. Monounsaturated fatty acid-enriched diet decreases plasma plasminogen acti- vator inhibitor type 1. Aterioscler Thromb Vasc Biol 16:82â88. Louheranta AM, Porkkala-Sarataho EK, Nyyssonen MK, Salonen RM, Salonen JT. 1996. Linoleic acid intake and susceptibility of very-low-density and low density lipoproteins to oxidation in men. Am J Clin Nutr 63:698â703. Louis-Sylvestre J, Tournier A, Chapelot D, Chabert M. 1994. Effect of a fat-reduced dish in a meal on 24-h energy and macronutrient intake. Appetite 22:165â172. Lovejoy JC. 1999. Dietary fatty acids and insulin resistance. Curr Atheroscler Rep 1:215â220. Lovejoy J, DiGirolamo M. 1992. Habitual dietary intake and insulin sensitivity in lean and obese adults. Am J Clin Nutr 55:1174â1179. Lovell CR, Burton JL, Horrobin DF. 1981. Treatment of atopic eczema with evening primrose oil. Lancet 1:278. Lubin F, Wax Y, Modan B. 1986. Role of fat, animal protein, and dietary fiber in breast cancer etiology: A case-control study. J Natl Cancer Inst 77:605â612. Lubin JH, Burns PE, Blot WJ, Ziegler RG, Lees AW, Fraumeni JF. 1981. Dietary factors and breast cancer risk. Int J Cancer 28:685â689. Ludwig DS, Majzoub JA, Al-Zahrani A, Dallal GE, Blanco I, Roberts SB. 1999a. High glycemic index foods, overeating, and obesity. Pediatrics 103:E26. Ludwig DS, Pereira MA, Kroenke CH, Hilner JE, Van Horn L, Slattery ML, Jacobs DR. 1999b. Dietary fiber, weight gain, and cardiovascular disease risk factors in young adults. J Am Med Assoc 282:1539â1546. Luhman CM, Beitz DC. 1993. Dietary protein and blood cholesterol homeostasis. In: Liepa GU, Beitz DC, Beynen AC, Gorman MA, eds. Dietary Proteins: How They Alleviate Disease and Promote Better Health. Champaign, IL: American Oil Chemistsâ Society. Pp. 57â76.
864 DIETARY REFERENCE INTAKES Lundgren H, Bengtsson C, Blohmé G, Isaksson B, Lapidus L, Lenner RA, Saaek A, Winther E. 1989. Dietary habits and incidence of noninsulin-dependent diabetes mellitus in a population study of women in Gothenburg, Sweden. Am J Clin Nutr 49:708â712. Lungershausen YK, Abbey M, Nestel PJ, Howe PRC. 1994. Reduction of blood pressure and plasma triglycerides by omega-3 fatty acids in treated hypertensives. J Hypertens 12:1041â1045. Luo J, Rizkalla SW, Boillot J, Alamowitch C, Chaib H, Bruzzo F, Desplanque N, Dalix A-M, Durand G, Slama G. 1996. Dietary (n-3) polyunsaturated fatty acids improve adipocyte insulin action and glucose metabolism in insulin-resistant rats: Relation to membrane fatty acids. J Nutr 126:1951â1958. Luo J, Rizkalla SW, Vidal H, Oppert J-M, Colas C, Boussari A, Guerre-Millo M, Chapuis A-S, Chevalier A, Durand G, Slama G. 1998. Moderate intake of n-3 fatty acids for 2 months has no detrimental effect on glucose metabolism and could ameliorate the lipid profile in type 2 diabetic men: Results of a con- trolled study. Diabetes Care 21:717â724. Maffeis C, Pinelli L, Schutz Y. 1996. Fat intake and adiposity in 8 to 11-year-old obese children. Int J Obes Relat Metab Disord 20:170â174. Maillard G, Charles MA, Lafay L, Thibult N, Vray M, Borys J-M, Basdevant A, Eschwège E, Romon M. 2000. Macronutrient energy intake and adiposity in non obese prepubertal children aged 5â11 y (the Fleurbaix Laventie Ville Santé Study). Int J Obes Relat Metab Disord 24:1608â1617. Männistö S, Pietinen P, Virtanen M, Kataja V, Uusitupa M. 1999. Diet and the risk of breast cancer in a case-control study: Does the threat of disease have an influence on recall bias? J Clin Epidemiol 52:429â439. Marckmann P, Grønbaek M. 1999. Fish consumption and coronary heart disease mortality. A systematic review of prospective cohort studies. Eur J Clin Nutr 53:585â590. Marckmann P, Raben A, Astrup A. 2000. Ad libitum intake of low-fat diets rich in either starchy foods or sucrose: Effects on blood lipids, factor VII coagulant activity, and fibrinogen. Metabolism 49:731â735. Marshall JA, Hamman RF, Baxter J. 1991. High-fat, low-carbohydrate diet and the etiology of non-insulin-dependent diabetes mellitus: The San Luis Valley Diabetes Study. Am J Epidemiol 134:590â603. Marshall JA, Bessesen DH, Hamman RF. 1997. High saturated fat and low starch and fibre are associated with hyperinsulinemia in a non-diabetic population: The San Luis Valley Diabetes Study. Diabetologia 40:430â438. Martin-Moreno JM, Willett WC, Gorgojo L, Banegas JR, Rodriguez-Artalejo F, Fernandez-Rodriguez JC, Maisonneuve P, Boyle P. 1994. Dietary fat, olive oil intake and breast cancer risk. Int J Cancer 58:774â780. Masironi R. 1970. Dietary factors and coronary heart disease. Bull World Health Organ 42:103â114. Mattes R. 1990. Effects of aspartame and sucrose on hunger and energy intake in humans. Physiol Behav 47:1037â1044. Mattson FH, Grundy SM. 1985. Comparison of effects of dietary saturated, monounsaturated, and polyunsaturated fatty acids on plasma lipids and lipo- proteins in man. J Lipid Res 26:194â202. Mayer EJ, Newman B, Quesenberry CP, Selby JV. 1993. Usual dietary fat intake and insulin concentrations in healthy women twins. Diabetes Care 16:1459â1469.
865 M ACRONUTRIENTS AND HEALTHFUL DIETS Mayer-Davis EJ, Monaco JH, Hoen HM, Carmichael S, Vitolins MZ, Rewers MJ, Haffner SM, Ayad MF, Bergman RN, Karter AJ. 1997. Dietary fat and insulin sensitivity in a triethnic population: The role of obesity. The Insulin Resis- tance Atheroscelrosis Study (IRAS). Am J Clin Nutr 65:79â87. McDevitt RM, Poppitt SD, Murgatroyd PR, Prentice AM. 2000. Macronutrient dis- posal during controlled overfeeding with glucose, fructose, sucrose, or fat in lean and obese women. Am J Clin Nutr 72:369â377. McDonald BE, Gerrard JM, Bruce VM, Corner EJ. 1989. Comparison of the effect of canola oil and sunflower oil on plasma lipids and lipoproteins and on in vivo thromboxane A2 and prostacyclin production in healthy young men. Am J Clin Nutr 50:1382â1388. McDowell MA, Briefel RR, Alaimo K, Bischof AM, Caughman CR, Carroll MD, Loria CM, Johnson CL. 1994. Energy and macronutrient intakes of persons ages 2 months and over in the United States: Third National Health and Nutrition Examination Survey, Phase 1, 1988â91. Adv Data 255:1â24. McGee DL, Reed DM, Yano K, Kagan A, Tillotson J. 1984. Ten-year incidence of coronary heart disease in the Honolulu Heart Program. Relationship to nutri- ent intake. Am J Epidemiol 119:667â676. McGill HC. 1968. Fatty streaks in the coronary arteries and aorta. L ab Invest 18:100â104. McGill HC, McMahan CA, Zieske AW, Sloop GD, Walcott JV, Troxclair DA, Malcom GT, Tracy RE, Oalmann MC, Strong JP. 2000a. Associations of coronary heart disease risk factors with the intermediate lesion of atherosclerosis in youth. Arterioscler Thromb Vasc Biol 20:1998â2004. McGill HC, McMahan CA, Zieske AW, Tracy RE, Malcom GT, Herderick EE, Strong JP. 2000b. Association of coronary heart disease risk factors with microscopic qualities of coronary atherosclerosis in youth. Circulation 102:374â379. McLennan PL. 1993. Relative effects of dietary saturated, monounsaturated, and polyunsaturated fatty acids on cardiac arrhythmias in rats. Am J Clin Nutr 57:207â212. Mensink RP, Katan MB. 1987. Effect of monounsaturated fatty acids versus com- plex carbohydrates on high-density lipoproteins in healthy men and women. Lancet 1:122â125. Mensink RP, Katan MB. 1992. Effect of dietary fatty acids on serum lipids and lipoproteins. A meta-analysis of 27 trials. Arterioscler Thromb 12:911â919. Mensink RP, Zock PL, Katan MB, Hornstra G. 1992. Effect of dietary cis and trans fatty acids on serum lipoprotein[a] levels in humans. J Lipid Res 33:1493â1501. Meyer KA, Kushi LH, Jacobs DR, Slavin J, Sellers TA, Folsom AR. 2000. Carbo- hydrates, dietary fiber, and incident of type 2 diabetes in older women. Am J Clin Nutr 71:921â930. Miles CW. 1992. The metabolizable energy of diets differing in dietary fat and fiber measured in humans. J Nutr 122:306â311. Miller AB, Kelly A, Choi NW, Matthews V, Morgan RW, Munan L, Burch JD, Feather J, Howe GR, Jain M. 1978. A study of diet and breast cancer. Am J Epidemiol 107:499â509. Miller GJ, Cruickshank JK, Ellis LJ, Thompson RL, Wilkes HC, Stirling Y, Mitropoulos KA, Allison JV, Fox TE, Walker AO. 1989. Fat consumption and factor VII coagulant activity in middle-aged men. An association between a dietary and thrombogenic coronary risk factor. Atherosclerosis 78:19â24.
866 DIETARY REFERENCE INTAKES Miller WC, Lindeman AK, Wallace J, Niederpruem M. 1990. Diet composition, energy intake, and exercise in relation to body fat in men and women. Am J Clin Nutr 52:426â430. Mooy JM, Grootenhuis PA, de Vries H, Valkenburg HA, Bouter LM, Kostense PJ, Heine RJ. 1995. Prevalence and determinants of glucose intolerance in a Dutch Caucasian population. The Hoorn Study. Diabetes Care 18:1270â1273. Mori TA, Vandongen R, Masarei JRL, Rouse IL, Dunbar D. 1991. Comparison of diets supplemented with fish oil or olive oil on plasma lipoproteins in insulin- dependent diabetics. Metabolism 40:241â246. Mori TA, Beilin LJ, Burke V, Morris J, Ritchie J. 1997. Interactions between dietary fat, fish, and fish oils and their effects on platelet function in men at risk of cardiovascular disease. Arterioscler Thromb Vasc Biol 17:279â286. Morris MC. 1994. Dietary fats and blood pressure. J Cardiovasc Risk 1:21â30. Morris MC, Sacks F, Rosner B. 1993. Does fish oil lower blood pressure? A meta- analysis of controlled trials. Circulation 88:523â533. Morris MC, Manson JE, Rosner B, Buring JE, Willett WC, Hennekens CH. 1995. Fish consumption and cardiovascular disease in the Physiciansâ Health Study: A prospective study. Am J Epidemiol 142:166â175. Morton JF, Guthrie JF. 1998. Changes in childrenâs total fat intakes and their food group sources of fat, 1989â91 versus 1994â95: Implications for diet quality. Fam Econ Nutr Rev 11:44â57. Murphy JL, Jones A, Brookes S, Wootton SA. 1995. The gastrointestinal handling and metabolism of [1-13C]palmitic acid in healthy women. Lipids 30:291â298. Nagata C, Takatsuka N, Kurisu Y, Shimizu H. 1998. Decreased serum total choles- terol concentration is associated with high intake of soy products in Japanese men and women. J Nutr 128:209â213. National Cholesterol Education Program. 2001. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). NIH Publication No. 01-3670. Bethesda, MD: National Institutes of Health. Nelson GJ, Schmidt PC, Kelly DS. 1995. Low-fat diets do not lower plasma choles- terol levels in healthy men compared to high-fat diets with similar fatty acid composition at constant caloric intake. Lipids 30:969â976. Nelson GJ, Schmidt PC, Bartolini GL, Kelley DS, Kyle D. 1997a. The effect of dietary docosahexaenoic acid on plasma lipoproteins and tissue fatty acid com- position in humans. Lipids 32:1137â1146. Nelson GJ, Schmidt PS, Bartolini GL, Kelley DS, Kyle D. 1997b. The effect of dietary docosahexaenoic acid on platelet function, platelet fatty acid composi- tion, and blood coagulation in humans. Lipids 32:1129â1136. Nelson M. 1991. Food, vitamins and IQ. Proc Nutr Soc 50:29â35. Newman TB, Garber AM, Holtzman NA, Hulley SB. 1995. Problems with the report of the Expert Panel on blood cholesterol levels in children and adolescents. Arch Pediatr Adolesc Med 149:241â247. Newmark HL. 1999. Squalene, olive oil, and cancer risk: Review and hypothesis. Ann NY Acad Sci 889:193â203. Nguyen VT, Larson DE, Johnson RK, Goran MI. 1996. Fat intake and adiposity in children of lean and obese parents. Am J Clin Nutr 63:507â513.
867 M ACRONUTRIENTS AND HEALTHFUL DIETS NHLBI/NIDDK (National Heart, Lung, and Blood Institute/National Institute of Diabetes and Digestive and Kidney Diseases). 1998. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults. The Evidence Report. NIH Publication No. 98-4083. Bethesda, MD: National Insti- tutes of Health. Nicklas TA, Webber LS, Koschak M, Berenson GS. 1992. Nutrient adequacy of low fat intakes for children: The Bogalusa Heart Study. Pediatrics 89:221â228. Nicklas TA, Myers L, Farris RP, Srinivasan SR, Berenson GS. 1996. Nutritional quality of a high carbohydrate diet as consumed by children: The Bogalusa Heart Study. J Nutr 126:1382â1388. Nicolosi RJ, Wilson TA. 1997. The anti-atherogenic effect of dietary soybean pro- tein concentrate in hamsters. Nutr Res 17:1457â1467. Niinikoski H, Viikari J, Rönnemaa T, Lapinleimu H, Jokinen E, Salo P, Seppänen R, Leino A, Tuominen J, Välimäki I, Simell O. 1996. Prospective randomized trial of low-saturated-fat, low-cholesterol diet during the first 3 years of life. The STRIP Baby Project. Circulation 94:1386â1393. Niinikoski H, Lapinleimu H, Viikari J, Rönnemaa T, Jokinen E, Seppänen R, Terho P, Tuominen J, Välimäki I, Simell O. 1997a. Growth until 3 years of age in a prospective, randomized trial of a diet with reduced saturated fat and choles- terol. Pediatrics 99:687â694. Niinikoski H, Viikari J, Rönnemaa T, Helenius H, Jokinen E, Lapinleimu H, Routi T, Lagström H, Seppänen R, Välimäki I, Simell O. 1997b. Regulation of growth of 7- to 36-month-old children by energy and fat intake in the prospective, randomized STRIP baby trial. Pediatrics 100:810â816. Nobukata H, Ishikawa T, Obata M, Shibutani Y. 2000. Long-term administration of highly purified eicosapentaenoic acid ethyl ester prevents diabetes and abnor- malities of blood coagulation in male WBN/Kob rats. Metabolism 49:912â919. Noguchi M, Rose DP, Earashi M, Miyazaki I. 1995. The role of fatty acids and eicosanoid synthesis inhibitors in breast carcinoma. Oncology 52:265â271. Norrish AE, Jackson RT, Sharpe SJ, Skeaff CM. 2000. Men who consume vegetable oils rich in monounsaturated fat: Their patterns and risk of prostate cancer (New Zealand). Cancer Causes Control 11:609â615. Obarzanek E, Schreiber GB, Crawford PB, Goldman SR, Barrier PM, Frederick MM, Lakatos E. 1994. Energy intake and physical activity in relation to indexes of body fat: The National Heart, Lung, and Blood Institute Growth and Health Study. Am J Clin Nutr 60:15â22. Obarzanek E, Velletri PA, Cutler JA. 1996. Dietary protein and blood pressure. J Am Med Assoc 275:1598â1603. Obarzanek E, Hunsberger SA, Van Horn L, Hartmuller VV, Barton BA, Stevens VJ, Kwiterovich PO, Franklin FA, Kimm SYS, Lasser NL, Simons-Morton DG, Lauer RM. 1997. Safety of a fat-reduced diet: The Dietary Intervention Study in Chil- dren (DISC). Pediatrics 100:51â59. Obarzanek E, Kimm SYS, Barton BA, Van Horn L, Kwiterovich PO, Simons-Morton DG, Hunsberger SA, Lasser NL, Robson AM, Franklin FA, Lauer RM, Stevens VJ, Friedman LA, Dorgan JF, Greenlick MR. 2001a. Long-term safety and efficacy of a cholesterol-lowering diet in children with elevated low-density lipoprotein cholesterol: Seven-year results of the Dietary Intervention Study in Children (DISC). Pediatrics 107:256â264.
868 DIETARY REFERENCE INTAKES Obarzanek E, Sacks FM, Vollmer WM, Bray GA, Miller ER, Lin P-H, Karanja NM, Most-Windhauser MM, Moore TJ, Swain JF, Bales CW, Proschan MA. 2001b. Effects on blood lipids of a blood pressure-lowering diet: The Dietary Approaches to Stop Hypertension (DASH) Trial. Am J Clin Nutr 74:80â89. Ogden J, Wardle J. 1990. Cognitive restraint and sensitivity to cues for hunger and satiety. Physiol Behav 47:477â481. OâHanesian MA, Rosner B, Bishop LM, Sacks FM. 1996. Effects of inherent respon- siveness to diet and day-to-day diet variation on plasma lipoprotein concentra- tions. Am J Clin Nutr 64:53â59. Ohta A, Ohtsuki M, Baba S, Adachi T, Sakata T, Sakaguchi E. 1995. Calcium and magnesium absorption from the colon and rectum are increased in rats fed fructooligosaccharides. J Nutr 125:2417â2424. Okita M, Yoshida S, Yamamoto J, Suzuki K, Kaneyuki T, Kubota M, Sasagawa T. 1995. n-3 and n-6 Fatty acid intake and serum phospholipid fatty acid composi- tion in middle-aged women living in rural and urban areas in Okayama Prefecture. J Nutr Sci Vitaminol 41:313â323. Olson RE. 2000. Is it wise to restrict fat in the diets of children? J Am Diet Assoc 100:28â32. Oomen CM, Feskens EJM, Räsänen L, Fidanza F, Nissinen AM, Menotti A, Kok FJ, Kromhout D. 2000. Fish consumption and coronary heart disease mortality in Finland, Italy, and the Netherlands. Am J Epidemiol 151:999â1006. Orencia AJ, Daviglus ML, Dyer AR, Shekelle RB, Stamler J. 1996. Fish consumption and stroke in men. 30-Year findings of the Chicago Western Electric Study. Stroke 27:204â209. Ostrowska E, Muralitharan M, Cross RF, Bauman DE, Dunshea FR. 1999. Dietary conjugated linoleic acids increase lean tissue and decrease fat deposition in growing pigs. J Nutr 129:2037â2042. Owen RW, Giacosa A, Hull WE, Haubner R, Spiegelhalder B, Bartsch H. 2000. The antioxidant/anticancer potential of phenolic compounds isolated from olive oil. Eur J Cancer 36:1235â1247. Parillo M, Rivellese AA, Ciardullo AV, Capaldo B, Giacco A, Genovese S, Riccardi G. 1992. A high-monounsaturated-fat/low-carbohydrate diet improves periph- eral insulin sensitivity in non-insulin-dependent diabetic patients. Metabolism 41:1373â1378. Pariza MW, Park Y, Cook ME. 2001. The biologically active isomers of conjugated linoleic acid. Prog Lipid Res 40:283â298. Park Y, Albright KJ, Liu W, Storkson JM, Cook ME, Pariza MW. 1997. Effect of conjugated linoleic acid on body composition in mice. Lipids 32:853â858. Park Y, Storkson JM, Albright KJ, Liu W, Pariza MW. 1999. Evidence that the trans-10,cis-12 isomer of conjugated linoleic acid induces body composition changes in mice. Lipids 34:235â241. Parker DR, Weiss ST, Troisi R, Cassano PA, Vokonas PS, Landsberg L. 1993. Rela- tionship of dietary saturated fatty acids and body habitus to serum insulin concentrations: The Normative Aging Study. Am J Clin Nutr 58:129â136. Parnaud G, Corpet DE. 1997. Colorectal cancer: Controversial role of meat con- sumption. Bull Cancer 84:899â911. Parrish CC, Pathy DA, Angel A. 1990. Dietary fish oils limit adipose tissue hypertrophy in rats. Metabolism 39:217â219. Parrish CC, Pathy DA, Parkes JG, Angel A. 1991. Dietary fish oils modify adipocyte structure and function. J Cell Physiol 148:493â502.
869 M ACRONUTRIENTS AND HEALTHFUL DIETS Pearce ML, Dayton S. 1971. Incidence of cancer in men on a diet high in poly- unsaturated fat. Lancet 1:464â467. Peiris AN, Struve MF, Mueller RA, Lee MB, Kissebah AH. 1988. Glucose metabo- lism in obesity: Influence of body fat distribution. J Clin Endocrinol Metab 67:760â767. Pelkman CL, Coval SM, Mauger DT, Zhao G, Kris-Etherton PM. 2001. A meta- analysis of low-fat versus high-MUFA diets. FASEB J 15:394. Pelletier DL, Frongillo EA, Schroeder DG, Habicht J-P. 1995. The effects of mal- nutrition on child mortality in developing countries. Bull World Health Organ 73:443â448. Perez-Jimenez F, Espino A, Lopez-Segura F, Blanco J, Ruiz-Gutierrez V, Prada JL, Lopez-Miranda J, Jimenez-Pereperez J, Ordovas JM. 1995. Lipoprotein con- centrations in normolipidemic males consuming oleic acid-rich diets from two different sources: Olive oil and oleic acid-rich sunflower oil. Am J Clin Nutr 62:769â775. Perez-Jimenez F, Catrso P, Lopez-Miranda J, Paz-Rojas E, Blanco A, Lopez-Segura F, Velasco F, Marin C, Fuentes F, Ordovas JM. 1999. Circulating levels of endothelial function are modulated by dietary monounsaturated fat. Athero- sclerosis 145:351â358. Perez-Jimenez F, Lopez-Miranda J, Pinillos MD, Gomez P, Pas-Rojas E, Montilla P, Marin C, Velasco MJ, Blanco-Molina A, Jimenez Pereperez JA, Ordovas JM. 2001. A Mediterranean and a high-carbohydrate diet improves glucose metabolism in healthy young persons. Diabetologica 44:2038â2043. Peterson S, Sigman-Grant M. 1997. Impact of adopting lower-fat food choices on nutrient intake of American children. Pediatrics 100:E4. Pfeuffer M, Ahrens F, Hagemeister H, Barth CA. 1988. Influence of casein versus soy protein isolate on lipid metabolism of minipigs. Ann Nutr Metab 32:83â89. Phillips RL. 1975. Role of life-style and dietary habits in risk of cancer among Seventh-Day Adventists. Cancer Res 35:3513â3522. Pietinen P, Ascherio A, Korhonen P, Hartman AM, Willett WC, Albanes D, Virtamo J. 1997. Intake of fatty acids and risk of coronary heart disease in a cohort of Finnish men. The Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study. Am J Epidemiol 145:876â887. Poppitt SD, Swann DL. 1998. Dietary manipulation and energy compensation: Does the intermittent use of low-fat items in the diet reduce total energy intake in free-feeding lean men? Int J Obes Relat Metab Disord 22:1024â1031. Poppitt SD, Swann DL, Murgatroyd PR, Elia M, McDevitt RM, Prentice AM. 1998. Effect of dietary manipulation on substrate flux and energy balance in obese women taking the appetite suppressant dexfenfluramine. A m J Clin Nutr 68:1012â1021. Popp-Snijders C, Schouten JA, Heine RJ, van der Meer J, van der Veen EA. 1987. Dietary supplementation of omega-3 polyunsaturated fatty acids improves insulin sensitivity in non-insulin-dependent diabetes. Diabetes Res 4:141â147. Porrini M, Crovetti R, Riso P, Santangelo A, Testolin G. 1995. Effects of physical and chemical characteristics of food on specific and general satiety. Physiol Behav 57:461â468. Prentice AM. 2001. Overeating: The health risks. Obes Res 9:234Sâ238S. Price JM, Grinker J. 1973. Effects of degree of obesity, food deprivation, and palatability on eating behavior of humans. J Comp Physiol Psychol 85:265â271.
870 DIETARY REFERENCE INTAKES Promislow JHE, Goodman-Gruen D, Slymen DJ, Barrett-Conner E. 2002. Protein consumption and bone mineral density in the elderly. The Rancho Bernardo Study. Am J Epidemiol 155:636â644. Proserpi C, Sparti A, Schutz Y, Di Vetta V, Milon H, Jéquier E. 1997. Ad libitum intake of a high-carbohydrate or high-fat diet in young men: Effects on nutri- ent balances. Am J Clin Nutr 66:539â545. Raben A, Macdonald I, Astrup A. 1997. Replacement of dietary fat by sucrose or starch: Effects on 14 d ad libitum energy intake, energy expenditure and body weight in formerly obese and never-obese subjects. Int J Obes Relat Metab Disord 21:846â859. Ramon JM, Bou R, Romea S, Alkiza ME, Jacas M, Ribes J, Oromi J. 2000. Dietary fat intake and prostate cancer risk: A case-control study in Spain. Cancer Causes Control 11:679â685. Rath R, Masek J, Kujalová V, Slabochová Z. 1974. Effect of a high sugar intake on s some metabolic and regulatory indicators in young men. Nahrung 18:343â353. Reaven GM. 1988. Banting lecture 1988. Role of insulin resistance in human dis- ease. Diabetes 37:1595â1607. Reaven GM. 1995. Pathophysiology of insulin resistance in human disease. Physiol Rev 75:473â486. Reaven GM. 2001. Insulin resistance, compensatory hyperinsulinemia, and coro- nary heart disease: Syndrome X revisited. In: Jefferson LS, Cherrington AD, Goodman HM, eds. Handbook of Physiology. Section 7: The Endocrine System. Vol- ume II: The Endocrine Pancreas and Regulation of Metabolism. Oxford: Oxford University Press. Pp. 1169â1197. Reaven P, Parthasarathy S, Grasse BJ, Miller E, Almazan F, Mattson FH, Khoo JC, Steinberg D, Witztum JL. 1991. Feasibility of using an oleate-rich diet to reduce the susceptibility of low-density lipoprotein to oxidative modification in humans. Am J Clin Nutr 54:701â706. Reaven P, Parthasarathy S, Grasse BJ, Miller E, Steinberg D, Witztum JL. 1993. Effects of oleate-rich and linoleate-rich diets on the susceptibility of low density lipoprotein to oxidative modification in mildly hypercholesterolemic subjects. J Clin Invest 91:668â676. Reaven PD, Grasse BJ, Tribble DL. 1994. Effects of linoleate-enriched and oleate- enriched diets in combination with alpha-tocopherol on the susceptibility of LDL and LDL subfractions to oxidative modification in humans. Arterioscler Thromb 14:557â566. Reddy BS. 1992. Dietary fat and colon cancer: Animal model studies. L ipids 27:807â813. Reddy BS, Burill C, Rigotty J. 1991. Effect of diets high in Ï-3 and Ï -6 fatty acids on initiation and postinitiation stages of colon carcinogenesis. Cancer Res 51:487â491. Reiser S, Handler HB, Gardner LB, Hallfrisch JG, Michaelis OE, Prather ES. 1979. Isocaloric exchange of dietary starch and sucrose in humans. II. Effect on fasting blood insulin, glucose, and glucagon and on insulin and glucose response to a sucrose load. Am J Clin Nutr 32:2206â2216. Rémésy C, Behr SR, Levrat M-A, Demigné C. 1992. Fiber fermentability in the rat cecum and its physiological consequences. Nutr Res 12:1235â1244. Renaud S, de Lorgeril M, Delaye J, Guidollet J, Jacquard F, Mamelle N, Martin JL, Monjaud I, Salen P, Toubol P. 1995. Creten Mediterranean diet for preven- tion of coronary heart disease. Am J Clin Nutr 61:1360Sâ1367S.
871 M ACRONUTRIENTS AND HEALTHFUL DIETS Ricketts CD. 1997. Fat preferences, dietary fat intake and body composition in children. Eur J Clin Nutr 51:778â781. Rissanen AM, Heliövaara M, Knekt P, Reunanen A, Aromaa A. 1991. Determinants of weight gain and overweight in adult Finns. Eur J Clin Nutr 45:419â430. Robertson WG, Peacock M. 1982. The pattern of urinary stone disease in Leeds and in the United Kingdom in relation to animal protein intake during the period 1960â1980. Urol Int 37:394â399. Robertson WG, Heyburn PJ, Peacock M, Hanes FA, Swaminathan R. 1979. The effect of high animal protein intake on the risk of calcium stone-formation in the urinary tract. Clin Sci 57:285â288. Roche HM, Zampelas A, Jackson KG, Williams CM, Gibney MJ. 1998. The effect of test meal monounsaturated fatty acid:saturated fatty acid ratio on postprandial lipid metabolism. Br J Nutr 79:419â424. Rodier M, Colette C, Crastes de Paulet P, Crastes de Paulet A, Monnier L. 1993. Relationships between serum lipids, platelet membrane fatty acid composition and platelet aggregation in type 2 diabetes mellitus. Diabete Metab 19:560â565. Rolland-Cachera MF, Deheeger M, Akrout M, Bellisle F. 1995. Influence of macro- nutrients on adiposity development: A follow up study of nutrition and growth from 10 months to 8 years of age. Int J Obes Relat Metab Disord 19:573â578. Rolls BJ, Hetherington M, Burley VJ. 1988. The specificity of satiety: The influence of foods of different macronutrient content on the development of satiety. Physiol Behav 43:145â153. Rolls BJ, Laster LJ, Summerfelt A. 1989. Hunger and food intake following con- sumption of low-calorie foods. Appetite 13:115â127. Rolls BJ, Kim-Harris S, Fischman MW, Foltin RW, Moran TH, Stoner SA. 1994. Satiety after preloads with different amounts of fat and carbohydrate: Implica- tions for obesity. Am J Clin Nutr 60:476â487. Romieu I, Willett WC, Stampfer MJ, Colditz GA, Sampson L, Rosner B, Hennekens CH, Speizer FE. 1988. Energy intake and other determinants of relative weight. Am J Clin Nutr 47:406â412. Rose DP, Connolly JM. 2000. Regulation of tumor angiogenesis by dietary fatty acids and eicosanoids. Nutr Cancer 37:119â127. Rugg-Gunn AJ, Hackett AF, Jenkins GN, Appleton DR. 1991. Empty calories? Nutrient intake in relation to sugar intake in English adolescents. J Hum Nutr Diet 4:101â111. Rush D, Stein Z, Susser M. 1980. A randomized controlled trial of prenatal nutri- tion supplementation in New York City. Pediatrics 65:683â697. Rustan AC, Hustvedt B-E, Drevon CA. 1993. Dietary supplementation of very long- chain n-3 fatty acids decreases whole body lipid utilization in the rat. J Lipid Res 34:1299â1309. Salmerón J, Manson JE, Stampfer MJ, Colditz GA, Wing AL, Willett WC. 1997. Dietary fiber, glycemic load, and risk of non-insulin-dependent diabetes mellitus in women. J Am Med Assoc 277:472â477. Salmerón J, Hu FB, Manson JE, Stampfer MJ, Colditz GA, Rimm EB, Willett WC. 2001. Dietary fat intake and risk of type 2 diabetes in women. Am J Clin Nutr 73:1019â1026. Salomon O, Steinberg DM, Zivelin A, Gitel S, Dardik R, Rosenberg N, Berliner S, Inbal A, Many A, Lubetsky A, Varon D, Martinowitz U, Seligsohn U. 1999. Single and combined prothrombic factors in patients with idiopathic venous thromboembolism. Prevalence and risk assessment. Arterioscler Thromb Vasc Biol 19:511â518.
872 DIETARY REFERENCE INTAKES Saltzman E, Dallal GE, Roberts SB. 1997. Effect of high-fat and low-fat diets on voluntary energy intake and substrate oxidation: Studies in identical twins consuming diets matched for energy density, fiber, and palatability. Am J Clin Nutr 66:1332â1339. Samaras K, Kelly PJ, Chiano MN, Arden N, Spector TD, Campbell LV. 1998. Genes versus environment. The relationship between dietary fat and total and central abdominal fat. Diabetes Care 21:2069â2076. Sanders TAB, Hinds A. 1992. The influence of a fish oil high in docosahexaenoic acid on plasma lipoprotein and vitamin E concentrations and haemostatic function in healthy male volunteers. Br J Nutr 68:163â173. Sanders TAB, Oakley FR, Miller GJ, Mitropoulos KA, Crook D, Oliver MF. 1997. Influence of n-6 versus n-3 polyunsaturated fatty acids in diets low in saturated fatty acids on plasma lipoproteins and hemostatic factors. Arterioscler Thromb Vasc Biol 17:3449â3460. Saris WHM, Astrup A, Prentice AM, Zunft HJF, Formiguera X, Verboeket-van de Venne WPHG, Raben A, Poppitt SD, Seppelt B, Johnston S, Vasilaras TH, Keogh GF. 2000. Randomized controlled trial of changes in dietary carbohydrate/fat ratio and simple vs complex carbohydrates on body weight and blood lipids: The CARMEN study. Int J Obes Relat Metab Disord 24:1310â1318. Sasaki S, Horacsek M, Kesteloot H. 1993. An ecological study of the relationship between dietary fat intake and breast cancer mortality. Prev Med 22:187â202. Sawaya AL, Fuss PJ, Dallal GE, Tsay R, McCrory MA, Young V, Roberts SB. 2001. Meal palatability, substrate oxidation and blood glucose in young and older men. Physiol Behav 72:5â12. Saynor R, Gillott T. 1992. Changes in blood lipids and fibrinogen with a note on safety in a long term study on the effects of n-3 fatty acids in subjects receiving fish oil supplements and followed for seven years. Lipids 27:533â538. Schmidt EB, Lervang H-H, Varming K, Madsen P, Dyerberg J. 1992. Long-term supplementation with n-3 fatty acids. I: Effect on blood lipids, haemostasis and blood pressure. Scand J Clin Lab Invest 52:221â228. Schønberg S, Krokan HE. 1995. The inhibitory effect of conjugated dienoic derivates (CLA) of linoleic acid on the growth of human tumor cell lines is in part due to increased lipid peroxidation. Anticancer Res 15:1241â1246. Schuurman AG, van den Brandt PA, Dorant E, Brants HAM, Goldbohm RA. 1999. Association of energy and fat intake with prostate carcinoma risk. Results from the Netherlands Cohort Study. Cancer 86:1019â1027. Seagle HM, Davy BM, Grunwald G, Hill JO. 1997. Energy density of self-reported food intake: Variation and relationship to other food components. Obes Res 5:78S. Serdula MK, Ivery D, Coates RJ, Freedman DS, Williamson DF, Byers TE. 1993. Do obese children become obese adults? A review of the literature. Prev Med 22:167â177. Severson RK, Nomura AMY, Grove JS, Stemmermann GN. 1989. A prospective study of demographics, diet, and prostate cancer among men of Japanese ancestry in Hawaii. Cancer Res 49:1857â1860. Shannon BM, Tershakovec AM, Martel JK, Achterberg CL, Cortner JA, Smiciklas- Wright HS, Stallings VA, Stolley PD. 1994. Reduction of elevated LDL- cholesterol levels of 4- to 10-year-old children through home-based dietary education. Pediatrics 94:923â927.
873 M ACRONUTRIENTS AND HEALTHFUL DIETS Shea S, Basch CE, Stein AD, Contento IR, Irigoyen M, Zybert P. 1993. Is there a relationship between dietary fat and stature or growth in children three to five years of age? Pediatrics 92:579â586. Sheppard L, Kristal AR, Kushi LH. 1991. Weight loss in women participating in a randomized trial of low-fat diets. Am J Clin Nutr 54:821â828. Shetty PS, Prentice AM, Goldberg GR, Murgatroyd PR, McKenna APM, Stubbs RJ, Volschenk PA. 1994. Alterations in fuel selection and voluntary food intake in response to isoenergetic manipulation of glycogen stores in humans. Am J Clin Nutr 60:534â543. Shide DJ, Rolls BJ. 1995. Information about the fat content of preloads influences energy intake in healthy women. J Am Diet Assoc 95:993â998. Shu XO, Zheng W, Potischman N, Brinton LA, Hatch MC, Gao YT, Fraumeni JF. 1993. A population-based case-control study of dietary factors and endometrial cancer in Shanghai, Peopleâs Republic of China. Am J Epidemiol 137:155â165. Shultz TD, Leklem JE. 1983. Dietary status of Seventh-day Adventists and non- vegetarians. J Am Diet Assoc 83:27â33. Shultz TD, Chew BP, Seaman WR, Luedecke LO. 1992. Inhibitory effect of conju- gated dienoic derivates of linoleic acid and β-carotene on the in vitro growth of human cancer cells. Cancer Lett 63:125â133. Sierakowski R, Finlayson B, Landes RR, Finlayson CD, Sierakowski N. 1978. The frequency of urolithiasis in hospital discharge diagnoses in the United States. Invest Urol 15:438â441. Simell O, Niinikoski H, Rönnemaa T, Lapinleimu H, Routi T, Lagström H, Salo P, Jokinen E, Viikari J. 2000. Special Turku Coronary Risk Factor Intervention Project for Babies (STRIP). Am J Clin Nutr 72:1316Sâ1331S. Singh RB, Rastogi SS, Verma R, Laxmi B, Singh R, Ghosh S, Niaz MA. 1992. Randomised controlled trial of cardioprotective diet in patients with recent acute myocardial infarction: Results of one year follow up. Br Med J 304:1015â1019. Singh RB, Ghosh S, Niaz AM, Gupta S, Bishnoi I, Sharma JP, Agarwal P, Rastogi SS, Beegum R, Chibo H. 1995. Epidemiologic study of diet and coronary risk factors in relation to central obesity and insulin levels in rural and urban populations of north India. Int J Cardiol 47:245â255. Singh RB, Niaz MA, Sharma JP, Kumar R, Rastogi V, Moshiri M. 1997. Random- ized, double-blind, placebo-controlled trial of fish oil and mustard oil in patients with suspected acute myocardial infarction: The Indian Experiment of Infarct Survivalâ4. Cardiovasc Drugs Ther 11:485â491. Siscovick DS, Raghunathan TE, King I, Weinmann S, Wicklund KG, Albright J, Bovbjerg V, Arbogast P, Smith H, Kushi LH, Cobb LA, Copass MK, Psaty BM, Lemaitre R, Retzlaff B, Childs M, Knopp RH. 1995. Dietary intake and cell membrane levels of long-chain n-3 polyunsaturated fatty acids and the risk of primary cardiac arrest. J Am Med Assoc 274:1363â1367. Skinner JD, Carruth BR, Moran J, Houck K, Coletta F. 1999. Fruit juice intake is not related to childrenâs growth. Pediatrics 103:58â64. Skov AR, Toubro S, Ronn B, Holm L, Astrup A. 1999. Randomized trial on protein vs carbohydrate in ad libitum fat reduced diet for the treatment of obesity. Int J Obes Relat Metab Disord 23:528â536. Slattery ML, Potter JD, Sorenson AW. 1994. Age and risk factors for colon cancer (United States and Australia): Are there implications for understanding dif- ferences in case-control and cohort studies? Cancer Causes Control 5:557â563. Slattery ML, Caan BJ, Potter JD, Berry TD, Coates A, Duncan D, Edwards SL. 1997. Dietary energy sources and colon cancer risk. Am J Epidemiol 145:199â210.
874 DIETARY REFERENCE INTAKES Sonko BJ, Prentice AM, Poppitt SD, Prentice A, Jequier E, Whitehead RG. 1994. Could dietary fat intake be an important determinant of seasonal weight changes in a rural subsistence farming community in The Gambia? In: Nestlé Foundation for the Study of the Problems of Nutrition in the World. Annual Report 1994. Lausanne, Switzerland: Nestlé Foundation. Pp. 74â87. Sonnenberg LM, Quatromoni PA, Gagnon DR, Cupples LA, Franz MM, Ordovas JM, Wilson PWF, Schaefer EJ, Millen BE. 1996. Diet and plasma lipids in women. II. Macronutrients and plasma triglycerides, high-density lipoprotein, and the ratio of total to high-density lipoprotein cholesterol in women: The Framingham Nutrition Studies. J Clin Epidemiol 49:665â672. Stamler J. 1979. Population studies. In: Levy R, Rifkind B, Dennis B, Ernst N, eds. Nutrition, Lipids, and Coronary Heart Disease. New York: Raven Press. Pp. 25â88. Stangl GI. 2000. Conjugated linoleic acids exhibit a strong fat-to-lean partitioning effect, reduce serum VLDL lipids and redistribute tissue lipids in food- restricted rats. J Nutr 130:1140â1146. Stary HC. 1989. Evolution and progression of atherosclerotic lesions in coronary arteries of children and young adults. Arteriosclerosis 9:I19âI32. Stefanick ML, Mackey S, Sheehan M, Ellsworth N, Haskell WL, Wood PD. 1998. Effects of diet and exercise in men and postmenopausal women with low levels of HDL cholesterol and high levels of LDL cholesterol. N Engl J Med 339:12â20. Steinberg D, Parthawarathy S, Carew TE, Khoo JC, Witztum JL. 1989. Beyond choles- terol. Modifications of low-density lipoprotein that increase its atherogenicity. N Engl J Med 320:915â924. Storlien LH, Kraegen EW, Chisholm DJ, Ford GL, Bruce DG, Pascoe WS. 1987. Fish oil prevents insulin resistance induced by high-fat feeding. S cience 237:885â888. Storlien LH, Jenkins AB, Chisholm DJ, Pascoe WS, Khouri S, Kraegen EW. 1991. Influence of dietary fat composition on development of insulin resistance in rats. Relationship to muscle triglyceride and Ï-3 fatty acids in muscle phospho- lipid. Diabetes 40:280â289. Straznicky NE, OâCallaghan CJ, Barrington VE, Louis WJ. 1999. Hypotensive effect of low-fat, high-carbohydrate diet can be independent of changes in plasma insulin concentrations. Hypertension 34:580â585. Strong JP, Malcom GT, Newman WP, Oalmann MC. 1992. Early lesions of athero- sclerosis in childhood and youth: Natural history and risk factors. J Am Coll Nutr 11:51Sâ54S. Stubbs RJ, Harbron CG, Murgatroyd PR, Prentice AM. 1995a. Covert manipulation of dietary fat and energy density: Effect on substrate flux and food intake in men eating ad libitum. Am J Clin Nutr 62:316â329. Stubbs RJ, Ritz P, Coward WA, Prentice AM. 1995b. Covert manipulation of the ratio of dietary fat to carbohydrate and energy density: Effect on food intake and energy balance in free-living men eating ad libitum. Am J Clin Nutr 62:330â337. Stubbs RJ, Harbron CG, Prentice AM. 1996. Covert manipulation of the dietary fat to carbohydrate ratio of isoenergetically dense diets: Effect on food intake in feeding men ad libitum. Int J Obes Relat Metab Disord 20:651â660. Sugano M, Tsujita A, Yamasaki M, Noguchi M, Yamada K. 1998. Conjugated linoleic acid modulates tissue levels of chemical mediators and immunoglobulins in rats. Lipids 33:521â527.
875 M ACRONUTRIENTS AND HEALTHFUL DIETS Swinburn BA, Boyce VL, Bergman RN, Howard BV, Bogardus C. 1991. Deteriora- tion in carbohydrate metabolism and lipoprotein changes induced by modern, high fat diet in Pima Indians and Caucasians. J Clin Endocrinol Metab 73:156â165. Swinburn BA, Metcalf PA, Ley SJ. 2001. Long-term (5-year) effects of a reduced-fat diet intervention in individuals with glucose intolerance. D iabetes Care 24:619â624. Takahashi M, Przetakiewicz M, Ong A, Borek C, Lowenstein JM. 1992. Effect of omega 3 and omega 6 fatty acids on transformation of cultured cells by irra- diation and transfection. Cancer Res 52:154â162. Talamini R, Franceschi S, La Vecchia C, Serraino D, Barra S, Negri E. 1992. Diet and prostatic cancer: A case-control study in Northern Italy. N utr Cancer 18:277â286. Tao SC, Huang ZD, Wu XG, Zhou BF, Xiao ZK, Hao JS, Li YH, Cen RC, Rao XX. 1989. CHD and its risk factors in the Peopleâs Republic of China. Int J Epidemiol 18:S159âS163. Tate G, Mandell BF, Laposata M, Ohliger D, Baker DG, Schumacher HR, Zurier RB. 1989. Suppression of acute and chronic inflammation by dietary gamma linolenic acid. J Rheumatol 16:729â733. Teixeira SR, Potter SM, Weigel R, Hannum S, Erdman JW, Hasler CM. 2000. Effects of feeding 4 levels of soy protein for 3 and 6 wk on blood lipids and apolipoproteins in moderately hypercholesterolemic men. A m J Clin Nutr 71:1077â1084. Terpstra AHM, Holmes JC, Nicolosi RJ. 1991. The hypocholesterolemic effect of dietary soybean protein vs. casein in hamsters fed cholesterol-free or cholesterol- enriched semipurified diets. J Nutr 121:944â947. Thomas CD, Peters JC, Reed GW, Abumrad NN, Sun M, Hill JO. 1992. Nutrient balance and energy expenditure during ad libitum feeding of high-fat and high-carbohydrate diets in humans. Am J Clin Nutr 55:934â942. Thomsen C, Rasmussen O, Christiansen C, Pedersen E, Vesterlund M, Storm H, Ingerslev J, Hermansen K. 1999. Comparison of the effects of a monounsaturated fat diet and a high carbohydrate diet on cardiovascular risk factors in first degree relatives to type-2 diabetic subjects. Eur J Clin Nutr 52:818â823. Tillotson JL, Grandits GA, Bartsch GE, Stamler J. 1997. Relation of dietary carbo- hydrates to blood lipids in the special intervention and usual care groups in the Multiple Risk Factor Intervention Trial. Am J Clin Nutr 65:314Sâ326S. Tobin J, Spector D. 1986. Dietary protein has no effect on future creatinine clear- ance (Ccr). Gerontologist 26:59A. Toft I, Bønaa KH, Ingebretsen OC, Nordøy A, Jenssen T. 1995. Effects of n-3 polyunsaturated fatty acids on glucose homeostasis and blood pressure in essential hypertension. A randomized, controlled trial. A nn Intern Med 123:911â918. Toniolo P, Riboli E, Shore RE, Pasternack BS. 1994. Consumption of meat, animal products, protein, and fat and risk of breast cancer: A prospective cohort study in New York. Epidemiology 5:391â397. Tonstad S, Sivertsen M. 1997. Relation between dietary fat and energy and micro- nutrient intakes. Arch Dis Child 76:416â420. Torun B, Chew F. 1999. Protein-energy malnutrition. In: Shils ME, Olson JA, Shike M, Ross AC, eds. Modern Nutrition in Health and Disease, 9th ed. Baltimore, MD: Williams and Wilkins. Pp. 963â988. Tremblay A, Plourde G, Despres J-P, Bouchard C. 1989. Impact of dietary fat con- tent and fat oxidation on energy intake in humans. Am J Clin Nutr 49:799â805.
876 DIETARY REFERENCE INTAKES Tremblay A, Lavallee N, Almeras N, Allard L, Despres J-P, Bouchard C. 1991. Nutritional determinants of the increase in energy intake associated with a high-fat diet. Am J Clin Nutr 53:1134â1137. Tremblay MS, Willms JD. 2000. Secular trends in the body mass index of Canadian children. Can Med Assoc J 163:1429â1433. Tremoli E, Maderna P, Marangoni F, Colli S, Eligini S, Catalano I, Angeli MT, Pazzucconi F, Gainfranceschi G, Davi G, Stragliotto E, Sirtori CR, Galli C. 1995. Prolonged inhibition of platelet aggregation after n-3 fatty acid ethyl ester ingestion by healthy volunteers. Am J Clin Nutr 61:607â613. Trevisan M, Krogh V, Freudenheim J, Blake A, Muti P, Panico S, Farinaro E, Mancini M, Menotti A, Ricci G. 1990. Consumption of olive oil, butter, and vegetable oils and coronary heart disease risk factors. The Research Group ATS-RF2 of the Italian National Research Council. J Am Med Assoc 263:688â692. Trichopoulou A, Katsouyanni K, Stuver S, Tzala L, Gnardellis C, Rimm E, Trichopoulos D. 1995. Consumption of olive oil and specific food groups in relation to breast cancer risk in Greece. J Natl Cancer Inst 87:110â116. Trinidad TP, Wolever TMS, Thompson LU. 1993. Interactive effects of Ca and SCFA on absorption in the distal colon of men. Nutr Res 13:417â425. Trinidad TP, Wolever TMS, Thompson LU. 1996. Effect of acetate and propionate on calcium absorption from the rectum and distal colon of humans. Am J Clin Nutr 63:574â578. Troiano RP, Flegal KM, Kuczmarski RJ, Campbell SM, Johnson CL. 1995. Over- weight prevalence and trend for children and adolescents: The National Health and Nutrition Examination Surveys, 1963 to 1991. Arch Pediatr Adolesc Med 149:1085â1091. Tsuboyama-Kasaoka N, Takahashi M, Tanemura K, Kim H-J, Tange T, Okuyama H, Kasai M, Ikemoto S, Ezaki O. 2000. Conjugated linoleic acid supplementation reduces adipose tissue by apoptosis and develops lipodystrophy in mice. Diabetes 49:1534â1542. Tucker LA, Kano MJ. 1992. Dietary fat and body fat: A multivariate study of 205 adult females. Am J Clin Nutr 56:616â622. Tuomilehto J, Lindström J, Eriksson JG, Valle TT, Hämäläinen H, Ilanne-Parikka P, Keinänen-Kiukaanniemi S, Laakso M, Louheranta A, Rastas M, Salminen V, Uusitupa M. 2001. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Eng J Med 344:1343â1350. Turini ME, Powell WS, Behr SR, Holub BJ. 1994. Effects of a fish-oil and vegetable- oil formula on aggregation and ethanolamine-containing lysophospholipid generation in activated human platelets and on leukotriene production in stimulated neutrophils. Am J Clin Nutr 60:717â724. Turner NC, Clapham JC. 1998. Insulin resistance, impaired glucose tolerance and non-insulin-dependent diabetes, pathologic mechanisms and treatment: Current status and therapeutic possibilities. Prog Drug Res 51:33â94. Uauy R, Mize CE, Castillo-Duran C. 2000. Fat intake during childhood: Metabolic responses and effects on growth. Am J Clin Nutr 72:1354Sâ1360S. Uematsu T, Nagashima S, Niwa M, Kohno K, Sassa T, Ishii M, Tomono Y, Yamato C, Kanamaru M. 1996. Effect of dietary fat content on oral bioavailability of menatetrenone in humans. J Pharm Sci 85:1012â1016. USDA (U.S. Department of Agriculture). 1996. The Food Guide Pyramid. Home and Garden Bulletin No. 252. Washington, DC: U.S. Government Printing Office.
877 M ACRONUTRIENTS AND HEALTHFUL DIETS Uusitupa M, Schwab U, Mäkimattila S, Karhapää P, Sarkkinen E, Maliranta H, à gren J, Penttilä I. 1994. Effects of two high-fat diets with different fatty acid compositions on glucose and lipid metabolism in healthy young women. Am J Clin Nutr 59:1310â1316. van Amelsvoort JM, van Stratum P, Kraal JH, Lussenburg RN, Houtsmuller UMT. 1989. Effects of varying the carbohydrate:fat ratio in a hot lunch on post- prandial variables in male volunteers. Br J Nutr 61:267â283. van Amelsvoort JM, van Stratum P, Dubbelman GP, Lussenburg RN. 1990. Effects of meal size reduction on postprandial variables in male volunteers. Ann Nutr Metab 34:163â174. van den Berg JJM, Cook NE, Tribble DL. 1995. Reinvestigation of the antioxidant properties of conjugated linoleic acid. Lipids 30:599â605. van den Brandt PA, vanât Veer P, Goldbohm RA, Dorant E, Volovics A, Hermus RJJ, Sturmans F. 1993. A prospective cohort study on dietary fat and the risk of postmenopausal breast cancer. Cancer Res 53:75â82. Van Dokkum W, Wesstra A, Schippers FA. 1982. Physiological effects of fibre-rich types of bread. 1. The effect of dietary fibre from bread on the mineral balance of young men. Br J Nutr 47:451â460. van Stratum P, Lussenburg RN, van Wezel LA, Vergroesen AJ, Cremer HD. 1978. The effect of dietary carbohydrate:fat ratio on energy intake by adult women. Am J Clin Nutr 31:206â212. vanât Veer P, Kok FJ, Brants HAM, Ockhuizen T, Sturmans F, Hermus RJJ. 1990. Dietary fat and the risk of breast cancer. Int J Epidemiol 19:12â18. Vartiainen E, Puska P, Pietinen P, Nissinen A, Leino U, Uusitalo U. 1986. Effects of dietary fat modifications on serum lipids and blood pressure in children. Acta Paediatr Scand 75:396â401. Veierød MB, Laake P, Thelle DS. 1997a. Dietary fat intake and risk of lung cancer: A prospective study of 51,452 Norwegian men and women. Eur J Cancer Prev 6:540â549. Veierød MB, Laake P, Thelle DS. 1997b. Dietary fat intake and risk of prostate cancer: A prospective study of 25,708 Norwegian men. Int J Cancer 73:634â638. Velie E, Kulldorff M, Schairer C, Block G, Albanes D, Schatzkin A. 2000. Dietary fat, fat subtypes, and breast cancer in postmenopausal women: A prospective cohort study. J Natl Cancer Inst 92:833â839. Vessby B. 2000. Dietary fat and insulin action in humans. Br J Nutr 83:S91âS96. Vessby B, Uusitupa M, Hermansen K, Riccardi G, Rivellese AA, Tapsell LC, Nälsén C, Berglund L, Louheranta A, Rasmussen BM, Calvert GD, Maffetone A, Pedersen E, Gustafsson I-B, Storlien LH. 2001. Substituting dietary saturated for monounsaturated fat impairs insulin sensitivity in healthy men and women: The KANWU study. Diabetologia 44:312â319. Visonneau S, Cesano A, Tepper SA, Scimeca JA, Santoli D, Kritchevsky D. 1997. Conjugated linoleic acid suppresses the growth of human breast adeno- carcinoma cells in SCID mice. Anticancer Res 17:969â974. Vobecky JS, Vobecky J, Normand L. 1995. Risk and benefit of low fat intake in childhood. Ann Nutr Metab 39:124â133. von Schacky C, Angerer P, Kothny W, Theisen K, Mudra H. 1999. The effect of dietary Ï-3 fatty acids on coronary atherosclerosis. A randomized, double- blind, placebo-controlled trial. Ann Intern Med 130:554â562. Walker AR, Walker BF. 1978. High high-density-lipoprotein cholesterol in African children and adults in a population free of coronary heart disease. Br Med J 2:1336â1337.
878 DIETARY REFERENCE INTAKES Walser M. 1992. The relationship of dietary protein to kidney disease. In: Liepa GU, Beitz DC, Beynen AC, Gorman MA, eds. Dietary Proteins: How They Alleviate Disease and Promote Better Health. Champaign, IL: American Oil Chemistsâ Society. Pp. 168â178. Ward MH, Zahm SH, Weisenburger DD, Gridley G, Cantor KP, Saal RC, Blair A. 1994. Dietary factors and non-Hodgkinâs lymphoma in Nebraska (United States). Cancer Causes Control 5:422â432. Waterlow JC. 1976. Classification and definition of protein-energy malnutrition. Monogr Ser World Health Organ 62:530â555. Weisburger JH. 1988. Comparison of nutrition as customary in the Western World, the Orient, and northern populations (Eskimos) in relation to specific disease risk. Arctic Med Res 47:110â120. West CE, Sullivan DR, Katan MB, Halferkamps IL, van der Torre HW. 1990. Boys from populations with high-carbohydrate intake have higher fasting triglyceride levels than boys from populations with high-fat intake. A m J Epidemiol 131:271â282. West KM, Kalbfleisch JM. 1971. Influence of nutritional factors on prevalence of diabetes. Diabetes 20:99â108. Westerterp KR, Verboeket-van de Venne WPHG, Westerterp-Plantenga MS, Velthuis-te Wierik EJM, de Graaf C, Weststrate JA. 1996. Dietary fat and body fat: An intervention study. Int J Obes Relat Metab Disord 20:1022â1026. Whigham LD, Cook ME, Atkinson RL. 2000. Conjugated linoleic acid: Implica- tions for human health. Pharmacol Res 42:503â510. Whiting SJ, Anderson DJ, Weeks SJ. 1997. Calciuric effects of protein and potassium bicarbonate but not of sodium chloride or phosphate can be detected acutely in adult women and men. Am J Clin Nutr 65:1465â1472. Willett WC. 1997. Specific fatty acids and risks of breast and prostate cancer: Dietary intake. Am J Clin Nutr 66:1557Sâ1563S. Willett WC. 1998. Is dietary fat a major determinant of body fat? Am J Clin Nutr 67:556Sâ562S. Willett WC, Stampfer MJ, Colditz GA, Rosner BA, Hennekens CH, Speizer FE. 1987. Dietary fat and the risk of breast cancer. N Engl J Med 316:22â28. Willett WC, Stampfer MJ, Colditz GA, Rosner BA, Speizer FE. 1990. Relation of meat, fat, and fiber intake to the risk of colon cancer in a prospective study among women. N Engl J Med 323:1664â1672. Willett WC, Hunter DJ, Stampfer MJ, Colditz G, Manson JE, Spiegelman D, Rosner B, Hennekens CH, Speizer FE. 1992. Dietary fat and fiber in relation to risk of breast cancer. An 8-year follow-up. J Am Med Assoc 268:2037â2044. Williams CL, Bollella M. 1995. Is a high-fiber diet safe for children? Pediatrics 96:1014â1019. Wisen O, Hellstrom PM, Johansson C. 1993. Meal energy density as a determinant of postprandial gastrointestinal adaptation in man. S cand J Gastroenterol 28:737â743. Wisker E, Maltz A, Feldheim W. 1988. Metabolizable energy of diets low or high in dietary fiber from cereals when eaten by humans. J Nutr 118:945â952. Wolfe BMJ, Piché LA. 1999. Replacement of carbohydrate by protein in a conventional-fat diet reduced cholesterol and triglyceride concentrations in healthy normolipidemic subjects. Clin Invest Med 22:140â148. Wolk A, Bergström R, Hunter D, Willett W, Ljung H, Holmberg L, Bergkvist L, Bruce à , Adami H-O. 1998. A prospective study of association of mono- unsaturated fat and other types of fat with risk of breast cancer. Arch Intern Med 158:41â45.
879 M ACRONUTRIENTS AND HEALTHFUL DIETS Wooley SC. 1972. Physiologic versus cognitive factors in short term food regulation in the obese and nonobese. Psychosom Med 34:62â68. Wu Y, Zheng W, Sellars TA, Kushi LH, Bostick RM, Potter JD. 1994. Dietary choles- terol, fat, and lung cancer incidence among older women: The Iowa Womenâs Health Study (United States). Cancer Causes Control 5:395â400. Yao M, Roberts SB. 2001. Dietary energy density and weight regulation. Nutr Rev 59:247â258. Yeomans MR, Gray RW, Mitchell CJ, True S. 1997. Independent effects of palatability and within-meal pauses on intake and appetite ratings in human volunteers. Appetite 29:61â76. Yost TJ, Jensen DR, Haugen BR, Eckel RH. 1998. Effect of dietary macronutrient composition on tissue-specific lipoprotein lipase activity and insulin action in normal-weight subjects. Am J Clin Nutr 68:296â302. Yu-Poth S, Zhao G, Etherton T, Naglak M, Jonnalagadda S, Kris-Etherton PM. 1999. Effects of the National Cholesterol Education Programâs Step I and Step II dietary intervention programs on cardiovascular disease risk factors: A meta- analysis. Am J Clin Nutr 69:632â646. Zambell KL, Keim NL, Van Loan MD, Gale B, Benito P, Kelley DS, Nelson GJ. 2000. Conjugated linoleic acid supplementation in humans: Effects of body composition and energy expenditure. Lipids 35:777â782. Zhang J, Sasaki S, Amano K, Kesteloot H. 1999. Fish consumption and mortality from all causes, ischemic heart disease, and stroke: An ecological study. Prev Med 28:520â529. Ziboh VA, Fletcher MP. 1992. Dose-response effects of dietary γ-linolenic acid- enriched oils on human polymorphonuclear-neutrophil biosynthesis of leukotriene B4. Am J Clin Nutr 55:39â45. Zock PL, Katan MB. 1992. Hydrogenation alternatives: Effects of trans fatty acids and stearic acid versus linoleic acid on serum lipids and lipoproteins in humans. J Lipid Res 33:399â410. Zock PL, Katan MB. 1998. Linoleic acid intake and cancer risk: A review and meta- analysis. Am J Clin Nutr 68:142â153. Zurier RB, Rossetti RG, Jacobson EW, DeMarco DM, Liu NY, Temming JE, White BM, Laposata M. 1996. Gamma-linolenic acid treatment of rheumatoid arthri- tis. A randomized, placebo-controlled trial. Arthritis Rheum 39:1808â1817.