3
Synthesis and Manufacturing: Creating and Exploiting New Substances and New Transformations
|
Some Challenges for Chemists and Chemical Engineers
|
|
GOALS
The long-term goal of the basic science in synthesis is to develop the ability to create all the substances and organized chemical systems and transformations that are possible under the limits of natural laws, not just those that occur in Nature. The importance of such an extension of Nature is clear in medicinal chemistry, for instance, but it is also part of the basic science of chemistry itself. In most cases, only enough of the new molecules will be created to permit an evaluation of their properties.
Of course, in reality new chemical substances are not synthesized at random with no purpose in mind—the numbers that have still not been created are too staggering for a random approach. By one estimate,1 as many as 10200 molecules could exist that have the general size and chemical character of typical medicines. Instead, chemists create new substances with the aim that their properties will be scientifically important or useful for practical purposes. As part of basic science, chemists have created new substances to test theories. For example, the molecule benzene has the special property of “aromaticity,” which in this context refers to special stability related to the electronic structure of a molecule. Significant effort has gone into creating new nonbenzenoid aromatic compounds to test the generality of theories about aromaticity. These experiments helped stimulate the application of quantum mechanical theory to the prediction of molecular energies.
Synthesis is achieved by performing chemical transformations, some of which are already known and some of which must be invented. Thus another goal of the basic science of synthesis is to invent new types of transformations, both because novel transformations are part of the universe of chemistry and also because they are the tools that make it possible to create interesting and useful new substances.
As another part of this field, it is also important to invent overall strategies to achieve multistep syntheses involving a sequence of transformations. Knowing how to perform simple transformations is like knowing the legal moves in chess; achieving the synthesis of a complicated molecule requires the strategic combination of many simple transformations, just as winning at chess requires a strategy to combine a sequence of legal moves. Some of the achievements in synthesis have involved the invention of general strategies to attack particular classes of difficult problems. These efforts will be directed toward the synthesis of many kinds of substances with a variety of descriptions, such as organic and inorganic compounds, polymers and ceramics, and hard materials and soft materials.
A special aspect of synthesis is the synthesis of natural products, chemical compounds found in nature. For example, once the chemical structure of penicillin had been determined, synthetic chemists set out to make it from simple chemicals using nonbiological techniques—even though the mold makes it very well. A synthetic procedure for making penicillin would also provide the opportunity for making analogous compounds. The structure of penicillin represented a challenge because it could not be made by the chemical transformations then known. Thus both new transformations and new strategies had to be invented. In this case, the result then allowed chemists to use these new transformations to make other related antibiotics, which are now used commonly as medicines. However, the original impetus for the synthesis was more basic. When chemists saw a structure in a natural compound that was too challenging for the current synthetic methodology, they created new methods and strategies that helped fill the chemical universe with more effective tools.
The goal of applied chemistry and chemical engineering is to learn how to manufacture useful new substances. Exploratory chemists often use methods that produce only small samples of the new material and are not suitable for large scale manufacturing, so entirely new schemes of synthesis must be invented. Also, manufacturing equipment and processes have to be developed that meet all the other criteria—efficient high-yield processes with inexpensive starting materials, easy isolation of pure products, and no environmental problems. A further more basic goal is to develop the general principles that can be applied to solve new problems.
In both synthesis and chemical manufacturing there is a general need to develop separation methods as well as synthesis methods. Until every synthetic reaction is perfect—with respect to atom economy, so no side-products are formed, and by performing the reaction without added solvents or catalysts—it is necessary to isolate the desired product and purify it. This is one of the most expensive aspects of some manufacturing processes, particularly in pharmaceutical manufacture. Thus there are two goals related to separations. One is to learn how to isolate and purify the desired compounds with high efficiency, and the other is to develop synthetic processes that do not need such separations. For manufacturing, a further goal is to eliminate solvents or at least replace them with
harmless, inexpensive, and easily removed or recovered solvents. One interesting approach is to substitute water for organic solvents; another is to use unusual solvents such as supercritical carbon dioxide or even supercritical water. Synthetic transformations often involve adding catalysts, substances that direct chemical reactions along less energetic but more efficient pathways. Solid catalysts that can be simply filtered away, or retained in a reactor as the other components pass through, are also desirable.
Atom economy is a goal only relatively recently understood. If all the atoms in the reactants are found in the desired product, we say that there is excellent atom economy. However, in many chemical reactions additional products are formed containing some of the atoms of the reactants. This is true in displacement and elimination reactions. It is also true if the reaction is not perfectly selective, and additional undesired products are formed. In most cases, the extra chemicals produced in displacement or elimination reactions or in nonselective reactions must be removed, and disposing of them adds cost and the potential for environmental problems (see Chapter 9 for further discussion of related matters).
PROGRESS TO DATE
Few known and thermodynamically feasible molecular structures are presently seen as impossible goals for synthesis. New transformations and effective strategies permit chemists to synthesize highly complex molecules, such as new natural compounds discovered in the continued chemical exploration of the natural world. Again, the point of such work is to develop new chemistry that permits an approach to structures of the type found in nature. This expands the power of chemistry and allows medicinal chemists to synthesize complex structures.
Synthetic efforts have also extended to compounds of theoretical and practical interest. A variety of novel cyclic, bridged, caged, interlocking, knotted, and otherwise topologically unusual structures have been prepared in both the organic and inorganic arenas. These studies have provided new insights into structure and reactivity: for example, new zeolitic materials have valuable properties as catalysts, and highly strained caged organic structures have shown intriguing energetic properties.
An interesting example is the work to synthesize the alkaloid strychnine. Robert Burns Woodward (Nobel Prize, 1965) was the first chemist to synthesize this very complicated structure, which has many places where the detailed spatial orientation of chemical bonds was important. The synthesis was accomplished by a team of students and postdoctoral associates that he directed. The world did not need a new source of highly toxic strychnine, which is widely available in nature, but the synthetic challenge required Woodward to develop new approaches to some synthetic problems. The general approaches—involving new reactions and new strategies—developed for strychnine and other biologically relevant molecules taught chemists how to tackle other challenging synthetic problems and
indicated the kind of complex molecule that was accessible through synthesis. Continuing developments of new strategies and new chemical reactions in the 1980s and 1990s led to several new syntheses of strychnine by other chemists, using less than half the number of steps of the original Woodward synthesis. Such work, expanding the tools of synthesis, has greatly increased our ability to synthesize useful medicines.
There are natural targets whose syntheses have important practical applications. For example, paclitaxel (marketed as Taxol by Bristol-Myers Squibb) is an anticancer compound originally isolated from the bark of a species of yew tree, but for a long time it seemed that natural sources would not be sufficient to meet the need. Consequently, many chemists developed synthetic approaches to paclitaxel from readily available materials, although these are not yet fully practical for manufacturing. The quest continues, and a semisynthetic route has been developed starting with a compound isolated from yew needles that can be harvested without destroying the tree. An alternative approach has employed plant-cell cultures in bioreactors to produce paclitaxel from yew needles (see Chapter 7 for discussion of related matters).
Some established methods to achieve particular transformations are not applicable in all situations, and some are not practical in large-scale manufacturing. Consequently, new methods are still being sought even for the simplest transformations. For example, the oxidation of an alcohol to a ketone or an aldehyde has long been performed in the laboratory by using certain compounds of chromium as the oxidant, but the aldehydes are themselves often oxidized further by the chromium reagents. Now, new oxidants have been developed that do not have this problem. Also, chromium oxidants are not practical in manufacturing. Their cost is high, and more seriously, the chromium-containing waste products are not environmentally tolerable. A better process for manufacturing would be to use the oxygen from air as the oxidant, resulting in water as a product. Catalysts for air oxidation already exist. However, it may be necessary to develop improved catalysts to perform this inexpensive and environmentally benign process.
This illustrates a general difference between the reactions used in manufacturing and those used in laboratory research. Whenever possible, manufacturing involves catalytic processes, with inexpensive reagents. In laboratory research, the immediate goal may be just to make a small sample of a new compound to determine its properties; consequently, there is less concern about the cost of reagents or the environmental effect that a large-scale version would create. The goal is to make the new compound as quickly as possible, to see if its properties make it worth manufacturing or simply to expand our basic knowledge.
Some manufacturing processes use the special properties of zeolites, inorganic substances that have interconnected cavities of well-defined size and shape. Reactions can be catalyzed inside the cavities, and they are normally selective for the shape of the substrate that can enter the cavities, and for the shape of the
products being formed. Selective absorption into the cavities also makes zeolites useful in separations.
Many laboratory syntheses of important structural types of compounds are too long or complex to work well in manufacturing. Chemists working in the process area are thus often engaged in inventing new approaches that use the most modern reactions, in order to develop compact synthetic schemes with small numbers of acceptable steps. The modern reactions that make this possible are being invented by chemists involved in basic discovery and creation, usually in universities. The pressure on industrial process chemists to develop practical schemes for manufacturing important products means that they do not normally have the time for the basic research that can lead to new chemical reactions.
How do synthetic chemists invent and create new reactions? After recognizing the challenge—a transformation that cannot be done easily with current methods—they look for inspiration in several places. One is biochemistry. If living cells already perform a transformation related to the desired one, it may be possible to invent an analog of the natural process—a biomimetic process. For example, in living cells there are enzymes that synthesize amino acids, and they do it with complete selectivity for the geometric orientation of the bonds. Chemists have now invented ways to do this without enzymes, but inspired by what is known about the natural process. For many practical purposes, the results are more useful and general for manufacturing.
Another inspiration comes from the chemists who study reaction mechanisms, learning how transformations occur (Chapter 4). The insights from such basic work often suggest new reactions that should be possible. For example, much work has gone on recently to understand the catalysis of chemical reactions by soluble complexes of certain metals such as palladium and platinum. They catalyze transformations, often of great importance in manufacturing, that otherwise may be impossible. Other work has helped us understand catalysis by metal particles, in which the special chemical properties of the metal surfaces play the key role. The insights from this work have stimulated the invention of other processes, while at the same time these applications have stimulated more work aimed at basic understanding. Work in synthesis is at least as important in showing chemists new parts of their universe as it is in solving practical problems.
Another important area is the use of photochemistry—chemistry that results from light absorption—to perform transformations that are not otherwise possible. The practical applications of photosynthesis were based on fundamental work to learn the new pathways that light absorption makes possible, but the work on these synthetic methods has also added to our basic understanding of the reaction mechanisms. The important natural process of photosynthesis also inspires some work in photochemistry, where the challenge is one of producing artificial photosynthetic systems that could use sunlight to drive the formation of energetic materials.
Finally, it is not possible to discount the component of intellectual creativity for such inventions. To quote George Bernard Shaw, “You see things; and you say, ‘Why?’ But I dream things that never were; and I say, ‘Why not?’” Chemists looking at their universe often see that some parts are missing, and they set out to create them. For example, a Nobel Prize was awarded to Herbert Brown and Georg Wittig in 1979 for the invention of new types of useful transformations that were unprecedented in nature. This effort to create goes beyond chemical reactions and extends to molecules and organized molecular systems that originate in the imagination of an individual chemist.
A Nobel Prize to Geoffrey Wilkinson and Ernest O. Fisher in 1973 recognized their creation, and understanding, of a new class of substances—unrelated to anything found in nature—called metallocenes. Originally, these organometallic sandwich compounds (and their “open-faced” versions) were of interest purely as new occupants of the chemical universe, as if a new type of planet had been created in the astronomical universe. However, some versions of such sandwich compounds have been shown more recently to act as catalysts for very significant reactions, and they are now applied in the manufacturing of polymers.
Synthetic inspiration can come from an attempt to make drastic improvements in the properties of existing substances. For example, some metals and metal compounds become superconducting at extremely low temperatures. Superconductors are attractive materials because they conduct electricity without any loss caused by resistance below a characteristic transition temperature, although above that temperature they are poor electrical conductors (and some are even insulators). More practical, high-temperature superconductors, first developed by Johannes G. Bednorz and Karl A. Muller, a mineralogist and a physicist, have great current and potential uses throughout science, including chemistry and chemical engineering. For example, most modern nuclear magnetic resonance (NMR) instruments have electromagnets wound with superconducting wires. Once an electric current is started in the wires, the source of electricity is disconnected, and the current keeps flowing as long as the temperature is kept very low by cooling the magnet with liquid helium. There is a great effort to invent new materials that would be superconducting at higher and more easily attained temperatures. Recently chemists and other scientists have been able to create new superconducting materials based on inorganic ceramics, which have considerably higher superconducting transition temperatures than the materials of Bednorz and Muller.
Since synthesis is such a central part of chemistry and chemical engineering, many other Nobel Prizes have recognized synthetic achievements. For example, a Nobel Prize in 1990 went to Elias J. Corey both for his contributions to the development of theory and methodology in organic synthesis and his application of them to the synthesis of important biologically active substances. It also recognized his formalization, and even computerization, of the strategies that successful multistep syntheses follow. Karl Ziegler and Giulio Natta received a Nobel
Prize in 1963 for inventing a new general method to synthesize important polymers, a method that uncovered much new basic science. A Nobel Prize in 1984 went to Robert Bruce Merrifield for his invention of a general approach to the synthesis of polypeptides and proteins, in a style directly reminiscent of the biological method used in such synthesis.
Although much of the preceding discussion involved the synthesis of new molecules by organic and inorganic chemists, there is another area of chemistry in which such creation is important—the synthesis of new atoms. The periodic table lists elements that have been discovered and isolated from nature, but a few have been created by human activity. Collision of atomic particles with the nuclei of existing atoms is the normal source of radioactive isotopes and of some of the very heavy elements at the bottom of the periodic table. Indeed nuclear chemists and physicists have created some of the most important elements that are used for nuclear energy and nuclear weapons, plutonium in particular.
An exciting goal has recently shown signs of progress. Theory has suggested that beyond the bottom of the usual periodic table, where the elements become quite unstable and have only a fleeting existence, there should be an “island of stability,” a group of elements more stable to nuclear decay than those coming earlier in the periodic table. Recently nuclear chemists have obtained evidence for such special stability in the predicted elements. They are not stable enough for long-term isolation, but their properties help confirm theories of nuclear structure and stability.
CHALLENGES AND OPPORTUNITIES FOR THE FUTURE
There is a healthy integration of the various branches of chemistry and chemical engineering. This integration brings together synthesis and measurement, theory and experiment, and design and manufacturing. The integration extends to the related disciplines with which chemistry and chemical engineering share the intellectual frontiers of science and technology—biology, physics, and electronics to name just a few. This has opened great new opportunities.
Within chemistry, synthetic chemists will increasingly make use of the special properties of metal-based catalysts, both solid and as soluble complexes, to devise new transformations that are selective and efficient. Chemical engineers are increasingly involved in product design, in addition to process design, requiring the integral participation of the chemical engineer in the molecular conception and design of the products. Physical chemists and chemical engineers will increasingly understand the surface chemistry and properties of solid catalysts; that understanding will help in the design of even better catalysts. Inorganic chemists will create many new types of molecules that combine inorganic and organic components, and that have applications and interest in both inorganic and organic chemistry. Chemical reactor modeling has advanced the field of electronic materials production significantly.
Theoretical chemists will increasingly be able to predict the properties of unknown molecules, and thus stimulate synthetic chemists to make the interesting ones. As chemical engineers open new frontiers in biological engineering, they team effectively with chemists and biologists to influence biosystems and processes from metabolism to cell adhesion. The synergy with biology is particularly strong in medicinal chemistry, where computer methods are used to design drugs. In every pharmaceutical company the computer-aided-design group works from the molecular structure of an important protein, that had been determined by structural chemists, and guides and rationalizes the work of the synthetic groups. Some important new medicines have been designed this way, and many more will come in the future.
Development of New Synthetic Methodology
In spite of the impressive progress achieved by synthetic chemists in the past several decades, the state of the art of methods for chemical synthesis remains at an early stage of development. There is a great need for new reactions and synthetic methods to permit any substance, of any complexity, be it organic or inorganic in structure, to be synthesized with high chemical efficiency, in a small number of steps, and with minimal cogeneration of any waste products. Synthetic methodology is the software of the chemical sciences. Just as there is an explicit expectation that there will be a constant evolution and improvement of the software and the operating systems that drive our computers, so should the chemical community expect that there will be constant improvement and evolution of the armament of methodologies available for the practicing preparative chemist and the chemical industries. An explicit goal of the chemical community should be to make continual improvements in synthetic methodology, thereby expanding the range of challenging molecules that are accessible to practical synthesis.
Catalysis
Some aspects of synthetic chemistry have changed in response to environmental needs. For example, in the pharmaceutical industry the classical methods produce, on the average, about nine times as much disposable waste as desired product. This has led to the demand for procedures that have atom efficiency, in which all the atoms of the reacting compounds appear in the product. Thus (as discussed earlier) the demand for atom economy offers additional opportunities for creative invention of transformations.
There are still numerous major important targets for synthesis. As a simple example, we currently manufacture ammonia—for use in fertilizers and other important products—by causing nitrogen to react with hydrogen at high temperature and pressure. Yet microorganisms in the roots of some legumes are able to convert nitrogen to ammonia at ordinary temperatures and pressures (although
not using gaseous hydrogen). We need to understand their chemistry, even if it is not as practical as is our current method of manufacturing ammonia. Similarly, the enzyme methane monooxygenase is able to use atmospheric oxygen to convert methane to methanol, whereas chemical systems tend to oxidize the methane further. We need to develop catalysts for this important process that could convert the methane in natural gas into a useful chemical. The catalysts for both of these processes will surely include the inorganic chemistry of metallic elements.
Combinatorial Chemistry
Some new aspects have been introduced into the style of synthesis recently, and these aspects hold great promise. One has to do with what are called combinatorial methods, where syntheses are carried out to make many different (but related) products at one time. Sometimes these are in a mixture, but with the different products attached to separate polymer beads, while at other times the simultaneous reactions are performed in many different small vessels. For example, a library of amide products might be produced by allowing a single reagent to react with a large group of different amines. For such libraries of products, different schemes have been devised to identify the individual components. Then the various products can be tested for a desirable property. In pharmaceutical chemistry, the library of products is put through biological screens, usually automated, to determine whether any of the products binds particularly well to an enzyme of interest or to another biological receptor. When one product has been identified as a good binder, normal synthetic procedures are used to make enough for thorough testing.
At one time, some people predicted that the ability of combinatorial methods to produce literally thousands of new products at one time would diminish the need for synthetic chemists in the pharmaceutical industry. However, the opposite has turned out to be true. The large number of leads produced by combinatorial methods requires many more synthetic chemists to continue research on these compounds in a more traditional way. Companies are increasing, not decreasing, their cadre of synthetic chemists. In addition, combinatorial methods have provoked radically new approaches to screening the libraries rapidly for good lead compounds, generating a new field called high throughput analysis and new opportunities for chemists.
Biomimetic Synthesis
As we increasingly understand the chemistry performed by living systems, in particular that catalyzed by enzymes, we will continue to develop biomimetic methods to achieve some of the special selectivities that enzymes show. Enzymes can selectively bind a particular molecule out of the mixture of substances in the cell, then hold it in such a way that the geometry of the enzyme-substrate com-
plex determines what happens next in a sequence of chemical reactions. For example, in an enzyme-substrate complex the reaction may take place at a particular part of a molecule even if that is not the most chemically reactive site—in contrast to normal synthetic chemistry, where changes take place at the reactive functional groups. Alternatively, enzymatic reaction might produce just one of several possible stereochemical consequences. For example, an enzyme might bind a natural L-amino acid but not the mirror image and thus distinguish the L -amino acids that human cells contain from the D-amino acids sometimes found in bacteria.
The selective introduction of chirality is a problem of much current interest in synthesis, and it is generally solved by using the same concept that governs enzymatic reactions. The nonchiral substrate interacts with a chiral reagent or catalyst, and the selective conversion of the substrate to a chiral product follows. Pioneering work to develop such methods was recognized in 2001 by Nobel prizes to William S. Knowles, Ryoji Noyori, and K. Barry Sharpless. In synthesis the binding often involves quite different forces than are used in enzymes, and the reagents and catalysts are much smaller than are protein enzymes. New procedures increasingly involve the formation of well-defined molecular complexes between substrate and catalyst, or substrate and reagent, that may allow chemists to overcome the classical domination of selectivity by the reactivity of functional groups. Donald J. Cram, Jean-Marie Lehn, and Charles J. Pedersen received Nobel prizes in 1987 for their work on molecular complexes.
There is another approach that is increasingly part of synthesis: the use of enzymes as catalysts. This approach is strengthened by the new ability of chemists and molecular biologists to modify enzymes and change their properties. There is also interest in the use of artificial enzymes for this purpose, either those that are enzyme-like but are not proteins, or those that are proteins but based on antibodies. Catalytic antibodies and nonprotein enzyme mimics have shown some of the attractive features of enzymes in processes for which natural enzymes are not suitable.
Self-Assembly
Historically, chemistry has been largely reductionist, breaking natural materials such as wood down to their pure components so they could be analyzed. It has since become more integrationist, putting together pure chemicals into complex organized structures. This is true in the new areas of nanoscience and nanotechnology, where synthesis is needed to make organized arrangements of many chemical components—nanostructures—with the distant goal of making tiny molecular machines and even molecular scale computers. Integration is also exploited as chemists begin to synthesize organized structures that imitate some of the properties of living cells. The processes carried out by living cells depend on the spatial organization of many different chemical components. Chemists, in
particular synthetic chemists, also need to start building such organization into structures with many different cooperating chemical components—both to understand them and to create useful microscopic devices. Such organization is most favorably based on spontaneous self-assembly of the chemical components, driven by their own selective molecular recognition and interactions. This is as if the components of an automobile would automatically fall into the correct places, rather than being placed there by machines. In chemistry such spontaneous self-assembly is possible on the molecular scale, but needs to be developed.
A vital aspect of chemical self-assembly is selection. In a mixture of compounds the correct pairings of chemicals must occur, so the system must have a way to choose those correct pairings. An exciting new challenge is to develop ways to impose a process of selection on the mixture of components, so that unwanted interactions are suppressed. In biology mutants are selected because they convey an advantage to a cell. Chemists need a similar tool, for example a library of possible catalysts from which the system would use their relative catalytic activities to self-select the one that is the most potent. This would remove the current need to screen the entire library for such activity. An even more desirable goal, difficult to reach currently, would be to develop chemical systems that could vary their own structures in a drive to produce the optimum of some desired property such as catalytic potency or strength of binding to a receptor. Such a chemical system could optimize itself according to the needs of humankind.
Manufacturing
Chemical manufacturing systems involve the implementation of chemical synthesis schemes to transform one composition of matter to another on scales from the very small (micrograms) to very large (hundreds of billions of kilograms per year). These materials and processes enable modern life. They may be inorganic, organic, or even biological, and they span the range from metals and concrete to glass, paper, and plastic; from advanced composites and electronic materials to fertilizers, agrochemicals, and dyes; from drinking water and fuels to safe refrigerants; and from pharmaceuticals to packaging for secure nuclear wastes. No matter what composition is produced, no matter what the purpose, and no matter what the scale, process systems engineers face relentless demands. These demands are for increased capital efficiency and reduced costs for materials, labor, and energy—all while producing material of high and consistent quality and doing so reliably, safely, sustainably, and with minimal environmental impact.
Specific new challenges to process systems engineering are both economic and social. Globalization of the chemical enterprise has opened new markets that are linked to the general increase in the standard of living throughout the world. At the same time, globalization has resulted in growing worldwide competition. This, along with the introduction of electronic commerce, is producing greater market efficiencies. It is also producing reduced manufacturing margins at a time
of increased investor demands for predictable earnings growth, despite the inherent cyclical character of very-large-scale capital-intensive industry.
Sustainability, hazard reduction, and protection of health and the environment remain great concerns for the process industries. Many of the raw materials used—especially those derived from oil, gas, and some plants and animals—have been and in some cases continue to be depleted at rates either large compared to known reserves or faster than replenishment. In addition, there is the desire for products, intermediates, solvents, catalysts, and other materials produced or selected for use in chemical manufacture to be as safe and nontoxic as possible during their use and to be recoverable or benignly degradable after their use.
Also, by the very nature of chemical transformations, there are almost always unused chemicals remaining. These chemical leftovers include contaminants in the raw materials, incompletely converted raw materials, unavoidable coproducts, unselective reaction by-products, spent catalysts, and solvents. There have long been efforts to minimize the production of such waste products, and to recover and reuse those that cannot be eliminated. For those that cannot be reused, some different use has been sought, and as a last resort, efforts have been made to safely dispose of whatever remains. The same efforts apply to any leftovers from the production of the energy from the fuels produced or consumed by the processing industries. Of particular immediate and increasing concern are the potential detrimental effects of carbon dioxide emissions to the atmosphere from fossil fuel combustion, as discussed further in Chapters 9 and 10.
The chemical enterprise must meet the societal and environmental challenges it faces. This will necessitate starting from different raw materials, producing new products, using new sources of energy, and paying much greater attention to the creation and disposal of waste. Taking these steps will require innovations in the chemistry used, in catalysts to facilitate this chemistry, in reactors to manufacture the products via this new chemistry, and in separations techniques to purify the resulting products and recover everything else.
The future will likely see greater use of more abundant or renewable raw materials, and greater reuse of materials such as carbon dioxide, salts, tars, and sludges that are currently discarded as waste. Exploiting some of these alternative raw materials and chemistries may involve greater energy input than required for raw materials presently in use, and the source and impact of any such increased energy requirements will need to be carefully considered. Many of these goals are incorporated in the principles of green chemistry.2
Chemistries such as gasification, carboxylation, carbonylation, partial oxidation, and salt splitting may see much greater emphasis in manufacturing. These chemistries will need concurrent development of more selective catalytic and biocatalytic systems and promoters, as well as processes requiring much less ex-
otic materials of construction than we now have available. Demands for greater capital, energy, and material efficiency will require the development of process systems that are more tightly integrated. Such systems will need greater mass and energy recycling, more by-product reuse, and advances in computer-aided, plantwide process control. Only with such advances will it be possible to maintain safety, reliability, flexibility, operability, and economic performance in highly integrated systems.
Some of the new process engineering technologies for manufacturing will involve new devices and the exploitation of new phenomena that will change the traditional way we look at unit operations in chemical engineering. In the area of reaction engineering, new units might include centrifugal or high shear reactors, very short contact time reactors, or rotating packed-bed reactors. In these units, process intensification (see box) is often a major driving force for reducing the size of the device or equipment. Other novel reactor technologies include microwave reactors that have the objective of reducing the energy use and bulk temperature of reaction mixtures by performing selective heating on the particles of the catalyst. Novel separations technologies include membrane absorption and membrane distillation, centrifugal distillation columns, and simulated moving bed adsorbers.
An interesting trend in process intensification is not only the reduction in the size of equipment, but the integration of different functions and phenomena. For instance, microreactors are devices of very small dimensions that have a structure consisting of different layers with micromachined channels (10 to 100 microns in diameter). The layers perform different functions such as mixing, catalytic reaction, heat exchange, and separation. The very high heat-transfer rates (e.g., 20,000 W/m2K) allow exothermic processes to be operated isothermally. Also, very low reaction-volume/surface-area ratios make microreactors attractive for processes involving highly toxic components.
Process intensification also can be achieved by reconfiguring standard equipment. A good example is the use of divided wall columns for distillation, in which various separations that are commonly performed in separate columns are integrated into one single column. This is accomplished through clever rearrangement of trays and divisions between them, yielding significant savings in both capital cost and energy. Another way of achieving process intensification is by expanding the functions in the device. An example is reactive distillation in which hold-up catalyst is added over a subset of trays in a column to perform simultaneous reaction and separation. The methyl acetate column developed by Eastman Chemical (see box) provides an excellent illustration of the dramatic cost reductions that can be achieved with this technology. Specifically, a single reactive distillation column replaced a large complex plant that consisted of one reactor and nine conventional columns. This not only reduced the required capital investment to one-fifth of the original cost, but it also greatly reduced the energy consumption.
Demands for greater capital, energy, and material efficiency will require the development of more integrated process systems involving greater mass and energy recycle, as well as advances in computer-aided process control to maintain safety, reliability, flexibility, and operability in such highly integrated systems. Capital efficiency will also drive the need for greater process intensification as discussed above, making more material more quickly and in less space. New methods will need to be developed exploiting new phenomena to increase the intrinsic rates of mass transfer (for reactors and separations devices), heat transfer (again associated with separations and reactions devices), and reaction kinetics. In addition, the synergistic combination of multiple tasks into single devices such as reactive distillation will become more widespread. Such advances could result in new reactor configurations and new separations and other processing equipment exploiting different phenomena than at present, resulting in smaller, simpler, less energy-demanding processes, and less environmental impact.
|
Emerging Platforms for Process Intensification and Miniaturization Chemical engineers have responded successfully for years to pressures to improve the efficiency and reduce the cost of chemical process operations and to design and construct chemical plants as safely and expeditiously as possible. In this regard, the unit operations concept has long been a useful organizing principle for the synthesis of chemical process flow sheets, while economies of scale and energy efficiency have dictated construction of the very large petrochemical plants that have dominated the scene for decades. However, this situation is hardly static. For instance, hybrid process methods and apparatus for performing chemical operations in a highly integrated fashion are continually being introduced that defy simple classification according to the traditional unit operations scheme—and render somewhat naive the view of the process flow sheet as an orderly network of discrete and well-defined steps. At the same time, the mix of technical, economic, and social factors that drive chemical plant siting, design, and capacity decisions is continually evolving to reflect emerging social issues related to environmental acceptability and public safety, especially where the transportation, storage, and use of hazardous chemicals are concerned. Bigger is not necessarily better, and the future may conceivably witness the construction of smaller, modular, less centralized, and more flexible chemical plants capable of “on-demand” chemical production. Several technological responses to these emerging trends can be gathered under the umbrella of process intensification. This term encompasses a wide range of methods and equipment for performing chemical processing steps more quickly and compactly, thereby increasing the |
|
volumetric productivity and reducing the footprint of the chemical plant. Efforts at process intensification, underway for several decades, have focused primarily on downsizing process equipment and boosting its throughput; static-mixer-based devices, compact heat exchangers, structured packings, catalyst monoliths, and centrifugal contactors have all been used for this purpose. In recent years, considerable energy has also been devoted to the design of multifunctional apparatus capable of conducting several process steps in a synergistic fashion. Such task integration is exemplified by the reactive distillation process for methyl acetate production. 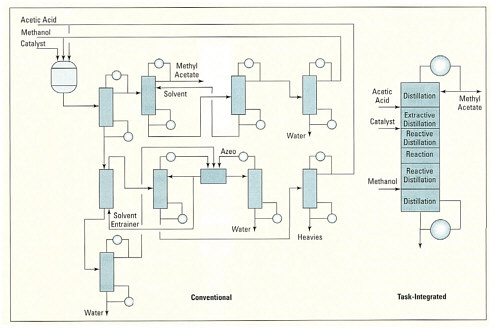 The task-integrated methyl acetate column is much simpler than a conventional plant. Courtesy of Eastman Chemical Company. A family of approaches to achieving high volumetric productivity that is receiving considerable attention in this age of miniaturization involves the use of apparatus with very small flow channels with dimensions of several microns and up. The steep concentration and temperature gradients realized in such equipment encourage rapid heat and mass transfer and promote greater uniformity of reaction conditions. Not only the rate but also the selectivity and yield of chemical conversions can, in principle, be significantly improved. Devices based on both silicon chip and microchannel architectures are currently being considered for performing rapid mixing, heat-exchange, and catalytic reaction in a highly compact and intrinsically safe manner. |
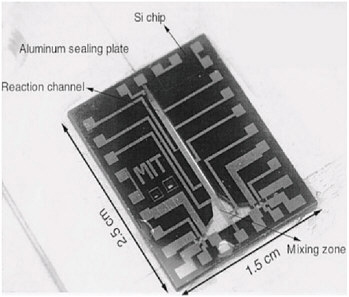 The silicon chip as a miniaturized chemical reactor. Courtesy of Felice Frankel.  Micro heat exchangers based on microchannel architecture. Top left and bottom: Reprinted with permission from W. Ehrfeld et al., Microreactors: New Technology for Modern Chemistry. Wiley-VCH: Weinheim, Germany, 2000. Top right: Courtesy of the Institut fuer Mikrotechnik Mainz, GmbH. Chemically functional membranes afford yet another intriguing platform upon which process-intensified chemistry can be performed. For example, an enzyme membrane reactor process is used to produce a |
|
chiral intermediate for the cardiac drug diltiazem. This membrane system performs a number of tasks in a highly integrated and effective fashion including:
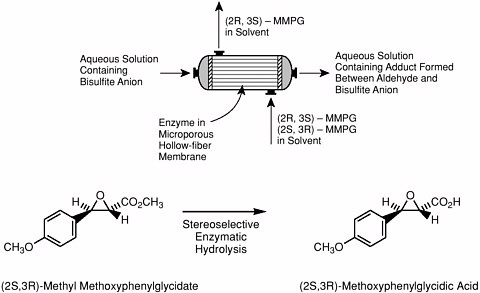 Enzyme membrane reactor for production of diltiazem intermediate. A solution of the racemic ester in organic solvent enters the port at the bottom of the reactor and flows past the strands of microporous, hollow-fiber membrane that contain an enzyme. The enzyme catalyzes hydrolysis of one enantiomer of the ester that undergoes decarboxylation to 4-methoxyphenylacetaldehyde (which in turn forms a water-soluble bisulfite complex that remains in the aqueous phase). The other enantiomer of the ester remains in the aqueous stream that leaves the reactor via the port at the top. Courtesy of Sepracor, Inc. Still other types of chemically functional membranes—particularly adsorptive microfiltration membranes containing affinity ligands or other complexants bound to interior pore wall surfaces—are capable of |
|
dramatically downsizing adsorptive separation equipment. Operation of these so-called “affinity membranes” in a flow-through mode sidesteps the rate-controlling diffusive mass transfer resistances characteristic of particulate adsorbents used in packed beds—and so improves volumetric productivity. Interestingly, nature abounds with examples of process intensification realized with the aid of adsorptive membranes and enzyme membrane reactors. |
WHY ALL THIS IS IMPORTANT
The most distinctive aspect of the chemical sciences and engineering is the ability to create new molecules and chemical systems—from minute to commodity quantities—without being limited to the study of those that already exist in nature. The expansion of our chemical universe through synthesis leads to great new scientific insights. Moreover, chemical synthesis is the activity that has made chemistry and chemical engineering so vital to human health and welfare. The opportunities and challenges for the future are unlimited. The promise of better medicines and better materials depends on the ability of synthetic chemists to create new transformations and to use them in the creation and manufacture of new substances. It is no surprise that synthesis is still the active concern of a large fraction of practicing chemists, and will remain so.
The development and implementation of techniques will be critical for all aspects of chemical manufacturing—from synthesis and analysis to optimization, evaluation, design, control, supply-chain management scheduling, and operation of chemical process systems. This will need to be done in a way that is consistent with societal and economic objectives and constraints. And it will be critical no matter what the chemistry or biochemistry, whether isolated or in supply-chain networks, dedicated or general purpose, batch or continuous, microscale or macroscale. These constraints will require the further creation and exploitation of a science base that includes novel representations of the underlying chemical and physical phenomena. Similarly, it will demand computationally efficient formulation and solution methods, as well as the expansion and incorporation of advanced process systems engineering expertise and judgment.



















