3
Background on Introduced Species
BRIEF OVERVIEW
Introduced species are organisms that have been intentionally or accidentally transported by human activities into regions where they were not previously found (i.e., nonindigenous; Carlton, 2001). While humans have been transporting marine species for hundreds of years, it has been exceedingly difficult to predict whether a species will become “invasive” (spread from the site of introduction and become abundant; Bergelson, 1994; Crawley, 1989; Kolar and Lodge; 2001; Lohrer et al., 2000; see Appendix J). Most nonnative species do not survive where they are introduced, and few become pests after they are transported to new environments. It has been estimated that about equal numbers of deliberate and inadvertent introductions to the United States of terrestrial vertebrates, fish, and molluscs have turned out to be harmful (U.S. Congress, 1993). Others have suggested that approximately 10% of nonnative species will become “invasive” (Williamson and Fitter, 1996). Low invasiveness is generally explained by a lack of favorable environmental conditions or by the lack of an “open” niche for the nonnative in a new habitat or region (Lohrer et al., 2000). Attempts have been made to predict life history traits of species that will be successful invaders, such as the number and size of propagules (progeny at early life history stages), age at first reproduction, and organism growth rate. Some studies have found correlations between life history traits and invasion success (e.g., O’Connor, 1986; Rejmanek and Richardson, 1996; Williamson and Fitter, 1996) and others have suggested that attributes of the invaded community (i.e., disturbance levels,
species diversity) affect its ability to resist nonindigenous species invasion (e.g., Hobbs, 1989; Case, 1990; Tilman, 1997; Stachowicz et al., 1999, 2002a). Often the most reliable predictor is the invasiveness of a species in situations with similar environmental characteristics (Forcella et al., 1986; Gordon and Thomas, 1997; Reichard and Hamilton, 1997).
Recent work has shown that relatively simple approaches may be used to address why some introduced species rapidly spread and flourish while others do not. Kolar and Lodge (2002) studied a number of nonindigenous fish species introduced into the Laurentian Great Lakes and found that different life history traits were important during different phases of the invasion process. Species that grew relatively quickly and had a wide range of temperature and salinity tolerances tended to be most successful during the establishment phase of the introduction. In contrast, species that rapidly spread only after becoming established typically had slower growth rates and a more limited range of temperature tolerance. Those species that eventually were categorized as “invasive” generally had wider salinity tolerances and were capable of surviving lower water temperatures than noninvasive species. These results demonstrate the importance of assessing stage-specific processes and patterns of successful and nonsuccessful invasions. Although the outcomes are habitat specific, the overall approach for predicting invasion success could be applicable to marine invaders. For example, a comparison of the survival patterns of different species as they are transported along a coastline provides useful information on the environmental tolerances of those species—information that is essential in studying the early phases of the invasion process.
Ecosystems that have reduced biodiversity or are under stress from environmental degradation and climate change appear to be more vulnerable to invasions. For example, Stachowicz et al. (2002b) demonstrated that enhanced species diversity directly increases the resistance of subtidal fouling assemblages to invasion and that surveys in a number of coastal habitats in southern New England also revealed an inverse correlation between resident species richness and the number of nonnative species in those habitats. Climate change has its greatest impact where invasive species occur at the southern and northern boundaries of ecosystems (see Root et al., 2003, for a recent meta-analysis). Temperate coastal regions appear to be particularly prone to invasion by nonnative species due to the effect of climate change on invasion by nonnative species. Stachowicz et al. (2002b) have correlated a doubling in the abundance of invasive ascidians in eastern Long Island Sound with a significant increase in seawater temperatures over the past two decades in that region. Sauriau (1991) correlated small increases in seawater temperatures with the spread of a gastropod, Cyclope neritea, initially introduced with shipments of the
Pacific oyster, Crassostrea gigas, along the Atlantic coasts of Portugal, Spain, and France. Several studies have reported the northward spread of the oyster parasite Perkinsus marinus (Dermo disease agent) along the U.S. East Coast in relation to the trend of increasing winter seawater temperatures during the past several decades (Ford, 1996; Cook et al., 1998).
Every major marine, freshwater, and terrestrial ecosystem in the United States contains introduced species, and in some ecosystems it is difficult to find a native species (e.g., Cohen and Carlton, 1998). Nonnative species typically make up a large fraction of the biota of marine and estuarine habitats, largely because of the use of these ecosystems for commerce, fishing, and recreation. Introduced species in coastal habitats arrived via several unintentional human-mediated mechanisms ranging from transport on hulls or in the ballast water of ships to intentional introductions for aquaculture, fisheries, and releases from pet and live seafood trades (e.g., Carlton, 2001).
The human-mediated movement of marine species within and between ocean basins as food items or for aquaculture purposes has occurred for millennia (Carlton, 1999a). For example, there is evidence that Vikings transported the soft-shell clam (Mya arenaria) to Europe in the 13th to 14th century (Carlton, 1999b), and the Japanese oyster (C. angulata) was brought to southern Europe in the 1500s by Portuguese explorers (Edwards, 1976). The transport of marine species for aquaculture purposes became far more pronounced during the 19th and 20th centuries and today aquaculture has become one of the fastest-growing components of the world’s food economy (Naylor et al., 2001). For example, the farming of marine molluscs (e.g., oysters, clams, mussels) is an important industry. In 2000, U.S. molluscan aquaculture was worth more than $75 million (National Marine Fisheries Service, 2002).
The intentional introductions of commercial marine species into coastal areas typically are to augment or replace depleted natural stocks or to diversify the number of species used in aquaculture operations. While the ecological impacts of molluscan aquaculture are relatively small in comparison to other types of seafood farming (Naylor et al., 2000), historically the activity has resulted in the introduction of many nonnative species throughout the world. Oysters have been intentionally transported more often than probably any other marine species (Sindermann, 1990). A wide array of plants and animal species has “hitchhiked” with introduced shellfish. Cohen and Carlton (1995) estimate that about 20% of the introduced species found in San Francisco Bay were the result of shipments of the Eastern and Japanese oysters for aquaculture purposes. The pace of shellfish introductions has greatly diminished with the switch to hatchery-propagated seed on the U.S. West Coast, which permits the application of International Council for the Exploration of the Sea (ICES)
protocols that greatly reduce the risk of cointroductions. Other researchers have estimated that the use of introduced species for fisheries activities is the second leading vector (the first is shipping) for the transport of nonnative species throughout the world (Naylor et al., 2001).
In some cases the introduced species have become significant competitors with native oysters. For example, there is evidence that the Japanese oyster, C. gigas, has outcompeted native oyster species in New Zealand (Dinamani, 1991a, b), Australia (Ayres, 1991) and France (Goulletquer and Héral, 1991). Some of the species transported with oyster shipments have become important predators and competitors of both cultured and wild molluscan stocks as well as other native species. For example, in the early to mid-1900s, transfers of C. virginica from the U.S. East Coast to the U.S. West Coast resulted in the introduction of oyster competitors (e.g., the slipper shell Crepidula fornicata) and predators (e.g., the oyster drill Urosalpinx cinerea) in those waters. Large-scale transfers of C. gigas to the U.S. west coast, British Columbia, Canada, and Western Europe in the early- to mid-1900s resulted in the establishment of a number of oyster predators (Barber, 1997). In addition to affecting economically important species, the invaders can pose significant threats to biodiversity, directly or indirectly alter local community composition, influence the performance of ecosystems, and cause significant economic impacts (e.g., Grosholz et al., 2000; Stachowicz et al., 1999, 2002a). A National Research Council (1995) report ranked invasive species and overexploitation of fisheries stocks as the most important threats to marine biodiversity. Considerable information cataloging and assessing the impacts of introduced species in coastal waters and reviews can be found in Carlton (1985, 1987, 1989), Grosholz (2002), and Ruiz et al. (1997, 1999, 2000).
Notwithstanding the positive impact that introductions of bivalves have had on economic development of local fisheries and aquaculture, there are no documented cases where an intentional introduction of an oyster species has resulted in an overall positive ecological impact on a U.S. coastal ecosystem. However, studies conducted in France have demonstrated that under intensive aquaculture conditions very high densities of the introduced oyster C. gigas have resulted in transient ecosystem impacts in several coastal embayments and lagoons by temporarily cropping primary production and increasing nutrient recycling rates (e.g., Bacher et al., 1997a, 1997b; Gangnery et al., 2001). In contrast, unintentional introductions of some species of suspension-feeding bivalves into U.S. waters have had pronounced positive and negative ecological impacts on the aquatic ecosystem. For example, millions of dollars are spent annually by Great Lakes cites and industries to unclog water intake pipes overgrown with the nonnative zebra mussel (Dreissena polymorpha), and
this invader has displaced some native species in certain areas. However, rapid population expansion of zebra mussels also has increased Lake Erie water quality as much as six-fold and has reduced some types of phytoplankton. Phelps (1994) reports that the invasion and subsequent population explosion of the nonnative Asian clam, Corbicula fluminea, in the late 1970s in the Potomac River estuary was likely responsible for enhanced water clarity and increased abundance and diversity of submerged aquatic vegetation and aquatic birds in the mid-1980s. In California, where Corbicula has become the most widespread and abundant freshwater bivalve, the species is regarded as an economic pest of industrial and municipal water delivery systems (Cohen and Carlton, 1995). In portions of San Francisco Bay, the filtration activities of several different species of nonnative bivalves have resulted in dramatically reduced phytoplankton abundance (Alpine and Cloern, 1992; Cloern, 1996) and may have altered the pelagic trophic structure in parts of the bay (Kimmerer et al., 1994). However, high abundance of the nonnative species has also greatly altered the diversity and biodiversity of the benthic communities (Nichols et al., 1990), and the presence of one species, Potamocorbula amurensis, has increased the bioavailablity and trophic transfer of the toxic metal selenium in certain parts of the bay ecosystem (Linville et al., 2002).
The remainder of this chapter reviews case studies of shellfish introductions in France, Australia, New Zealand, and the North American Pacific and Atlantic coasts. In many of these cases the introduction of a nonnative shellfish species was intentional, in response to the severe depletion of native stocks and fisheries. The variety of consequences observed in these case studies, even those employing the same species in different localities or environments, demonstrates the uncertainty of predicting ecological outcomes of shellfish introductions.
CASE STUDIES OF SHELLFISH INTRODUCTIONS
France
Commercial oyster landings by French oyster farmers currently rank fourth in the world, behind China, Japan, and Korea with 150,000 metric tons harvested per year. The landings are valued at $280 million and represent 25% of the total gross value of French seafood production. Landings reached these levels in the early 1990s and have remained stable. Further expansion is limited by the availability of leasing grounds. Two species are cultivated in France: the endemic native flat oyster, Ostrea edulis, and the introduced Pacific cupped oyster, C. gigas (Goulletquer and Héral, 1997). Culture of the flat oyster has been drastically affected by diseases, with production collapsing to less than 1% of past harvests
(1,500 metric tons a year). The overall French production is stabilized and supplies almost all the yearly consumption. Therefore, exports are rather limited (less than 3%), as are imports from European countries. Presently, the oyster industry is valued at 500 million euros, with more than 4,000 companies and 20,000 direct employees and occupying more than 50,000 acres of state leasing grounds and 10,000 acres of private oyster ponds. Because oyster culture is concentrated in bays and estuaries, usually in rural areas, it has played a critical role in shaping the landscape and stabilizing populations in otherwise economically depressed areas. The oyster industry plays a critical role in coastal zone management and employs environmentally sound practices in wetlands areas. Ninety percent of the production is based on the natural, reliable spatfall that occurs yearly in the southwest part of France. Abnormally low recruitment was observed in only four of the past 30 years. Spat is collected by carefully timed deployment of various types of spat collectors when larval density peaks during the spawning season (July-August). Although their market share is increasing, at present the five private hatcheries only produce about 10% of the total seed (triploids and selected strains) required at the national level to sustain production.
Over the past century the oyster industry experienced major upheavals with C. gigas being only the most recent of three species cultured by farmers (see Figure 3-1). Based on past problems, reliance on a single species creates a structural weakness in the industry. This has prompted research to anticipate future problems and assess options for diversifying the culture stock.
Historical Background
Demand for oysters increased significantly during the 19th century, leading to overfishing. Natural flat oyster beds distributed along the French coastline were physically destroyed and overfished, prompting state managers to establish and enforce new regulations based on quotas and seasonal openings. A commercial trade was initiated with Portugal to market the oyster C. angulata to meet increasing demand. Beginning in 1860, spat were imported to compensate for the lack of flat oyster spat collected on overfished beds. The first natural spatfall of C. angulata occurred in the Gironde estuary after a boatload of Portuguese oysters was jettisoned overboard during a storm. Because some of the oysters were still alive and environmental conditions were highly favorable, this species became established in the area. Later, natural reproduction facilitated invasion of this nonnative oyster from the southwest of the country north to the Loire estuary. At that time a large debate among farmers led to a temporary halt in transfers of oysters into northern Brittany waters. Even-
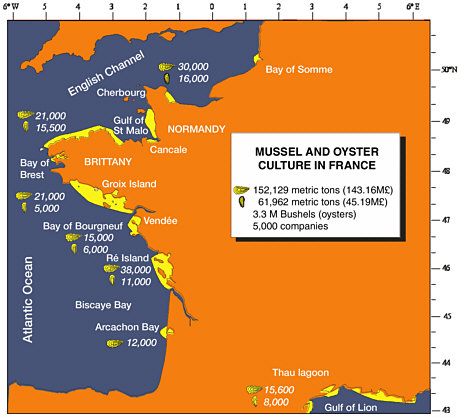
FIGURE 3.1 Mussel and oyster culture in France.
SOURCE: Modified from Goulletquer and Héral (1997).
tually, the Portuguese oysters were cultured outside their natural reproductive range in northern beds (e.g., Cancale) by transplanting seed. After 1920, production increased significantly when spat collectors began to be used systematically. C. angulata and O. edulis production reached 85,000 and 28,000 metric tons, respectively, in 1960, despite a major disease outbreak affecting the flat oyster in 1920 and low C. angulata recruitment in the 1930s. C. angulata replaced the native species in the main French rearing areas except Brittany, where oyster farmers were most successful in obtaining spat settlement from C. angulata at the same time they were having difficulty obtaining sets of the flat oyster. Although the disease agent was never identified, the massive die-off of flat oysters affected the main European populations simultaneously (Orton, 1924) and therefore has been considered the first oyster epizootic event in Europe.
In the late 1970s, two major disease-causing organisms, Marteilia refringens and Bonamia ostreae, spread and drastically reduced production
of O. edulis in almost all rearing areas in France, as well as in Europe. Despite new management practices and an intensive repletion program, O. edulis production remains low 30 years later, with no anticipation of a rebound in the foreseeable future. The present approach to rebuilding the population is introduction of a disease-tolerant strain from an ongoing mass selection research program. The strain has been selected for more than 20 years and shows significantly higher survival and growth rates compared to natural populations. However, several questions remain to be addressed, including the potential genetic consequences of using a limited brood stock to restore wild populations. It should be noted that the parasite B. ostreae is an exotic species accidentally introduced to Brittany with a small batch of flat oysters originating from California (Grizel, 1997; Cigarria and Elston, 1997).
Concomitant with the increase in C. angulata production through the 1960s, abnormal mortality and reduced growth rates were both reported in the main rearing areas (Marennes-Oléron Bay and Arcachon Bay). This resulted from overstocking such that the carrying capacity of the ecosystem was exceeded, a consequence of poor management. From 1966 to 1969 the first outbreak of “gill disease” affected already-weakened oyster populations, leading to a decline in C. angulata production. Drastic mortalities from a second viral disease occurred between 1970 and 1973 and caused the total and rapid disappearance of this species from the French coast. Thirty years later, attempts to reintroduce C. angulata are still unsuccessful due to high mortality rates. The C. angulata collapse led to a major economic crisis, affecting 5,000 oyster farmers and generating a $90 million yearly loss. Therefore, an urgent solution was sought to prevent unemployment and sustain the ailing shellfish industry. A massive introduction of C. gigas originating from Japan and British Columbia was organized to provide spat to the industry and brood stock to establish new reefs and sanctuaries along the French Atlantic coastline.
Introduction of the Japanese Oyster C. gigas
The Pacific oyster, C. gigas, is native to Japan, far-eastern Russia, Korea, Taiwan, and China and has been introduced to all continents except Antarctica (Mann, 1979). Spat of C. gigas were first imported from Japan in 1966 by an oysterman looking for growth rate improvements at a time when C. angulata production was declining significantly because of overstocking. The nonofficial aspects of these imports and the concomitant increased mortality of C. angulata prompted the state agencies to ban further imports and to assume control of the issue. Scientists from Institut français de recherche pour l’exploitation de la mer (IFREMER) were in charge of developing the expertise to (1) evaluate the quality of oyster
beds in Japan with regard to zoosanitary and sanitary status, fouling organisms, and oyster quality and (2) study how an introduction could be done to revitalize the industry. Experts concluded that C. gigas appeared healthy, and no relationship was established between the pathology observed in France, the mortality of C. angulata, and the status of oysters in Japan. Moreover, the environmental conditions sustaining C. gigas populations in Japan were considered similar enough to those on the French oyster beds to expect natural colonization after the introduction. Eventually, the introduction of C. gigas was approved because the whole industry was collapsing. The introduction was performed through large imports and massive release into the environment in two stages aimed at first building sanctuaries to establish brood stock and then supplying the oystermen with spat.
Sanctuaries—“RESUR” (meaning “resurrection”)
After evaluating the sanitary quality of C. gigas beds in British Columbia, adult oysters were imported to constitute brood stocks. Histological controls were used for each transfer to ensure oyster zoosanitary status. This operation lasted from 1971 to 1973. The last additional batch was introduced in 1975 in the Marennes Oléron Bay. The sites were free from dying C. angulata populations, and environmental conditions were highly favorable (large unused carrying capacity, available habitat) resulting in successful establishment of populations in three areas, all located south of La Rochelle. A large natural spatfall following the introduction resulted from high larval survival rates under favorable temperature and salinity conditions. Presently, the sanctuaries are not maintained because the rearing and wild populations are sufficient to sustain spat supply. In contrast, the carrying capacity is now limiting, prompting the state managers to reduce those sanctuaries.
Spat Supply
From 1971 to 1977, spat was imported from Japan to sustain aquaculture. Each imported batch of spat was inspected and certified for origin, health status, and presence of predators. For disease, histological analyses were performed and an index of meat quality was established. Each batch was immersed in fresh water to reduce the risk of importing species such as the flatworm Pseudostylochus (Table 3.1).
The brood stock and spat that generated the present population may be more precisely estimated to be 456 metric tons of brood stock and 3,394 million spat. It should be emphasized that the latter represents 7 years of French hatchery production (500 million spat/year). Therefore, if a simi-
TABLE 3.1 Quantities of Spat and Brood Stock Imported During the Massive Introduction Effort in France from 1971 to 1976
|
C. gigas stage |
1971 |
1972 |
1973 |
1974 |
1975 |
1976 |
TOTAL |
|
Adult (metrictons) |
117.5 |
236.5 |
173 |
0 |
35 |
0 |
562 |
|
Spat (estimated in millions) |
1,226 |
2,413 |
365 |
965 |
34 |
5 |
5,008 |
|
SOURCE: Modified from Maurin and LeDantec (1979). |
|||||||
lar introduction were to be done again, hatchery production would technically be able to sustain such an operation under full compliance with the ICES protocol.
Reasons for the Success of the Introduction
The extensive introduction of C. gigas was so successful that production exceeded the record for C. angulata in less than 10 years. There are several reasons for the rapid increase in production. C. gigas has the biological advantage of resistance to the disease-causing organisms affecting C. angulata (viruses), O. edulis (parasites B. ostreae, M. refringens), and clams (Perkinsus spp.). Environmental conditions favored C. gigas expansion to such an extent that the natural spat supply quickly became sufficient for the entire industry. The ecological cost of this introduction has been limited because few areas were colonized outside the traditional rearing grounds. As early as 1975, spat originating from the sanctuaries and cultured populations were sufficient to sustain the industry. Yearly production is now estimated to be around 15 trillion spat in both Marennes-Oléron Bay and Arcachon Bay. No competition occurs between the Portuguese oyster, C. angulata, and C. gigas or with other species. Although hybridization has been observed experimentally, it was not a problem because of the very low brood stock of C. angulata and the differences in spawning pattern. Where oyster habitat was fully available and carrying capacity was high, optimal C. gigas growth was obtained (>70 g in 18 months). No inbreeding problem has been observed despite the relatively limited number of individuals contributing to the population. At least four invertebrate and three algal species were accidentally introduced during the massive operations even with stringent management. The introduced species have limited biomass and distribution and have negligible impact compared to other invasive species such as the limpet C. fornicata or species brought in ballast waters (Goulletquer et al., 2002). Similarly, Haplosporidium sp. was likely introduced with C. gigas as the
healthy carrier, but it has remained at low prevalence and does not harm the current shellfish populations. It is unlikely that Marteilia spp. were introduced with C. gigas as once suggested, based on new genetic information on M. maurini and M. refringens as well as the worldwide status of the genus Marteilia (Berthe et al., 2000; Berthe, 2002). After 30 years of C. gigas production, the main management question is how to optimize stocking biomass with available carrying capacity and avoid overstocking effects. In specific areas, overstocking led to decreased growth and increased mortality rates. Where natural spatfall occurs (southwest coastline), management practices have been developed to limit expansion of natural beds that can compete for food with farmed populations. Sanctuaries are no longer in use because the farmed population is sufficient to sustain yearly recruitment.
Human Component
Economic pressure to carry out the species introduction was very high at that time and would be similarly high if the situation were to happen again. A critical step for the success of the introduction was that the government assumed control rather than allow unregulated importation of oysters by farmers. The massive introduction required a defined schedule and preliminary scientific investigations to maximize benefits and limit associated risks. Because C. angulata and C. gigas share many similarities, the existing aquaculture practices were directly applicable to C. gigas without major changes. Scientific advice to optimize recruitment (e.g., timing for spat collector deployment) contributed to the success of the introduction. The new oyster benefited from an overall acceptance by consumers, as in previous disease outbreaks when a new species was substituted. Oyster consumption has increased with production and a slight decrease in price.
Shellfish Introduction into French and European Waters: The Case of Manila Clam
During the 1970s, the shellfish industry was looking to diversify and carried out experimental trials using the Manila clam, Ruditapes philippinarum. Rather than a massive old-fashioned introduction, the first clams (150 individuals from Seattle) were obtained from hatchery production to reduce the risk of introducing disease and exotic species. The Manila clam showed larger growth rates than the native species R. decussatus, a high value, and a high acceptance by consumers. Because no natural spat were available, hatchery spat and scientific rearing techniques were used to increase production under a variety of environmen-
tal conditions. In 1985, clam culture practices using diploids were fully operational and production reached hundreds of metric tons in several European countries. In 1986, abnormal shell calcification affected several intertidal clam-rearing areas, causing abnormal mortality rates (Goulletquer et al., 1989). The Brown ring disease was later attributed to a vibrio infection (Vibrio tapetis; Paillard et al., 1989). The origin of Brown ring disease is unknown, but large international transfers of clams may have facilitated spread of the disease. Although no cases of the disease were observed in ponds, most of the intertidal clam culture areas were impacted, drastically reducing the landings. It should be noted that the endemic species R. decussatus has not been affected by V. tapetis. However, the main factor affecting clam culture was the species’ invasive pattern. R. philippinarum extensively colonized natural beds in several areas in Europe, leading to an extensive public fishery (65,000 metric tons in 2000) that overtook aquaculture production. Since complete geographic overlap does not occur between the native and exotic clams, both populations are exploited. However, hybridization may occur in specific geographic areas.
Summary
-
French oyster production (O. edulis and C. angulata) suffered from overfishing and five disease events during the 20th century that led to the decision to introduce two exotic species (C. angulata and C. gigas).
-
Scientific and management efforts to reverse the declines of indigenous species were uniformly unsuccessful.
-
The introduction of exotics facilitated the survival and expansion of the industry, generating employment and economic benefits.
-
Ecological impacts from those introductions have been limited, although the ICES protocols were not followed (ICES, 1994).
-
The French shellfish research effort is now focused on disease and its prevention, shellfish ecosystem management to optimize stocking biomass, and genetic studies to diversify and develop selected (and disease-resistant) strains to avoid future population declines (Figure 3.2).
Australia
In Australia shellfish have been commercially harvested since the early 1800s (Biosecurity Australia, 2002). Present production is mainly from aquaculture. Recent research on disease-resistant selected strains and new varieties has promoted expansion of hatchery products. The
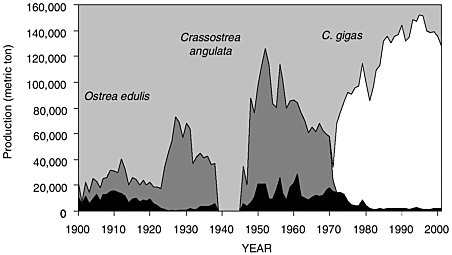
FIGURE 3.2 French oyster landings
SOURCE: Modified from Goulletquer and Héral (1997).
edible oyster species currently grown in Australia include the Sydney Rock oyster, Saccostrea glomerata (S. commercialis); the Pacific cupped oyster, C. gigas; and the flat oyster, O. angasi. The Sydney rock oyster is a native species cultured in estuarine areas and rivers, as well as embayments along the coast of New South Wales and Southern Queensland. Since 1988, landings of S. glomerata have declined from 7,919 metric tons to 5,000 metric tons in 1997. The seasonal occurrence of the parasite QX (M. sydneyi) has reduced the tidal areas where the Sydney rock oyster can be grown. Most of the production relies on natural spatfall and is located in New South Wales with 4,300 hectares of leasing grounds distributed among 41 estuaries. More recently, QX disease has shown a dramatic increase. Tests carried out in 2002 demonstrated that QX has doubled its range, affecting more than 10 major estuaries in New South Wales. Over $1 million has been spent in the past 10 years to come up with disease-resistant strains and fast-growing oysters.
The Pacific cupped oyster (C. gigas) is exotic to Australia. The first recorded importation in 1940 was seized and destroyed on the recommendation of the New South Wales state fisheries (Ayres, 1991). However, consignments were imported in 1947, 1948, and 1952 and planted in South and Western Australia and Tasmania. Only oysters planted in Northern Tasmania became established and were spread by larval transport. In 1955, stocks were moved to Victoria and South Australia for aquaculture. Those stocks survived and spread progressively northward to New South Wales and became established. In Tasmania the success of hatchery pro-
duction led to a decline of the original local industry based on the flat oyster, O. angasi. The Tasmanian industry now relies entirely on hatchery production of C. gigas and represents 70% of total Australian production.
In New South Wales, Pacific oysters were first found in 1967, and to a greater extent in 1973. The Fisheries Department acted to eradicate the species. However, in 1985 significant numbers of C. gigas were found in Port Stephens, the main S. glomerata oyster-rearing area, likely resulting from an intentional introduction effort. Since then C. gigas has spread rapidly, and all efforts to eradicate the species have been unsuccessful. Although considered a marine pest in this traditional rearing area, the C. gigas landings have increased, from 1,516 metric tons in 1988 to 6,000 metric tons in 1997. Joint cultivation of S. glomerata with C. gigas is unworkable because C. gigas overgrows the native rock oyster.
Australian oyster production has increased (10,574 metric tons during the 1998-1999 season), although significant decreases in Sydney rock oyster landings have occurred, mainly due to diseases losses and reduced spat settlement due to C. gigas spat overset.
New Zealand
Exploitation of rock oyster S. glomerata beds under government control began in 1877 in the northern part of the North Island based on a licensing system (Dinamani, 1991b). Although several attempts were made to develop oyster culture at the turn of the 20th century, the results were disappointing. Between 1908 and 1960, government efforts were focused on maximizing spat settlement surface for recruitment and on managing natural oyster beds, including removing predators, to sustain oyster development. In 1964, the Rock Oyster Farming Act was passed for the purpose of establishing farms on leased areas. It was followed in 1971, by the Marine Farming Act to facilitate the culture of any marine species under a leasing and licensing system. Natural spatfall was irregular, and spat collector deployment suffered from space competition with other species. Although rock oyster production reached more than 500 metric tons in the mid-1970s, it began to be overwhelmed by the Pacific oyster, C. gigas, which was first found in 1971 (Dinamini, 1991a). Irregular spatfall during the 1970s prompted farmers to consider hatchery production. When C. gigas was introduced to New Zealand, there were some concerns about its impacts on the native species. C. gigas colonized much larger areas than the endemic species, especially in northern New Zealand where it outgrew S. glomerata on oyster leasing grounds (Smith et al., 1986). The C. gigas introduction displaced the active fishery based on the native species. By 1978, rock oyster production was eliminated and the total present oyster landings reached 3,500 metric tons in 2001 (Hay, 1999). No disease
has been associated directly with this introduction, but it is having an impact on the coastal ecology; the native species has become so rare as a result of the spread of C. gigas that harvest of the native oyster is prohibited in some places. There are reports that the rock oyster can be found higher in the exposed intertidal zone than C. gigas (Hay, 1999), providing some refuge for the native species.
Summary
Positive economic benefits have been realized from introduction of the Pacific oyster into several areas of Australia and New Zealand, but there have been negative impacts on the native oyster S. glomerata. The fishery for the native oyster has declined or been closed in parts of New South Wales and New Zealand where C. gigas is considered a marine pest.
Western North America
The oyster industry and resource management agencies in the western United States and Canada have introduced a variety of nonnative shellfish species over the past 134 years. A review of these introductions provides insight for evaluating both the negative and positive consequences associated with the proposed introduction of C. ariakensis to the Chesapeake Bay. In addition, it provides an example of a shellfish industry largely dependent on hatchery-produced seed that is relevant to the development of aquaculture in the Chesapeake Bay.
Historical background
Native Oyster Beds Depleted by Fishery
The oyster industry on the West Coast of North America started with harvest of natural populations of the native oyster, Ostreola conchaphila. Native Americans harvested them where large concentrations were found. European settlers gathered native oysters for food and sale (Steele, 1957). Commercial exploitation began in earnest to supply hungry (and wealthy) gold miners during the California Gold Rush following the discovery of gold at Sutter’s Mill in 1848. Beds of native oysters in San Francisco Bay were depleted within 3-years of the Sutter’s Mill strike (Gordon et al., 2001; Shaw 1997; Barrett, 1963).
The range of O. conchaphila extends from Baja California to Sitka, Alaska, but the most abundant resources were discovered in Shoal-Water Bay (today referred to as Willapa Bay) in the Washington territory. A local settler in Shoal-Water Bay, Captain Charles J. W. Russel, reportedly
traveled to San Francisco with several sacks of oysters in 1851. This marked the beginning of 20-plus years of a lucrative fishery, where oyster baskets costing $1 at the source could command as much as $30 in San Francisco’s wholesale markets. Oysters were shipped by schooner, with one captain making 74 trips between 1875 and 1880 (Gordon et al., 2001). As many as 200,000 bushels per year were shipped from Shoal-Water Bay to the San Francisco markets (Scholz et al., 1984). The Shoal-Water Bay resource was depleted by the end of the 1870s, with little effort directed at conservation or enhancement.
Washington State Oyster and Tideland Laws
Washington became a state in 1890, and some of the first laws passed by the legislature were related to oysters. On March 26, 1890, a law was passed that provided for the appraisal and disposal of state tide and shorelands. It provided for planting of natural oyster beds, declaring them “natural oyster beds reserved.” The beds were not to be sold or leased and were intended to provide seed for oyster farmers and stock for wild fishermen. Outside these beds, individuals could purchase up to 80 acres if developed for planting oysters. In 1903 another law was passed to “care for, protect, reseed, and replant the oyster reserves.”
In 1895 two other oyster-related laws were passed by the Washington state legislature that allowed the state to dominate U.S. West Coast shellfish production. The laws, known as the Bush Act and the Callow Act, provided for the sale of tidelands into private ownership, specifically for the purpose of culturing oysters. The legislature recognized that an oyster cultivation industry could be encouraged to replenish the depleted wild stocks of native oysters through provisions for private ownership of the tidelands and shellfish grown there. The land returned to the state if the owner used the property for purposes other than oyster cultivation. In 1919 the Clam Act was passed to allow cultivation of clams and other edible shellfish on privately owned tidelands. The practice of selling state tidelands for shellfish culture stopped in the 1970s; however, land already purchased remained in private ownership. Because Washington’s shellfish growers held clear title to the tidelands, they were able to obtain working capital through bank loans that were used to acquire more land that was then leveled and diked. With dikes, seawater remained over the oysters at low tide and served as a buffer against lethal temperature extremes.
After passage of the Bush and Callow Acts in 1895, the Washington oyster industry began to aggressively culture the native oyster. Efforts were particularly successful in southern Puget Sound, where more than 1,000 acres of tidelands were leveled and diked for native oyster culture (Lindsay and Simons, 1997).
Another consequence associated with private ownership of the tidelands is that Washington state shellfish growers have become aggressive defenders of water quality in the watersheds in which they farm. Sulfite waste liquor from a South Sound pulp mill, though never conclusively determined to be the cause, likely hastened the demise of the native oyster culture industry and stimulated the transition to the hardier Pacific oyster. Oyster growers brought suit against the pulp mills in the 1930s and 1940s, and litigation over water quality issues continues today with regard to nonpoint pollution sources.
Shellfish growers in other West Coast states rely on being able to lease tidelands or bedlands from their respective states. Washington state growers also lease tidelands from the state; however, the bulk of the production is from privately owned lands. In considering restoration of the oyster resources in the Chesapeake Bay, changing laws to allow private ownership may not be practical. However, long-term leasing for aquaculture could have a similar stimulatory effect on restoration efforts as the Bush and Callow Acts had in Washington state.
California C. virginica
Northern California oystermen made the first nonnative oyster introduction to the West Coast. Jealous that their Washington territory counterparts had taken over the San Francisco oyster markets and aided by the completion of the Union-Central Pacific transcontinental railroad, Californians brought the Eastern oyster, C. virginica, to San Francisco Bay. The October 22, 1869, issue of the San Francisco tabloid Alta California announced the arrival of the first shipment.
Early records of C. virginica imports to San Francisco Bay are sketchy and difficult to interpret since they are recorded under different measures, including pounds, gallons, bushels, barrels, shucked meats, and shells. An 1875 issue of the Manufacturer and Builder observed: “Every year some 500 carloads of small oysters are transported across the continent, to be brought up in the Pacific.” The article further noted, “There appears to be a limit to the growth of any kind of Eastern oyster in Pacific waters; after a certain period, a year and half at the utmost, for some reason as yet not well understood—perhaps the meat becoming too large for the shell planted—the oyster dies.”
Between 1887 and 1900, 33,480 bushels of Eastern oysters were shipped annually, constituting 80% of the total oyster production from San Francisco Bay during these years (Gordon et al., 2001). The availability of more popular Eastern oysters in San Francisco markets severely depressed the value of local oysters shipped from Washington. Prior to completion of the transcontinental railway, San Francisco buyers were
paying $16 per sack of 1,000 native oysters. By the mid-1870s that price was down to $4 per sack and by the 1880s it was down to $2.50 per sack or about half the price being paid for Eastern oysters (Gordon et al., 2001).
From 1872 until the early 1900s the Eastern oyster dominated West Coast oyster production, peaking in 1899 with an estimated 1,134 metric tons of shucked oyster meat (Conte et al., 1996). Urbanization of the San Francisco Bay area degraded water quality, and by 1908 Eastern oyster production had fallen 50%. The industry stopped importing Eastern oyster seed in 1921, and the last of the San Francisco oysters were commercially harvested in 1939 (Conte et al., 1996).
Washington C. virginica
Washington oystermen who wanted to participate in the Eastern oyster business had to wait for a northern spur of the transcontinental railroad. After an unsuccessful planting of 80 barrels in 1895, the state fish commissioner’s staff planted an assortment of C. virginica from Connecticut, the Chesapeake Bay, New York’s East River, Massachusetts, and Maryland’s Princess Bay with a $7,500 appropriation from the Washington state legislature in 1899. With proper handling and planting in appropriate locations in Shoal-Water Bay, the oysters thrived.
Within a decade Washington’s C. virginica industry employed several hundred people and was valued at around $1 million. Production climbed from 4,000 gallons in 1902 to 20,000 gallons in 1908. As a 1906 issue of the Willapa Harbor Pilot said: “Many will remember and smile at the great interest, not to say excitement, manifested over the shipment of 80 barrels of eastern oysters to this bay in 1895, while now the importation of from 50 to 100 carloads annually excites little comment” (Gordon et al., 2001).
Transplantation of Eastern oysters to the West Coast in the late 1800s and early 1900s marked a significant transition in the industry from an extractive to a culture-based fishery. Washington’s production of C. virginica continued to grow until 1919 when the stock collapsed due to an unknown cause. Some speculate that a seed shortage in 1913 coupled with a large harvest in 1918 led to the collapse (Gordon et al., 2001). Today there are no natural established populations of C. virginica in Washington state although the industry continues to cultivate them in limited numbers with hatchery-reared seed.
British Columbia C. virginica
In 1903, C. virginica was introduced to British Columbia in Boundary Bay, Esquimalt, and Ladysmith harbors and Hammond and Nanoose bays. The only site with good yield was Boundary Bay, where C. virginica contin-
ued to be introduced until around 1940. Because of poor results obtained with transplanted seed, 3- to 4-year old oysters were transplanted instead and kept for about a year in Boundary Bay prior to marketing. A mortality of roughly 25% occurred even with the larger oysters (Quayle, 1988). Successful breeding of C. virginica occurred only in the estuaries of the Nicomekl and Serpentine rivers that flow into Boundary Bay.
Washington C. gigas
The first documented introductions of the Pacific oyster, C. gigas, to Washington took place in the early years of the 19th century. The first large commercial planting was in 1919 by the Pearl Oyster Company in Samish Bay. Four hundred cases of mature adult C. gigas (approximately 800 bushels) from the Miyagi Prefecture were shipped in early April 1919 (Lindsay et al., 1997). The oysters were covered with rice matting and given frequent showers of seawater until they arrived 18 days later. Many were dead on arrival, but the young oyster spat attached to the adults survived.
The discovery that spat were hardier than adults shifted the importation strategy to shipments of spat. The wooden cases for shipping seed could contain 12,000 to 30,000 spat averaging 6 to 18 mm in size depending on the size of shell cultch. Pacific oyster seed imports began slowly, reached a peak of nearly 72,000 cases in 1935 and then declined. Oyster seed imports ceased during World War II but began again in 1947 and continued yearly with the exception of 1978 and 1979 (Chew, 1984).
During the summer of 1936, Pacific oysters bred successfully in Dabob Bay on the Hood Canal, southern Puget Sound, and Willapa Bay. The resulting adult oysters provided a large brood stock as well as an important source of market oysters to carry through World War II when Japanese seed shipments were halted. This 1936 spawning by many accounts was huge, particularly in Willapa Bay. Al Qualman in his personal history, Blood on the Half Shell (1990), wrote: “There was no place to run, no place where the water would not be filtered and refiltered a thousand times over by the fantastic numbers of hungry little mouths.” The overabundance of oysters that resulted from this massive spawning forced the industry to expand its markets. Canning proved to be the answer because it extended shelf life and allowed oysters to be shipped by rail or airplane. The War Department recognized the nutritional value of oysters and in October 1943 approached the industry about supplying the troops. In response, Willapa Bay production numbers nearly doubled from 1943 to 1946 and reached a historical peak of 43,000 metric tons.
After the war the industry was almost driven from the canned oyster market by competition from Korea and Japan. At the time the wholesale
price for a carton (48 cans, 10 ounces each) of Willapa Bay oysters was about $14, while cartons of Korean oysters were selling on the open market for $5.65, including shipping costs and import duty (Gordon et al., 2001). With this price advantage, foreign imports might have wiped out the West Coast industry’s fresh-shucked and live markets were it not for provisions in the Lacey Act that banned the importation of any insects or other wildlife that might threaten crop production. Because Korean oysters were diagnosed as disease carriers, the importation of fresh-shucked or live oysters into the United States was banned. This ban protected the domestic industry by reducing the level of foreign competition in the fresh-shucked and live markets, thereby ensuring a market niche for the domestic industry.
Subsequent warm years in 1942, 1946, and 1958 resulted in excellent spat sets again in Willapa Bay, Dabob Bay, and southern Puget Sound. In 1942 the Washington Department of Fisheries initiated efforts to study spawning to better predict spatfalls for the industry. Spatfall predictions by the department continued into the 1990s, with many growers putting down cultch every year to supplement Japanese seed and later hatchery seed. Today, natural sets in these bays continue to provide a substantial percentage of Washington state’s oyster production.
Production of Pacific oysters on the West Coast has been measured traditionally in terms of gallons. Between 1937 and 1993 in Washington production ranged from 458,000 to 1,553,000 gallons. This would translate to roughly 12,700 to 43,000 metric tons of live oysters, assuming a gallon of shucked oysters weighs 8.75 pounds and live oysters are seven times heavier than shucked meats. Washington state oyster production was estimated by the Pacific Coast Shellfish Growers Association in 2000 at 35,000 metric tons live weight. Most of the production consisted of C. gigas. There are no statistics documenting what percentage of this amount is attributed to natural production in the Hood Canal and Willapa Bay versus hatchery-reared seed.
British Columbia C. gigas
C. gigas was first introduced to British Columbia around 1912 or 1913, when a few were planted in Ladysmith Harbor and Fanny Bay. By 1925 there was evidence of successful breeding in the harbor. The first significant importation occurred in 1926 with approximately 2,000 adult oysters from Samish Bay, Washington, and 20 cases of seed from Japan. Small numbers of naturally set oysters were found as a result of spawnings in Ladysmith Harbor in 1926, 1930, and 1931. The first major spawning of Pacific oysters in British Columbia occurred in 1932 in Ladysmith Harbor, where natural spawning continues periodically. Subsequently, plantings
of imported Pacific oyster seed took place throughout the Strait of Georgia, and in 1942 massive spawning in these areas resulted in a “catch” of oyster seed on many beaches particularly in the northern Strait of Georgia and Gulf Islands. As in Washington, this large set in 1942 carried the industry through World War II when Japanese seed shipments halted.
In 1958, widespread successful spawning of the Pacific oyster throughout the Strait of Georgia basically supplanted the need for importing Japanese seed. Pendrell Sound was established as a seed catching area (no leases allowed), and importation of Japanese seed ended around 1961. As in Washington state and the rest of the North American West Coast, the British Columbia industry today has transitioned to a significant reliance on hatchery-produced Pacific oyster seed. According to the British Columbia Shellfish Growers Association, 1999 production of Pacific oysters totaled 5,800 metric tons.
California C. gigas
When the Eastern oyster industry collapsed in the 1920s and 1930s, the industry and state explored the option of using C. gigas. In 1929 the California Department of Fish and Game (CDFG) and the industry made experimental plantings of C. gigas in Tomales Bay and Elkhorn Slough. In the 1930s additional experiments were carried out in Drakes Estero, Bodega Lagoon, and Morro, Newport, and San Francisco bays. Humboldt Bay was not included in the areas planted with C. gigas because CDFG was working there to reestablish the native oyster, O. conchaphila (Conte et al., 1996).
Many of these C. gigas plantings were successful, and seed shipments continued through the 1930s, halted during World War II (1940-1946), and then expanded again in 1947. Seed was inspected prior to shipment from Japan by CDFG personnel for organisms considered harmful to California waters. The CDFG lifted restrictions on C. gigas seed in Humboldt Bay in 1953 (Conte et al., 1996), and for the next 30 years seed shipments of C. gigas from Japan revived the California industry. Today approximately 90% of California’s oyster production comes from Humboldt Bay and Drakes Estero. The Pacific Coast Shellfish Growers Association estimates California’s 2000 C. gigas production at 4,500 metric tons.
Washington C. sikamea
After World War II, Washington’s supplies of Pacific oysters were depleted. The seed oyster industry in Miyagi and Hiroshima were set back by damages sustained in the war. In 1946, the first seed in 6 years was shipped, containing 30 cases of juvenile C. sikamea oysters from the Kumamoto Prefecture. The Kumamoto oysters are less likely to spawn
and recruit in the northwestern United States because they originate from a more southern and warmer prefecture than C. gigas. In 1960 and 1961, 1,004 and 1,200 cases, respectively, were imported (Steele, 1964). Careless brood stock management allowed hybridization with C. gigas, such that pure stocks of the Kumamoto oyster were hard to find in the early 1990s, when molecular markers for distinguishing the two species were developed (Banks et al., 1993; Hedgecock et al., 1993). Genetic testing of brood stock and more careful management has allowed the industry to rebuild productive Kumamoto oyster stocks. Kumamoto oysters are petite (50 cm) and have a deep cup that has been extremely popular in the live oyster market. Production continues to increase, and the industry is currently considering expanding the gene pool with oysters from Kumamoto Prefecture in Japan.
West Coast Transition to Hatchery-Based Production
During the 1980s there was a significant shift in the entire U.S. West Coast shellfish industry away from Japanese seed to seed produced in West Coast hatcheries and nurseries. Today, virtually all of the oysters grown in California, Oregon, and Alaska are reared from larvae produced in Washington and Oregon hatcheries. Washington and British Columbia have areas where natural spatfall occurs. In these areas (Willapa Bay and Hood Canal in Washington, Pendrell Sound and the Strait of Georgia in British Columbia) the industry captures seed and harvests these naturalized oysters. In areas with natural recruitment, the resource supports a substantial recreation harvest.
Currently, many West Coast companies are expanding production of single “cultchless” oysters for live or processed “half shell” markets and reducing “cultched” oyster production for the shucked market. This transition has been market driven. Taylor Shellfish Company, one of the largest producers of oysters on the West Coast over the past 8 years, has reversed production from 80% shucked, 20% live to 20% shucked, 80% live.
The transition to hatchery-produced seed has facilitated the use of genetically enhanced oyster stocks. Since the early 1990s, oyster yields have increased due to the success of crossbreeding and selective breeding programs. Growers have transitioned to triploid oyster production for higher growth rates, improved yields, and firm, glycogen-filled oysters during the summer when reproductive oysters are depleted from spawning. Of the 37.5 billion eyed larvae currently produced in West Coast hatcheries, about 12 billion are triploid (Nell, 2002).
Also, hatchery technology has allowed the West Coast shellfish industry to expand and diversify through culture of more species and pro-
vision of reliable seed resources to ensure harvests in available growing areas. Hatcheries provide a stable source of seed at predictable prices, enabling sound business planning, bank financing, and market development. Finally, hatchery production of seed has substantially reduced the risk of unintentional species introductions that were common in the early years when seed on cultch was transported within the United States and internationally. Species diversification helps buffer temporary price drops and losses from species-specific diseases.
Unintentional Species Introductions Attributed to the Intentional Introduction of C. gigas and C. virginica to the West Coast of North America
Lack of adequate regulatory protections associated with the movement of live C. gigas from Japan and C. virginica from the East Coast resulted in the introduction of 27 species of Asian and Atlantic molluscs to the Pacific Coast of North America (Carlton, 1992b). In 2000 a survey for nonnative species in Elliot Bay (Seattle), Totten and Eld inlets (southern Puget Sound), and Willapa Bay suggests that oyster seed shipments from Japan and the U.S. East Coast may be responsible for 35 of 40 exotic species collected (Cohen et al., 2001).
The consequences of an unintentional species introduction became apparent to West Coast oystermen with the introduction of Ceratostoma inornatum, the Japanese oyster drill, in cases of oyster seed from Japan. The drill is particularly damaging to newly seeded Pacific oyster crops. The Japanese drill also attacks exposed Manila clams, and predation by this drill has hampered recent efforts to restore beds of native oysters. An adult C. inornatum requires about 2 weeks to drill through a 5-cm-length Pacific oyster and only a day or so for seed oysters up to 2.5 cm in diameter (Quayle, 1988). A major inspection program was established in 1947 with state biologists from Washington and California checking oyster seed shipments in Japan for pest species prior to seed shipment. Additional inspections were made on arrival from Japan. Regulations have subsequently been adopted in West Coast states to prevent accidental introductions of nonnative species or transfer of disease. Washington initiated regulations in 1945 to prohibit transfer of Japanese drills among oyster plantings in the state. These regulations, together with the transition to hatchery-produced seed, now prevent the unintentional introduction of nonnative species due to the activities of the shellfish industry.
A couple of unintentional species introductions have been economically valuable. Most notable is the Manila clam, Venerupes philippinarum. The Manila clam was introduced into the Puget Sound during the 1930s and 1940s with shipments of Pacific oyster seed from Japan (Chew, 1989;
Quayle, 1938, 1988). Since its introduction it has become established throughout the Puget Sound region. It reproduces successfully in a number of areas in Puget Sound, Willapa Bay, and British Columbia. Production in 2000 in Washington state alone was estimated by the Pacific Coast Shellfish Growers Association to be in the neighborhood of 7 million pounds valued at $14 million. Second only to the native geoduck clam, the Manila clam is one of Washington’s most valuable commercially harvested shellfish.
The Eastern soft shell clam, Mya arenaria, is another unintentional species introduction of economic value. The soft shell clam was apparently brought to the West Coast in shipments of the Eastern oyster in the early 1900s. M. arenaria became established in a number of West Coast bays at higher tidal elevations. The species has done particularly well in Skagit Bay and Port Gardner in northern Puget Sound, where in the 1980s the natural population was sustaining annual harvests in the 135 to 180 metric tons range. These were all being shipped to East Coast markets. Recently, the resource has been largely inaccessible to commercial harvest because pollution has forced the Washington State Department of Health to downgrade the areas.
Nonbeneficial or harmful species introductions have been more common. Several examples of detrimental unintentional species introductions are described below.
Urosalpinx cinerea, the Atlantic oyster drill introduced to the West Coast with shipments of Eastern oyster seed, was primarily especially a problem when Eastern oysters were raised. It preys on Pacific oysters as well and thus remains a problem for the West Coast shellfish industry. Its distribution extends from British Columbia, Canada, to Newport Bay in Southern California. It is a predatory snail that feeds on barnacles and mussels as well as oysters. Both the Eastern and Japanese oyster drills may prove problematic to current efforts to restore populations of the native West Coast oyster, Ostreola conchaphila.
Pseudostylochus ostreophagus is a flatworm that is thought to have been introduced with Japanese oyster seed shipments. It is predatory on juvenile mussels and oyster seed.
Mytilicola orientalis is a parasitic copepod originating in Japan. It occurs in the lower intestine of oysters and mussels. It appears to cause no damage other than reduced condition of the meat tissue. M. orientalis was probably brought to Europe in shipments of C. gigas. Its occurrence in Pacific oysters was first reported by His (1977). However, another copepod (M. intestinalis) was already present, and the impact of M. orientalis was only evident at high infestation rates. M. orientalis is considered a parasite because it sometimes induces metaplasia in oysters (Deslous, 1981). The zoosanitary monitoring network in France and Europe does not report infestations of M. orientalis
and the overall impacts of this introduced species are not considered significant (Leppäkoski et al., 2002).
Crepidula fornicata was introduced with the importation of Eastern oyster seed. It has become well established in Totten and Eld inlets in Southern Puget Sound and has also been documented in Willapa Bay (Cohen et al., 2001). This species competes for food with oysters, clams, and other filter feeders that consume phytoplankton. Crepidula has not caused problems where phytoplankton resources are abundant such as in southern Puget Sound. However, in the right circumstances, this species could become invasive. In France, populations have become invasive and the oyster industry must control them due to the competition for limited phytoplankton resources.
Sargassum muticum, a Japanese seaweed commonly referred to as wireweed, was introduced with Japanese oyster seed. It first gained a solid foothold in British Columbia waters between 1941 and 1945 (Quayle, 1988). It is thought to have been introduced to the Puget Sound in the 1930s with Pacific oyster seed. A highly invasive species, today it has established itself from southern Southeast Alaska to Baja California, Mexico (O’Clair et al., 2000). It requires rocky bottom in the lower intertidal and has been known to attach to Pacific oysters with its holdfast. The mature alga has thick fronds reaching up to 2 m in length. During storm events, rough seas may dislodge the seaweed and attached oyster causing mortality if they wash ashore. The Monterey Bay Aquarium Research Institute examined the epibiont community associated with S. muticum in northern Puget Sound (San Juan Islands) from May to September 1997 (Osborn, 1999). This examination found that epibiont diversity and abundance increased in areas invaded by S. muticum because of the increased habitat, productivity, and complexity that S. muticum provides. Based on the positive effects on the epifauna, their recommendation was to not attempt to eliminate the species but to further study potential negative effects on water movement, light penetration, sediment accumulation, and anoxia at night.
Zostera japonica, Japanese eelgrass, is thought to have been introduced with shipments of Pacific oyster seed from Japan. First reported in Willapa Bay, Washington, in 1957, it is colonizing midintertidal tideflats (+0.6 to +1.2 m above mean lower low water), which previously lacked macrophyte cover (Bigley and Harrison, 1986). Its range currently extends from southern British Columbia to Coos Bay, Oregon (Posey, 1988). Posey researched the effects of Z. japonica invading Coos Bay and found a general positive effect on local species diversity and abundance. In Boundary Bay, British Columbia, where Z. japonica is well established, it was found to be a much more suitable food for dabbling ducks than the native Z. marina (Posey, 1988). In concluding his paper assessing the Z. japonica
introduction, Posey offers advice relevant to the C. ariakensis question. He suggests “consideration should be given to the possibility that an established exotic may not necessarily be detrimental to native organisms.”
In Washington state, Z. japonica is present throughout Puget Sound, Hood Canal, and Willapa Bay. Anecdotally, shellfish growers claim it is currently on the increase. It causes problems when it heavily infests Manila clam beds primarily by reducing drainage and obstructing clam harvest. It is interesting to note that the sand/mud substrate in parts of Willapa Bay was not stable enough to allow Manila clams to recruit and survive until Z. japonica invaded the area (W. J. Taylor, Taylor Shellfish Company, Shelton, Washington, personal communication, 2003).
Spartina alterniflora, or Smooth Cord Grass, is thought to have arrived on the West Coast when it was used as packing in freight cars carrying loads of Eastern oysters to Willapa Bay. It established itself in the bay over 100 years ago but only became invasive in recent years. Today it is estimated to be filling in the higher intertidal mudflat regions of Willapa Bay at an average rate of 1 acre per day. As Spartina invades shallow mudflats, it traps sediments that build up and allow it to expand farther out away from shore. The Spartina is taking over critical habitat for shellfish, salt marsh plants, migratory birds, juvenile fish, and other wildlife. Massive efforts are being undertaken to eradicate it at great expense. Spartina has also infested parts of Puget Sound but was more likely deliberately introduced by duck hunters as a duck blind or by cattle operations as an alternative food that would grow well in the salty soil recovered by building dikes in the bays.
Although many nonnative species were probably introduced to the West Coast with foreign oyster seed, only one significant disease is thought to have resulted from such shipments. Mikrocytos mackini, a parasite that lives in the glycogen storage cells of the Pacific oyster and causes Denman Island Disease (Elston, 1990), is suspected to have been introduced, but its foreign origin has not been confirmed. First reported in 1960 in Henry Bay on Denman Island, British Columbia, it has since been noted at other locales in the Strait of Georgia. In 2001 it was discovered in oysters in Sequim Bay in Washington state, triggering emergency regulations restricting live oyster movements from potentially infected areas. Mortality rates up to 53% in a single season have been reported, but severity fluctuates from year to year (Elston, 1990). The disease mainly affects older oysters that occur at lower tidal elevations.
Summary
-
Driven by depleted natural stocks of the native oyster, O. conchaphila, and aided by the transcontinental railroad, massive introductions of C. virginica were made initially in California (1869 to 1921) and
-
later in Washington state (1894 to 1919) and British Columbia (1903 to 1940). The Eastern oyster naturalized (became self-populating) for a time in Willapa Bay, in southwest Washington, and Boundary Bay, British Columbia. C. virginica continues to be farmed with hatchery-produced seed on a small scale on the West Coast.
-
C. gigas was introduced first to Washington state (1904), then British Columbia (1912 to 1913), and later California (1929). Massive amounts of seed attached primarily to shells imported annually between 1919 and 1979 from Japan, with a short break during World War II. Successful breeding was noted first in Ladysmith Harbor, British Columbia, in 1932 and Dabob Bay, Southern Puget Sound, and Willapa Bay, Washington, in 1936. In notably warm years successful breeding continues to occur in certain areas of Washington and British Columbia; however, the Pacific oyster fails to breed over most of the West Coast of North America, limiting its ability to invade new areas.
-
There were few or no protections to prevent unintentional species or disease introductions with oyster seed imports from the East Coast or Japan until 1945. As a consequence, numerous nonnative species were introduced and became established. Many of these introduced species cause ecological and economic harm. At least two introduced clam species have resulted in economic benefit through the establishment of lucrative aquaculture and fishing industries.
-
State regulations restricting interstate and international seed importation coupled with the transition to hatchery-produced seed have dramatically reduced, though not eliminated, the risk of unintentional species or disease introduction.
-
Hatcheries and nurseries capable of supporting the majority of the North American West Coast oyster industry (estimated 48,000 metric tons annually) evolved over a period of 20 to 30 years. Roughly a third of this production is triploid.
-
The transition to hatchery production on the West Coast has facilitated diversification of the industry and supported breeding programs for superior aquaculture stocks.
-
Laws allowing the private ownership of tideland in Washington state have served as a critical foundation for a stable and expanding industry.
Eastern and Gulf Coasts of North America
The Pacific oyster, C. gigas, and the European or flat oyster, O. edulis, have been introduced to the East Coast of North America multiple times (see Table 3.2). Before 1950 some of these introductions
were documented in the scientific literature by the scientists who deliberately introduced the new species. This included scientists associated with state fish and wildlife departments. These introductions were small scale and made with the purpose of testing whether the species would be suitable for commercial production. Survival, growth, and reproduction were monitored. Since 1950, two experimental introductions of C. gigas in Maine (Dean, 1979) and Massachusetts (Hickey, 1979) have been reported officially. Others reported have been anecdotal accounts of C. gigas introductions (Andrews, 1980; Mann et al., 1991; Carlton, 1992b).
Importation of limited numbers of O. edulis into New England in the 1940s and 1950s led to the establishment of small, naturally reproducing populations in Maine and Rhode Island, where the species is now also grown in aquaculture. In contrast, introductions of O. edulis into the Canadian Maritime Provinces apparently did not naturalize (Medcof, 1961).
In contrast to the establishment of O. edulis, no naturalized populations of C. gigas have been reported to exist on the East or Gulf Coasts of North America, despite the known and suspected introductions (the latter being undoubtedly more numerous than the literature indicates), including one or two large-scale plantings in Chesapeake Bay (Table 3.2). With a single exception, all of the reported introductions of C. gigas have been to the mid-Atlantic, New England, and Canadian Maritime Provinces. A single published account exists for the southeastern United States and Gulf of Mexico and describes the testing of C. gigas in Louisiana and Alabama. In this environment the oysters were emaciated and heavily infested with a polychaete worm ( Polydora sp.) that caused mud-filled blisters on the inner shell; they eventually died (Kavanagh, 1940).
It is probable that water temperatures are too low for successful reproduction of C. gigas in the northeastern North America. This is not true of the mid-Atlantic, however, where experimentally held C. gigas underwent gametogenesis and spawned in laboratory tanks that received ambient water (Barber and Mann, 1994). It is possible that the numbers of oysters introduced into the mid-Atlantic were simply too low to establish a wild population. It should be noted that the establishment of C. gigas in France occurred after an estimated 562 metric tons of adults and several billion spat were imported and planted between 1971 and 1975 (Grizel and Héral, 1991). Further, naturally occurring populations of C. gigas on the West Coast of North America developed only after numerous introductions of seed from Japan (Carlton, 1992b), although reproduction is definitely limited by low temperatures in this region.
TABLE 3.2 Reported Introductions of Nonnative Oysters to the East and Gulf Coasts of North America Prior to the Recent Testing of C. ariakensis
|
Date |
Species |
Location |
Comments |
Reference |
|
Early 1930s |
C. gigas |
New Jersey |
One bushel, placed in Barnegat Bay, grew rapidly during first 2 weeks, then stopped and gradually died out without further growth. The poor performance may have been due to the relatively low salinity (12-20 ppt), with low dissolved oxygen and stagnation. |
Nelson (1946) |
|
1930s |
C. gigas |
Louisiana and Alabama |
At least two introductions of several “cases,” presumably from the U.S. West Coast, were made into Mobile Bay, Alabama, and later transferred to Louisiana. All died. A smaller lot imported later and placed in a flow-through system for closer study rapidly lost meat condition and acquired heavy mud blisters (Polydora sp.). |
Kavanagh (1940) |
|
1949 |
C. gigas |
Massachusetts |
Six bushels of spat from Washington state, grown in Barnstable Harbor on Cape Cod over 1.5 years, grew to 5 inches with little evidence of mortality or spawning in the field. |
Turner (1949) |
|
1949 |
C. gigas |
Maine |
6 bushels of spat from Washington State suffered 40% mortality from starfish but otherwise no losses. |
Turner (1949; 1950) |
|
1949 |
C. gigas |
Maine |
A “few bushels” were introduced; gonadal maturation occurred, but no spat were found. |
Dow and Wallace (1971) cited in Dean (1979) |
|
1949 and 1955 |
O. edulis |
Connecticut and Maine |
In 1949 9,000 oysters in three year-classes were brought from Holland: 3,000 to Booth Bay Harbor, Maine; 6000 to Milford, Connecticut. |
|
|
|
|
|
In 1955 another batch of 4,000 from Holland were imported into Maine. Survival was good in both Connecticut and Maine. Successful reproduction and persistence of population was observed in Maine, where small populations remain. Some individuals were later found in Rhode Island, including at borders of Connecticut and Massachusetts. |
Loosanoff (1955), Welch (1963), Zabaleta and Barber (1996), R. Rheault, (Moonstone Oysters, Pt. Judith, RI pers com 2002) |
|
1957, 1958, 1959 |
O. edulis |
Prince Edward Island and New Brunswick, Canada |
About 5,000 individual wild-set oysters were imported each year from England. The shipments were examined macroscopically for parasites and hitchhikers. The oysters did well during the summer but suffered heavy losses during severe winters. |
(Medcof, 1961) |
|
Pre-1950 |
C. gigas |
Long Island Sound |
No information was available other than “numerous attempts at introduction.” |
Davis (1950) and personal communication in Dean (1979) Andrews (1980) |
|
Early 1960s |
C. gigas |
Delaware |
An unknown quantity of oysters was held in trays in Rehoboth Bay for several years without serious mortality or apparent reproduction. |
|
|
1971-1973 |
C. gigas |
Maine |
Oysters were imported for limited controlled experiments on the growth of juveniles in Maine waters. Fast growth and good gonadal development but no spawning was reported. |
Dean (1979) |
|
1976-1977 |
C. gigas |
Massachusetts |
Some 100,000 seed (6-8 mm) from West Coast hatchery were placed in an enclosed pond off Buzzards Bay. About 80% were lost shortly thereafter during a hurricane and presumed buried in mud as they were never found. The oysters grew well, and spawning occurred “more or less continuously” during the summer of 1977. Larvae were found in the water, but no spat were ever recovered even though cultch was placed in the pond. |
Hickey (1979) |
|
Date |
Species |
Location |
Comments |
Reference |
|
1970s? |
C. gigas |
Maryland |
“Recently, C. gigas from the west coast of North America was planted in Maryland waters (of Chesapeake Bay] by a seafood dealer which resulted in a specific law in that state prohibiting the species. The oysters were recovered as completely as possible by scuba diving.” |
(Andrews, 1980) |
|
~1988-1990 |
C. gigas |
Maryland or Virginia |
Thousands of bushels were reported to have been planted in Chesapeake Bay. |
(Carlton, 1992a) |
|
1990s |
C. gigas |
Lower Chesapeake Bay |
Testing of sterile triploids at three salinities for growth, survival, and resistance to Dermo and MSX diseases was conducted by the Virginia Institute of Marine Science. They were deemed unsuitable for Chesapeake Bay because of poor performance in low salinity and the acquisition of heavy mud (Polydora sp.) blisters. |
(Calvo et al., 1999) |
CHAPTER SUMMARY
-
Estuarine habitats frequently contain large percentages of nonnative species because these areas are heavily used for shipping, fishing, and recreation. Introductions of nonnatives occur mainly through human activities. Nonnative shellfish have been transported from region to region for hundreds of years, and oysters have been intentionally transported more than any other marine species.
-
The incentive for introduction of commercial marine species has been to replace depleted natural stocks or to diversify the types of species used in aquaculture. Although molluscan aquaculture typically has relatively small environmental impacts relative to culture of fish and shrimp, diseases and “hitchhikers” have been transported along with the shellfish. A number of these unintentional introductions have caused severe impacts on shellfish production and ecosystem structure and function.
-
While there are no documented cases that an intentional introduction of a marine bivalve species in U.S. waters has caused pronounced environmental change, there are a number of well-documented cases of how unintentional introductions have resulted in both positive and negative impacts.
-
It is exceedingly difficult to predict whether a marine species has the potential to become an “invasive” or a “nuisance” species. Species that exhibit fast growth and high reproductive rates and are tolerant of a wide range of environmental conditions generally tend to be viewed as candidate “invasive” species.
-
Ecosystems with reduced biodiversity or that are environmentally degraded may be more vulnerable to invasion. In addition, the southern and northern boundaries of faunal provinces may be more susceptible to invasion by nonnative species as a consequence of climate change.
-
A review of a number of case studies of intentional shellfish introduction indicates that there are both benefits and risks dependent on the region. In France, introduction of the Pacific oyster, C. gigas, led to revitalization of the oyster industry, but in New Zealand and Australia, the Pacific oyster outcompeted the commercially viable native oyster in some areas.
-
The oyster industry on the U.S. West Coast relies almost exclusively on nonnative species. The success of this industry is largely based on culture of several types of molluscs and regulations that allow private ownership of tidelands.
































