Panel II
SBIR at the National Institutes of Health
Moderator:
James Turner
House Science Committee
Mr. Turner introduced the panel by relaying the Chinese term for a perfect plan as a “garment made in heaven,” because “that’s the only place a garment can be seamless.” He acknowledged that the SBIR was not yet either perfect or seamless; nor was it “a patchwork quilt” of disparate elements, though it might have become such during its creation by five separate committees of Congress.
He said that his own committee, the House Science Committee, was one of those five, and that Congress had re-examined the SBIR program three times since its inception in 1982. Thanks to the efforts of the program managers responsible for each agency’s SBIR activities, he said, the program had worked quite well and produced strong results. Mr. Turner noted that the SBIR program itself is not in doubt: The Congress had not questioned the program’s value during its last authorization, and it is not expected to do so during the next authorization. Further, the upcoming study by The National Academies is not expected to question whether the program should exist. “We’re 20 years into the SBIR now,” he said. “It’s a proven entity; it’s going to be with us. The question is how to get as close as possible to a garment made in heaven, here on earth.”
Mr. Turner said that the program has faced, and will continue to face, many challenges, including “its schizophrenic origins,” rapid changes in the small business community, broad challenges to the R&D system stemming from terrorism, and the substantial economic and technological changes that occurred since the program’s origin. He suggested that appropriate goals for the study would be to look ahead and craft a series of sound suggestions on how to improve the program so that, when it is time for its next review by Congress, good advice will be available to committees on what legislative changes, if any, are necessary.
ACHIEVEMENTS, OPPORTUNITIES, AND CHALLENGES
Jo Anne Goodnight
National Institutes of Health
Ms. Goodnight introduced herself as the SBIR/STTR coordinator for the National Institutes of Health (NIH). She expressed her pleasure at having the opportunity to participate in this symposium to discuss the “congressionally mandated, comprehensive study of the SBIR program.” She said she would discuss the achievements and goals of the SBIR program, the role the small business research community plays in the mission of the NIH, and the opportunities and challenges of the upcoming SBIR study.
NIH is comprised of 27 institutes and centers, 23 of which participate in the SBIR program. Each of the components that awards SBIR grants has a mandate with well-defined priorities that address science and health issues from a specific perspective, such as minority health issues, health disparities, particular disease areas, such as cancer, and broader areas of concern, such as aging. Because NIH is primarily a granting organization, about 95 percent of its SBIR awards are made through its grant mechanism. Some 4 to 5 percent of awards are made by contract and a few are made through cooperative agreements.
The individual institutes and centers of the NIH develop topics that relate to their missions and could be considered “NIH-generated ideas.” But NIH also seeks to encourage “investigator-initiated” ideas that fall within the mission of any of the awarding components.
The NIH mission, she said, is to improve human health through biomedical and behavioral research, research training, and communications. In carrying out this mission, the NIH supports basic, applied, and clinical research to better understand the complex processes underlying human health and to acquire new knowledge that will help prevent, diagnose, and treat human diseases and disabilities.
“From the Test Tube to the Medicine Cabinet”
The SBIR program plays an integral role in the NIH mission, said Ms. Goodnight, particularly the goal of translating scientific findings from concept to societal benefit. She said that one might think of this process as “moving from the test tube to the medicine cabinet.” Small technology firms are prolific innovators which are recognized as unique resources not only for the development of enabling technologies, but also for creating “disruptive technologies”—those that displace entrenched techniques and have the potential to create new industries.
She said that SBIR is a perfect program to allow the entrepreneurial research community to “go out on a limb and challenge paradigms.” In the health arena, disruptive technologies are often capable of changing the landscape of health care. Because of these technologies, for example, nurse practitioners, general
practitioners, and even patients can perform procedures in less expensive, decentralized settings that could once be performed only by specialists. While these disruptive technologies may face initial resistance from doctors, insurance companies, and hospitals, they clearly have the potential to bring enormous benefits to society.
SBIR Achievements
The SBIR program at NIH has achieved much since its inception in 1983, she said, highlighting the following acheivements:
-
The program is well integrated into the overall scientific programs and goals of the NIH, as is the case for many agencies. It continues to receive positive support from the NIH leadership. As it has evolved, it has benefited from an enhanced collaborative effort among the SBIR agencies and the research community.
-
Agencies are collaborating more closely to develop trans-agency initiatives in areas such as nanotechnology, assistive technology, and biodefense. The small business community can respond quickly to the changing nature of science because of its own flexibility and adaptability. It also encourages multidisciplinary approaches, which are needed to address some of the more complex research questions.
-
Another achievement is that the SBIR program and certainly the STTR program have each created avenues for connecting basic knowledge to the marketplace through university-industry partnerships, which is an important dimension in our rapidly evolving economy. Another achievement is that the SBIR has assisted NIH, and indeed all federal agencies, in addressing agency-critical technology research areas and in responding to national priority areas. Examples of such areas include biodefense and nanotechnology, as well as imaging, bioengineering, bioinformatics, and biomaterials.
-
The flexibility of the Small Business Administration allows different ways to support various agency mission outcomes. This latitude, which is supported by the SBA, allows companies to propose research and development in fields that have the most biological promise, rather than limiting ideas to those that can be conducted under prescribed budgets and timelines.
The 1982 legislation that initially authorized the SBIR program included four primary goals:
-
To encourage small businesses to stimulate technological innovation;
-
To encourage small businesses to meet federal R&D needs;
-
To foster and promote additional participation by minorities and disadvantaged individuals in creating technological innovation; and
-
To increase commercialization of these federally funded R&D innovations in the private sector.
Reauthorization Brought Changes
As a result of the December 2000 SBIR program reauthorization, there was now greater emphasis on outputs, outcomes, and commercialization. There is also continued support for outreach and technical assistance for the small businesses. One mandate of the current evaluation was to study this new emphasis on outcomes. This study presents a number of opportunities to evaluate the program’s effectiveness across the ten federal agencies that make SBIR awards. These agencies have collectively invested more than $12 billion in SBIR projects since 1983. The NIH investment exceeds $2.5 billion.
With the current structure of the SBIR, most small companies are able to view the federal government as a transparent “technology clearing house.” This allows the companies to consider SBIR opportunities across agency boundaries and align their technical competencies with the needs of multiple agencies.
The SBIR structure also allows new companies an excellent chance of gaining awards. Based on the experience of the Department of Defense SBIR, about one-third of the companies entering the program each year are first-time recipients of awards. NIH data show that 75 to 80 percent of the companies receiving the awards have earned between one and three previous awards.
Opportunities for the Study Panel
The plan for the comprehensive SBIR study, said Ms. Goodnight, presents a number of desirable opportunities. She hoped that the study will:
-
Describe the trans-agency program while highlighting how each agency uses the program to accommodate its particular culture;
-
Provide hard information on how well legislative goals are linked to program outcomes;
-
Assess the degree to which each agency’s operational and administrative activities support SBIR goals;
-
Offer a means to quantify both the economic or direct commercial benefits and the non-economic or societal benefits of the program.
Given the enormity and complexity of this study, she said, a number of considerations are worth highlighting. First, it is critical that the evaluation framework incorporate all SBIR legislative intents across all the agencies—not merely the mission and goals of a select subset. “Important outcomes could easily be over-
looked,” she said, “if this study does not take into consideration the investments made by all 10 agencies.” She stated that the synergistic benefits of the national SBIR program as a whole are greater than the sum of its parts, and hoped that the study would verify this. A second consideration, said Ms. Goodnight, is that the methodology of the study must address how agencies incorporate their own culture and processes in achieving the congressionally mandated goals of the program.
Challenges for the Study Panel
While the SBIR study brings numerous opportunities, she said, it also poses a number of challenges, including the following:
-
Identifying commonalities and unique features of each agency’s programs;
-
Developing a systematic and comprehensive methodology that adheres to the goals of the study;
-
Reconciling existing knowledge bases within agencies while maintaining an independent and unbiased global study;
-
Appreciating redundancies and avoiding undue burdens on respondent companies, where appropriate.
Some additional challenges, she said, included identifying the actual direct and indirect value of SBIR-supported R&D in creating commercial and societal benefits. In some cases, for example, it might be shown that multiple awards were needed to bring a product to market. Or it might emerge that SBIR funding was only one step in bringing a product to market, which also required other federal or nonfederal funding.
Technologies Progress at Different Rates
An important topic for the study to consider is the rate at which various companies move toward commercialization, from Phase I to Phase III. It is already known that these rates vary for some technologies and agencies. For example, NIH-supported projects to develop pharmaceuticals require an average of 12 years and hundreds of millions of dollars to complete, while DoD-supported military products are developed for more immediate use. The development timeline for NSF-supported information technology products is relatively short, while the timeline to plant and grow crops under a USDA-supported SBIR can be considerably longer. In studying these varying commercialization timelines, it is likely that the progression from Phase I to II to III is not always linear. That is, discoveries or questions in the developmental or commercialization phases may stimulate new or additional research, while new research may suggest new applications.
Ms. Goodnight said in closing that she fully appreciated the importance of an objective and comprehensive evaluation of the SBIR program. In light of the R&D investments made by NIH over the last 20 years, during which some 50,000 awards have been made to some 12,500 firms, the study represents a unique opportunity to evaluate the economic and societal benefits of the SBIR program. She concluded that the agencies had worked thoughtfully, responsibly, and collaboratively for the past year to take full advantage of this opportunity, and expressed pleasure at the prospect of working out the next steps with the National Research Council.
THE NIAID PERSPECTIVE
Carole A. Heilman
National Institute of Allergy and Infectious Diseases
Dr. Heilman, Director of the Division on Microbiology and Infectious Diseases at the National Institute of Allergies and Infectious Diseases, focused on the long-term relationship of NIAID with public-private partnerships. Within that context, she addressed both the role of the SBIR within the broader range of partnerships and within the nation’s biodefense research needs.13
High Hurdles for Companies
She began by emphasizing a point made by Ms. Goodnight–that many of the products developed by the National Institutes of Health require an extremely long time and a large amount of money to discover, develop, test, and approve. NIH is involved in both basic and, to a lesser extent, applied research that attracts the interest of both large and small companies that develop products for the marketplace. Among the most important DHHS agencies with respect to product development are the FDA, which is responsible for approving biological or intervention products for marketing, and the CDC, which is responsible, in the case of newly licensed vaccines, for deciding who is recommended to receive them. For products developed for the commercial market, both steps are high hurdles that must be overcome. For products developed for biodefense, the Office of Emergency Preparedness, within DHHS, plays a key decision-making role with respect to the purchase of products.
NIAID supports both basic and applied research, but unlike many of NIH’s component institutes, a major part of its mission is to focus on applied translational research, which it has done for about four decades. Thus NIAID,
even before the SBIR began, had had considerable experience interacting with industry and translating the information developed by basic research into products.
Three Mechanisms to Help Companies
The Institute used three general mechanisms to help translate discoveries into products. Dr. Heilman illustrated these mechanisms by three examples from the post-September 11 rush to develop biodefense products. Three of the initiatives it was asked to undertake concerned smallpox: (1) a smallpox vaccine dilution study, including a determination of whether the vaccine already stored was of good quality and whether it could be diluted so as to protect everybody in the United States; (2) whether the Institute had any compounds that could be used as therapeutic agents in the event of a smallpox attack; and (3) whether the Institute could enlist industry as well as academia in moving immediately into the biodefense field. She used these three examples to show how the public-private partnerships of NIAID have worked.
Three Examples of Public-Private Partnerships
-
Launching the vaccine study in record time required an infrastructure that had been in existence for over 40 years. It had been used routinely to assist companies in deciding whether to pursue particular products. The infrastructure had a core of clinical research capability, but to get to that point requires a huge investment in both basic research and product development, including preliminary information that the product may indeed be of value. “No company is going to invest hundreds of millions of dollars in a concept,” she said, “until there is a good idea how that concept works.” In order to speed concept development, the Institute offers a contract process that includes opportunities to test a certain product at a preclinical stage. NIAID tests the product for the company, which then decides internally if the product is worth pursuing. The Institute can also assist the company in approaching the FDA and applying for investigational new drug (IND) status for the product. Further, it can help the company do NIH Phase I and Phase II clinical studies, and in some cases Phase III through Phase IV studies, which entail most of the expense of product development. The Institute also makes available to the company its extensive academic expertise.
-
For the task of surveying existing, already-licensed compounds as well as compounds under development that might be useful against smallpox, the Institute used a second existing mechanism. This mechanism, which is a series of in vitro and in vivo antiviral screening contracts, is used for evaluating a compound a company may have developed or is considering developing to determine if it has value. These contracts can provide information as to whether compounds
-
inhibit certain viruses. If the product candidate performs well in tissue culture, the Institute can assume responsibility for moving it to animal studies, which are more elaborate and expensive. The resulting data is given to the company, which again decides internally whether to proceed to the next stage. During the post-9/11 period, this survey process produced a promising candidate in the drug cidofovir—drug that had already been licensed for the relatively rare application of combating CMV (cytomegalovirus) retinitis in AIDS. It was found also to be very active against smallpox. The Institute worked with the company to develop an IND application for the FDA that would allow the use of this drug in case of a smallpox event.
-
A third general mechanism of NIAID is to prompt companies as well as academia to invest effort in areas considered to be important (see Table 1). As an example, within about two months of September 11 the Institute published a request for applications in a variety of areas of high priority. A specific request was targeted at the small business community, asking companies to select certain platform technologies they had been developing and move them in the direction of the Institute’s Category A biodefense activities. At that point, very few investigators in the academic or business worlds were working on biodefense products, and the request was intended as a quick stimulus to the non-military research community.
TABLE 1 FY2002 NIAID Biodefense Initiatives
|
Initiative Title |
Target Audience |
|
NIAID Small Business Program on Bioterrorism-Related Research (SBIR/STTR) |
Small Business |
|
Rapid Response Grant Program on Bioterrorism-Related Research |
Academia |
|
Exploratory/Developmental Grants: Technology Applications |
NIAID-Funded Researchers |
|
Partnerships for Novel Therapeutic, Diagnostic, and Vector Control Strategies in Infectious Diseases |
Small and Large Businesses |
|
Investigator-Initiated Small Research Grants |
New Researchers/changing fields |
|
U.S.-Based Collaboration in Emerging Viral and Prion Diseases |
Academic and Industrial Researchers |
|
Development & Testing of Vaccines Against Anthrax |
Industry |
The High Quality of Small-business Research
For a small firm, movement into such a new area would require not only commitment but also the flexibility that small businesses uniquely have. NIAID put out the request under the SBIR, asking companies to respond within six weeks. They knew that responding to their request would require a huge amount of effort on the part of companies: redirecting research in rapid fashion, learning basic principles of biodefense, and asking consultants to determine whether a company’s approaches would be amenable to peer review. Nonetheless, the Institute received 184 applications within the short published time limit, revealing both a commitment toward biodefense and very high quality of small-business research. NIAID was able to fund 40 of these applications—a high (22 percent) approval rate, given the stringency of peer review and the requirement that only appropriate topics could be funded. “Usually,” said Dr. Heilman, “only a small business is able to shift that quickly and be responsive to a request like that.”
For FY2003, the Institute expected a “huge” increase in the President’s budget for biodefense (see Figure 3). Because some 80 to 90 percent of NIH’s dollars go outside the agency to support academic and industrial research, there was an urgent need for this community to be invested in the new emphasis on biodefense. NIAID accomplished this by bringing people from both communities to NIH and enlisting their help in identifying the critical knowledge and developmental gaps that needed to be filled. This “NIAID Research Agenda” explained its strategic plans and research needs to these academic and industry researchers allowing
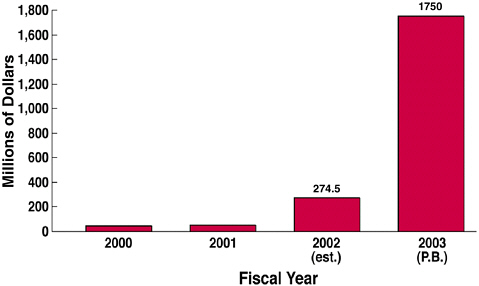
FIGURE 3 NIH Biodefense research funding, FY 2000-2003.
them to better plan for the kinds of expertise and approaches they wanted to invest in. Copies of this Research Agenda can be found on the NIAID web site.
Helping Outside Partners
Summarizing the Institute’s strategy for public-private partnerships, Dr. Heilman said that NIAID needed to be able to understand enough about the pathogens that are involved in biodefense to be able to develop new products. The Institute had recognized that it needed to be able to help outside partners in the development of new products by making available the right research resources, such as a vaccine infrastructure or screening process. This allows companies to avoid duplicating or developing processes that are very expensive, and instead to focus their attention on the part of the process they do best, which is research application. She said that the Institute had planned to issue 28 new requests of this nature in FY2003 and would also be expanding features of its existing infrastructure.
In the areas most relevant to industry, the Institute was asking firms to begin looking at some of the targets that recent advances in genomic information have brought into focus. A goal was to allow scientists to expand the roster of effective drugs, vaccines, and diagnostics. NIAID also wants to partner with industry to help them move toward new targets identified in initial testing to see if these targets are feasible for development by industry.
A New Model: The Partnership Grant
Dr. Heilman described a new model created by NIAID called the partnership grant, which allowed larger amounts of money to go to industry. These grants required that industry make a serious commitment to working in a particular area, in terms of the number of people who would work on a product or a particular suite for the process development of the product. If a company could demonstrate this commitment, the Institute was willing to take the risk of spending more money to help the company by means of a development partnership. The industry would need to understand that the partnership grant would be milestone-driven, but that the Institute would work closely with the industry to decide whether the project would be a “go” or “no go” at every step of development. Partnership grants are much more cooperative than other types of grants, and the Institute leverages all of its resources to make the company as successful as possible.
Agreements with Industry
The Institute already exercises many agreements with industry. At the time of the symposium, it held more than 100 agreements for research on investigational new drugs (INDs), and at any particular time it was performing about that
number of clinical protocols. The Institute was approximately the size of a medium-sized company, and included an IND group, clinical groups, and other resources typical of a commercial research organization. NIAID already had many behaviors, she said, by which it “thinks” as a company. It developed pre-clinical agreements with companies, agreements to test and hold confidential the kind of information that is required in pre-clinical agreements, and clinical agreements that allowed companies to test their clinical products in NIAID’s clinical system “in ways they are comfortable with.” The Institute had many resources to expedite clinical testing, from proper reagents to standardized tests and various specimens. The Institute had also had CRADAs (cooperative research and development agreements) with both large and small companies, most of which are now large companies. All of these steps encouraged companies to work with NIAID to achieve company objectives.
Larger Issues Requiring Public-Private Collaboration
She listed several larger issues on which NIAID was prepared to work collaboratively with industry:
-
Liability: In the case of interventions, especially for vaccines that are to be given to healthy people, the potential for liability suits becomes a substantial barrier for companies that might want to invest in product development. She said that it costs a company nearly a billion dollars to develop a vaccine, and that such products have not historically had a high potential for economic return. When liability risk is added to that “huge, huge investment,” the choice to pursue a vaccine product adds up to a considerable economic risk to the company. NIAID’s help in planning, testing, and consulting can help reduce this risk.
-
The question of control: Each company has a different philosophy on the ownership of their products. The Institute was prepared to work out well-defined agreements that specify exactly what the company wants to do and how the Institute can help—while staying within the customs, abilities, and restrictions of government behavior.
-
Building a clinical infrastructure: Many small companies make the reasonable decision not to make substantial investments in clinical infrastructure until they actually have a product with commercial promise. By that point, however, it may be too late to begin building that infrastructure. A company is best positioned if it has already begun thinking about how it wants to develop a product to the clinical stage. NIAID is prepared to help the company to do its planning early by being a partner and advising about the steps that must be taken to comply with regulations.
She emphasized that public-private partnerships can succeed only when both sides understand them and are able to keep to their commitments. NIAID had initiated CRADA agreements that had been broken by companies that unexpectedly changed organizational structure. For its part, NIAID has also encountered difficulties in staying with an agreement when government goals or responsibilities changed with a new annual budget cycle. Some breakdowns are inevitable, she conceded, but “we each have to be as fair and committed as we can.”
Dr. Heilman concluded by mentioning a potential difficulty for partnerships that has been brought to her attention by her industrial colleagues. The FTC regulations presently bar certain kinds of cooperation and sharing among companies, and companies have expressed their concern that this might constitute an impediment to public-private partnerships of the kind envisioned by NIAID. She suggested that this may be of importance to the committee.
DISCUSSANTS
Henry (Pete) Linsert, Jr.
Martek Biosciences Corporation
Pete Linsert, chairman and CEO of Martek, said he would illustrate the value of the SBIR program to his company by summarizing the history of Martek and the commercial success of the two compounds it markets. Martek is headquartered in Columbia, Maryland, and has research facilities in Boulder, Colorado, and a fermentation plant in Winchester, Kentucky.
“Martek is prospering these days,” he began, “largely as a result of four SBIRs. I doubt if Martek would be here without them.”
The Story of Martek
The story of Martek began in the mid-to-late 1980s, when scientists were studying the physiological value of two fatty acids: arachidonic acid (ARA) and docosahexaenoic acid (DHA). About 60 percent of the brain is composed of fats, and the most abundant of those are DHA and ARA, in that order. DHA also makes up about 60 percent of the components of the retina, especially the rods and cones; it is also found in heart, blood and muscle cells and, in males, in the testes and sperm. ARA is the principal omega-6 fatty acid in the brain and is abundant in other cells throughout the body. ARA is important for proper brain development in infants and is a precursor to a group of hormone-like substances called eicosanoids, which are important in immunity, blood clotting and other vital functions.
Most humans ingest ARA in common foods, such as meat, eggs and milk, but DHA is found in only a few foods, such as fatty fish and organ meat. Both
DHA and ARA occur naturally in breast milk and exhibit health benefits that extend from prenatal development through adult life.
In the late 1980s, scientists at the National Institutes of Health issued a call for proposals for new sources of these fatty acids through the SBIR program. Martek, a spin-off of Martin-Marietta, had previously worked, as part of Martin, on a NASA contract to study the use of algae in the space station, both to reduce carbon dioxide in the station’s atmosphere and to provide a nourishing food source for its inhabitants. The Martek scientists, working in suburban Baltimore, knew that algae could make DHA and fungus, ARA. They proposed the development of a process to do this commercially. The company won a Phase I SBIR grant for DHA in 1988, and after meeting the milestones required by the contract, won a Phase II grant to continue development. A similar sequence of events occurred for ARA. The DHA project was later awarded the 1995 SBIR Award of the Year for both biotechnology and for all SBIR classes.
Capturing a New Market
Today Martek is the sole large-scale commercial manufacturer of these fatty acids, which are sold in various forms, including brands of infant formula, in 70 countries. According to Mr. Linsert, double-blind, placebo-controlled studies show that infants who are fed formula with DHA show an IQ that is six to seven points higher than infants fed formula without DHA, and they also develop better eyesight. The United States approved the use of DHA in formula only this year—making it one of the last countries to do so—and formula containing DHA from Martek is now sold nationwide. The company had also applied for the use of DHA as supplements for pregnant and lactating women as studies done on DHA supplemented women in pregnancy and lactation show improved mental development in their infants.
Mr. Linsert concluded by saying that the success his company derived from two Phase I and two Phase II grants from the NIH “had consequences beyond anyone’s expectations.” “Babies’ lives will be improved, future humans will be improved, and we will be touching millions of lives around the world.”
Gail Cassell
Eli Lilly and Company
Dr. Cassell said that she had served as Vice-President for Scientific Affairs at the University of Alabama at Birmingham School of Medicine for 30 years and chaired the department for 10 years. In academic life, she said, she had collaborated with small businesses that received SBIRs, and that she had become “a fan of what this program can do to enhance technology transfer.” As a department chair in a medical school, she saw the impact of faculty who were able to transfer a technology to the private sector, start small companies, and apply for SBIR
grants that led to the success of the companies. She had served on the board of the university research foundation from its outset, where she “developed a feel for what it takes to make small businesses progress and thrive.” After moving to Lilly, she had the opportunity to end-license a number of potential products that were the outcomes of funding through the SBIR program, especially those of the NIH, primarily NIAID.
The Economic Impact of Licensing
During the fall of 2002 she was asked to give a presentation in Tokyo to the U.S. ambassador and a group of people from the U.S. and Japan on the role of biotechnology. She talked about not only scientific advances but also the economic impact of biotechnology in the United States, and specifically the economic impact of universities. She used the University of Alabama at Birmingham (UAB) as an example to describe technology transfer from universities and the impact she had observed on local economies. UAB is relatively small compared to some biotech powers, such as the University of California at San Francisco (UCSF), where approximately 400 patents in biotechnology had resulted in licensing and royalty fees totaling approximately $495 million in income for the university. UCSF is now the second largest employer in the Bay Area, where it had an economic impact amounting to about $2 billion. The economic impact of UAB was also considerable, she said, and the university had grown to be the largest employer in the state, with an economic impact on the local economy of about $1 billion each year.
The Importance of Bayh-Dole
Dr. Cassell suggested that it was important, in trying to assess the impact of SBIR grants, not to consider them in isolation, but as part of a continuum. In her Tokyo talk, she said that she described the Bayh-Dole Act of 1982 and its impact on technology transfer.14 At the time of her talk, she did not know that Japan had recently revised national policies in favor of larger support for basic research; she said that Japan had come to invest considerably more in basic research than any other country, including the United States. Japan had not, however, succeeded in translating that support into global competitiveness. As a result, the country has recently changed its legislation to mimic the Bayh-Dole Act, setting aside money for interactions among small companies, large companies, universities, and gov-
ernment labs. She said that many other countries, recently including Ireland and Korea, had visited the U.S. to learn more about Bayh-Dole and its impact.
As a result of the well-studied impact of Bayh-Dole, she recommended that the upcoming SBIR study make careful attempts to quantify the impact of technology transfer from academic institutions, calculate how many small companies actually result from academic technology transfer, and estimate the overall impact of SBIR funding. Doing this properly, she suggested, would require a better understanding of the continuum and the synergies among granting mechanisms. She seconded Dr. Heilman’s assertion that the needs of biodefense have placed unprecedented importance on the need for interaction between institutions and creation of public-private partnerships to stimulate new research and tools to counter terrorism. Because of the complexity of biodefense, and the urgent need for countermeasures, she said, the nation needs to engage the entire spectrum of industry.
Setting Realistic Expectations for “Success”
Her final remarks concerned the need to think about metrics for SBIRs. Most of the SBIRs she had reviewed personally while on an NIH study section had been related to the development of diagnostics, products, medicines, biologics, and vaccines. In these areas, she said, the failure rate for industry—from target identification to the launch of a product—was approximately 90 percent. This held true even under the best of conditions, and even for projects in which large companies had invested billions of dollars. In setting metrics for SBIR projects, she said, it was important to give a realistic expectation of “success,” especially for small companies investing in biotech. Most of these small companies that NIH would be funding are narrowly focused, so that the failure rate is likely to be especially high.
Even in this area of risky research, however, Dr. Cassell asserted that “the successes will more than pay for the failures.” She recalled a suggestion by the Ad Hoc Group for Biomedical Research and the Joint Economic Commission that a new cancer drug that would only improve life expectancy in one-third of cancer patients would be worth a trillion dollars to Americans—a figure larger than the national debt. In light of the expected outcomes of SBIR-supported companies, she concluded, “I would argue that investment in NIH overall is well worth it, and that investment in the SBIR program also is well worth it.”
Maryann Feldman
Johns Hopkins University
Dr. Feldman began by soliciting the “advice and counsel” of all participants as the complex SBIR study goes forward. She noted that in studying 10 different agencies, it would require extensive input to understand the differing contexts of the SBIR program within each agency. As an example, she cited the “NIH umbrella” which comprises 27 different centers and institutes, 23 of which participate in the SBIR with many differences and also “a lot of synergies.”
A Mission Relevant to SBIR
The mission of NIH, as she described it, was to “expand fundamental knowledge of living systems.” The Institutes provide a comprehensive program of intramural research, extramural research, and grants that together strengthen the biomedical capability in this country. NIH is not only the largest funder of basic research in the world, but also develops strategies for improving the diagnosis, treatment, and prevention of diseases. It is these strategies that lead most directly to commercialization and have greatest relevance to the SBIR.
Despite the diversity of the NIH centers, she said, there are commonalities that would assist in studying the program. One is a uniform peer review process for the SBIR program run by the Center for Scientific Review. This center is “very comfortable” for academic scientists, who are accustomed to using peer review in many aspects of their work. There are also common NIH databases (CRISP, IMPAC)15 that provide resources and common criteria for judging success that transcend different programs.
The Challenge of Measuring “Invisible” Activities
She stressed the unique challenge of studying the success of the NIH in commercializing various products. That is, both drug products and commercial drug companies vary widely in their payoffs and outcomes. Under the best of conditions, the lag time between discovery and commercial realization of a drug candidate is very long. She cited a study estimating that it takes 10 to 15 years to develop a new drug and costs $800 million to take it through the FDA approval process. Even after approval, the path to market is usually circuitous and uncertain. A firm might win an SBIR award, but many firms merge, fail, and change their names before a product reaches the market. When firms change their names, it is difficult to track their progress. In addition, while companies often fail, key individuals tend to move on to other firms where their success in the previous firm may translate into valuable but “invisible” activity. In short, many aspects of the process of discovery and experimentation that are key to the biomedical enterprise are difficult to track.
Dr. Feldman said that the study panel, through its understanding of this complexity, would try to understand in the broadest sense how the NIH SBIR program has contributed to expanding our fundamental knowledge of health. This would include not only the metrics of products, patents, licenses, sales, and other traditional SBIR measures of commercial success, but also publications, citations of those papers, the contribution of companies in training human capital, and other ways of measuring the larger spillover effects of the program.
Beginnings of the Biotech Industry
She said that the biotech industry essentially had begun when the Cohen-Boyer Patents, filed in 1973, established the industry’s commercial potential. Subsequently, the concept of patenting life forms was upheld in court cases. The industry grew through academic breakthroughs, the work of “star scientists in dedicated biotech firms,” and the use of intellectual property licenses from universities. The NIH SBIR program, which began in 1983, provides a mechanism to translate academic science into commercial discoveries.
The current SBIR study, she said, could bring fascinating and useful information about the biomedical revolution and the role of the NIH SBIR program in advancing that revolution. It would also provide an important new perspective on how individual researchers move from the support of university research grants toward the support of SBIR grants, using this new mechanism to translate their academic science into useful products.
Tracking Award Recipients
As a test of the kinds of data that might enhance the value of the study (after all, she said, “to a researcher, having data is as fundamental as having air”), she had asked her research assistant to review any available information about the 1983 SBIR award winners. Using the CRISP on-line data, she found that 120 companies had received SBIR Phase I awards in 1983, and asked the question, Where are they now? It turned out that seven (6 percent) were publicly traded companies—“names you would recognize” (see Table 2). Twenty-one (18 percent) had changed their identities through merger or acquisition, but could still be followed; 38 were still independent. Fifty-four (45 percent) had ceased to exist as companies, but for 80 percent of these, interim data existed that showed their last SBIR award or patent application.
TABLE 2 Publicly Traded Companies—1983: NIH SBIR Awards
|
Company |
Year Founded |
Employees |
Revenues ($) |
|
Genzyme Corporation |
1981 |
5500 |
1 billion |
|
Maxwell Laboratories, Inc. |
1965 |
483 |
78 million |
|
Enzon |
1981 |
127 |
76 million |
|
Bioanalytical Systems, Inc. |
1975 |
260 |
25 million |
|
Surmodics, Inc. |
1979 |
143 |
23 million |
|
Bioqual, Inc. |
|
|
13 million |
|
PAR Technologies Corporation |
|
|
118 million |
In conclusion, she said that this kind of “industrial forensics” offered a basis for thinking “that we may be able to get in and look at these companies in more detail.”
DISCUSSION
Mr. Turner, who played a central role in framing each of the amendments to the SBIR legislation in 1982, applauded the efforts and understanding of the research team so far, and called it “quite an undertaking—a massive charge, looking at such diverse programs.” He also affirmed the difficulty of tracking companies that change rapidly. He cited the case of Mr. Linsert and his company Martek as an example of a successful firm that emerged around a single individual whose initial work on a product had begun for quite different reasons. “One trick,” he said, “is how we find the Pete Linsert’s out of all the SBIR applicants. When he got his first SBIR grant he did not know what he had. His company would not have made it had he not gotten the grant, but it did, and we have a wonderful company because of that. How can we direct more of our funds in that direction?”
A Few Successes Outweigh Many Failures
He agreed with Dr. Cassell that the study panel should not be overly concerned about the high failure rate of SBIR companies because “a handful of successes in these programs more than compensate for all the failures. He echoed Dr. Feldman’s view that determining the value of an SBIR company is a complex undertaking, as illustrated by the frequency of company changes: “Just because a company disappears doesn’t mean we don’t gain benefit from its research.”
In reviewing the meaning of the original two-page statute that Congress issued in 1982, Mr. Turner said that many of its rules were based on the assumption of the linear model of research and development—the idea that knowledge moves in a straight line from the laboratory to the marketplace. Echoing Ms. Goodnight’s earlier comment, he noted that we now understand that knowledge moves in more complex and circular fashion, with numerous feedback loops between the marketplace and the laboratory. A challenge for the study panel would be to recommend ways to amend some of the “statutory impediments” that reflected the earlier, linear model, that we now know impact the overall program effectiveness, such as narrow time frames, fixed amounts of money, and a “one-size-fits-all” program. “How do we sort of bend or change the rules,” he asked, “to nurture the best ideas and make winners into tremendous winners?”
Congress Would Like to Improve Effectiveness
He said that one thing Congress anticipates in the study is a baseline review. The purpose of such a review would be to raise basic questions about the quality of the research, its value to the federal government, its economic benefits, and small business’s “share of the pie.”
In addition, Congress would like to see two sets of recommendations. The first set would respond to the Government Performance and Results Act (GPRA). That is, how efficient and effective is the program? What improvements should be made to improve efficiency and effectiveness? One of the goals of GPRA is to measure the outcomes of government programs. Even though the SBIR program does not receive an annual review by the Office of Management and Budget, Congress does consider it a program that falls under the GRPA statutes. He asked the study panel for “anything you can do to help us understand how to improve efficiency” and also suggested that there was no reason to wait until the end of the study to use a result that becomes apparent early.
Strengthening Phase III
The second set of recommendations that Congress would like to see, he said, is “bottom-line recommendations.” Small business has much to offer, and yet the small-business sector in general is not respected in some parts of government, especially in the procurement arena. He noted that his colleagues on the Small Business Committee had tried for years to raise the awareness of the potential contributions of small business and to “force more doors open for small business.” He said that anything the study panel could do to help strengthen “Phase III” and make it as important as the first two phases may be the most valuable contribution it could make to the SBIR program.
He elaborated on the premise that the statute itself had been written during a different era, and that it might very well contain ideas that should not be pursued today. The statute was also written as part of a political process, which meant that it contained ideas that were generated by a small minority of participants—ideas that had to be included to ensure passage through the legislature. He suggested that the current study would provide an excellent opportunity to reinforce the best of the statute and to add positive correctives.
The Challenge of Many SBIR Programs
Finally, Mr. Turner addressed the challenge of studying all facets of a complex and multi-faceted program. The study panel was charged with examining the five agencies with large SBIR programs, as well as the five with much smaller programs. And yet in practical terms, Mr. Turner suggested, there are probably 40 to 50 SBIR programs in all. This is because NIH alone has 23 SBIR “sub-
programs,” and some of them are larger than the entire SBIR programs of several agencies; similarly, a few other agencies have a large number of separate programs. He conceded that there was no practical way to study all 40 or 50. The most effective compromise would probably be to spend most of the panel’s time on the largest programs, searching for suggestions on better ways to run them, and to spend at least some time on the smaller programs in the event that good suggestions are to be found among them as well.
“It will be great if you have suggestions for all the programs,” he concluded, “but we realize that resources are limited. We know that we have a wonderful team and we’re looking forward to the results.”
Stimulating Translational Research
Greg Millman of the National Institutes of Health commented that a key aspect of the NIH SBIR program was its ability to stimulate translational research. He noted that formation of companies like Martek Biosciences was an outcome, but not the overall objective. The objective was to have as much translational research as possible, of which only a few will lead to the Martek-like successes; many will fail.
To measure translational research, he suggested, one could do a simple experiment: first, find the funding of NIH by ZIP code, which is available on the NIH web site. Second, plot the nation’s major academic centers. Third, look at the distribution of SBIR companies. They tended to cluster around the same ZIP codes and academic centers, which suggested a great deal of interaction between the NIH SBIR companies and major academic centers. That interaction, he said, is a truer measure of success than a tally of functional companies.
Gail Cassell of Eli Lilly and Company emphasized that the SBIR program performed an essential function in providing “a great training mechanism for young investigators” who would stay in industry. Even if their company failed, she said, they would go to other companies. Smaller companies were more willing to take risks in hiring young people, which is something that should be considered by the study panel in trying to assess the impact of SBIR.
Agency Studies of SBIR
Michael Borrus of the Petkevich Group, LLC, returned to Ms. Goodnight’s point that some of the panel’s evaluation would necessarily be less quantitative and more qualitative. He noted that she had described some of the broader goals of the research program at NIH, specifically the SBIR component, as “translating medicine from the test tube to the medicine cabinet,” generating disruptive technologies, and meeting specific national needs that are high priority areas, such as biodefense. He recalled that DoD had done considerable self-assessment of its own program, and asked whether the NIH had also collected quantitative data to
measure outcomes at NIH against its specific goals. He also asked whether the NIH had analyzed such data and whether it was available to the public.
Jo Anne Goodnight of NIH responded to the three questions with “Yes, yes, and yes.” She said that a number of studies had already been done on the program, basically measuring it against the goals identified in the initial legislation. Those studies were in report form and available from the GAO, SBA, and others. The studies also showed that NIH, which is rarely if ever the customer, had one of the highest rates of commercialization among SBIR agencies.
In addition, the NIH did collect extensive data on its own R&D investments, and was at the end of a study of its Phase II awardees. She said that the agency would be happy to share those data and outcomes. Some other agencies, including some of the smaller ones, had also done their own SBIR evaluations. She suggested that their data should be factored into the recommendations that emerge from the study, because those recommendations would affect all ten SBIR agencies as well as the SBA—not just the five largest. The other reason to look at all ten, she said, was that it was now possible to fund a Phase II award to a company whose Phase I funding came from a different agency. She demonstrated how this new practice could lead to erroneous study results unless all agencies are included. For example, one might see that a company received only a Phase I grant from a particular agency and conclude that it failed to get a Phase II grant. In fact, that company might have received a Phase II award from a different agency and gone on to be successful.
“We do have data like this,” she reiterated, and “we have success stories. We are more than happy to share them. That’s the point of assimilating the data the agencies have already collected and not going back to the companies to ask the same questions.”
Extending the Study
James Gallup, SBIR program manager for the U.S. Environmental Protection Agency, referred to Mr. Turner’s comment that it might be impractical for the study panel to evaluate all ten SBIR agencies. He said that he agreed with Ms. Goodnight’s reason for trying to look at all of the ten. As the EPA SBIR program manager, he said, he could see “really significant” differences between the large and small agencies, and saw “some things the small agencies can do that will help us greatly.” He called for clear guidance from the very beginning of the study about just how many agencies should be included.
Jim Turner of the House Science Committee responded that “we’ve thrown an impossible job at the committee, and I’m sure it will be sorted out in a wise way, probably through a compromise.” He did point out that the largest programs are some 200 to 300 times the size of the smallest programs, and that it would be impractical to spend equal amounts of time on the smallest ones. He thanked the panel for taking on this challenge.





















