3
Overview of Health Effects, Air Quality, and Emissions
INTRODUCTION
New Source Review (NSR) programs are an important element in a large, complex, and evolving regulatory system. To understand how changes in NSR programs are likely to affect air quality and human health, it is necessary to examine the broader air pollution issues and the contributions that facilities subject to NSR may make.
This chapter begins with a brief discussion of the National Ambient Air Quality Standards (NAAQS) process for the criteria pollutants and then describes what is known about the health effects of criteria pollutants and volatile organic compounds (VOCs) regulated through NSR. It also describes progress that has been made toward NAAQS attainment and where further work is needed. The chapter discusses the inventory of emission sources, including stationary sources subject to NSR and the emissions contributed by the various industrial sectors. Because it is important to assess which existing stationary sources are affected by the NSR changes under review by this committee, the vintage of the facilities is considered.
NATIONAL AMBIENT AIR QUALITY STANDARDS FOR CRITERIA POLLUTANTS
As discussed in Chapter 2, the Clean Air Act (CAA) requires the U.S. Environmental Protection Agency (EPA) to set primary NAAQS for
selected air pollutants considered harmful to public health, including the health of “sensitive” populations such as people with asthma, children, and the elderly. Secondary NAAQS are intended to protect public welfare, such as crops and other vegetation. EPA has, for the most part, set the secondary standards at the same levels as the primary standards.1
EPA has set NAAQS for six pollutants (see Table 3-1): nitrogen dioxide (NO2), ozone (O3), carbon monoxide (CO), sulfur dioxide (SO2), lead (Pb), and particulate matter (PM; PM10 and PM2.5).2 Units of measure for the standards are parts per million (ppm) by volume, milligrams per cubic meter of air (mg/m3), and micrograms per cubic meter of air (µg/m3). Pollutants for which there are NAAQS are known as criteria pollutants because EPA prepares “criteria documents” for them describing their sources and effect.
Each NAAQS is composed of an indicator (e.g., PM2.5), a level or concentration (e.g., 65 µg/m3), an averaging time (e.g., 24 hours), and a statistical form (e.g., 3-year average of the 98th percentile of 24-hour concentrations at each population-oriented monitor). Although the concentration of the standard tends to receive the most public attention, the averaging time and statistical form are also important determinants of stringency.
The CAA mandates that the NAAQS be reviewed at 5-year intervals, although in practice, the interval often takes much longer—sometimes more than a decade. A typical review cycle for each pollutant involves developing a criteria document, a compendium of current knowledge about the pollutant, followed by development of a “staff paper,” which summarizes knowledge considered most pertinent to decision making and proposes a range of actions for the EPA administrator’s consideration. The range of actions and preferred options may include no change to the existing standard. Both documents are made available to the public for comment and are formally reviewed by the independent Clean Air Scientific Advisory Committee (CASAC) of the EPA Science Advisory Board, which complements the Committee with additional experts to form a review panel for each pollutant. The EPA administrator, after a lengthy period of public comment, makes the final decision on the standard to be promulgated. Although CASAC approval of (“closure on”) the two documents is sought before the EPA administrator proposes
TABLE 3-1 National Ambient Air Quality Standards
|
Pollutant or Indicator |
Primary Standards |
Averaging Times |
Secondary Standards |
|
Carbon monoxide (CO) |
9 ppm (10 mg/m3) 35 ppm (40 mg/m3) |
8-houra 1-houra |
None None |
|
Lead (Pb) |
1.5 µg/m3 |
Quarterly average |
Same as primary |
|
Nitrogen dioxide (NO2) |
0.053 ppm (100 µg/m3) |
Annual (arithmetic mean) |
Same as primary |
|
Particulate matter <10 µm aerodynamic diameter (PM10) |
50 µg/m3 150 µg/m3 |
Annualb (arithmetic mean) 24-houra |
Same as primary |
|
Particulate matter <2.5 µm aerodynamic diameter (PM2.5) |
15 µg/m3 65 µg/m3 |
Annualc (arithmetic mean) 24-hourd |
Same as primary |
|
Ozone (O3) |
0.08 ppm 0.12 ppm |
8-houre 1-hourf |
Same as primary Same as primary |
|
Sulfur oxides (SO2) |
0.03 ppm 0.14 ppm —— |
Annual (arithmetic mean) 24-houra 3-houra |
—— —— 0.5 ppm (1,300 µg/m3) |
|
aNot to be exceeded more than once per year. bTo attain this standard, the expected annual arithmetic mean PM10 concentration at each monitor within an area must not exceed 50 µg/m3. cTo attain this standard, the 3-year average of the annual arithmetic mean PM2.5 concentrations from single or multiple community-oriented monitors must not exceed 15 µg/m3. dTo attain this standard, the 3-year average of the 98th percentile of 24-hour concentrations at each population-oriented monitor within an area must not exceed 65 µg/m3. eTo attain this standard, the 3-year average of the fourth-highest daily maximum 8-hour average O3 concentrations measured at each monitor within an area over each year must not exceed 0.08 ppm. f(a) The standard is attained when the expected number of days per calendar year with maximum hourly average concentrations above 0.12 ppm is ≤ 1 based on a 3-year average. (b) EPA will revoke the 1-hour NAAQS 1 year after the effective date of designating attainment and nonattainment areas for the 8-hour O3 NAAQS. The effective designation date for most areas is June 15, 2004 (40 CFR 50.9; see 69 Fed. Reg. 23996 [2004]). Source: EPA 2004d. |
|||
a standard, CASAC does not “approve” the standard promulgated by the agency.
Successful promulgation of a NAAQS typically is followed by a multiyear period before the standard is fully implemented. Monitoring data typically are used to determine areas of nonattainment, which may take some time if the pollutant indicator or averaging time differs from that of the preceding standard. EPA designates an area as one of three categories:
-
Nonattainment—any area that does not meet the NAAQS or that contributes to violations of a NAAQS in a nearby area.
-
Attainment—an area that meets the NAAQS and does not contribute to the violation of a NAAQS in a nearby area.
-
Unclassifiable—an area that cannot be classified on the basis of available information as meeting or not meeting the NAAQS for a pollutant.3
HEALTH EFFECTS OF AIR POLLUTION
This section provides brief descriptions of the status of health concerns associated with the criteria pollutants, excluding lead.4 In addition, summary information is given for VOCs, which are also regulatory targets but are managed differently from the criteria pollutants.5
A large and growing body of published epidemiologic and toxicologic research establishes that the criteria pollutants are associated with a variety of health effects, including increased occurrence of cardiopulmonary morbidity, cardiopulmonary and cancer mortality, effects on birth outcomes (low birth weight, small for gestation age), and impaired growth of lung function in children. The associations are most consistent for children (particularly those with asthma) and for the eld-
erly (particularly those with underlying cardiovascular diseases and diabetes). In contrast to the clarity of the overall associations, the extent to which the various health effects can be attributed to any given pollutant or combinations of pollutants is less clear; this limitation must be considered when examining the quantitative estimates of association attributed to any single pollutant. In the case of PM, the problem is compounded by the fact that PM is a complex physical-chemical mixture whose components likely interact with other criteria pollutants (e.g., O3) in a number of ways that may affect health outcomes. Characterizing the overall pollutant mixture with respect to its health implications is a major future challenge for air-pollution-related health effects research.
For two of the pollutants (NO2, SO2), the discussion that follows focuses on what is known about their direct associations with health outcomes. However, each also makes a substantive contribution to the formation of PM2.5 in the atmosphere (also known as secondary PM2.5) and undoubtedly contributes to some portion of the health effects attributed to PM2.5. In addition, NO2 plays a central role in the chemistry of O3 formation (see Box 3-1).
The committee refers only briefly to the problem of investigating and estimating threshold effects from population data, which is important in determining whether health effects would be anticipated in NAAQS attainment areas. There is general agreement that detection of thresholds for various pollutants is difficult with population data because such data represent weighted averages (weights unknown) of pollutant-host interactions. By and large, the body of personal exposure data is insufficient to precisely determine pollutant thresholds.
Carbon Monoxide
Concerns for the health effects of inhaled CO historically have focused on the various manifestations of its greater affinity for binding to blood hemoglobin than oxygen’s, thus reducing blood oxygen concentrations. Accordingly, the current NAAQS for CO is based on preventing significant health impacts by limiting blood carboxyhemoglobin concentrations (in normal nonsmokers) to approximately 2% or less (EPA 2000). The health effect driving the current NAAQS is angina (chest pain) in exercising individuals with coronary artery disease. The onset of angina signals ischemia (lack of sufficient oxygen) in heart muscle and not only limits the level and duration of exercise but also can increase the risk for arrhythmias and death.
|
BOX 3-1 Air pollutants are often characterized by how they originate: pollutants emitted directly into the atmosphere are called primary pollutants; those formed as a result of chemical reactions within the atmosphere are called secondary pollutants. Many precursors are known to form secondary pollutants within a day or two of emission, while being transported downwind in the atmosphere. The formation of O3 and PM in the atmosphere, as secondary pollutants, involves complex chemical and physical interactions of multiple pollutants from natural and anthropogenic sources. This complexity has resulted in ongoing difficulties in measuring and regulating those pollutants. O3 is formed during the production of NO from NO2 in the presence of products of photochemical reactions of VOCs (labeled RO2 in Figure 3-1). The reaction is self-perpetuating (or catalytic) in the presence of sunlight because NO2 is photochemically reformed from NO. In this way, O3 is controlled by both NOx and VOC emissions. The complexity of these interacting cycles of pollutants means that incremental decreases in one emission may not result in proportional decreases in O3. Although these reactions and interactions are well understood, variability in emission source operations and meteorology creates uncertainty in the modeled O3 concentrations to which downwind populations may be exposed. Ambient PM is produced by a variety of emission sources. Some particles are emitted directly into the atmosphere from sources (such as metals from industrial processes). Secondary particles are produced by reactions that involve SO2, NOx, and VOCs and ammonia. Oxidizing agents, produced by O3, VOCs and NOx, convert SO2 to sulfuric acid and then to sulfate; they also convert NOx to nitric acid and then to nitrate. These stable products form into particles and typically are neutralized by ammonium-forming salts. The same reactions in which RO2 compounds are produced and O3 is formed also make organic components that condense into particles. Aqueous-phase reactions in fog and cloud droplets also produce organic and inorganic products that contribute to mass in the condensed phase. Figure 3-1 summarizes the role that these four types of emissions play in producing PM. |
Epidemiologic research since the last review of the CO NAAQS has provided some evidence of associations between increases in CO concentrations and increased rates of cardiopulmonary mortality and morbidity. Several studies focused on PM, but multipollutant models also appeared to show significant relationships between CO and mortal ity (EPA 2004e). Some studies suggest that in such cases CO serves as a
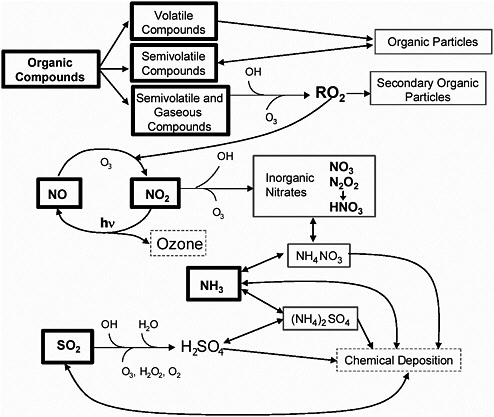
FIGURE 3-1 Chemical links between O3 and PM formation processes. Major precursors are shown in the boxes with thick sides. Secondary particle components are shown in the boxes with thin-sided solid sides. Source: NARSTO 2004. Reprinted with permission; copyright 2004, Cambridge University Press.
marker for mobile source contributions of several pollutants and perhaps even as a surrogate for toxic fine particles (putatively, those emitted directly from combustion sources). Whether CO affects mortality directly or serves as an indirect indicator of exposure to other toxicants is not resolved; however, the statistical link between CO and mortality cannot be disregarded.
Recent research has also demonstrated statistical associations between current ambient CO levels and low birth weight (Ritz and Yu 1999). This finding has not been replicated sufficiently to confidently evaluate either exposure-response relationships or the specificity of the effect of CO, but there is sufficient evidence to warrant concern.
CO can also exert central nervous system effects, including reductions in hand-eye coordination (driving or tracking) and in attention and
vigilance. Present data, however, suggest that ambient concentrations are unlikely to cause such effects unless ambient exposures are superimposed on exposures from other sources, such as heavy smoking or hot spots caused by extreme traffic emissions (EPA 2000).
Nitrogen Oxides
The term nitrogen oxides (NOx) commonly refers to the sum of nitric oxide (NO) and nitrogen dioxide (NO2). The symbol NOy commonly refers to the sum of NOx and other oxidized nitrogen compounds, such as nitric acid (HNO3), nitrogen trioxide (NO3), dinitrogen trioxide (N2O3), dinitrogen tetroxide (N2O4), dinitrogen pentoxide (N2O5), and peroxyacetylnitrate. Nitrous oxide (N2O) is commonly excluded. NO and NO2 have numerous sources, especially combustion emissions, and are present in ambient air in greater concentrations than the other species. NO2 is toxic by virtue of its oxidation potential and has received the greatest health research and risk assessment attention. NO has lower biological toxicity but is increasingly recognized as a mediator or intermediate of cellular signaling and biochemical processes, both beneficial and detrimental. NOy is of concern not only for its toxicity but also because of its participation in the formation of O3 (together with VOCs and sunlight) and secondary PM (nitrates).
NOx constituents are oxidants and thus may contribute to the wide range of respiratory health impacts thought to be associated with inhaling oxidants or mediated by oxidation pathways. Experimental and accidental inhalation of high NO2 concentrations have been shown to cause respiratory symptoms (e.g., cough and wheezing), reduced lung function, and increased airway responsiveness of both normal and asthmatic individuals. The evidence for these effects at common ambient concentrations (less than 0.05 ppm based on an annual average) is equivocal (EPA 1995a). Among the many recent epidemiologic studies of the effects of particles and co-pollutants on respiratory and cardiovascular outcomes, NOx has seldom arisen as a significant factor. Prolonged high-level exposures of animals (e.g., 10 ppm and higher) have caused emphysema-like changes in some, but not all, studies. Tissue changes are unlikely at ambient exposure concentrations; however, the recent finding that ambient exposure to O3, also an oxidant gas, affects lung development, suggests the possibility that NOx may contribute to similar effects. High indoor exposures of children (often associated with gas stove usage) have been linked to reduced resistance to respiratory infections and increased
likelihood of respiratory illness (Hasselblad et al. 1992), including wheezing and persistent coughing (van Strien et al. 2004). In addition, recent evidence suggests that NO2 exposures increase the severity of virus-induced asthma exacerbations (Chauhan et al. 2003). Although unconfirmed, it is possible that NOx emissions may contribute to the associations between respiratory illness in children and proximity to heavily traveled roadways (Wjst et al. 1993; Oosterlee et al. 1996; Brunekreef et al. 1997).
Research since the last review of NOx NAAQS indicates that NOx may retard lung growth in children (Gauderman et al. 2004). Differences between the rates of lung growth in children living in areas of relatively greater and lesser pollution were statistically associated with NO2 and also with airborne acid and fine particles (PM2.5), substances to which NOx contributes. NO2 (along with other air pollutants) also may increase airway responsiveness to allergens (Rusznak et al. 1996; Jenkins et al. 1999).
Although it is both plausible and probable that NOx contributes to the impacts of air pollution on public health, it is likely that NOx species affect health more strongly via their contribution to the formation of O3 and secondary PM than via their direct effects.
Ozone
O3 (along with PM) currently is one of the criteria pollutants of greatest health concern. There is little doubt that public health is measurably affected at exposure levels that exist in many areas in the United States (EPA 1996).
O3 is an oxidant gas emitted directly from sources in only small amounts (e.g., electric motors); ambient O3 is overwhelmingly formed in the atmosphere from reactions involving NOx, VOCs, and ultraviolet light (sunlight) (see Box 3-1). It is one of several photochemical oxidants. In the respiratory tract, O3 interacts with polyunsaturated fatty acids, electron donors (e.g., ascorbate, vitamin E), and the thiol, aldehyde, and amine groups of low-molecular-weight compounds and proteins. O3 is very reactive, and it is likely that most harmful biological effects result from the products of initial reactions of O3 with surface fluids and cell walls rather than from direct interactions between O3 and intracellular targets. For people with chronic obstructive pulmonary disease or asthma, the antioxidant content of the lung lining can be reduced,
potentially contributing to increased sensitivity to O3 (Mudway and Kelly 2000).
O3 has been associated with a spectrum of adverse effects, but the effects primarily driving the current standard are pulmonary function decrements in exercising children, adolescents, and susceptible (e.g., asthmatic) individuals. O3 causes a neurogenic (reflex) inhibition of maximal inspiration that reduces maximal forced expiratory volume during lung function tests and ventilatory capacity during exercise. O3 also causes increased airway responsiveness (constriction) to other materials (e.g., airway constricting drugs, allergens). Although the degree of increased responsiveness may not differ much between healthy people and people with asthma, the much greater baseline responsiveness of people with asthma makes the O3-induced additional decrement potentially more serious. High O3 levels are associated with more clinic visits and hospitalization of people with asthma and other preexisting respiratory diseases; there is also some evidence for increased deaths among these subpopulations (Thurston and Ito 2001, Bell et al. 2004). The National Morbidity, Mortality, and Air Pollution Study (Bell et al. 2004) finds a similar relative risk for O3 above and below the NAAQS, although differences between personal O3 exposures and ambient concentrations make it difficult to detect thresholds (Zhang and Lioy 1994, Brauer et al. 2002).
O3 also acts as a respiratory and eye irritant, giving rise to noticeable, if not life threatening, discomfort that limits activity and reduces quality of life. O3 may impair defenses against respiratory pathogens and inhaled particles. There is evidence (EPA 1996) that O3 slows the clearance of particles from the lungs by damaging cilia and retarding the activity of macrophages; however, it is not clear whether this occurs in humans at actual exposure concentrations. Furthermore, Pope et al. (2002) observed a positive association between long-term exposure to summertime O3 and increased risk of premature death in cohort studies, although the relationship was not statistically significant. Bell et al. (2004) found a statistically significant association between short-term changes in O3 and mortality on average for 95 large U.S. urban communities.
Research since the 1996 O3 Criteria Document (EPA 1996) reviews indicates that O3 retards lung growth. Exposures of animals have produced alterations in the development of lung structure during growth (Schelegle et al. 2003). Epidemiologic studies have demonstrated that entering college students from areas with high O3 concentrations have
smaller lungs and less lung function than students from areas with low O3 levels (Kunzli et al. 1997).
Particulate Matter
PM encompasses a physically and chemically diverse class of ambient air pollutants of both anthropogenic and biological origin. The PM standard is the only NAAQS that does not target a specific chemical or family of chemical species. PM concentration is defined functionally as the change in mass of a filter through which ambient air is drawn under standardized conditions. PM can be solid, liquid, or composed of semivolatile species (partitioned between the PM and vapor phases depending on ambient conditions), and it can be either directly emitted from sources (primary; e.g., fly ash) or formed in the atmosphere (secondary; e.g., sulfate) from precursor gases. Regardless of composition, the aerodynamic size of PM determines its inhalability (ability to enter the mouth or nose) and the distribution of fractional deposition throughout the respiratory tract (EPA 2004e). There is no exclusive difference in the ability of PM at any aerodynamic diameter less than 10µm to reach different parts of the respiratory tract; both a 10-µm and a 10-nm particle could be deposited in the nose or the deep lung. However, the probability of deposition in different locations varies considerably with size. Most inhaled PM mass of a typical ambient size distribution is not deposited at all but is exhaled (EPA 2004e); however, there is clear evidence that the portion that does deposit can exert adverse health effects if the exposure level is sufficient.
A large range of health effects has been associated with exposure to PM in both epidemiologic and laboratory studies. The current PM2.5 NAAQS (promulgated in 1997) was based on epidemiologic evidence of mortality. The range of human health effects associated with ambient PM levels or demonstrated in laboratory studies has expanded from earlier concerns for total mortality and respiratory morbidity to include cardiac mortality and morbidity, blood vessel constriction, stroke, premature birth, low birth weight, retarded lung growth, enhancement of allergic responses, reduced resistance to infection, degenerative lesions in the brain, and lung cancer (EPA 2004e).
Both short-term increases in PM exposure and longer-term exposure concentrations are of concern. Numerous time-series studies have correlated variations in ambient PM levels with concurrent or lagging variations in health outcomes; other studies have demonstrated differ-
ences in mortality and morbidity between populations with different long-term PM exposures (Dockery et al. 1993; Pope et al. 1995, 2002, 2004). The relationship between the effects of short-term spikes in exposure and the cumulative effects of longer-term exposure to both spikes and baseline levels is uncertain.
The quantitative relationship between PM exposure levels and health effects remains uncertain, as does the proportion of the criteria pollutant health effects attributable to PM versus co-pollutants. Despite the uncertainties and the attendant debates, it seems clear that current concentrations in some areas and at some times in the United States are associated with health burdens that warrant concern, with a causal interpretation of the epidemiologic evidence implying a public health burden from PM exceeding that of other criteria air pollutants (EPA 2004e). A critical uncertainty involves determining a population threshold for PM (a concentration below which no health effects would occur). Although epidemiologic evidence to date has not detected a threshold for mortality effects (Pope et al. 2002; Daniels et al. 2004), epidemiologic studies lacking accurate personal exposure data have a limited ability to detect thresholds were they to exist. Furthermore, although animal studies have provided insight into mechanisms of response and confirmed the toxicity of certain PM components, few statistically significant responses have been demonstrated in the laboratory at ambient exposure concentrations. Given this uncertainty, regulatory impact analyses typically evaluate the sensitivity of conclusions to the no-threshold assumption (EPA 1999).
Similarly, the relationship between PM composition (particle-size fractions and chemical components) and adverse health effects remains uncertain. Current PM NAAQS assume that all PM mass is of equal toxicity, but it is unlikely that the mechanisms of action and relative toxicities do not differ across constituents. Different PM components (e.g., metals, organics) drive different types of toxicity (some components are more toxic than others for certain effects), and the same PM composition administered in different particle sizes has different potencies. However, there has been insufficient systematic study of the impacts of composition and size differences on the different effects of concern upon which to base composition-specific standards. In part, size is incorporated into the NAAQS as a rough surrogate for composition (because most fine PM stems from the combustion of fossil fuel or biomass, whereas coarse particles tend to arise from fugitive dust, pollens and spores, and sea salt). However, particles of similar size can have vastly different origins and composition.
Sulfur Dioxide
SO2 is of concern both because it is a direct respiratory irritant and because it contributes to the formation of sulfate and sulfuric acid in PM. The current NAAQS for SO2 is based primarily on preventing airway constriction in exercising, unmedicated asthmatic individuals (EPA 1994a). People with asthma are of particular concern, both because they have increased baseline airflow resistance and because their SO2-induced increase in resistance is greater than in healthy people, and it increases with the severity of their asthma. SO2 is thought to induce airway constriction via neural reflexes involving irritant receptors in the airways. The NAAQS is aimed at preventing stimulation of these reflexes in people with asthma.
Epidemiologic research since the last review of the SO2 NAAQS (Krewski et al. 2000; Pope et al. 2002) has repeatedly demonstrated associations between ambient SO2 and cardiopulmonary morbidity (illness) and mortality. Studies focused primarily on PM have often demonstrated significant or near-significant statistical associations between SO2 and the health outcomes. The implications of these findings are somewhat unclear. As a respiratory irritant, SO2 likely contributes directly to respiratory morbidity, and it might contribute directly to mortality with a respiratory origin. Through neural reflexes, it is possible that SO2 also might contribute directly to cardiac arrhythmia and thus cardiac morbidity and mortality, but this possibility has not been explored in controlled studies.
To the extent that ambient concentrations of SO2 are linked to concentrations of particulate sulfate and airborne acid at the same location, SO2 also might be serving as a marker for ambient sulfate (and thus ambient fine PM), which might cause the effects. However, until we have a better understanding of the specific physical-chemical air-pollutant species causing cardiopulmonary morbidity and mortality, we cannot ignore the possible direct contribution from SO2.
Volatile Organic Compounds
In addition to the NAAQS pollutants, emissions addressed by the NSR program include a diverse group of air contaminants termed for regulatory purposes, volatile organic compounds (VOCs). VOCs encompass a range of chemical species that have in common a fundamental
organic structure (primarily hydrocarbons, including oxidized and substituted hydrocarbons), a presence primarily or entirely in the gas phase under ambient conditions (vapor pressure generally greater than 10-4 millimeters of mercury, below which most organic compounds typically are considered semivolatile) and a reactivity leading to participation (together with NOx and sunlight) in atmospheric reactions resulting in the formation of O3. Because of research on the role VOCs played in the formation of O3 and efforts to identify and control emissions of its most reactive precursors, VOCs have long been targeted as a group to be regulated (EPA 1996). VOC emissions also contribute to the formation of airborne PM. The major classes of VOCs are alkanes, alkenes, aromatics, carbonyls, alcohols, and ethers. The substances included in VOC measurements can vary; some measurements target specific species and others (e.g., total nonmethane hydrocarbons) encompass multiple substances.
Although VOCs are addressed by NSR because of their contributions to health and welfare impacts as precursors of O3, many members of the group are also of concern because of their toxicity. Those considered to have the most important primary health implications are also included within the regulatory group termed “hazardous air pollutants” (HAPs, also known as “toxic air pollutants” or “air toxics”). The CAA lists 188 HAPs species or classes. In its most recent National-Scale Air Toxics Assessment, EPA reviewed 32 of the 33 HAPs it identified in 1999 to be of greatest concern in urban areas, plus diesel PM (EPA 2002d). EPA terms the 33 key HAPs “urban air toxics” (64 Fed. Reg. 38706 [1999]); these are listed in Table 3-2, in which the 21 VOC members are indicated. Examples of VOC HAPs include benzene, which is found in gasoline; methylene chloride, which is used as a solvent and paint stripper by a number of industries; and perchloroethlyene, which is emitted from some dry cleaning facilities. The health effects of VOC HAPs differ among the species; summaries can be found at EPA (2004f). Most effects of concern involve mutagenicity, cancer, irritation (inflammation), neurotoxicity, or modulation of immune responses.
ATTAINMENT STATUS OF NATIONAL AMBIENT AIR QUALITY STANDARDS
SO2 and CO are directly emitted by sources (primary pollutants). Portions of NO2, PM2.5, and PM10 are directly emitted (primary pollutants), and other portions form as a result of chemical reactions in the at-
mosphere (secondary pollutants). Very little O3 is emitted directly; most forms from chemical reactions of oxides of nitrogen (NOx, which is the sum of NO and NO2, expressed as NO2) and VOCs (see Box 3-1).
O3, PM10, and PM2.5 concentrations typically result from the contributions that are spread out over global (10,000 kilometers [km]), continental (1,000 km), regional (about 100 km), urban (about 10 km), neighborhood (1 km), and middle (intraneighborhood) (about 0.1 km) scales (Chow et al. 2002). Primary emissions from a single source may contribute large portions of primary emittants at the middle to neighborhood scales, but their relative contributions decrease with distance from the source.
There are currently no nonattainment areas for NO2 (EPA 2004i). There are 10 designated nonattainment areas for CO, all near heavily traveled roadways, and only a few of them have experienced NAAQS exceedances in recent years (AirData 2004). None of the 17 designated nonattainment areas for SO2 has experienced NAAQS exceedances during the past 10 years (EPA 2004i; Fred Dimick, EPA, unpublished data, November 2, 2004). The reduction in exceedances are mostly because of modifications or closures of nearby smelters and steel mills. Several years of ambient measurements that attain the NAAQS are required before EPA grants the attainment designation. CO and SO2 NAAQS exceedances were often caused by, and traceable to, nearby sources that were reasonably identifiable as the cause of the exceedance. This can be
TABLE 3-2 Urban Air Toxics
|
acetaldehydea acroleina acrylonitrilea arsenic compounds benzenea beryllium compounds 1, 3-butadienea cadmium compounds carbon tetrachloridea chloroforma chromium compounds coke oven emissions 1,3,-dichloropropenea dioxin ethylene dibromidea ethylene dichloridea ethylene oxidea formaldehydea hexachlorobenzenea hydrazinea lead compounds manganese compounds mercury compounds methylene chloridea nickel compounds perchloroethylenea polychlorinated biphenyls (PCBs) polycyclic organic matter propylene dichloridea quinolinea 1,1,2,2-tetra chloroethanea trichloroethylenea vinyl chloridea |
|
aAlso a volatile organic compound (VOC). Source: 64 Fed. Reg. 38706 (1999); EPA 2004g. |
important because state and local pollution control authorities can more easily take steps to achieve attainment when the emission sources are within their jurisdiction.
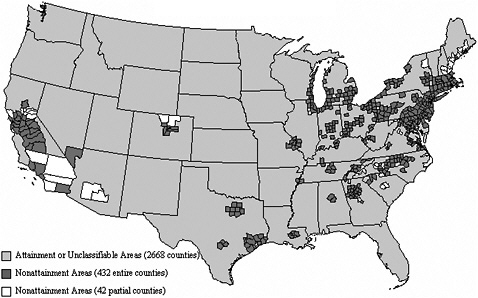
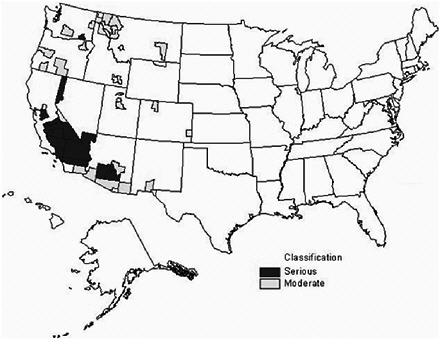
Figures 3-2 through 3-4 show the extent of U.S. nonattainment areas for O3, PM10, and PM2.5, respectively. Areas where the O3 standard is exceeded cover multiple counties in central and southern California; Las Vegas, Nevada; Phoenix, Arizona; along the Colorado Front Range; the Houston, Dallas, and San Antonio areas in Texas; nearly the entire Midwest and Southeast; and the Washington to Boston corridor (Figure 3-2). O3 can seldom, if ever, be reduced meaningfully by controlling single sources of precursors; reduction strategies need to encompass many different sources of NOx and VOCs. These groups of sources may be hundreds of kilometers from the locations where exceedances are measured and outside the jurisdictions of local air-quality authority (Seinfeld 1988; Sillman 1993, 1999; NARSTO 2000; Placet et al. 2000; Russell and Dennis 2000; Solomon et al. 2000; Watson et al. 2001). The rate of formation of O3 can be limited by the amount of either NOx or VOCs. The critical precursor to target to reduce O3 formation varies with location. For example, the rate of formation is often VOC-limited in urban areas and NOx-limited in rural areas. Therefore, it is usually more important to reduce VOCs and NOx emissions in urban and rural areas, respectively. Indeed, NOx reductions in some urban areas may even increase O3 levels, as evidenced by weekend versus weekday comparisons, where weekend NOx emissions are lower, and the resulting O3 concentrations are higher (Fujita et al. 2003).
Figure 3-3, which shows the nonattainment areas for the annual average PM10 NAAQS, indicates that most of the nonattainment areas are in the western United States. In the past, exceedances that occurred outside of California were often dominated by fugitive dust from disturbed land, construction, wintertime road sanding, and suspended road dust. PM10 state implementation plans (SIPs) developed in the early 1990s implemented wood-burning and dust-reduction measures, so many of these areas currently are not showing excessive PM10 concentrations. Several areas also had industrial sources that reduced emissions to comply with SO2, PM10, and Pb NAAQS requirements.
Figure 3-4 shows that nonattainment areas for the annual average PM2.5 NAAQS are more numerous and are located primarily in California and the eastern United States. In addition, many areas of the eastern United States have PM2.5 concentrations that approach the annual NAAQS of 15 µg/m3.
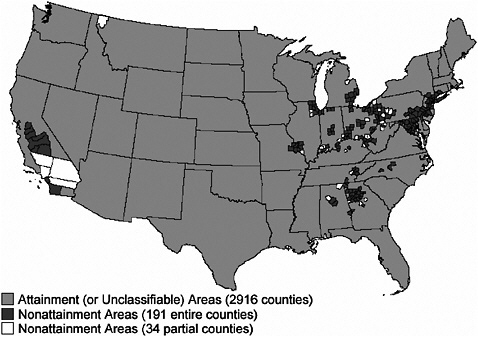
FIGURE 3-4 Nonattainment areas for annual average PM2.5 NAAQS. Source: EPA 2004l.
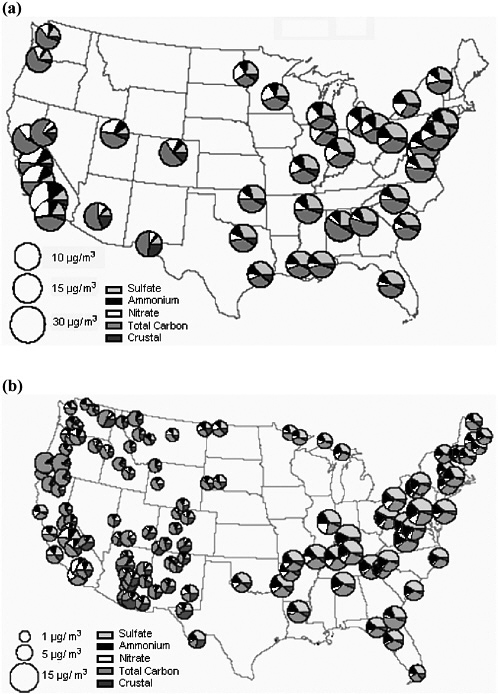
Unlike O3, which is a single compound, PM is composed of multiple chemical components, and the PM2.5 fraction displays more variability in its composition than does the PM10 fraction. Figure 3-5 shows how this composition differs among different parts of the United States and among urban-scale and regional-scale monitors. Comparing Figure 3-5a and 3-5b shows that many of the urban sites contain a larger component of carbonaceous particles than the nonurban sites, probably due to more abundant primary emissions from traffic and fuel use. The exception is the large nonurban (and urban) carbon fraction in the Pacific Northwest due to extensive wildfires during the summer of 2002. Sulfate levels are much higher in the eastern United States than in the west, and the levels are similar at nearby urban and regional sites, consistent with a regional distribution. Nitrate constitutes a larger fraction of PM2.5 in the west, especially in California. Sulfate and nitrate are usually found in the form of ammonium sulfate and ammonium nitrate, so ammonia emissions are also relevant to excessive PM2.5 concentrations. Even though SO2 and NO2 NAAQS are largely attained, SO2 and NOx emissions are relevant to PM2.5, and further reductions are needed to attain the PM2.5 NAAQS. Similarly, although VOCs and ammonia are not regulated by NAAQS,
VOCs are precursors of PM2.5 and O3, and ammonia is a precursor of PM2.5. Therefore, their emissions must be considered as part of PM2.5 and O3 SIPs. Figure 3-6 shows the number of people in the United States who are exposed to exceedances of the NAAQS. More than half the U.S. population lives in counties that exceed the O3, PM2.5, or PM10 NAAQS (see Figure 3-6).
EMISSION SOURCES AND TRENDS
EPA prepares a national database of air emissions based on input from state and local air agencies, Indian tribes, and industry. The information is compiled and made available to the public as the National Emissions Inventory (NEI). Box 3-2 provides an overview of the NEI based on information from EPA’s website (EPA 2004m).
In this section, the committee presents data from the EPA emissions inventory in relation to the air-quality issues presented earlier. The committee recognizes that the EPA emissions inventory data used in this report are under continual review and revision by the agency. However, the committee has made great efforts to present the most currently available information.
In general, the EPA inventory is based on data from states for major point-source emissions, whereas area and mobile source emissions are estimated by using surrogates such as fuel consumed, population density, vehicle-miles traveled, and land use. As noted in Box 3-2, EPA has developed a number of tools to ease the burden of states in estimating emissions for many source categories and to provide for the use of more uniform estimation methods across the country. For mobile and area sources, methods are used to simplify the process of estimating emissions by providing default emission factors and activity data. However, by necessity, these defaults tend to average out the local variations because they rely on the use of surrogates (e.g., household and population density, acres of farmland) available on a national basis to apportion the NEI.
In contrast, point source data, which rely on state or facility inputs for emission estimates, are considered more representative of the actual source emission characteristics than are the area- and mobile-source data discussed above. For some point sources, regulations require continuous emission monitoring (CEM) to document emissions. Therefore, the committee considers the point source data the most accurate of the emis-
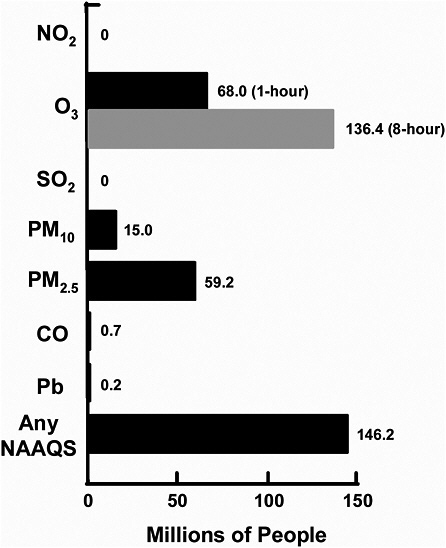
FIGURE 3-6 Population exposure to pollutants in nonattainment areas. The people studied lived in counties with air-quality concentrations above the level of the NAAQS in 2002. The bar for PM2.5 is based on EPA suggested nonattainment areas before designations were made. Source: EPA 2004n.
sions inventory categories, particularly for estimates of NOx and SO2. In the absence of CEM data, states and facilities estimate emissions by multiplying emission factors (pounds per unit consumption) times an activity parameter (e.g., number of units consumed or produced). For example, a cement kiln might calculate its emissions by multiplying an emission factor for fuel oil by the amount of fuel consumed. A problem arises,
|
BOX 3-2 The NEI database contains information on stationary and mobile sources that emit criteria air pollutants and their precursors as well as hazardous air pollutants (HAPs). The database includes estimates of annual emissions, by source, of air pollutants in each area of the country, on an annual basis. The NEI includes emission estimates for the following pollutants: carbon monoxide, nitrogen oxides, sulfur dioxide, particulate matter (PM10 and PM2.5), volatile organic compounds, and ammonia. The NEI database defines three classes of criteria air pollutant sources: —Point Sources: large stationary sources of emissions, such as an electric power plant, that can be identified by name and location. A major source emits a threshold amount (or more) of at least one criteria pollutant and must be inventoried and reported. Many states also inventory and report stationary sources that emit amounts below the thresholds for each pollutant. —Area sources: small stationary sources, such as a home or office buildings, or a diffuse stationary source such as wildfires or agricultural tilling. These sources do not individually produce sufficient emissions to qualify as point sources. Dry cleaners are one example—a single dry cleaner within an inventory area typically will not qualify as a point source, but collectively the emissions from all the dry cleaning facilities in the inventory area may be significant and therefore must be included in the inventory. —Mobile sources: any kind of vehicle or equipment with a gasoline or diesel engine, airplanes, or ships. The main sources of criteria pollutant emissions data for the NEI are —For electric generating units: EPA’s Emission Tracking System/ Continuous Emissions Monitoring Data (ETS/CEM) and Department of Energy fuel-use data. —For other large stationary sources: state data and older inventories where no state data were submitted. —For on-road mobile sources: the Federal Highway Administration’s (FHWA’s) estimate of vehicle miles traveled and emission factors from EPA’s MOBILE Model. —For nonroad mobile sources: EPA’s NONROAD Model. —For stationary area sources: state data, EPA-developed estimates for some sources, and older inventories where no state or EPA data were submitted. State and local environmental agencies supply most of the point source data. EPA’s Clean Air Market program supplies emissions data for electric power plants. More information about the NEI database and the compilation of criteria pollutant and HAP emissions inventories, and links to the database, are available on the CHIEF NEI webpage (EPA 2004m). |
however, from the difficulty in accounting for large emission spikes from stationary sources related to upsets, which are not captured in the average emission estimate for each source and may result in increased overall emissions (TNRCC 2003). The emission inventories developed from a compilation of various emission estimates are generally considered to have an uncertainty of about a factor of 2 or more (see NARSTO 2000), although the uncertainty factor is poorly defined (NRC 2004). A number of recent studies have aimed to quantify uncertainty in emission inventories for utility NOx emissions (e.g., Frey and Zheng 2002; Abdel-Aziz and Frey 2003a,b) and for hazardous air pollutants (e.g., Frey and Zhao 2004; Zhao and Frey 2004). The NRC and other organizations have often recommended that uncertainties in emission factors and inventories should be quantified in practice (e.g., NRC 2000; IPCC 2000).
EPA has been reporting emissions by source categories since the early 1970s (EPA 2004m). A variety of regulations controlling emission sources of pollution have been implemented over the past several decades and have been associated with declining emissions for some pollutants in some source categories. Because NSR is directed at large stationary sources, the focus is on these sources.
Figure 3-7 shows the trend of point source emissions since 1970 for NOx, VOCs, PM10, and SO2. Point source NOx emissions have remained nearly constant over the last 30 years, with power-generating point sources dominating the emissions. Total point source SO2 emissions have decreased over the last 30 years. Much of the early decreases in point source SO2 emissions are the result of decreases in the metal smelting sector, whereas later decreases are due to reductions from the electric utility sector. Nevertheless, point sources remain a large fraction of SO2 emissions, and point source emissions are responsible for a significant fraction of PM. PM10 emissions declined substantially from 1970 to 1985 because of industrial emission controls; however, there have been no major changes since 1985. PM2.5 has remained largely unchanged (not shown in the figure). VOC emissions declined over the last 30 years, for a variety of reasons (e.g., O3 SIPs).
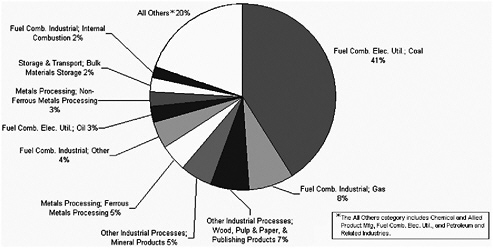
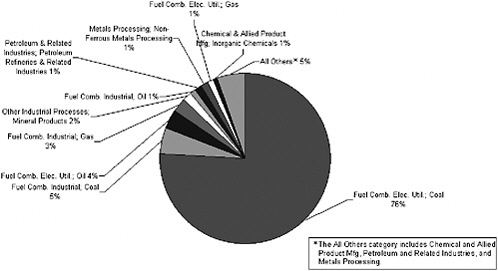
To examine the contributions of specific industries to total point source emissions (Figure 3-7), data by pollutant and by industry are presented below for 1999 (see Figures 3-8 through 3-11). For PM2.5, about 50% of point source emissions is due to fuel combustion in the electric utility industry; is about 40% of point source emissions is due to coal combustion. Other important source categories include wood, pulp, and
paper; metals; and mineral processing (Figure 3-8). Based on emissions data for 1999, point sources compose about 18% of PM2.5 emissions and about 7% of PM10 emissions (EPA 2003c). Primary PM2.5 and PM10 emissions compose a minor fraction (<10%) of measurable ambient PM concentrations (Chow and Watson 2002; EPA 2004o).
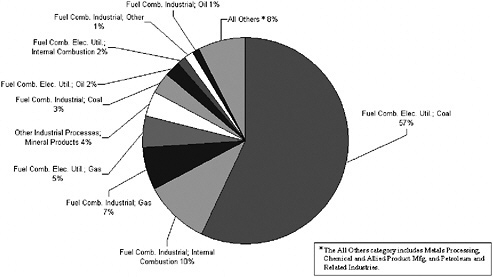
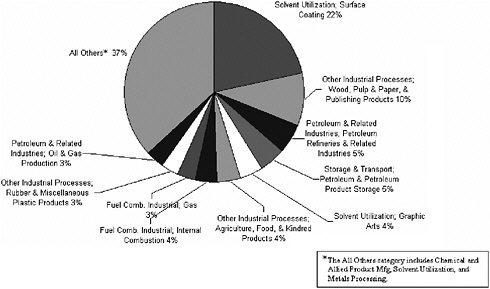
Point sources are important contributors to pollutants (SO2, NOx, and VOCs) that participate in the formation of secondary aerosols. For SO2, 75% of point source emissions are attributable to coal combustion by the electric utility industry, with the remaining contributions due to fuel combustion from other industrial sources (Figure 3-9). In contrast, only about 50% of NOx point source emissions are due to coal combustion from electric utilities, with the remainder divided equally among other fuel combustion activities (Figure 3-10). In contrast to the predominance of fuel combustion activities for NOx and SO2, point sources of VOCs are primarily attributable to petroleum and related industries, chemical manufacturing, and other industrial processes, including pulp and paper, food, rubber, and plastics manufacturing (Figure 3-11). VOC-derived organic aerosol may represent an important fraction of PM2.5 in U.S. regions when photochemical transformation predominates (Cabada et al. 2004; Dechapanya et al. 2004). Individual VOCs can differ substantially in the rates at which they react in the atmosphere and the ef-
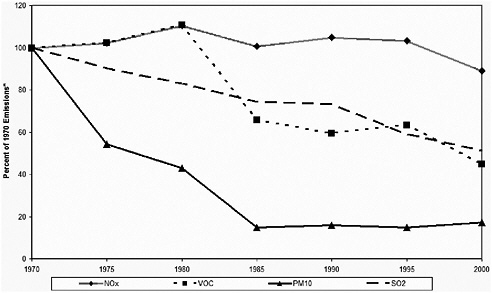
FIGURE 3-7 Criteria pollutant emission trends for point sources. Source: Data from EPA 2004n.
fects that they have on the formation of O3 and PM (Carter 1994; Atkinson 2000; NARSTO 2004). Another important contributor to secondary aerosol formation is ammonia emissions. Nationally, point sources contribute only a minor fraction of ammonia emissions compared with agricultural sources (EPA 2004o).
Point source emissions are not evenly distributed across the United States. Large industrial sources are located on the periphery of urban areas and near convenient transport facilities or near their sources of raw materials. Figures 3-12 through 3-14 show the point source emissions of SO2, NOx, and VOCs by county across the United States overlaid on the nonattainment areas discussed previously.
As shown in Figure 3-12, SO2 emissions from point sources are located predominantly in the eastern United States, with particularly high concentrations in the Ohio River Valley, in the southern Appalachians, and along the northeastern seaboard. NOx point sources also are predominantly observed in the eastern United States (Figure 3-13) and mirror the spatial pattern of SO2 emissions, although there are fewer counties with major point sources of NOx emissions in the west. For VOCs (Figure 3-13), the pattern is completely different and is determined primarily by the location of petrochemical industries along major waterways—California, the Gulf Coast, the Eastern seaboard, the Great Lakes, and the Ohio River Valley.
As discussed earlier, sulfates, nitrates, and organics are important contributors to observed ambient particle concentrations. In viewing Figures 3-12, 3-13, and 3-14, it is apparent that PM and O3 nonattainment areas are correlated with emissions of SO2, NOx, and VOCs. With respect to Figure 3-14, it is also important to note that organic materials compose a substantial component of observed aerosols. The VOC emissions, which are primary contributors to organic aerosols, may be associated with natural biogenic sources from non-point-source emissions but also may be due to VOC emissions from point sources as depicted in the NEI or from emissions not currently reflected in the point source VOC emission inventory.
As shown above, point sources contribute substantially to NOx, SO2, PM, and VOCs in areas of the United States that are experiencing air-quality concentrations at levels associated with adverse health effects. Therefore, it is important to assess which of the point sources are most affected by the NSR changes under review by this committee. Also, some point sources may not be subject to NSR requirements because of their relatively small size and location.
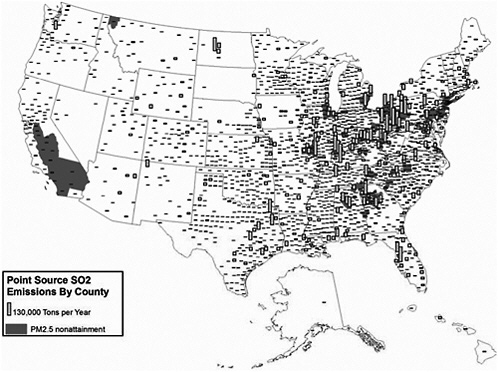
FIGURE 3-12 Point source SO2 emissions and PM2.5 nonattainment, 1999. Nonattainment areas are EPA suggested and not final. Point source emission inventory date, 1999. Sources: AirData 2004; EPA 2004l. Map drawn by Sean M. Raffuse, Sonoma Technology, Inc.
VINTAGES OF EMISSION SOURCES
Information on vintages of different facilities within most of the major NSR-affected industries is difficult to obtain.6 Linking these emissions data to information about vintage is difficult to do without access to the facility-level information from the U.S. Census.
The one industry for which vintage data are more readily available and linkable to the emission data is the electricity sector. Data on vintage are collected and maintained by the U.S. Energy Information
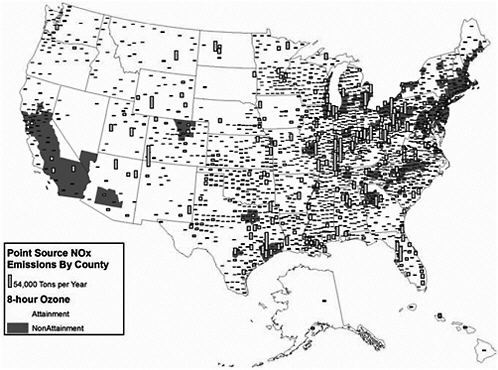
FIGURE 3-13 Point source NOx emissions and 8-hour ozone nonattainment, 1999. Nonattainment areas effective date May 2004. Point source emission inventory date 1999. Sources: AirData 2004, EPA 2004j. Map drawn by Sean M. Raffuse, Sonoma Technology, Inc.
Administration (EIA) and emissions data are available from the EPA’s CEM database. Table 3-3a and 3-3b provides summaries of NOx and SO2 emission rates, respectively, from coal-fired generators in 2002 by vintage of generating unit, where vintage is assigned based on the year that a power plant came into service. The table also provides information on how much each vintage class (with classes arranged by decade) contributed to both total generation and total capacity of coal-fired generation in 2002. The SO2 table (Table 3-3b) includes information on average capacity factor (actual generation divided by potential generation) of generators by vintage and the average heat rate (British thermal units of heat input from fuel combustion per kilowatt hour of electricity generated) for generators within each vintage class.
Table 3-3a and 3-3b clearly shows that older facilities have higher emission rates than newer facilities, and older facilities tend to contribute more to total emissions than they do to total electricity generation from coal units. Older generators also have lower capacity factors than newer
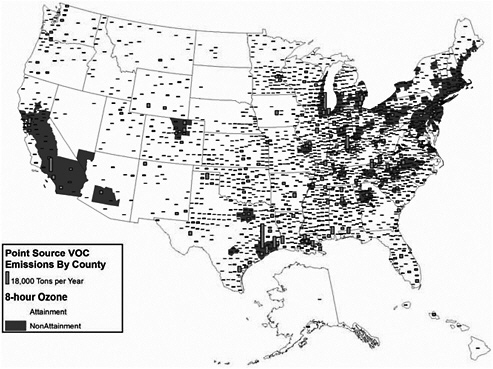
FIGURE 3-14 Point source VOC emissions and 8-hour ozone nonattainment, 1999. Nonattainment areas effective date May 2004. Point source emission inventory date 1999. Source: AirData 2004; EPA 2004j. Map drawn by Sean M. Raffuse, Sonoma Technology, Inc.
generators, which are run more often. The data also show that, with perhaps one exception, heat rates are lower for newer power generators than they are for older units. This piece of data needs to be interpreted with caution because older generators have lower capacity factors than newer generators and, if those generators were operated more, their average heat rates likely would be lower. There is a selection bias in the data whereby the generators with lower heat rates of any vintage typically are the ones that are operated more.
Table 3-3c and 3-3d displays the same data organized a different way. Coal-fired generating units are classified by a New Source Performance Standards (NSPS) category according to information from EIA Form 767 for 2002 (EIA 2004a). Most of the capacity and generation falls into the category that is not subject to NSPS standards. Generators in this category typically have much higher SO2 and NOx emission rates (in units of pounds of pollutant per megawatt-hour of electricity generated) than generators that were permitted under the NSPS standards that
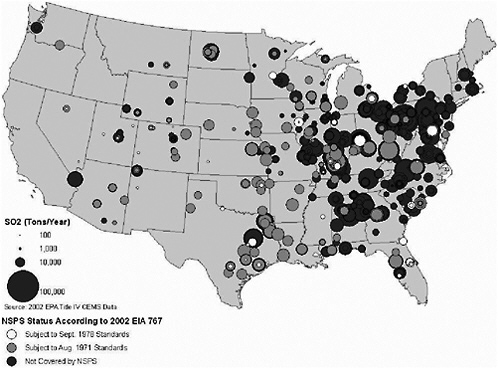
FIGURE 3-15 2002 SO2 emissions for utility-scale coal-fired generators that operated in 2002. Source: EIA 2004a; EPA 2004p. Map drawn by Sean M. Raffuse, Sonoma Technology, Inc.
came into effect in 1971. The generators in the post-1978-standard category have substantially lower SO2 emission rates than those subject to earlier standards but NOx emission rates virtually identical to the earlier group, reflecting the fact that new source standards for NOx did not change much in 1978 (Burtraw and Evans 2004). Note that NSPS regime depends on when construction was started, whereas the vintages in Tables 3-3a and 3-3b are determined by when a generator comes into service.
Figures 3-15 and 3-16 show the annual emissions of SO2 and NOx, respectively, in 2002 from generating facilities categorized by NSPS status according to the EIA 767 data. The coal`-fired generating units not covered by NSPS are located predominantly in the eastern United States, with large concentrations in Pennsylvania and Ohio, upwind of major East Coast population centers, and with other clusters in Kentucky, southern Illinois and Indiana, the Carolinas, and Alabama. These units tend to have higher annual emission rates of SO2 than plants permitted under the 1971 and 1978 NSPS standards. There is less variation in total
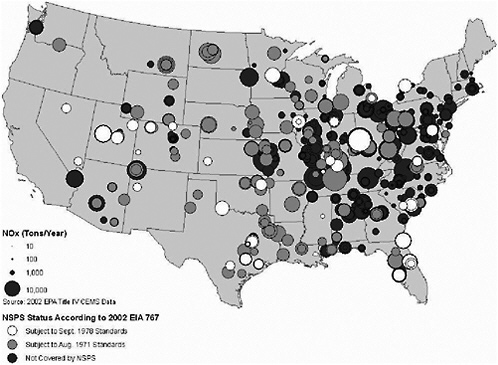
FIGURE 3-16 2002 NOx emissions for utility-scale coal-fired generators that operated in 2002. Source: EIA 2004a; EPA 2004p. Map drawn by Sean M. Raffuse, Sonoma Technology, Inc.
annual emissions of NOx across the different categories of NSPS status, which is consistent with the smaller differences in average NOx emission rates across these different classes of generating units. Most of the emitting units are in the east, but new units can have total annual emissions as high as older units, reflecting in part the higher capacity factors at the newer units. Geographic concentrations of NOx emissions typically are found in the same locations as they are for emissions of SO2.
SUMMARY
The committee draws the following conclusions:
-
Large stationary sources (point sources) are substantial emitters of NOx, SO2, and VOCs into areas of the United States that are experiencing concentrations of criteria pollutants at levels associated with an increased risk of adverse health effects. Because large stationary
-
sources are not distributed evenly throughout the United States and because many different industries contribute to stationary source emissions, it is important to assess a variety of types and locations of large stationary sources that are affected by the NSR changes. The age of the facilities may be an important consideration because older facilities are more likely than newer facilities to undergo maintenance, repair, and replacement of key components.
-
Because of the long-range transport of some pollutants, important emission sources may be far from the locations where measured pollutant concentrations exceed the NAAQS. Thus, for areas experiencing higher ambient concentrations of pollutants, such as fine PM and O3, controlling emissions of those pollutants and their precursors is typically a regional, often multistate, problem not a local one.
-
Data presented from electricity-generating facilities (coal plants) show that older facilities have higher emission rates than newer facilities, and older facilities tend to contribute proportionately more to total emissions than they contribute to total electricity generation.
Although this chapter broadly describes the connection between emissions and health effects, in many places, it relies on incomplete data about the consequences of air pollution, its causes, and the specific contributions that can be attributed to individual sources. The approaches needed to address these areas of uncertainty will be developed further as the committee goes forward with its charge.
TABLE 3-3 Emissions from Coal-Fired Electricity Generation by Vintage
a) 2002 NOx Emissions and Share of Generation of Coal-Fired Capacity by Vintage
|
Power Plant Vintage |
Avg. NOx Emission Rate (lb/MWh) |
% Total NOx Emitted |
% of Coal-Fired Electricity Generation |
% of NOx Emitted per % of Electricity Generateda |
% of Coal-Fired Electricity Capacity |
|
Pre-1950 |
5.51 |
0.65 |
0.50 |
1.31 |
0.92 |
|
1950-1959 |
5.07 |
15.11 |
12.56 |
1.20 |
14.32 |
|
1960-1969 |
4.56 |
21.27 |
19.65 |
1.08 |
20.51 |
|
1970-1979 |
4.28 |
39.31 |
38.76 |
1.01 |
38.13 |
|
1980-1989 |
3.53 |
21.74 |
25.97 |
0.84 |
23.84 |
|
Post-1990 |
3.15 |
1.92 |
2.56 |
0.75 |
2.27 |
b) 2002 SO2 Emissions and Performance of Coal-Fired Capacity by Vintage
|
Power Plant Vintage |
Avg. SO2 Emission Rate (lb/MWh) |
% of Total SO2 Emitted |
% of Coal-Fired Electricity Generation |
% of SO2 Emitted per % of Electricity Generateda |
Average Capacity Factor (%)b |
Average Heat Rate (Btu/kWh generated) |
|
Pre-1950 |
20.58 |
1.02 |
0.50 |
2.04 |
36.35 |
12,549 |
|
1950-1959 |
15.78 |
19.64 |
12.56 |
1.56 |
58.93 |
10,668 |
|
1960-1969 |
13.92 |
27.12 |
19.65 |
1.38 |
64.37 |
10,150 |
|
1970-1979 |
9.31 |
35.75 |
38.76 |
0.92 |
68.29 |
10,270 |
|
1980-1989 |
6.02 |
15.49 |
25.97 |
0.60 |
73.17 |
10,401 |
|
Post-1990 |
3.88 |
0.98 |
2.56 |
0.38 |
75.80 |
9,982 |
c) 2002 NOx Emissions and Share of Generation of Coal-Fired Capacity by NSPS Standardc
|
NSPS Status According to EIA 767 |
Avg. NOx Emission Rate (lb/MWh) |
% Total NOx Emitted |
% of Coal-Fired Electricity Generation |
% of NOx Emitted per % of Electricity Generateda |
% of Coal-Fired Electricity Capacity |
|
Unknown |
2.93 |
0.16 |
0.23 |
0.69 |
0.27 |
|
Not Affected by NSPS |
4.67 |
65.90 |
59.51 |
1.11 |
62.62 |
|
Subject to Aug. 1971 Standards (D) |
3.57 |
26.73 |
31.58 |
0.85 |
29.56 |
|
Subject to Sept. 1978 Standards (Da) |
3.50 |
7.21 |
8.68 |
0.83 |
7.56 |
d) 2002 SO2 Emissions and Performance of Coal-Fired Capacity by NSPS Standardc
|
NSPS Status According to EIA 767 |
Avg. SO2 Emission Rate (lb/MWh) |
% of Total SO2 Emitted |
% of Coal-Fired Electricity Generation |
% of SO2 Emitted per % of Electricity Generateda |
Avg. Capacity Factorb (%) |
Average Heat Rate (Btu/kWh generated) |
|
Unknown |
4.56 |
0.10 |
0.23 |
0.45 |
56.58 |
11,247 |
|
Not Covered by NSPS |
12.93 |
76.25 |
59.51 |
1.28 |
63.85 |
10,250 |
|
Subject to Aug. 1971 Standards (D) |
6.66 |
20.86 |
31.58 |
0.66 |
71.79 |
10,519 |
|
Subject to Sept. 1978 Standards (Da) |
3.23 |
2.78 |
8.68 |
0.32 |
77.17 |
10,185 |
|
aIf the generators of a particular vintage (or in a particular NSPS category) emitted a particular pollutant in proportion to its share of total electricity generation, the value would be 1. bCapacity factor of units that operated that are strictly associated with boilers in CEMS database. cThe Subpart D standards are those that apply to fossil-fuel-fired steam boilers for which construction began after August 17, 1971. The Subpart Da standards affect those boilers that began construction after September 18, 1978. For those boilers not covered by NSPS construction commenced before August 17, 1971. Notes: All quantities, including percentages of emissions and generation capacity, are calculated with reference only to coal-fired generating units. Percentages (taking account of rounding) add to 100% because other types of generating capacity are not considered. These tables and the associated data set were constructed by David Evans of Resources for the Future. Data used to make these tables come from three sources; emissions data are from EPA’s Continuous Emissions Monitoring System Database; generation and capacity data are from the EIA’s 767 data set; and information on vintage of generating units is from the EIA's 860 data set. Abbreviations: EIA, Energy Information Agency; Btu/kWh, British thermal unit per kilowatt-hour; lb/MWh, pound per megawatt-hour. Source: EIA 2004a,b; EPA 2004p. |
||||||