Appendix B
A Comparison of the Risk of Skin Cancer with the Risk of Lung Cancer from Exposure to Radon Decay Products in Underground Mines
It has been suggested that ambient conditions in underground mines might cause cancers at sites other than the lung (Tomasek et al., 1993; Denman et al., 2003), specifically, that radon in the ambient air might be responsible for an excess risk of skin cancer and leukemia. The committee has reviewed the relevant literature and has compared the risk of skin cancer with the risk of ling cancer.
The nobel gas radon-222 (222Rn) is produced naturally in rocks and underground formations. Because of its mobility, it can escape from the geologic matrix and accumulate in the atmosphere. It can reach high concentrations particularly in caves and underground mines. Radon is radioactive; when it decays, it initiates a series of radioactive transitions. The short-lived radioactive descendents of 222Rn—polonium-218 (218Po), lead-214 (214Pb), bismuth-214 (214Bi), and polonium-214 (214Po)—have historically been called radon daughters, radon progeny, or radon decay products. Their kinetic behavior is complicated. They are not inert and do adhere to other objects when contact is made. Many of the daughters “plate out” on surfaces, where they become immobile and decay in situ. Others attach to aerosols and remain suspended in the atmosphere until they decay.
Risks of lung cancer are related to inhalation of radon daughters, not radon itself, because almost all radon inhaled is immediately exhaled, but inhaled radon daughters mostly become resident in the airways of the lung. The radioactive half-lives of the daughters are shorter than the respiratory clearance time, so they decay in the lung. The alpha particles released by 218Po and 214Po have sufficient penetration to reach and deliver high doses to sensitive cells in the bronchial epithelium.
The concentration of radon daughters suspended in air is measured in working levels (WL) or equilibrium equivalent concentration (EEC). Human exposure to radon daughters depends on the concentration at a specific location and the time that the person spends at that location; it is measured in working level months (WLMs). The risk increases as the exposure in WLM increases. Numerical estimates of risk have been reviewed extensively by the committee on health risks of exposure to radon (BEIR VI) (NRC, 1999) and International Commission on Radiation Protection (ICRP, 1981; 1991; 1993).
The risk of skin cancer in radon-rich atmospheres is not associated with inhalation or other intake of radioactivity. It is related to the plate out of radon daughters on the skin. The dose to sensitive cells in the skin depends on the concentration of radioactivity on the surface skin and the ability of alpha particles from 218Po and 214Po to penetrate to the location of the sensitive cells. Previous studies of that process have used a nominal value of 70 mm as the depth of the sensitive basal cells at the base of the epidermis. Recent measurements have indicated large variation in the depth of the basal cells, and this could lead to higher doses than previously expected (Eatough, 1997). We have assessed the pathway to determine whether the projected risk of skin cancer is comparable with the risk of lung cancer posed by the same exposure in WLM.
The dose delivered by an alpha particle as it passes through tissue depends on the stopping power, dE/dx. Values of that quantity have been complied by the National Institute of Standards and Technology and can be downloaded directly from its Web site (http://physics.nist.gov/PhysRefData/contents-radi.html). Those data can be used to compute the dose as a function of depth for radioactivity uniformly deposited on the surface. Figure B.1 shows the results of the computation for 218Po (6 MeV alpha particle) and 214Po (7.7 MeV alpha particle). The data clearly illustrate that the alpha particle from 214Po will not reach basal cells at a depth of more than 50 µm.
The epidermis is not a flat organ located uniformly below the skin. There are undulations that are responsible for large variations in depth. Eatough has published distributions of epidermal thickness in various locations on the body (Eatough, 1997). We have simulated those data as a lognormal distribution (median = 50, σg = 1.5) to represent the epidermal thickness on the exposed face of a person in Figure B.2.
The mean dose to basal cells per disintegration on the basis of the data in Figures B.1 and B.2 is shown in Table B.1.
The next step is to determine the amount of radioactivity that will plate out on the skin for a given concentration of radon daughters suspended in the air. Denman et al. (2003) and Sevkova et al. (1978) have reported measurements of plate out on exposed skin. We have adopted a value of 2 (Bq/m2)/(Bq/m3) which is consistent with the reported results. Combining those results and assuming a steady-state condition with equal concentrations of 218Po and 214Po on the skin, we obtain a result of 200 mSv/WLM for the effective dose to the skin.
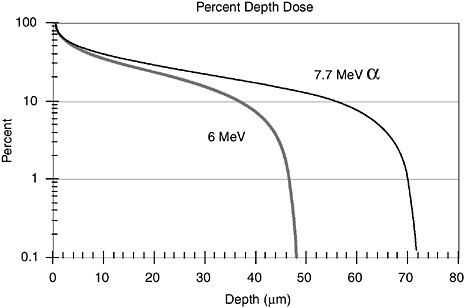
FIGURE B.1 Percent depth dose distribution for 6 MeV and 7.7 MeV alpha particles that emitted isotropically emission from a source that is uniformly distributed on surface.
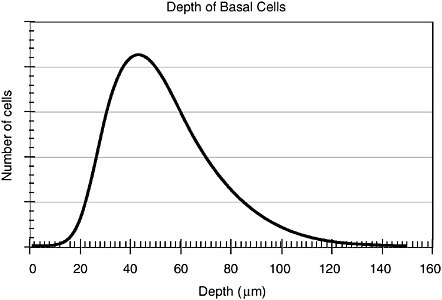
FIGURE B.2 The distribution of basal cells as a function of depth below surface of skin. Diagram has been normalized such that the area under the curve is equal to 1.0.
TABLE B.1 Mean Dose to Basal Cells per Disintegration
|
Isotope |
Gy·cm2/dis |
µSv·cm2/dis |
|
218Po |
1 × 10−8 |
0.2 |
|
214Po |
4 × 10−8 |
0.8 |
Mining activity is physically rigorous and some perspiration is expected while underground. That adds a layer of water on the skin and increases the thickness of material between the radioactivity and the basal cells.
We assumed that prolonged activity in the mine results in a water loss of 5 liters per day through the skin. The water will be released uniformly over a surface area for a standard adult of 1.7 m2. It will evaporate from exposed surfaces at about 10% per minute. The result is a steady-state layer of water that is about 20 mm thick. In addition to perspiration, fumes and dust will accumulate on the surface of the skin, and we have assumed that this will provide a thickness of material equivalent to another 20 mm of water.
We have computed the reduction of dose from alpha particles as a function of the thickness of water-equivalent material on the surface of the skin, assuming that the activity is on the top surface of the water. The attenuation factor is shown in Figure B.3.
It can be seen that 40 µm of material will reduce the dose to the basal cells by a factor of about 100. Thus, the equivalent dose is reduced to 2 mSv/WLM.
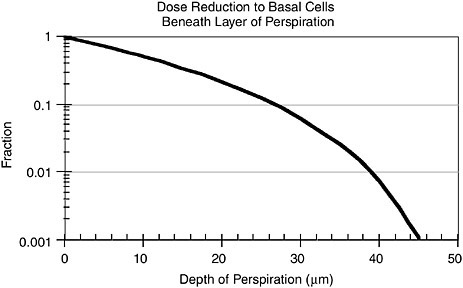
FIGURE B.3 Reduction of dose to basal cells from water-equivalent material on skin. Activity is assumed to be on surface of water above skin.
Uranium miners wear protective clothing while underground, so only their arms, hands, heads, and necks are directly exposed to the atmosphere. That represents less than 25% of the surface area of the skin. Using that value, the effective dose to the exposed portion of a person’s body 0.5 mSv/WLM.
The lifetime risk of fatal skin cancer after whole-body exposure to the skin is 2 × 10−4 /Sv (ICRP, 1991 p 132). The risk from radon decay products deposited on the skin would be about 1 × 10−7/WLM, that is, (5 × 10−4 Sv/WLM) or (2 × 10−4 /Sv). The lifetime risk for lung cancer from inhalation of radon daughters is ~2 × 10−4/WLM (ICRP, 1991 p 139). From this dosimetric analysis, the risk of lung cancer is greater than 2000 times the risk of skin cancer from the same exposure to radon daughters.
We have repeated the calculation assuming that the radon decay products are uniformly distributed throughout the layer of perspiration on the skin. The average attenuation factor for this case increased from 0.01 to 0.32. The effective dose to the skin for the exposed portion of the body is then 16 mSv/WLM. The associated risk of lung cancer in this case is more than 60 times the risk of skin cancer.
Epidemiologic studies have evaluated the incidence of skin cancer in underground miners (Darby et al., 1995) and have concluded that high concentrations of radon are not associated with excess mortality from cancers other than lung cancer. A cohort of underground miners in the Czech Republic did show some increased incidence of skin cancer (Sevcova et al., 1978). It was not related to accumulated radon exposure, but it did seem to be associated with length of exposure (i.e., 10 years or longer). The pathologic evaluation diagnosed the lesions as basal cell carcinomas. In all cases, the tumors were excised surgically, and there were no signs of recurrence.
On the basis of dosimetry and epidemiology, the risk of lung cancer is significantly higher than the risk of fatal skin cancer in persons working in an environment where radon decay products are suspended in air.
REFERENCES
Darby, S. C., Whitley, E., Howe, G. R., Hutchings, S. J., Kusiak, R. A., Lubin, J. H., Morrison, H. I., Tirmarche, M., Tomasek, L., Radford, E. P., R. J., Samet, J. M., Shu, X. Y. Radon and Cancers other than Lung Cancer in Underground Miners: A Collaborative Analysis of 11 Studies. J. Natl. Cancer Inst. 87(5):378-384. 1995.
Denman, A. R., Eatough, J. P., Gillmore, G., Phillips, P. S. Assessment of Health Risks to Skin and Lung of Elevated Radon Levels in Abandoned Mines. Health Physics 85(6):733-739. 2003.
Eatough, J. P. Alpha-Particle Dosimetry for the Basal Layer of the Skin and the Radon Progeny 218-Po and 214-Po. Phys. Med. Biol. 42(10):1899-1911. 1997.
ICRP (International Commission on Radiological Protection). Limits for Inhalation of Radon Daughters by Work. ICRP Publication 32, Annals of the ICRP 6(1). Elmsford, NY: Pergamon Press. 1981.
ICRP (International Commission on Radiological Protection). 1990 Recommendations of the International Commission on Radiological Protection. ICRP Publication 60, Annals of the ICRP 21(1-3). Elmsford, NY: Pergamon Press. 1991.
ICRP (International Commission on Radiological Protection). Protection Against Radon-222 at Home and at Work. ICRP Publication 65, Annals of the ICRP 23(2). Elmsford, NY: Pergamon Press. 1993.
NRC (National Research Council). Health Effects of Exposure to Radon: BEIR VI. Washington, DC: National Academy Press. 1999.
Sevcova, M., Sevc, J., Thomas, J. Alpha Irradiation of the Skin and the Possibility of Late Effects. Health Physics 35(6):803-806. 1978.
Tomasek, L., Darby, S. C., Swerdlow, A. J., Placek, V., Kunz, E. Radon Exposure and Cancers Other than Lung Cancer among Uranium Miners in West Bohemia. Lancet 341(8850): 919-923. 1993.






