6
New Approaches
The committee’s review of current toxicity-testing strategies revealed a system that is approaching a turning point. Most toxicity-testing frameworks were developed decades ago and may not adequately reflect today’s science, let alone the emerging challenges and new approaches of the future. Agencies have responded by altering individual tests and by adding tests to the existing regimens to incorporate end points and mechanistic evaluations newly recognized to be of potential importance. Those patches have not provided a fully satisfactory solution of the fundamental problem.
The core of the problem appears to be tension among four objectives of regulatory testing schemes that are difficult to meet simultaneously: depth, providing the most accurate, relevant information possible for hazard identification and dose-response assessment; breadth, providing data on the broadest possible universe of chemicals, end points, and life stages; animal welfare, causing the least animal suffering possible and using the fewest possible animals; and conservation, minimizing the expenditure of money and time on testing and regulatory review (see Figure 6-1). The committee initially noted that decreasing animal use and decreasing costs may pull the testing programs in similar directions, such as toward the use of in vitro methods. However, those two objectives may not always be aligned; initial efforts to reduce animal suffering and animal use may increase costs in some situations. Thus, approaches designed to move toxicity testing toward one of the objectives frequently move it away from one or more of the others. The Environmental Protection Agency (EPA) and other agencies that perform or require toxicity
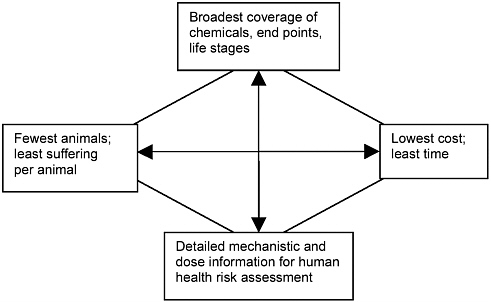
FIGURE 6-1 The four objectives of toxicity testing.
testing are constantly being challenged to meet all four objectives and are often caught between competing priorities.
Setting priorities among the competing objectives is more than a scientific issue. Individuals and organizations can have different, sometimes strongly held beliefs about which of the four is most important, and trying to satisfy the objectives has driven different efforts to reform toxicity testing. For example, legislation requiring creation of an endocrine-disruptor screening program was driven by an effort to increase breadth, whereas the Interagency Coordinating Committee on the Validation of Alternative Methods (ICCVAM) was created in an effort to conserve animals. Rather than attempting to set priorities among the objectives, this committee recognized that all four are important. The committee acknowledges, however, that embracing all four poses a difficult challenge. This chapter and the next review some selected approaches that may ultimately help to move toxicity testing toward one or more of the objectives.
In this chapter, the committee summarizes and comments on some strategies proposed by others for near-term improvements in existing toxicity-testing approaches, including the EPA review of toxicity data available for establishing reference doses (RfDs) and reference concen-
trations (RfCs), the International Life Sciences Institute (ILSI) Health and Environmental Sciences Institute (HESI) draft reports proposing modifications of EPA’s approach for pesticide testing, the National Toxicology Program (NTP) Roadmap for the Future, and the European Union (EU) strategy in the proposed REACH (Registration, Evaluation and Authorisation of Chemicals) testing scheme for existing industrial chemicals.
APPROACHES FOR IMPROVING EXISTING TOXICITY-TESTING STRATEGIES
Toxicity-testing guidelines and strategies described in the previous chapters are the results of the gradual evolution of testing requirements and risk-assessment approaches that took place as the field of toxicology advanced. However, agencies have struggled to incorporate recent scientific and technologic advances in toxicology, basic human biology, molecular biology, pharmacokinetics, dose-response modeling, imaging, computation, and other relevant fields, so many of the current requirements are still based on approaches that originated more than 40 years ago. In addition, more sophisticated exposure assessments have identified different durations and routes of exposure for various populations, such as residential exposures of toddlers, that require more toxicology data for risk assessment.
Proposed strategies for improving toxicity testing can be difficult to compare directly. Some strategies aim at meeting regulatory mandates and therefore focus on specific needs. For example, the ILSI-HESI documents described below focus on toxicity-testing strategies for pesticides, whereas the REACH program attempts to address the numerous industrial chemicals that have been inadequately studied. The different purposes of those testing strategies contribute to major differences between them. However, there can also be important differences between testing strategies and approaches of initiatives and proposals that try to fulfill the same risk-management needs.
The committee elected to focus primarily on the major aspects of the reports reviewed, rather than critiquing the details. Most of the reviewed reports describe initiatives or proposals that are still under development, some of which are sometimes presented with few details; some reports were available to the committee only as drafts. The committee
reviewed the documents primarily in an effort to compare various testing strategies proposed and to evaluate their potential to move toward the overall objectives of broadening coverage, increasing depth, addressing animal-welfare issues, and conserving resources. In general, the committee did not engage in detailed assessments of individual bioassays, although a few observations related to the alternatives presented by EPA for discussion are included.
Environmental Protection Agency Review of Data Needs for Risk Assessment
A Review of the Reference Dose and Reference Concentration Processes (EPA 2002), written by the EPA Risk Assessment Forum’s RfD/RfC Technical Panel, provides recommendations for improvements in deriving RfDs and RfCs (see Chapter 5 of the present report for a discussion of RfD and RfC derivation). The committee focused on aspects of the EPA document related to toxicity testing, particularly its Chapter 3, which reviews the adequacy of tests in EPA guidelines (EPA 2005) for deriving RfDs and RfCs for chronic, acute, and other less-than-lifetime exposures.
EPA reviewed information generated from currently required acute, short-term, chronic, and specialized toxicity studies. It then identified data gaps with regard to the assessment of life stages, end points, route and duration of exposure, and latency of response and made recommendations to fill the gaps. Options for alternative testing systems were also presented. EPA (2002) was careful to point out that
the intent of this review is not to suggest that additional testing be conducted for each and every chemical in order to fill in the information gaps identified for those organ systems evaluated. Nor is it suggested that the alternative testing protocols discussed in this chapter be conducted for every chemical or become part of current toxicology testing requirements or that these alternative protocols are the only options available. Rather, it is the goal of this document to provide a basis for the development of innovative alternative testing approaches and the use of such data in risk assessment.
Specifically, EPA made a number of observations about the generation of toxicity data under the test guidelines for exposures at different life stages. Most of the standard adult toxicity-testing guidelines for acute, subchronic, chronic, and carcinogenicity testing were not designed to evaluate different life stages, and that has led to substantial gaps (see Figure 6-2). Acute and short-term testing is done only in prenatally exposed animals and in young adults, not in postweaning young animals or aged animals. In addition, only a few toxicologic end points are evaluated in the EPA-required acute toxicity (lethality) studies. EPA discusses how data typically collected in subchronic studies—such as hematologic, clinical, and histologic data—could augment acute studies so that acute RfDs could be developed on the basis of end points other than lethality.
Subchronic and chronic toxicity studies are conducted in young adult animals, and exposure in the rodent chronic–carcinogenicity studies continues to the age of 2 years, considered by EPA to be into old age.
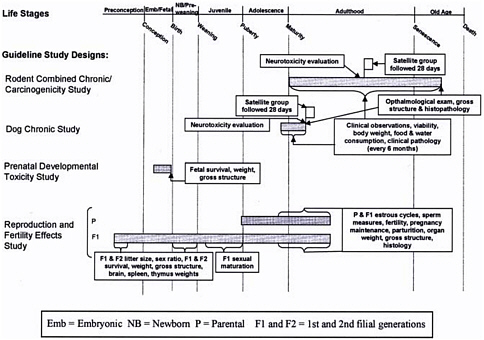
FIGURE 6-2 Guideline study designs used to derive the oral reference dose. Life stages during which exposure occurs (gray), times at which observations are made, and end points evaluated are indicated. Source: EPA 2002.
No subchronic or chronic toxicity studies include exposures beginning in the prenatal period, in the early postnatal period (before weaning), or in animals younger than 6-8 weeks old. Tests for reproductive effects provide data on subchronic exposures of animals that are exposed from before birth up to mating of the F1 males and females and through pregnancy of the F1 young adult females. No subchronic-toxicity evaluations are conducted in aged animals.
EPA emphasized the need to collect pharmacokinetic data that would help define the internal dose of the active agent to the target site. That information could be used to improve study design, study interpretation, dose scaling, and route extrapolation. EPA also notes that there are no guideline protocols for pharmacokinetic evaluations related to exposures and outcomes during infant development or later in old age. The pharmacokinetic and pharmacodynamic datasets are described as useful in determining the interspecies and intraspecies uncertainty factors and in calculating human equivalent exposure concentrations and doses.
As a result of its review, EPA identified several important gaps in the current toxicity-testing framework and concluded that there was minimal evaluation of the following: aged animals in general, but especially after early exposures; some systems and end points (for example, cardiovascular and immunologic) in terms of both structure and function; latency and reversibility; effects of acute and short-term exposure, which are needed to determine acute and short-term RfDs and RfCs; pharmacokinetics; and portal-of-entry effects, especially for substantial dermal exposure.
EPA’s recommendations address two main objectives for testing strategies—evaluating a broader array of end points and life stages and increasing information on mechanism or mode of action to improve the human relevance of risk assessment—and include the following:
-
Develop a strategy for alternative approaches to toxicity testing, with guidance on how and when to use existing and newly recommended guidelines.
-
Develop guidelines or guideline study protocols that will provide more systematic information on pharmacokinetics and pharmacodynamics (that is, mechanism or mode of action), which includes information at different life stages.
-
Develop protocols for acute and short-term studies that provide more comprehensive data for setting reference values.
-
Modify existing guideline study protocols to provide more comprehensive coverage of life stages for both exposure and outcomes.
-
Collect more information on less-than-lifetime exposures to evaluate latency and reversibility of effect.
-
Develop guidelines or guideline study protocols to assess immunotoxicity, carcinogenicity, and cardiovascular toxicity at different life stages.
-
Explore the feasibility of setting dermal reference values for direct toxicity, including sensitization, at the portal of entry.
EPA explored, but did not endorse, different testing protocols for acute, subchronic, and chronic toxicity testing to address gaps pertaining to life stage, duration of exposure, and latency. Specifically, EPA described an alternative acute-toxicity testing protocol and two alternative chronic toxicity-testing protocols. The purpose of the alternative acute-toxicity testing protocol is to provide hazard and dose-response information after a single acute exposure. That protocol uses a control group and at least three dose groups with 10 animals per sex per group. Clinical signs of toxicity are recorded daily, and food consumption and body weights are recorded on days 1-4, 8, and 14. Five animals per sex per group are killed 3 days after dosing, and the remaining animals are killed 2 weeks after dosing. At both times, urinalysis and hematologic and clinical-chemistry analyses are conducted to address potential reversibility and latency of effects within 14 days of dosing. In addition, the animals are necropsied, organ weights are recorded, and the organs are examined histologically. On the basis of other toxicologic data, this study may be conducted with animals at different life stages and include other end points.
EPA described two chronic protocols with continuous exposure through all life stages. The first is essentially the in utero carcinogenicity evaluation used by the Food and Drug Administration (FDA) for food additives but with yearly interim kills and study termination when the animals reach the age of 3 years rather than 2 years. It involves exposure before mating and then continuous exposure of offspring. In addition to routine clinical pathology and histopathologic evaluations, unspecified neurotoxicity and immunotoxicity testing would be conducted yearly. The second protocol is a unified screening study that has at least four segments: a two-generation reproduction and fertility study, an expanded chronic–carcinogenicity study (Figure 6-3), a developmental-toxicity
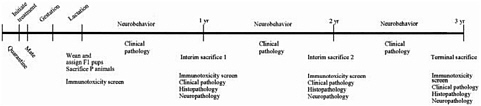
FIGURE 6-3 Expanded chronic-carcinogenicity study. Source: EPA 2002.
study, a developmental-neurotoxicity study, and an optional continuous-breeding study.
EPA identified important challenges to the chronic protocols, including the total number of animals needed and the feasibility of conducting studies as complex and large as the two chronic studies without increasing experimental error. EPA presented the chronic protocols “to demonstrate the advantages (and disadvantages) of exploring nontraditional testing paradigms” and noted that using them in a regulatory setting would require thorough discussion between EPA and registrant scientists.
Committee’s Evaluation of Environmental Protection Agency Review of Data Needs for Risk Assessment
Overall, the committee agrees with EPA’s analysis and conclusions regarding data gaps. For the most part, EPA’s recommendations, if implemented, would improve RfD and RfC development. They encourage the development of innovative toxicity-testing protocols, and following EPA’s recommendations for any one chemical would enhance the depth of information on pharmacokinetics, life stages, and end points available for hazard identification and dose-response assessment. However, such an intensive toxicity-testing approach would probably be applied to only a small fraction of chemicals in commerce, would increase the number of animals used to study one chemical, and might even reduce the numbers of chemicals tested. The EPA analysis and recommendations do not address the overall goals of increasing the breadth of coverage of chemicals, conserving animals, and reducing costs and other expenditures. Ultimately, the EPA recommendations need to be evaluated within a lar-
ger strategy that considers the uses of toxicity testing for purposes other than to support quantitative risk assessments (for example, chemical-class testing to guide broader screening and coverage).
EPA also made recommendations for research on uncertainty factors used in dose-response assessment. The committee agrees with EPA’s conclusion that it is important to research the basis of uncertainty factors. For example, research on the intraspecies uncertainty factor is needed to evaluate whether it adequately addresses the full range of variability of response due to different life stages, genetic susceptibility, and other factors. However, the committee is reserving its detailed comments on the issues surrounding uncertainty factors for its second report.
Specifically, the committee identified five major issues raised in the EPA review that require evaluation and comment: (1) the presence of data gaps in current toxicity-testing approaches, (2) a possible need to refine acute-toxicity testing protocols to support short-term risk assessments, (3) concerns about methods to incorporate pharmacokinetic and pharmacodynamic data into toxicity-testing approaches, (4) questions regarding incorporation of data on direct dermal toxicity into RfD development, and (5) a need to reconsider current toxicity-testing strategies systematically with an eye to improving efficiency and effectiveness. In addition to comment on those major issues, the committee offers some general observations on the acute and chronic toxicity-testing protocols that are explored in Section 3.3 of the EPA report as alternatives to guideline studies.
Presence of Data Gaps
The committee agrees with EPA that there are numerous data gaps in life stages and end points covered in current testing approaches and in functional assessments of some organ systems. However, there are insufficient data to determine the degree to which those data gaps have practical significance in risk assessment or whether they are primarily of theoretical or academic concern. Depending on how the data are used in risk assessment, some of the data gaps may have little effect on the final outcome, whereas others may be very important. The committee cautions against adding testing requirements only for the sake of thoroughness, because such an approach can result in a substantial waste of animals and resources with little gain. The challenge is to try to cover a broader range
of end points and life stages with improved sensitivity to subtle or functional effects without indiscriminately expanding current toxicity-testing approaches.
The committee favors a two-pronged approach in the near term. Such an approach includes relatively modest enhancements of current protocols designed to improve screening for a somewhat wider array of end points in a somewhat broader array of life stages. Such enhanced protocols could trigger more in-depth testing. Adequate second-tier evaluation of such end points as immunotoxicity and cardiovascular toxicity may require the creation of new test guidelines with indications of what would trigger such testing. Research is needed to determine the practical effect of filling or not filling some of the data gaps. In the meantime, it is presumed that current uncertainty and adjustment factors applied by EPA to derive reference values are sufficiently large, although that conclusion has not yet been established. The magnitude of uncertainty factors used in deriving reference values can be reassessed once the enhanced protocols are developed, implemented, and validated and a comprehensive review of outcomes is conducted for a set of test chemicals.
It is also important to look beyond animal toxicity data for help in resolving some of the questions. Epidemiologic studies with reliable exposure assessment can shed some light on the likelihood that current toxicity-testing data are missing important end points or are insufficiently sensitive to be applied to life stages not studied. For example, the prospective National Children’s Study1 or other prospective cohort studies that include children and the elderly may be helpful in ascertaining differences in susceptibility. Studies of workers who handle various agents can be of great help in assessing effects on the immune system, cardiovascular system, and functional end points that are not now well assessed with animal studies. However, human data will be available on only chemicals that are already in use and, because of problems with exposure assessment and other difficulties in epidemiologic studies, can be collected only on a small subset of existing chemicals. In addition, legal and ethical issues limit the design of human studies that can be conducted and integrated into toxicity-testing strategies.
|
1 |
The National Children's Study can be found at http://nationalchildrensstudy.gov/. |
Refinement of Acute-Toxicity Testing Protocols
The committee agrees with EPA that the current protocols for acute toxicity testing focus on lethal effects and gross observations and generally do not provide adequate information for acute and short-term RfDs and RfCs. EPA has identified a regulatory need for improved data for setting acute RfDs and RfCs, and such data are within reach of current toxicity-testing approaches. Improved acute-toxicity assessment may be particularly important in situations where there is high worker exposure and for the development of acute-exposure guideline levels (AEGLs) used to design emergency response plans for accidental releases of chemicals. However, conducting acute protocols that address latency, reversibility, and differential susceptibility for all toxicity end points currently required in subchronic and chronic protocols (such as hematology, clinical chemistry, pathology, functional tests, and detailed clinical observations) will lead to very complex animal studies. Before such complex protocols are conducted, acute LD50 studies, repeated-dose toxicity studies, and human data should be evaluated to determine the need for these studies and ultimately to guide the design of such studies.
Pharmacokinetics2 and Pharmacodynamics
The committee agrees with EPA that generally little information is available on pharmacokinetics, including possible differences across life stages. Although there is a need to develop more information on pharmacokinetics and pharmacodynamics, it is critical to define the purpose of such studies to avoid the creation of data that are unlikely to be used and that constitute a waste of animals, time, and resources. Data on absorption, distribution, metabolism, and excretion (ADME) should be used in guiding toxicity testing, including identifying the most relevant routes of exposure. Beyond the ADME studies, additional data should not be required routinely but instead case by case. It is important to use caution in drawing major conclusions on the basis of relatively few pharmacokinetic and pharmacodynamic data. For example, such data may not always be sufficiently explanatory to support major conclusions about
concordance or extrapolations to other routes of exposure. It is often tempting to assume that one mode of action that has a strong hypothetical basis supported by some data fully explains the toxicity of a given chemical. However, many chemicals have multiple modes of action, and these can depend on exposure route, concentration, and duration. Furthermore, there can be competing plausible hypotheses that may not have been considered or subjected to study.
Dermal Portal of Entry
The committee considered EPA’s conclusion that there is a need to improve the assessment of portal-of-entry effects, especially on the skin, in the risk-assessment process. EPA emphasized dermal effects because oral and inhalation effects are already recognized as important routes of exposure and require specific test data for risk assessments in, for example, its pesticide testing programs. Current protocols for dermal exposure, when they are applied, do evaluate dermal toxicity, including sensitization, but the overall assessment of systemic and portal-of-entry effects after dermal exposure is limited. The committee agrees with EPA that there is a general need for better dermal-uptake data and for a better understanding of uptake after injury to the skin. However, it is important to consider whether skin is an important route of exposure before triggering an effort to set a dermal RfD. Again, human data can be useful in assessing the effects of dermal exposure. Worker data and clinical reports have been among the main sources for identifying dermal irritants and sensitizers. Such data could be collected more systematically and used preferentially in setting dermal RfDs for existing chemicals. In addition, there should be a more systematic effort to gather postmarketing data on chemicals to ascertain, for example, whether dermal toxicity is reported.
Toxicity-Testing Strategy
The first recommendation of Chapter 3 of the EPA review is that EPA “develop a strategy for alternative approaches to toxicity testing, with guidance on how and when to use existing and newly recommended guidelines” (EPA 2002). The committee agrees that such a new strategy is needed to improve efficiency, reduce animal use, increase the number
of chemicals that are screened for toxicity, and address some of the data gaps noted above. Such an improved strategy could include setting priorities for screening and testing of agents and incorporating such factors as use, exposure, pharmacokinetic data, and new screening tools. The committee’s second report will be designed to help to create just such a new strategy.
Specific Testing Protocols
EPA explored alternative testing protocols for acute and chronic toxicity testing. In making the following comments on those protocols, the committee recognizes that EPA offered them primarily to stimulate new ideas. In addition, the committee recognizes the difficulty of evaluating specific protocols out of the context of the overall testing strategy.
Acute-Toxicity Testing Protocol
EPA proposes an alternative acute-toxicity testing protocol that provides more-comprehensive toxicity data for derivation of the acute RfD than its current acute-toxicity test guidelines (EPA 1998a,b,c) that provide data to calculate acute LD50s and LC50s and to select doses for use in other studies. Taken in isolation, the alternative toxicity study may not fully address all the important data gaps raised by EPA in its analysis of acute toxicity tests, such as reversibility, latency, and end-point assessment in detail and at different life stages. The animal group size of five is small for conducting hematology, clinical chemistry, and pathology evaluations, and this limits the capacity to detect effects, particularly subtle ones. For example, statistical significance of dichotomous findings of effects in a group of 10 animals requires that 40% of the animals be affected if the effect rarely occurs in treated animals; greater proportions are required for more common effects or smaller groups. Even so, compared with the current acute toxicity tests, the protocol is relatively resource-intensive with respect to animal use and histopathologic examination.
Designing comprehensive acute toxicity testing could be guided by existing data, including data from repeated-dose studies. For example, acute risk assessment could first be conducted on the basis of short-term repeated-dose studies and developmental-toxicity studies. When human
exposures are well understood, the margin of exposure could be examined. If the margin of exposure is too small, sensitive acute toxicity studies could be designed and performed to focus on the more sensitive end points of concern. Indicators of acute toxicity could also be added to repeated-dose studies, such as an early assessment of hematology and clinical-chemistry end points and more frequent clinical observations on the first few days of repeated-dose oral, inhalation, or dermal toxicity studies. The limitations of adding early assessments to repeated-dose studies are that not all acute end points may be identified and the dose-response relationships may not be adequately characterized, especially those which might result from higher concentrations that cannot be used in longer-term repeated-dose toxicity studies.
Alternative Chronic Testing Protocols
EPA also presented two chronic protocols that address continuous exposure through all life stages as alternatives to current test procedures. The chronic–carcinogenicity study would evaluate the potentially increased sensitivity of both developing and aging animals to chronic and carcinogenic effects of chemicals. It would be relevant to chemicals to which there may be substantial chronic exposure of the general population or of workers. Studies of that type have already been conducted by industry and contract laboratories.
According to current combined chronic–carcinogenicity test guidelines (EPA 1998d), 50 rats per sex per dose are required, for a total of 400 animals. The expanded study specifies 25 rats per sex per dose for each annual interim kill, for a minimum of 500 animals. In practice, more animals may be necessary to account for the possibility of decreased survival. EPA addressed the issue of increased animal use, in part, by suggesting fewer animals per segment (for example, 20 per sex per group) or only one interim kill (for example, at 1.5 years). EPA acknowledged that reducing the number of animals to 20 per sex per group decreases the statistical power to detect tumors but did not suggest further study changes to resolve the issue satisfactorily.
Another challenging aspect of the study is setting the doses to produce sufficient toxicity while yielding an adequate survival rate by the age of 3 years. Setting doses too low results in a study that is not sufficiently sensitive to detect an effect; setting doses too high can increase mortality, leaving the group size reduced and thereby compromising the
statistical power to detect effects. Also, the feasibility of conducting a study of a specified duration will depend on the survival characteristics of the strain chosen, which can range from less than 2 years to 4 years for different rat strains. Instead of selecting a predetermined termination time, the study could end when some prespecified fraction of control or treated animals is surviving (for example, 20%).
Another limitation of the expanded chronic–carcinogenicity study is that it may not provide an adequate basis for quantifying increased susceptibility to carcinogenicity at different life stages. That information is important for risk evaluations if people are exposed at different doses at a given age (for example, while breast-feeding and while being bottle-fed). For those reasons, alternative study designs should be considered, such as designs that include dose groups that are exposed briefly and followed for long periods. As alluded to by EPA, the study design makes difficult or impossible the assessment of whether toxicity is due to repeated chronic exposures or to shorter-term prenatal exposures.
Although the chronic–carcinogenicity study is a challenging study design, the committee supports the development of bioassays that address differential sensitivity to a chemical’s immunologic, neurologic, and cancer effects during different life stages. However, this study is limited in that it may be more difficult to attribute effects to any one life stage (that is, prenatal development period, the postnatal period before weaning, adolescence, and old age) and would not be useful for shorter-term risk assessment for different populations.
EPA presented the Unified Screening Study to stimulate ideas on how studies could be combined to limit animal use while expanding the toxicity evaluation across multiple generations to study all life stages and transgenerational effects. Further development will require considerable resources and expertise in the fields addressed by the different arms of the study. It would require development and pilot experimentation by such a group as the NTP before such a proposal could be implemented. The committee’s review below highlights a few important issues that may arise from conducting such a study.
The Unified Screening Study attempts to economize animal use in a multigeneration study so that fewer naïve animals would be used than if all the studies that would potentially be replaced by it were done separately. Substantially more animals would be needed than are now required in a two-generation study to ensure that sufficient numbers of animals were available for both the chronic study and the reproduction study and to guarantee survival of enough animals to the age of 3 years.
EPA (2002) states that animals “assigned to the chronic/carcinogenicity segment should be genetically diverse within each dose group and should … not be siblings.” For that segment, only one male and one female from each litter could be used to meet the need for 100 males and females per dose group. Thus, at least 100 litters per dose group would be needed. That is 5 times the number of litters required by the standard reproduction study (EPA 1998e) and would make such an experiment difficult to conduct. Further evaluation is needed regarding the importance of the requirement of genetic diversity in the cancer-bioassay segment of an expanded study like this one.
As with the expanded chronic–carcinogenicity study described above, a major challenge is dose selection. It is compounded by the fact that there are many arms to the study. Because many more animals per litter would be assigned to either the chronic study or the reproduction study, there is less room for error in setting doses. EPA (2002) states that “although it is assumed that treatment levels and route of administration will remain constant across all study segments, this approach to dose-setting and route selection may not always be optimal for every phase.” That may be especially true for dietary exposures in which chemical exposure of pups around the time of weaning can be much higher than exposure of adults and result in unacceptable toxicity in pups. Insufficient animals would then be available for both the two-generation reproduction study and the chronic–carcinogenicity study. Selection of dose is difficult for each of the studies when considered separately and will be exceedingly difficult for such a complex study. Setting doses either too high or too low could lead to repeat studies or addition of dose groups. Keeping doses the same in the different arms would generally mean lowering them to preclude toxicity in the most sensitive study arms, and that would reduce study sensitivity overall and decrease one’s ability to characterize the dose-response relationship.
As noted by EPA, another challenge in the development of the Unified Screening Study is the selection of strain. Sprague-Dawley and Wistar rats (outbred strains) are typically used in toxicity studies, including those of developmental toxicity, reproductive toxicity, and developmental neurotoxicity. Inbred Fischer 344 rats are often used in standard chronic–carcinogenicity studies. Thus, the Unified Screening Study would at a minimum require a change in the standard strain used (that is, for either carcinogenicity or reproductive and developmental-toxicity end points). EPA noted that changing the strain used in any type of study would compromise the use of historical data. Furthermore, a sensitive
strain to detect carcinogenicity (NTP 1984, 2005) may not be the optimal strain for developmental and reproduction studies or the optimal strain for longevity. The NTP is reviewing the issue of strains used in chronic testing, and results of this effort should be useful to EPA as it considers alternative test protocols (NTP 2005).
The idea of obtaining animals for the developmental-neurotoxicity segment from the reproduction and fertility study segment’s second generation (F2) is more complex than is presented by EPA. The major difficulty in the developmental-neurotoxicity segment is the need to maintain tight control of mating to manage logistically the multiple behavioral testing required during lactation and immediately after weaning. Lack of tight control over mating can easily cause behavioral testing for one time (for example, postnatal day 13) to spread out over many days and overlap into other testing periods, resulting in increased variability of the data. Such a study can be conducted, but it is not optimal for developmental neurotoxicity.
A protocol of the magnitude and complexity of the Unified Screening Study would require substantial development and evaluation before it could be implemented as a test requirement. EPA does not indicate exactly how the protocol would be applied. Some committee members found that routine use of the study as a first-tier screening test would be problematic for both scientific and practical reasons. They believed that potential disadvantages of the study outweigh its advantages, especially because it appears to preclude the use of toxicity results from one study to trigger or prevent additional evaluation in other studies in that all studies would be conducted simultaneously. Other members of the committee found that the study protocol could function as a component in a toxicity-testing strategy as applied to either selected chemicals with widespread human exposure or chemicals with initial indications of developmental toxicity. Some segments of the study could be applied in a tiered fashion, in which selected end points could be examined in followup studies if triggered by observations in earlier segments.
In addition to the scientific and strategic concerns, there are practical concerns that the studies are extremely resource-intensive and complex. The separate components are temporally tied to each other intimately, thereby decreasing flexibility of study starts in any laboratory that might be capable of conducting all arms of the experiment. Furthermore, few laboratories have the capability (for example, trained personnel and appropriate computer systems) to conduct multigeneration, developmental-neurotoxicity, and immunotoxicity evaluations. Hence, widespread re-
placement of current approaches by the Unified Screening Study could reduce the number of chemicals evaluated during the development of laboratory capacity.
In conclusion, EPA has explored alternative testing protocols but does not articulate how they would be incorporated into a testing strategy. The committee supports the notion of expanded tests that combine studies to limit animal use and provide more in-depth evaluations of end points and life stages. However, considerable development and evaluation may be required to ensure that tests are feasible, do not compromise study sensitivity, produce the desired data, and reduce the use of animals. The committee also points out that subchronic and chronic studies might be expanded to obtain better data for establishing acute reference values. The expanded bioassays may ultimately have a role as part of a broader testing strategy applied selectively to high-priority chemicals, but such approaches may not be amenable to widespread application.
International Life Sciences Institute Draft Proposals3
ILSI-HESI developed three draft white papers (ILSI-HESI 2004a,b,c) proposing a tiered-testing scheme that includes several tests that are currently part of EPA’s testing scheme for pesticides but also deletes some that are required. Modified protocols for some EPA guideline toxicity tests are also included. ILSI-HESI focused on EPA toxicity-testing approaches for pesticides, but in principle the approaches could apply to other compounds that are subject to toxicity testing.
One ILSI-HESI paper focuses on assessing systemic toxicity in young adult animals. For the purposes of the project, ILSI-HESI (2004a) defines systemic toxicity as “the potential adverse effects of agricultural chemicals on ‘young adults.’ ” The evaluation of effects of chemicals on different life stages—including reproduction, development, adolescence, and old age—is presented in a second paper. The third paper focuses on the acquisition and application of pharmacokinetic data in agricultural-chemical safety assessments. The overall testing scheme incorporating the three aspects is outlined in the second draft paper, on life stages.
The discussion of the testing scheme below focuses on the general concepts and approaches rather than the technical details of the draft papers.
Systemic-Toxicity Testing
ILSI-HESI proposes a tiered approach to systemic-toxicity testing that considers the use profile, the resulting exposure, and the toxicologic profile of the pesticide in identifying test requirements (ILSI-HESI 2004a). The goal is to improve the adequacy of the dataset for the variety of risk assessments required while reducing test artifacts and the number of animals used in testing and increasing the efficiency and accuracy of the safety-assessment process. The overall process used by ILSI-HESI included (1) identifying exposure durations that are important for pesticide risk assessment, (2) evaluating a database of EPA guideline test results on representative pesticides to determine the necessity of particular tests by evaluating how EPA used test results in setting the RfD, (3) developing or modifying toxicity tests to address risk-assessment needs, and (4) developing a flexible tiered-testing scheme that would be guided by pharmacokinetic data and the known toxicologic profile of the chemical.
Originally, the assessment of agricultural chemicals focused on continuous dietary exposure; that led to an emphasis on lifetime exposures and acute accidental exposures. Today, EPA conducts risk assessments to evaluate a variety of agricultural and residential pesticide exposures of different durations and at different life stages. ILSI-HESI identified the following human exposure durations for which risk assessments may be required: 1 day, 2-28 days, 1-6 months, greater than 60 months, and intermittent. ILSI-HESI compared those durations with current toxicity-testing requirements and concluded that the tests did not meet all the needs of risk assessors.
ILSI-HESI evaluated the toxicology database of results of subchronic and chronic pesticide studies to examine which studies were used most often in deriving the RfD or in classifying carcinogenicity. The analysis was used to differentiate between study types that should be retained or developed further for future use and types that should not. Of the hundreds of pesticides in the EPA database, data on 65 pesticides representing different classes were entered into a toxicity database developed specifically for the ILSI-HESI evaluation. Of the 65 pesticides selected, data on 28 were sufficiently robust to allow the kinds of comparisons that ILSI-HESI intended to make. Results of subchronic and
chronic studies were compared to examine the relative sensitivity of different test species and different exposure durations: 90-day studies in rats versus dogs, 90-day versus 1-year studies in dogs, 90-day versus 2-year studies in rats, 2-year rat versus 1-year dog studies, and rat versus dog studies as the basis of the chronic RfD. The effect of mouse carcinogenicity studies on cancer-hazard determinations, but not dose-response assessment, was also assessed.
For the dataset of 28 chemicals, the chronic RfD was based half the time on the rat and the other half on the dog. Significant quantitative differences were noted; most often, the dog was more sensitive in the 90-day study. It was concluded that both species should be used in pesticide testing. In the rat, the no-observed-adverse-effect levels (NOAELs) in 2-year studies were often lower (by more than a factor of 2) than those in the 90-day studies; in the dog, that was also the case for the 1-year versus 90-day study but somewhat less in frequency and magnitude. On the basis of those data, ILSI-HESI concluded that the 1-year chronic dog study did not affect the RfD assessment in an important way. Finally, the mouse carcinogenicity study was proposed for elimination because it was found to add little to the findings of the rat carcinogenicity study.4
After considering possible test redundancies and risk-assessment needs, ILSI-HESI selected core tests for initial toxicity evaluation. For assessing short-term exposures, a new 28-day rat study with recovery period and a modified 90-day dog study were proposed. ILSI-HESI proposed that single-exposure acute studies be performed if the NOAELs from those studies were too close to the predicted single-day human exposures.
The results of the 90-day dog and 28-day rat studies are compared, and the more sensitive species is taken as the more relevant unless compelling scientific data indicate otherwise. If the rat is considered the relevant species, the new 28-day rat study is used to assess the effects of human exposures of less than 6 months, and a 1-year rat study for exposures of more than 6 months. The 1-year rat study would be conducted as part of an expanded 2-year carcinogenicity assay; an interim kill would occur at 1 year. If the dog is found more relevant, the 90-day dog study would be used to evaluate effect of human exposures over 2 days
|
4 |
One important inconsistency between the draft papers is that the life-stages paper appears to eliminate carcinogenicity testing, but the systemic-toxicity paper includes the rat carcinogenicity test in the first tier (see Figures 6-4 and 6-5). That may have been an oversight in the draft life-stages paper. (Since completion of this interim report, the life-stages figure in the final paper [Cooper et al. 2006] was revised and now includes carcinogenicity testing in the first tier.) |
and up to 6 months; for exposures beyond 6 months, the 1-year rat or 90-day dog study would be used. A 24-month carcinogenicity study would also be available. The 28-day rat or 90-day dog studies would be considered the most relevant for human effects of exposures of less than 6 months. Performance of the longer-term or acute studies would be contingent on predicted human exposure scenarios. Figure 6-4 shows the tiered-testing strategy proposed by ILSI-HESI.
The pivotal studies of the testing strategy are the 28-day rat study and the 90-day dog study. In addition to a 14-day recovery group, the rat study would include satellite groups for neurotoxicity and immunotoxicity evaluations as indicators of specific effects to trigger additional specialized studies. If the indicators were affected in that study, second-tier specialized testing would be performed to investigate indicated end points further or to characterize mode of action. Tissues would be examined as specified in the EPA guidelines for the oral 90-day rat study (EPA 1998f). The 90-day dog study would extend the existing guideline study to include cardiac and pulmonary evaluations and would include
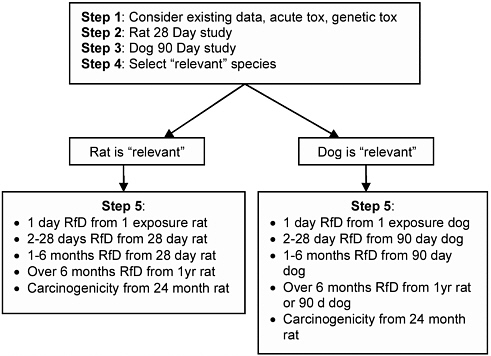
FIGURE 6-4 ILSI-HESI draft proposed tiered approach for systemic-toxicity evaluation. Source: ILSI-HESI 2004a. Reprinted with permission. This figure appears in the final paper of Doe et al. (2006) in Critical Reviews in Toxicology; copyright 2006, Taylor & Francis Group, LLC.
blood draws for pharmacokinetic analyses. Treatment-group size would be increased in the dog study from four to six animals.
The ILSI-HESI tiered-testing strategy assumes that genotoxic chemicals would not be developed as pesticides and emphasizes the use of pharmacokinetic data from basic ADME studies to support the design of toxicity studies (for example, using those data to determine the highest dose to test and the duration of the recovery period). ILSI-HESI also acknowledges the potential future use of novel high-throughput screens that can predict potential adverse effects of concern and thereby indicate relevant end points to incorporate into the tier I studies. The new technologies would be derived from in vitro screening, computational biology, molecular biology, and systems biology. ILSI-HESI envisions that high-throughput screening could potentially include in vivo and in vitro biologic systems in which the active ingredient is tested, RNA or protein isolated from the cell culture or target tissues is analyzed, and genomic or proteomic assays are used to identify markers of toxicity that suggest the potential for particular adverse health effects.
Life Stages
ILSI-HESI proposes a tiered-testing approach for “assessing the potential adverse effects of agricultural chemicals on preconception, embryo/fetal and newborn/pre-weaning life stages and on adults of all ages, including the young and aged” (ILSI-HESI 2004b).
The centerpiece of the proposed life-stages paradigm (Figure 6-5) is a new testing protocol for an F1 extended one-generation study. In that study, mature parental males are treated for 4 weeks and parental females for 2 weeks before mating. Treatment is continued for males and females during mating, for females during gestation and lactation, and for males until it is confirmed as not needed for a second mating. Selected F1 pups (three males and three females per litter) are treated continuously until postnatal day 70 and are given a much more comprehensive histopathologic evaluation at postnatal days 21 and 70 than is now required in the multigeneration study (EPA 1998e). Developmental-neurotoxicity assessments (motor activity, functional observational battery, and neuropathology), developmental-immunotoxicity assessments (antibody plaque-forming cell response), and additional toxicity assessments based on the results of a 28-day systemic-toxicity test also would be conducted.
The proposed life-stages tier 1 would include a rabbit developmental-toxicity study and the extended one-generation rat reproduction study.
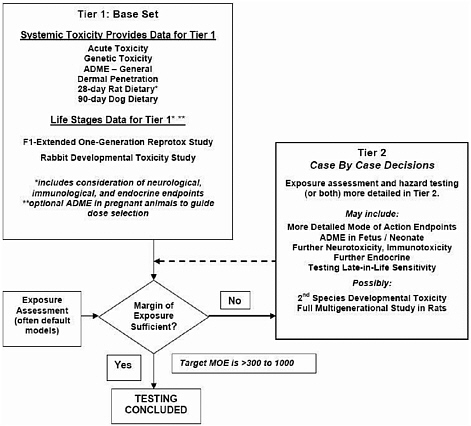
FIGURE 6-5 ILSI-HESI draft proposed tiered life-stages testing scheme for pesticides. Source: ILSI-HESI 2004b. Reprinted with permission. This figure has been modified and appears in the final paper of Cooper et al. (2006) in Critical Reviews in Toxicology; copyright 2006, Taylor & Francis Group, LLC.
The first tier incorporates a number of features from the current EPA reproduction and developmental test guideline studies, such as evaluation of comprehensive reproductive and developmental end points. It also includes end points and evaluations not required by EPA guidelines, such as more comprehensive postnatal histopathologic evaluation; neurologic, immunologic, and endocrine end points after exposure during gestation and lactation; and pharmacokinetic observations at key stages of gestation and lactation.
EPA now requires a two-generation rat reproduction study and developmental-toxicity studies in two species. The ILSI-HESI draft paper discusses the potential concerns regarding removal of the second-generation part of the multigeneration study and the rat developmental-toxicity study. ILSI-HESI concluded, on the basis of an unpublished EPA evaluation of the existing pesticide database, that the second gen-
eration had relatively minor effects on risk assessment. It hypothesized that rat developmental toxicity may be adequately addressed with the extended one-generation rat study. Additional developmental-toxicity or multigeneration studies in the second tier could be triggered if the margin of exposure were not sufficiently large (for example, less than 300), if there were a substantial difference between the rat and rabbit NOAELs, or if there were effects in rabbit fetuses of a type unlikely to be detected postnatally in rat pups. In addition, extension of the one-generation study into a multigeneration study would be triggered on the basis of potential concerns about possible effects on reproduction, such as alterations in anogenital distance, timings of vaginal opening or preputial separation, or altered estrous cycles.
The ILSI-HESI approach was designed to reduce the number of animals used, the cost, and the time involved in testing while improving the data on several specific end points available for risk assessment. The proposed tiered approach also incorporated an evaluation of margin of exposure with hazard and dose-response assessment to determine whether additional testing would be needed. Finally, the ILSI-HESI draft paper concluded that the intraspecies uncertainty factor of 10 was sufficient to address risks to the elderly on the basis of few toxicity data on aged rats and human data. ILSI-HESI argued that pharmacokinetic differences due primarily to reduced renal function are what drive susceptibility. Relevant triggers for further unspecified animal studies in aging animals include effects on renal function, the immune system, the nervous system, the cardiovascular system, and the liver. The proposal did not specify how and when additional tests would be triggered.
Pharmacokinetics
The ILSI-HESI report The Acquisition and Application of Absorption, Distribution, Metabolism and Excretion (ADME) Data in Agricultural Chemical Safety Assessment contains recommendations for generating and using pharmacokinetic information in risk assessment (ILSI-HESI 2004c). It suggests a tiered approach that consists of basic, intermediate, and advanced studies: basic studies to support dose selection and evaluation of half-life; intermediate studies to guide study interpretation and to provide information on absorbed dose, tissue persistence, and tissue accumulation; and advanced studies to aid in understanding of mode of
action and to support extrapolations across routes of exposure and among species.
ILSI-HESI argues that it is important to base the dose, route, and species used in toxicity testing on an understanding of a compound’s behavior—such as saturation of absorption, elimination pathways, and metabolic profiles—pover a range of doses. The proposed basic tier includes studies to collect blood time-course data after single oral and intravenous exposure and other pharmacokinetic studies to establish the extent and rate of absorption, bioavailability, general distribution of the chemical, extent of metabolism, routes and rates of excretion, half-life, tissue persistence, and clearance. The proposed second tier includes routine metabolite identification and pharmacokinetic sampling during toxicity studies and studies of tissue distribution, serum protein binding, and in vitro metabolism in rodents and other species. If first-tier findings indicate biliary elimination or enterohepatic recirculation, followup studies in the second tier would be conducted. The third tier could include studies aimed at better understanding of route-specific pharmacokinetic differences and further studies to support the application of physiologically based pharmacokinetic (PBPK) models in risk assessment.
Committee’s Evaluation of International Life Sciences Institute Draft Proposals
Through a tiered-testing scheme, ILSI-HESI proposes substantive modifications of EPA’s toxicity-testing approach for pesticides. In developing its proposal, ILSI-HESI identified some potential omissions and redundancies and possible improvements in existing tests. Recommendations include changing required toxicity-testing durations, removing some currently required guideline studies, modifying other studies to increase end-point coverage, triggering specialized studies on the basis of findings in the core set of toxicity tests, and generating chemical-specific pharmacokinetic data to inform study design and data interpretation. Several recommendations address the goals of conserving animals and other resources and increasing the depth of some particular areas, such as neurotoxicity. The new study designs would result in coverage of a broader array of end points, improved sensitivity to subtle or functional effects, and information on reversibility and latency after 28-day exposures. However, more rigorous evaluation is needed to determine whether the expanded designs compensate for the elimination of particu-
lar studies, and some important data gaps with regard to life stages were not adequately discussed.
Several studies that are required in EPA pesticide testing are not included in the first tier of the ILSI-HESI proposal: the rat teratology, 1-year dog, mouse carcinogenicity, and two-generation reproduction and fertility studies. The following discussion provides committee comments on the removal of those studies and highlights the committee’s concerns.
As pointed out by ILSI-HESI, no single species has been shown to be the most predictive of human teratogenicity. ILSI-HESI proposes removing the rat teratology study and using the extended one-generation study and rabbit teratology study to evaluate developmental effects. Although the proposed one-generation study substantially improves postnatal evaluation of many nonreproductive end points after in utero and postnatal exposures, it is unclear whether the proposed study would be as sensitive as a rat teratology study for prenatal developmental-toxicity end points or would adequately reveal the potential hazard and trigger a followup study. Furthermore, EPA often bases acute reference values on the rat teratology study. In contrast, postnatal effects in a one-generation study are not typically used for deriving acute reference values. The effect of eliminating the rat teratology study on hazard identification and setting of the acute reference value should be evaluated if the proposal is pursued. Careful validation using known development toxicants is needed.
On the other hand, the proposed extended 28-day rat study, with improvements as discussed above, should provide a better means of assessing short-term exposures than the 90-day rat study and the existing acute toxicity studies used to establish LD50s. The 28-day rat study is also being proposed to assess risks posed by human exposures as long as 6 months. The extent to which the 28-day study improves on the 90-day rat study for exposures longer than 1 month is unclear, and further effort to assess this issue is needed. In a similar vein, the extended 90-day dog study with its improved evaluation of critical end points provides a better subchronic dataset than the current guideline 90-day dog study but does not offer substantial advantages over the current study regarding exposure-duration data gaps. Because the 90-day dog study and the 28-day rat study are proposed to provide core data for assessing human exposures of up to 6 months, further assessment of their use for long-term human exposures is needed; this is critical for the dog study because in cases where the rat is not a good model the 90-day dog study potentially could serve as the basis of the RfD for lifetime human exposure.
The proposal to eliminate the chronic 1-year dog study and the mouse carcinogenicity study is based in part on a review of a dataset on only 28 pesticides. The committee agrees with the overall approach of reviewing the redundancy and importance of tests with retrospective analyses of pesticide-study findings, but it finds that the review in the proposal is too limited to support firm conclusions. For example, for the 21 cases in which comparisons could be made, there were eight cases (38%) in which the NOAEL from the 1-year dog study was less by more than a factor of 2 than that from the 90-day dog study. Although some of the differences may be due to dose spacing in the studies, the differences indicate the ambiguous nature of the comparison and the need for a larger, systematic evaluation. Similarly, the carcinogenicity comparison was based on only 23 chemicals for which rat and mouse carcinogenicity studies were conducted—five showing tumors only in the rat, four only in the mouse (three in liver), six in both species, and eight without tumors—again too few comparisons to support firm conclusions. Quantitative comparisons of dose-response characteristics on the basis of the benchmark dose or cancer potency were not made. Thus, the retrospective review was not sufficiently extensive or rigorous to support study removal.
Nonetheless, the committee strongly supports the general approach used by ILSI-HESI of evaluating existing databases in considering testing strategies, and it recommends a further review based on a larger, more robust dataset. A database of test results from guideline and special studies of the numerous pesticides in the EPA database could be used to evaluate the redundancies and gaps in pesticide testing further. Shorter-term studies, such as 28-day studies (for example, OECD Test Guideline 407), should be evaluated in addition to 90-day and carcinogenicity studies. It would also be informative in assessing study sensitivity to compare benchmark doses in addition to NOAELs and lowest-observed-adverse-effect levels (LOAELs). When comparisons of relative sensitivity across species are made, those evaluations should consider what the most appropriate dose scale (for example, mg/kg3/4 per day vs mg/kg per day) should be. Ideally, such evaluations would be conducted blind, without knowledge of the particular pesticide being reviewed. The committee endorses further broad retrospective reviews extending beyond the pesticide database to evaluate study reproducibility and predictability among and between in vivo and in vitro findings. In conducting and assessing retrospective analyses, it should be recognized that the power of human studies is often too low to be of value for
evaluations of predictability and study validation. Ultimately, reviews should consider how regulatory decision-making might change with the elimination of some test protocols and the role that test redundancy plays in safety assessments.
The EPA (1998g) approach in its guidelines for pharmacokinetic testing of pesticides is tiered and focuses on the generation of data to aid in interpreting toxicity tests and risk-assessment applications. The ILSI-HESI proposal also is tiered and focuses on generation of similar data, but it emphasizes the generation of pharmacokinetic data to aid in dose selection and other aspects of study design, which is also an important objective. The ILSI-HESI proposed first tier is similar to EPA’s first tier, which provides a minimal dataset from standard chemical disposition ADME studies. EPA’s second-tier testing is to address specific questions of importance for risk assessment that arise from the toxicology database on the chemical and the tier 1 testing. It can include data generation for PBPK modeling. EPA’s second-tier testing requirements are not fixed and can be developed through cooperative agreements with industry. Unlike EPA’s, the ILSI-HESI second tier includes routine metabolite identification and pharmacokinetic sampling during some toxicity tests. To ensure study relevance, both EPA and ILSI-HESI schemes emphasize an iterative process of data collection and use of data analysis in designing pharmacokinetic studies for specific compounds; no single prescribed series of studies is likely to provide necessary and adequate pharmacokinetic datasets on every compound.
The possibility of increases in susceptibility to cancer at particular life stages was acknowledged. The ILSI-HESI proposal assumes that genotoxic chemicals will not be developed as pesticides and that the end points added in the one-generation study will provide adequate indication of toxicity and carcinogenicity to trigger necessary testing involving perinatal exposure; however, ILSI-HESI did not identify the indicators that might trigger a perinatal carcinogenicity study. Some nonmutagenic compounds, such as estrogen-receptor agonist and antagonists, may elicit effects in critical windows of susceptibility, and the possibility of tier 2 testing was raised if concerns were triggered by mode-of-action information, but no specific proposals were made. Similarly, no specificity was given regarding when to follow up on possible enhanced sensitivity in aging animals.
The tailoring of the testing regime to meet risk-assessment needs is a strength of the ILSI-HESI proposal. Exposure considerations, such as the margin of exposure between doses that produce effects in animals
and expected human exposures to pesticides, also provide a conceptual framework for guiding the selection and extent of testing. The ILSI-HESI proposal to trigger further testing when the margin of exposure falls below some threshold value is a reasonable approach to limiting testing to issues of public-health importance. Still, although using exposure to guide selection of studies may work well for chemicals to which exposure can be relatively well defined, it may not work as well for industrial chemicals, because the degree and circumstances of human exposures can be difficult to predict or adequately assess.
To conclude, the proposed changes in the scheme for pesticide testing may affect the probability of finding some effects and change the volume of evidence available to an assessor in judging the presence or importance of an effect. The changes include eliminating the second species in teratogenicity and carcinogenicity testing, using the 90-day dog study as an indicator of chronic toxicity, and altering dose selection. Cumulatively, it is unclear how the different aspects of the proposal would affect the overall fidelity of the testing process. Thus, another important issue for evaluation is the overall effect of the changes on the sensitivity and reliability of the testing process. The committee notes that redundancy of testing is a critical part of the weight-of-evidence approach that may have been overlooked in the ILSI-HESI evaluation. More-limited testing and less redundancy could mean less confirmatory evidence and greater potential overall for reduced sensitivity of the testing strategy. Making decision-making more conservative, erring in the direction of false positives, or using greater uncertainty factors may address those issues, as would corresponding adjustments of risk-assessment guidance documents that emphasize positive results of multiple studies for confirmatory evidence.
REACH Program
The European Union’s REACH program was adopted by the European Commission on October 29, 2003. Its primary goal is to obtain data on and appropriately regulate some 30,000 chemicals that are produced or imported in excess of 1 metric ton per year and on which there is little toxicity and environmental information. In eliminating the practical distinction between “new” and “old” chemicals, it differs from the toxicity testing and risk assessment of chemicals under the U.S. Toxic Substances Control Act.
TABLE 6-1 REACH Testing Strategy
|
Test |
1 metric ton or more |
10 metric tons or more |
100 metric tons or more |
1,000 metric tons or more |
|
Skin irritation or skin corrosion |
Ra |
Ra |
Ra |
Ra |
|
In vivo skin irritation |
NR |
Ra |
Ra |
Ra |
|
Eye irritation |
Ra |
Ra |
Ra |
Ra |
|
In vivo eye irritation |
NR |
Ra |
Ra |
Ra |
|
Skin sensitization |
Ra |
Ra |
Ra |
Ra |
|
In vitro gene mutation in bacteria |
R |
R |
R |
R |
|
In vitro cytogenicity in mammalian cells |
Rb |
Ra |
Ra |
Ra |
|
In vitro gene mutation in mammalian cells |
Rb |
Rc |
Rc |
Rc |
|
In vivo mutagenicity |
Pa |
Pa |
Pa |
Pa |
|
Acute toxicity by oral route |
NR |
Ra |
Ra |
Ra |
|
Acute toxicity by inhalation |
NR |
Ra |
Ra |
Ra |
|
Acute toxicity by dermal route |
NR |
Ra |
Ra |
Ra |
|
Repeated-dose toxicity |
NR |
NR |
NR |
Pb |
|
Short-term repeated-dose toxicity (28 days) |
NR |
Ra |
Ra |
Ra |
The REACH program has four major components (EU 2004). First, a registration process requires manufacturers and importers of chemicals to obtain relevant information on their substances and to use the data to manage chemicals more safely. Second, unnecessary testing is avoided by having government regulators evaluate industry testing proposals and check compliance with registration requirements; evaluation also enables agencies to investigate chemicals that potentially pose risks. Third, substances with properties of very high concern will be subject to authorization; applicants will have to demonstrate that risks are adequately controlled, and uses of high-risk substances may be authorized if socioeconomic benefits outweigh the risks and there are no suitable al ternative substances or technologies. Fourth, reducing testing in vertebrate animals is encouraged, and toxicity testing is subject to data-sharing. The committee focuses its evaluation on the overall toxicity-
|
Test |
1 metric ton or more |
10 metric tons or more |
100 metric tons or more |
1,000 metric tons or more |
|
Subchronic toxicity (90 days) |
NR |
Pb |
Pb |
Pb |
|
Long-term repeated-dose toxicity |
NR |
NR |
NR |
Pb |
|
Reproductive toxicity |
NR |
Ra |
Pb |
Pb |
|
Screening for reproductive and developmental toxicity |
NR |
Ra |
Ra |
Ra |
|
Developmental toxicity |
NR |
Ra |
Ra |
Ra |
|
Two-generation reproductive toxicity if 28-day or 90-day exposure indicates adverse effects on reproductive organs or tissues |
NR |
Pb |
Pb |
Ra |
|
Two-generation reproductive toxicity |
NR |
NR |
NR |
Pb |
|
Pharmacokinetics |
NR |
R |
R |
R |
|
Carcinogenicity |
NR |
NR |
NR |
Pb |
|
Abbreviations: NR, not required; R, required; Ra, required with possible exception; Rb, required if test 6 positive; Rc, required if test 6 and 7 negative; Pa, proposed if test 6 or 7 or 8 positive; Pb, proposed if appropriate. |
||||
testing strategy and is not commenting on the legal and regulatory procedural implementation of the legislation.
The legislation, if enacted, will require that all chemicals used in commerce at over 1 metric ton per year have basic toxicity and risk information generated within an 11-year period and that chemicals of very high concern be treated like drugs—only uses approved by government authorities would be permitted. The toxicity testing that would be required is outlined in Table 6-1. The exposure route is generally specified to be the one most relevant for potential human exposure.
Toxicity testing to be conducted is based primarily on the quantity of chemical manufactured or imported, with cutoffs at 1, 10, 100, and 1,000 metric tons. Toxicity-testing requirements for chemicals manufactured in quantities of 1-10 metric tons per year include only skin irrita-
tion or corrosion and sensitization, eye irritation, and several in vitro gene-mutation and cytogenicity studies. Positive results may trigger a hazard classification or additional testing. More substantial toxicity testing is required for substances manufactured or imported in quantities of 10 metric tons per year or more. Those chemicals must have a chemical safety assessment (CSA) as part of a registration dossier. In the CSA, the derivation of the hazard classification must be documented with the assessment of persistence, bioaccumulation, and toxicity (PBT) and the assessment of whether the substance is very persistent and very bioaccumulative (vPvB). The CSA also includes the derived no-effect levels and predicted no-effect concentrations.
The European Centre for the Validation of Alternative Methods (ECVAM) is involved in facilitating the implementation of the REACH program, which calls for the use of alternative testing strategies where feasible. ECVAM is directly targeting the animal tests to be replaced. For example, the REACH legislation calls for data on acute lethal toxicity of chemicals. ECVAM is validating a five-tier testing strategy that would reduce the number of animals being used for acute lethality. Stage 1 involves a quantitative structure-activity relationship (QSAR) model, stage 2 a basal cytotoxicity test (such as the 3T3 neutral red uptake test), stage 3 a computer model and a biotransformation test for metabolism, and stage 4 a cell-specific toxicity test and a prediction model. In each of the four stages, a finding of “very toxic” would indicate that no further testing is done. A fifth stage—in vivo testing—may be needed when the previous stage indicates the absence of significant acute lethal potential that should be confirmed.
Committee Comments on REACH Program
The REACH program is designed primarily to broaden coverage by ensuring that all chemicals that are manufactured in or imported into the EU will undergo at least some evaluation. In this strategy, tonnage cutoffs are used as rough surrogates for potential human exposure to the chemical agents, thereby triggering different levels of toxicologic scrutiny. Tonnage may be an initial rough surrogate, but additional information regarding potential for human exposure (for example, whether the chemical is an intermediate that has very low exposure) may also be helpful in guiding toxicity testing. Furthermore, classification of chemi-
cals according to their PBT and vPvB may be a useful and simple adjunct to priority-setting and hazard assessment.
The committee made several general observations about the REACH program. First, REACH specifies categories of tests or end points that require evaluation at each level without being prescriptive about how the test will be done. Such an approach enhances flexibility but potentially affects comparability of results and indicates the need for the tests to be validated against each other. Second, as discussed above, the process is iterative with an evaluation step by government agencies and a data-sharing requirement to try to ensure that unnecessary or redundant animal testing is not performed. That step reflects an effort to decrease animal use in toxicity testing. Third, the REACH program focuses more on screening a large number of chemicals than on attaining the depth that is often needed for quantitative dose-response assessment. However, the REACH program does allow for greater depth of testing to be triggered on the basis of initial results. Lastly, the REACH program is more directly comparable with the voluntary U.S. high-production-volume screening program than with the proposals for improvements in the EPA RfD-RfC review or the ILSI-HESI proposals. REACH has the advantage of generating at least some toxicity data on chemicals that in the United States are not subject to testing requirements.
National Toxicology Program: Roadmap for the Future
Since its inception in 1979, the NTP has provided data for identifying the health hazards posed by environmental agents, and the data have been used extensively by regulatory agencies in risk assessment. The NTP’s recently released Roadmap for the Future (NTP 2004) has emphasized several ways in which it expects to make substantial progress in the next few years. First, the NTP envisions that studies of gene and protein expression and of metabolic profiling in tissues of exposed animals will help to characterize responses to environmental agents and therefore lead to a better understanding of the mechanisms by which such agents alter disease incidence and progression. Second, the NTP is exploring the use of nonmammalian in vivo assays in toxicity testing. For example, one project, which is expected to be completed in 2007, involves the potential use of Caenorhabditis elegans as a screen for developmental, neurologic, and behavioral toxicity. Finally, increasing emphasis will be
placed on developing alternative assays in which critical molecular and biologic events that occur early in the causation of adverse effects can be targeted, quantified, and placed within the toxicity-testing framework. The systems developed may help in setting priorities among substances for further study or in refining biologically based dose-response (BBDR) models.
In the Roadmap, NTP efforts are directed toward three main goals: refinement of traditional toxicology assays; development of rapid, mechanism-based predictive screens for environmentally induced diseases; and improvement in the overall utility of NTP toxicity-testing assays for public-health decisions. Those goals and the NTP’s plan to train scientists conducting NTP studies in the practice of humane science indicate the NTP’s commitment to reducing the pain and distress of animals and to decreasing the number of animals used in toxicity testing. The Roadmap describes the following current NTP research initiatives:
-
Review existing protocols for carcinogenicity, reproductive toxicity, developmental toxicity, and immunotoxicity and make appropriate changes in the protocols to increase efficiency and to improve the quality of data generated. For example, the NTP has initiated a review of its cancer bioassay and is using public scientific workshops as one mechanism to receive input. The first workshop is focusing on stocks and strains used in the rodent cancer bioassay to seek scientific input on whether they should be continued or replaced or multiple strains should be used. Future workshops will address other cancer-bioassay design issues. Reviews of noncancer assays—such as developmental toxicity, immunotoxicity, and neurotoxicity studies—are expected within the next 3 years.
-
Devise a strategy for routine incorporation of genomic analysis into current toxicity studies within the next year. The goal is to provide mechanistic information for improved understanding of toxicity. Resulting data on the identification of mechanistic targets would be used in the overall toxicity evaluation and in the development of high-throughput systems. The NTP expects such data also to permit the application of toxicity findings on a chemical to other chemicals that act by the same mechanisms. Data collection for some genomic analyses is already under way.
-
Improve the use of pharmacokinetic information in toxicologic evaluations made by the NTP. The effort seeks to strengthen
-
NTP work in pharmacokinetics by establishing a minimal pharmacokinetic dataset on agents evaluated by the NTP, developing and validating PBPK models for agents of concern, building a multidisciplinary capacity within the NTP to do the work, and developing training and user-friendly tools.
-
Continue to explore nonmammalian in vivo alternatives to toxicity testing as tools for medium-throughput screening (MTS) of specific toxicity end points.
-
Expand the use of imaging technologies for detecting and quantifying molecular and cellular lesions and for improving the speed and precision of pathology reviews. This approach could increase the statistical power of NTP studies and reduce the number of animals used in the cancer bioassay and other studies. In the near term, the NTP plans to capture digital images from all pathology slides from the cancer bioassay routinely and to develop new image-analyzing techniques to guide the review and evaluation of lesions. A long-term goal is to provide a digital archive of such images from NTP studies that is accessible via the NTP’s website.
Most of the above actions are intended to refine and increase the efficiency of the NTP’s toxicity-testing strategy and study portfolio. The NTP Roadmap notes, however, the time-consuming and resource-intensive nature of traditional toxicity testing and the consequently large volume of newly introduced and existing chemicals in commerce that have been inadequately assessed for toxicity. The Roadmap outlines the NTP’s long-range research strategy to develop rapid screening systems for providing toxicity information on large numbers of chemicals. The NTP plans to develop further MTS systems, such as in vivo nonmammalian systems, including C. elegans. High-throughput screening (HTS) may enable the evaluation of thousands of agents rapidly with in vitro biologic systems and robotics technology. The NTP’s emphasis is on cellular targets known to influence disease or data interpretation. The NTP expects initially to target genotoxicity, cytotoxicity, cell proliferation, apoptosis, and some receptor-mediated activities and to run all relevant agents previously tested by the NTP through HTS with a set of other identified chemicals.
The databases created with the HTS and MTS can be used to determine the predictive value of the higher-throughput screens. In the short term, the NTP expects HTS to help to set priorities among agents for more extensive testing. In the longer term, the NTP hopes that such
screens will become sufficiently developed and validated to be usable more directly in public-health decision-making. The NTP has staged its work in this field in phases, as outlined in Box 6-1.
Committee Evaluation of the NTP Roadmap
The NTP Roadmap is designed both to refine existing toxicity-testing approaches and to develop new approaches for eventual incorporation into toxicity testing. Although the NTP strategy has been presented publicly in bold brushstrokes rather than in detail, the general approaches are of great interest. The NTP’s near-term efforts to refine and extend its toxicity tests and to improve the generation and use of pharmacokinetic and mechanistic data promise to increase the depth of toxicity information on chemicals assayed and to provide greater insight in applying test findings to humans. Furthermore, the NTP’s stated objective of reducing animal use and increasing efficiency may be partly realized. However, as acknowledged by the NTP, the result-
|
BOX 6-1 Short-Term Activities
Medium-Term Activities
Long-Term Activities
|
ing test portfolio would still be resource-intensive and incapable of addressing large numbers of chemicals that require some level of toxicity assessment. In the relative near term, the alternative—in vivo nonmammalian MTS—holds promise for improvements in cost, animal use, resources, and throughput, but it generally received little attention in the Roadmap. The extent of the NTP MTS effort is unclear. The NTP’s development of such systems to the point where they may be used in public-health decision-making (for example, NTP identifications of chemicals as reproductive toxicants) would be a major advance. In the long term, HTS has the potential to increase the breadth of chemical assessment substantially while preserving the goal of limiting animal testing. Development of improved biomarkers of effect with new -omics approaches would contribute to all four goals of increasing breadth, increasing depth (if biomarkers provide valid mechanistic information), conserving animals, and reducing costs. A specific focus on developing nonanimal models by an agency, such as the NTP, is needed if such approaches are to become a useful alternative to traditional toxicity testing in animals.
COMMITTEE EVALUATION OF SUGGESTED IMPROVEMENTS IN TESTING STRATEGIES
Several government agencies and nongovernment organizations have recognized important gaps and inefficiencies in toxicity testing. EPA, ILSI-HESI, the NTP, and the EU have assessed or undertaken initiatives to build on and improve testing strategies. There is much to learn from their varied assessments and strategies.
EPA evaluated its testing requirements for pesticides and toxic substances in the context of establishing RfDs and RfCs, found substantial data gaps in life stages studied and end points assessed, and made recommendations for improvements in the near term by using existing toxicity-testing methods and techniques. ILSI-HESI convened a panel of scientists to evaluate EPA’s approaches to toxicity testing for pesticides and developed its own set of recommendations. EPA made ambitious recommendations that would increase the breadth of toxicity testing in two key ways: coverage of different life stages and improved coverage of different end points, such as cardiovascular effects and immunotoxicity. In doing so, EPA explored alternative test protocols that would increase the complexity of some tests but ulti-
mately conserve animal use while increasing critical toxicity information. ILSI-HESI emphasized potential test redundancies and test elimination. It recommended eliminating the mouse carcinogenicity study and 1-year dog study and making the rat acute toxicity study optional, thereby reducing animal use, the robustness of the database, and the potential to confirm findings. It also included proposals to increase the breadth of testing by modifying tests to evaluate more end points. It did not propose specific protocols to address effects of exposures in the elderly, although it did suggest that special studies could be triggered by findings in first-tier studies in young adult animals. Both EPA and ILSI-HESI recommended increased efforts to generate pharmacokinetic data, a practice already in place at the NTP, which increases the depth of available toxicity data. Both proposals also suggested reductions in group sizes in toxicity testing. Although in some cases the evaluation of end point or life stage may be more detailed, the small group size (for example, 20 vs 50 in the unified study and six vs eight dogs in the systemic-toxicity evaluations) can significantly reduce the statistical power of a study to detect effects.
The EU is engaged in a bold effort to restructure its approach to toxicity testing to screen the backlog of tens of thousands of industrial chemicals that have not been adequately assessed. It has taken the approach of attempting to broaden the screening of chemicals maximally while minimizing animal use in existing protocols. It is unclear whether the EU approach will provide adequate depth of information for dose-response assessment, especially with respect to the breadth of coverage of relevant end points and life stages. The approach to obtaining test data for detailed risk assessment is still under development, although for some chemicals (pesticides) schemes similar to those used in the United States are in place. It is therefore difficult to compare the REACH approach directly with the EPA and ILSI-HESI approaches because they are designed primarily to focus on different subsets of chemicals (tens of thousands of existing industrial chemicals vs hundreds of pesticides). The REACH approach includes greater depth of information and coverage of end points and life stages than the current EPA approach to testing of existing or new industrial chemicals under the Toxic Substances Control Act.
The NTP has not proposed specific changes in toxicity tests but has initiated a process, involving scientific workshops and public input, that could in the near term lead to fundamental changes in the design and conduct of the bioassays of cancer, reproductive and developmen-
tal toxicity, and other end points. The NTP is beginning by evaluating and considering changes in cancer-bioassay design. Also in the near term, the NTP seeks to improve testing by capitalizing on recent advances in imaging technologies, devising strategies for routine incorporation of toxicogenomic analyses, further developing nonmammalian test systems, and using transgenic animals. This effort goes considerably beyond the proposals and recommendations of ILSI-HESI and EPA. That is in part understandable in that some of the NTP effort involves technology transfer and the development and validation of new approaches; the changes are not yet sufficiently well defined to be incorporated into mandated test requirements, such as the EPA pesticide test guidelines. However, specific study designs and protocols should be given consideration for such purposes as they emerge from the NTP program. The NTP also is taking a longer view through its initiative to develop rapid screening systems for providing toxicity information on large numbers of chemicals.
The NTP approach is the only one that incorporates strategies that have potential for satisfying all four key goals of increasing breadth and depth and decreasing animal use and costs. Yet the NTP HTS of large numbers of chemicals relies in the long term on the development of new methods that are as yet unproved and will have to be evaluated. The NTP HTS approach is many years away from being practical for adoption in testing requirements by a regulatory agency, such as EPA. In the meantime, some of the near-term initiatives at the NTP, as well as strategies proposed in the EPA review and perhaps in the ILSI-HESI papers and in the REACH proposal, may be of use for improving current regulatory toxicity testing.
The committee identified several recurring themes and questions in the various reports that it was asked to review:
-
Which environmental agents should be tested? All new and existing environmental agents should be evaluated; however, the intensity and depth of testing should be decided according to practical needs, including the use of the chemical, the likelihood of human exposure, and the scientific questions that such testing must answer to support a reasonable science-policy decision. Fundamentally, the design and scope of a toxicity-testing approach needs to reflect risk-management needs. Regulatory agencies are pursuing different testing strategies depending on whether their goal is generating detailed data for optimal dose-response assessment of pesticides or screening exist-
-
ing industrial chemicals. Hence, neither the EPA testing recommendations in its RfD-RfC review nor the EU REACH program is “correct” or “incorrect,” because they are designed for different purposes.
-
How should priorities for testing chemicals be set? Priority-setting should be a key component of any testing strategy that is designed to address a large number of chemicals, and a well-designed scheme is essential for systematic testing of industrial chemicals on which there are few data. It makes sense to consider exposure potential in designing test strategies. Chemicals to which people are more likely to be exposed or to which some populations may receive relatively high exposures—whether they are pesticides or industrial chemicals—should undergo more in-depth testing. This concept is embedded in several existing and proposed strategies. In some strategies, production volume is the primary measure of potential human exposure. But production volume alone may not be the best surrogate of human exposure. Other important factors to consider are use, exposure patterns, and a chemical’s environmental persistence and bioaccumulation, which is important because of the potential for increasing exposure over time and continuing exposure even after use has ceased. Indicators of potential toxicity from existing toxicity data or structural analogues and computational approaches, such as structure-activity relationships, may help to refine priorities further. In addition, there has been some investigation of high-throughput in vitro methods as a possible priority-setting tool. The committee will discuss possible priority-setting approaches in greater depth in its second report.
-
What strategies for toxicity testing are the most useful and effective? Existing test strategies include test batteries, tiered-testing strategies, tailored approaches, and strategies that combine various approaches. The committee finds that there are pros and cons of various approaches but leans toward tiered testing with the goal of focusing resources on the evaluation of the more sensitive adverse effects of exposures of greatest concern rather than full characterization of all adverse effects irrespective of relevance for risk-assessment needs. A tiered-testing approach would require that EPA have clear regulatory authority to require additional testing of pesticides or industrial chemicals beyond the first tier when it is indicated. Such a strategy would also require a priori stopping rules to prevent all chemicals from going through all tiers. For example, some observers have expressed the concern that some potentially “positive” result may often pop up in tiered testing and trigger the next tier of testing of nearly all chemicals, which
-
would erase the benefit of tiering. Other observers have expressed the concern that in tiered strategies, strong positive results may simply trigger moving to ever-higher and more time-consuming tiers of testing while delaying risk-management action based on the findings. Both concerns are related to the need to have clear rules that move environmental agents out of the tiered-testing strategy into either a “stop” category or a preliminary risk-management category. One important point regarding the rules is that one should determine whether additional testing is likely to make a difference in human risk assessment. For example, the margin of exposure may be sufficiently large that further testing is not needed to make a decision or the end point in question is unlikely to be more sensitive than other end points of concern, in which case no further testing regarding that end point would be needed.
-
How can toxicity testing generate data that are more useful for human-relevant dose-response assessment? Many observers have criticized existing approaches to toxicity testing on the grounds that it is difficult to use their results in risk assessment. In particular, observed results are often difficult to extrapolate with confidence to humans at environmentally relevant doses. As a result, such extrapolations are often made with little scientific justification, and conventional uncertainty factors are used to bridge the gaps.
Current approaches to toxicity testing could be enhanced in some cases by the use of pharmacokinetic data from basic ADME studies to derive dose information before embarking on toxicity testing and by the judicious use of some pharmacokinetic data to aid in extrapolation. In addition, adding lower doses to some protocols could help with obtaining more environmentally relevant information, although to maintain adequate statistical power increased group size at lower doses may be needed, with attendant increases in cost. Using benchmark dose extrapolations in place of NOAELs is another strategy that has been shown to be useful in maximizing the use of the existing dataset. However, the current menu of approaches to toxicity testing is unlikely to solve the fundamental problem. The committee cautions against indiscriminately generating large amounts of data on single chemicals in an effort to generate the optimal dose-response dataset. Such an approach entails the substantial use of animals and often comes at the expense of broader screening that may also be useful. Newer approaches to toxicity testing may help to address this problem and will be discussed in
-
the committee’s second report. In the interim, uncertainty factors must still be used to address scientific uncertainties.
-
How can toxicity testing be applied to a broader universe of chemicals, life stages, and end points? It is clear that there are major gaps in current toxicity-testing approaches. The real importance of the gaps is a matter of debate and depends on whether effects of public-health importance are being missed by current approaches. Historically, an important end point or life-stage susceptibility has in many cases been missed in initial toxicity testing. However, it is neither practical nor desirable to attempt to test every chemical (or mixture) against every end point during a wide range of life stages. The committee recommends toxicity screening of every agent to which there is a strong potential for human exposure. A well-designed tiered strategy could help to set priorities among environmental agents for screening and could identify end points or mechanisms of action that would trigger more in-depth testing for various end points or in various life stages. Newer methods of screening that incorporate such tools as -omics or computational toxicology may help to screen more chemicals rapidly and trigger appropriate further testing where necessary. Those approaches are discussed briefly in Chapter 7 of this report and will be discussed in more depth in the committee’s second report.
-
How can environmental agents be screened with the minimal use of animals and minimal expenditure of time and other resources? One strategy that is useful to reduce animal use is the grouping of chemicals of similar structural class and the in-depth testing of only one or a few representative chemicals; risk assessments of all chemicals in the class would then be based on the resulting data. In grouping chemicals, known modes of action (for example, nicotinic agonists) should be emphasized. Such strategies should address any data needed to support application of study findings on the representative chemicals to other chemicals in the group. Newer approaches also have great promise. Chapter 7 discusses current developments in reduction, refinement, and replacement of animals in toxicity testing and newer technologies that hold promise for greatly reducing the reliance on animal testing.
-
How should tests and testing strategies be evaluated? An important consideration in evaluating test strategies is the risk-management context in which they are being applied. For example, intensive study of untested industrial chemicals is of little use if it means that few chemicals can be addressed. However, further explora-
-
tory and intensive study of a relatively well-tested chemical can be of great value if costs of controlling exposure are high, there is widespread and relatively high exposure, and study is likely to refine the risk assessment substantially. Test strategies may be evaluated in terms of the value of information they provide in light of the cost of the testing and the animal resources expended. That should be kept in mind in considering testing strategies in terms of the four testing objectives—increasing depth of knowledge for more accurate risk assessment; increasing coverage of chemicals, life stages, and end points; preserving animal welfare; and minimizing cost.
In evaluating new tests and test strategies, there remains the difficult question of what is to serve as a “gold standard” for performance. Simply comparing the outcomes of new tests with the outcomes of current tests may not be the best approach; whether it is will depend on the reliability and relevance of the current tests. Another consideration is how test results will be used in the assessment. Even if a test strategy provides robust and informative data, the risk assessor may be unable, because of legal constraints or risk-assessment guidelines, to use the data. Ideally, regulations and risk-assessment guidelines will evolve with testing capabilities and scientific understanding. That issue will increase in importance with greater use of screening approaches that produce indirect evidence (in vitro tests, gene arrays, and mode-of-action screens), for both cancer and noncancer end points.
REFERENCES
Cooper, V.L., J.C. Lamb, S.M. Barlow, K. Bentley, A.M. Brady, N.G. Doerrer, D.L. Eisenbrandt, P.A. Fenner-Crisp, R.N. Hines, L.F.H. Irvine, C.A. Kimmel, H. Koeter, A.A. Li, S.L. Makris, L.P. Sheets, G.J.A. Speijers, and K.E. Whitby. 2006. A tiered approach to life stages testing for agricultural chemical safety assessment. Crit. Rev. Toxicol. 36 (1):69-98.
Doe, J.E., A.R., Boobis, A. Blacker, V.L. Dellarco, N.G. Doerrer, C. Franklin, J.I. Goodman, J.M. Kronenberg, R. Lewis, E.E. McConnell, T. Mercier, A. Moretto, C. Nolan, S. Padilla, W. Phang, R. Solecki, L. Tilbury, B. van Ravenswaay, and D.C. Wolf. 2006. A tiered approach to systemic toxicity testing for agricultural chemical safety assessment. Crit. Rev. Toxicol. 36 (1):37-68.
EPA (U.S. Environmental Protection Agency). 1998a. Health Effects Test Guidelines OPPTS 870.1100. Acute Oral Toxicity. EPA 712-C-98-190. Office of Prevention, Pesticides, and Toxic Substances, U.S. Envi-
ronmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/opptsfrs/OPPTS_Harmonized/870_Health_Effects_Test_Guidelines/Series/870-1100.pdf [accessed March 15, 2005].
EPA (U.S. Environmental Protection Agency). 1998b. Health Effects Test Guidelines OPPTS 870.1200. Acute Dermal Toxicity. EPA 712-C-98-192. Office of Prevention, Pesticides, and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/opptsfrs/OPPTS_Harmonized/870_Health_Effects_Test_Guidelines/Series/870-1200.pdf [accessed Oct. 7, 2005].
EPA (U.S. Environmental Protection Agency). 1998c. Health Effects Test Guidelines OPPTS 870.1300. Acute Inhalation Toxicity. EPA-712-C-98-193. Office of Prevention, Pesticides, and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/opptsfrs/OPPTS_Harmonized/870_Health_Effects_Test_Guidelines/Series/870-1300.pdf [accessed Oct. 7, 2005].
EPA (U.S. Environmental Protection Agency). 1998d. Health Effects Test Guidelines OPPTS 870.4300. Combined Chronic Toxicity/Carcinogenicity. EPA 712-C-98-212. Office of Prevention, Pesticides, and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/opptsfrs/OPPTS_Harmonized/870_Health_Effects_Test_Guidelines/Series/870-4300.pdf [accessed March 15, 2005].
EPA (U.S. Environmental Protection Agency). 1998e. Health Effects Test Guidelines OPPTS 870.3800. Reproduction and Fertility Effects. EPA 712-C-98-208. Office of Prevention, Pesticides, and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/opptsfrs/OPPTS_Harmonized/870_Health_Effects_Test_Guidelines/Series/870-3800.pdf [accessed March 15, 2005].
EPA (U.S. Environmental Protection Agency). 1998f. Health Effects Test Guidelines OPPTS 870.3100. 90-Day Oral Toxicity Study in Rodents. EPA 712-C-98-199. Office of Prevention, Pesticides, and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/docs/OPPTS_Harmonized/870_Health_Effects_Test_Guidelines/Series/870-3100.pdf [accessed Oct. 26, 2005].
EPA (U.S. Environmental Protection Agency). 1998g. Health Effects Test Guidelines OPPTS 870.2400. Acute Eye Irritation. EPA 712-C-98-195. Office of Prevention, Pesticides, and Toxic Substances, U.S. Environ-mental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/opptsfrs/OPPTS_Harmonized/870_Health_Effects_Test_Guidelines/Series/870-2400.pdf [accessed April 7, 2005].
EPA (U.S. Environmental Protection Agency). 2002. A Review of the Reference Dose and Reference Concentration Processes. Final Report. EPA/630/P-02/002F. Risk Assessment Forum, U.S. Environmental Protec-
tion Agency, Washington, DC [online]. Available: http://www.epa.gov/iris/RFD_FINAL%5B1%5D.pdf [accessed March 11, 2005].
EPA (U.S. Environmental Protection Agency). 2005. OPPTS Harmonized Test Guidelines. Series 870 Health Effects Test Guidelines-Final Guidelines. Office of Prevention, Pesticides, and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/opptsfrs/OPPTS_Harmonized/870_Health_Effects_Test_Guidelines/Series/ [accessed May 5, 2005].
EU (European Union). 2004. The REACH Proposal Process Description. REACH (Registration, Evaluation and Authorisation of Chemicals), Enterprise and Industry, European Union [online]. Available: http://europa.eu.int/comm/enterprise/reach/docs/reach/reach_process_description-2004_06_15.pdf [accessed March 8, 2005].
ILSI-HESI (International Life Sciences Institute Health and Environmental Sciences Institute). 2004a. Systemic Toxicity White Paper. Systemic Toxicity Task Force, Technical Committee on Agricultural Chemical Safety Assessment, ILSI Health Sciences Institute, Washington, DC. November 2, 2004.
ILSI-HESI (International Life Sciences Institute Health and Environmental Sciences Institute). 2004b. Life Stages White Paper. Life Stages Task Force, Technical Committee on Agricultural Chemical Safety Assessment, ILSI Health Sciences Institute, Washington, DC. November 2, 2004.
ILSI-HESI (International Life Sciences Institute Health and Environmental Sciences Institute). 2004c. The Acquisition and Application of Absorption, Distribution, Metabolism, and Excretion (ADME) Data in Agricultural Chemical Safety Assessments. ADME Task Force, Technical Committee on Agricultural Chemical Safety Assessment, ILSI Health Sciences Institute, Washington, DC. November 2, 2004.
NTP (National Toxicology Program). 1984. Report of the NTP Ad Hoc Panel on Chemical Carcinogenesis and Testing Evaluation. NTP Board of Scientific Counselors, U.S. Department of Health and Human Services. August 17, 1984 [online]. Available: http://ntp-server.niehs.nih.gov/files/NTPAdHocPanel84.pdf [accessed Oct. 26, 2005].
NTP (National Toxicology Program). 2004. The NTP Vision for the 21st Century. National Toxicology Program, National Institute for Environmental Health, Research Triangle Park, NC [online]. Available: http://ntp-server.niehs.nih.gov/ntp/main_pages/NTPVision.pdf [accessed March 11, 2005].
NTP (National Toxicology Program). 2005. Presentations at the Workshop: Animal Models for the NTP Rodent Cancer Bioassay: Strains and Stock- Should We Switch? June 16-17, 2005, National Institute of Environmental Health Sciences, Research Triangle Park, NC [online]. Available: http://ntp-server.niehs.nih.gov/files/Agenda_Presentations.pdf [accessed Oct. 26, 2005].













































