4
Strategies for Toxicity Testing
Toxicity-testing strategies designed to generate information on potential hazards or risks posed by environmental agents have evolved in response to legislative mandates, scientific developments, and public concerns. Accordingly, testing strategies have been developed or mandated to evaluate pesticides, food additives, high-production-volume (HPV) industrial chemicals in the United States and the European Union, and risks associated with endocrine-disrupting chemicals, developmental toxicants, and carcinogens. The widespread inclusion of genetic-toxicity tests in testing schemes followed scientific advancements that led to the understanding and general recognition that chemicals could cause mutations and mutations could cause cancer. Concerns over other specific effects have also led to modification and refinement of testing strategies so that chemicals posing those hazards would be identified. For example, the thalidomide and diethylstilbestrol disasters emphasized the need for testing strategies to assess chemically mediated effects on reproduction and development. Furthermore, concerns over the contribution of environmental agents to the development of neurobehavioral disorders, neurodegenerative diseases, and respiratory disorders have highlighted the need for testing strategies to address those possible effects (Peters et al. 1999; Gauderman et al. 2000; Schettler 2001). In addition to identifying hazards, toxicity-testing strategies can be designed to provide a basis for dose-response assessment (for establishing exposures expected to pose no risk or for estimating the relationship between risk and exposure). The ideal testing strategies would provide a systematic approach to gathering the data necessary for hazard identification and dose-response assessment thoroughly, rapidly, at low cost, and with few animals.
In this chapter, the committee characterizes testing strategies used by federal and international agencies to gather the data used to identify and evaluate human health hazards and risks. Several examples of testing strategies that are currently used or have been proposed are presented. The testing strategies identified are not meant to be exhaustive but to illustrate the array of toxicity tests that may be required under different circumstances. Furthermore, presentation of the examples in this chapter is not meant to be a committee endorsement of any given strategy. The chapter concludes with committee observations on the current or proposed strategies.
TYPES OF TOXICITY-TESTING STRATEGIES
In practice, testing strategies vary considerably, although they can often be described by three basic testing approaches: battery, tiered, and tailored. A battery is a specific set of toxicity tests applied to all chemicals in a group. Testing batteries are sometimes intended to provide the minimal dataset necessary for risk-informed regulation or risk management.
In tiered testing, the results of a specific set of toxicity tests and risk-management needs are used to guide decisions about the nature and extent of further testing. Often, a substance is first assigned to categories (for example, based on structure or exposure) that guide testing sequences. The chemical then moves through a series of tests sequentially with the data from each test informing the next step in the process.
In tailored testing, information on exposure circumstances, suspected adverse effects, and knowledge of mechanism of action is used to determine the scope of tests to be conducted on a given chemical or class of chemicals. The strategies are thus tailored to the nature of the substance under consideration, its likely use or the likely exposure to it, and the extent of the information available and information needed. Tailored testing strategies may start with a flexible test battery and evolve to different tiers or types of testing in an iterative manner based on scientific judgment.
Characterizing an overall testing strategy as a battery, tiered, or tailored approach is often not possible, because testing strategies are typically combinations of these three basic elements. The examples that follow illustrate that point and demonstrate that testing strategies differ based on the concerns that they were meant to address. The examples address three applications: testing for registration of pesticides and food
additives, screening of large numbers of chemicals to develop basic hazard and risk information, and screening of chemicals for specific effects of concern, such as endocrine disruption.
TOXICITY-TESTING STRATEGIES FOR PESTICIDES AND FOOD ADDITIVES
To protect the food supply, a pesticide cannot be sold or distributed in the United States without being licensed by the Environmental Protection Agency (EPA), and food additives must be formally approved for use by the Food and Drug Administration (FDA). The exceptions are food additives that had already been sanctioned as safe when the 1958 food-additive amendments were adopted or that were generally recognized as safe by FDA. To provide a basis for evaluating the safety of pesticides and food additives, FDA and EPA require a series of tests from applicants and petitioners, as discussed in the following sections.
Federal Insecticide, Fungicide, and Rodenticide Act Testing Program
The EPA Office of Pesticide Programs regulates the use of pesticides under the authority of the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA). Through FIFRA, EPA mandates a battery of toxicity tests of conventional chemical pesticides to assist in determining the precautionary language that is required on the label, the type of personal protective equipment that is required for appliers, and the types of uses and use rates to allow. The tests (Table 4-1) are grouped in five main categories: acute tests, subchronic tests, chronic tests, mutagenicity tests, and special tests. Most of the tests are required for pesticides that can end up as food residues or potentially have widespread exposure of the general population, such as those which have residential use. For other pesticides, only acute and mutagenicity testing may be required. Additional studies—for example, dermal penetration, 21-day dermal, subchronic dermal, subchronic inhalation, acute and subchronic neurotoxicity, acute and subchronic delayed neurotoxicity, and developmental neurotoxicity—may be triggered by some special characteristic of a pesticide (such as its chemical class), by potential use and exposure patterns (such as residential uses), or by the results of routinely required studies.
TABLE 4-1 Battery of Tests Required by EPA for New Pesticide Chemicals
EPA has the authority to impose data requirements on pesticides beyond what is required routinely if it determines that more data are needed to characterize the hazard potential of a particular pesticide, including potential hazards to infants and children.
Microbial Pest-Control Agents Testing Program
Microbial pest-control agents (MPCAs) are natural and strain-improved bacteria, algae, fungi, viruses, and protozoa that act as biologic pesticides (40CFR152.20[2002]). MPCAs typically have unique or nontoxic modes of action and are often naturally occurring. They are most appropriately characterized for health and environmental safety with testing schemes that take their unique characteristics into account. Unlike chemical pesticides, MPCAs may survive and reproduce in the environment and may infect or cause disease in other living organisms. Consequently, basic testing protocols are designed specifically to detect any of those characteristics. Protocols for further testing emphasize exposure or environmental expression in addition to infectivity and pathogenicity.
Toxicity-testing requirements, as described by EPA (2004), are set forth in two tiers (see Table 4-2). Tier I consists of a battery of short-term tests designed to evaluate toxicity, infectivity, and pathogenicity. Tier II is designed to evaluate the particular situation when either toxicity or infectivity but not pathogenicity is observed in tier I (EPA 1996).
FDA Testing Strategies for Food Additives
FDA provides guidance to industry and the public concerning the procedures and methods for assessing the safety of direct and indirect food and color additives. FDA published its guidelines, Toxicological Principles for the Safety Assessment of Food Ingredients, also known as the Redbook, in 1982 and revised them in 1993, 2000, and 2004. The
TABLE 4-2 Toxicity Tests for Microbial Pest-Control Agents
|
Tier I |
Tier II |
|
Mammalian toxicity, pathogenicity, infectivity Acute oral toxicity, pathogenicity Acute dermal toxicity, pathology Acute pulmonary toxicity, pathogenicity Acute injection toxicity, pathogenicity Hypersensitivity incidents Cell culture |
Acute toxicology Subchronic toxicity, pathogenicity Reproductive and fertility effects |
|
Source: EPA 2004. |
|
Redbook contains general guidelines for toxicity studies, including such issues as good laboratory practices, test-animal housing and maintenance, species selection, age, diet, observations, clinical tests, and histopathologic examinations. The Redbook also takes a prescriptive approach, defining in detail how to test various food ingredients, how the agency will review the data, and how decisions will be made. It stipulates specific study designs, what data to collect and report, and how pathologic and statistical analyses should be used in data interpretation. Traditional toxicologic methods are emphasized. FDA states that sponsors may use alternatives to methods that are contained in the Redbook as long as they satisfy applicable regulations and statutes.
The process of evaluating a direct food additive begins with assignment of the additive to one of three concern levels on the basis of chemical structure and expected concentrations of the chemical in the diet. Specifically, the additive is first assigned to structure category A, B, or C (FDA 1993). Structure category A includes compounds associated with low toxic potential or identified as normal cellular constituents, such as alkanes, complex carbohydrates, and fatty acids. Structure category B includes compounds associated with noncancer adverse effects in animals or humans, such as certain amino acids, carboxylic anhydrides, peptides, and proteins. Structure category C includes compounds associated with mutagenicity or carcinogenicity, such as benzofurans, epoxides, and phenols. Potential exposure based on dietary concentration determines the final concern level. This process is illustrated in Table 4-3. Indirect food additives—chemicals that become part of food in trace amounts because of packaging, storage, or other handling—are categorized for testing only according to dietary concentration.
TABLE 4-3 Concern Levels for Direct Food Additivesa
As shown in Table 4-4, specific tests are required on the basis of concern level for direct food additives or dietary concentration for indirect food additives. For example, a short-term feeding study in rodents and short-term tests for carcinogenic potential would be required for direct food additives with the lowest concern (concern level I). For an indirect food additive expected to occur at less than 0.05 ppm in the diet, only an acute oral toxicity study in rodents would be required. The tests increase in complexity and duration as the level of concern or dietary concentration increases. As Table 4-4 indicates, some tests are contingent on other test findings, but there is no formal guidance on proceeding
TABLE 4-4 Testing Required for Direct and Indirect Food Additives
|
Direct Food Additives |
Toxicity Tests |
|
|
Concern Levela |
Testing Required |
|
|
I |
B, K |
|
|
II |
A***, D, E, I, J*, K |
|
|
III |
A***, D***, F, G, H, I, J*, K, L** |
|
|
Indirect Food Additives |
||
|
Dietary Concentration |
Testing Required |
|
|
<0.05 ppm |
A |
|
|
>0.05 ppm |
A***, C, I*, K**, E, J* |
|
|
>1.0 ppm |
A***, D***, F, G, H, I, J*, K**, L** |
|
|
* If indicated by available data or information. ** Suggested. *** If needed as preliminary to further study. |
||
|
|
The Redbook contains references to current guides on these tests. |
|
|
aThe concern level is determined by chemical structure and dietary concentration (see Table 4-3). Source: Adapted from FDA 1997. |
||
from one test to another on the basis of negative or positive results. FDA may request studies that go beyond its guidance and does mention epidemiologic studies for assessing safety, but it is vague as to when they could best be used.
FDA’s approach to the hazard assessment of drug candidates is somewhat different from its approach for food additives. Although FDA expects a package of toxicity studies similar to those required for food additives, it is far more willing to modify pharmacologic and toxicologic studies so that they answer questions peculiar to the proposed therapeutic entity. Thus, safety assessments of drugs are less prescriptive than those of food ingredients. They lack detail on study designs but focus on the principles that need to be addressed.
TOXICITY-TESTING STRATEGIES FOR SCREENING OF INDUSTRIAL CHEMICALS
In addition to the vast number of chemicals already in commerce, many chemicals are introduced each year. The following describes testing strategies used in the United States and Europe to screen and provide the basis for toxicity assessment of new and existing industrial chemicals.
The Toxic Substances Control Act and the High-Production-Volume Chemical Testing Program
The Toxic Substances Control Act (TSCA) was passed in 1976 and gave EPA the authority to collect information and issue regulations on new and existing industrial chemical substances. When EPA began evaluating chemicals under TSCA in 1979, about 62,000 chemicals were in commerce (GAO 2005). Today, 82,000 chemicals are in commerce, and about 700 chemicals are introduced each year (GAO 2005).
TSCA does not prescribe or detail a testing strategy to evaluate the large volume of existing and new chemicals. Companies that manufacture or process new chemicals for commercial purposes must submit to EPA a premanufacturing notice (PMN), which includes information on chemical structure, production process, expected production volume, intended uses, possible exposure and release levels, disposal procedures, and other data “concerning the chemical’s environmental or health effects known to or reasonably ascertainable by the chemical company”
(GAO 2005). To register a new chemical, companies are not required to conduct any specific toxicity tests, and EPA estimates that only about 15% of PMNs contain health or safety data (GAO 2005). EPA typically uses models to predict a chemical’s toxicity on the basis of its structure. A more detailed review is conducted only on about 20% of the chemicals (GAO 2005). EPA does have the authority to require manufacturers, importers, and processors of chemical substances to submit new data on existing chemicals, and it can issue a test rule that indicates specific tests to be conducted. However, the process is burdensome, and “EPA has used its authority to require testing for fewer than 200 of the 62,000 chemicals in commerce when EPA began reviewing chemicals under TSCA in 1979” (GAO 2005).
To address the lack of data on existing industrial chemicals, EPA implemented a voluntary program in negotiation with the American Chemistry Council, the American Petroleum Institute, and Environmental Defense known as the HPV chemical testing program. The program’s purpose is to ensure that basic toxicity data are available on all organic, nonpolymeric chemicals produced or used in the United States in excess of 1 million pounds per year. EPA focused attention on HPV chemicals because it considered such chemicals to have a higher potential for environmental and workplace exposure than low-production-volume chemicals (65 Fed. Reg. 81661[2000]). The HPV testing program is intended to support the development of screening-level hazard and risk characterizations with a battery of tests for basic toxicity testing end points (see Box 4-1). EPA is implementing the program to be consistent with the HPV program developed by the Organisation for Economic Co-operation and Development (OECD) in that it includes the
|
BOX 4-1
|
same end points and extent of testing as the OECD screening information dataset (SIDS) (65 Fed. Reg. 81686 [2000]).
The international community has agreed that SIDS is the minimal dataset required to screen HPV chemicals for toxicity (OECD 2004a). In the United States, if SIDS toxicity data are not available or are incomplete for a particular chemical, a test plan is proposed and reviewed by EPA and other organizations. Test plans can be designed for categories of chemicals that have common physiochemical characteristics, common functional groups, and common toxic properties. Specific tests are conducted according to EPA or comparable OECD test guidelines (65 Fed. Reg. 81686 [2000]). Tests are sponsored by private organizations, and the results are made publicly available. EPA (65 Fed. Reg. 81686 [2000]) notes that the results of the SIDS testing will support preliminary risk assessment, anticipating that for some chemicals of lower concern the results will be adequate to evaluate the hazards and risks posed. The results may indicate the need for further testing of other chemicals. Conceptually, the SIDS battery is an initial battery in an overall tiered approach to testing for hazard and risk assessment.
Canada has a program similar to the U.S. and OECD HPV testing programs (GAO 2005).
European Union Testing Strategies
Toxicity testing of environmental agents to inform human health risk assessments occurs in the European Union (EU) under Commission Directives 93/67/EEC and 98/8/EC and Commission Regulation 1488/94, which direct the risk assessment of new substances, existing substances, and biocidal products. A human health risk assessment must be carried out for all existing substances (substances marketed before September 18, 1981) and for new substances that are identified because of their toxic or physiochemical properties and possible human exposure via workplace, product consumption, or indirect environmental exposure.
The EU issued a technical guidance document (TGD) on risk assessment (EC 2003) for use by authorities to help in carrying out toxicity testing. EC (2003) describes a process whereby chemicals in the EU are tested according to their annual manufactured quantity; thus, it is a tonnage-driven testing program. That strategy is based on the assumption that adequate risk assessment of chemicals with low general exposure might not require as much toxicity-testing data as risk assessment of substances with higher exposure. The scheme is intended to assess the risk
posed to humans by individual substances; additive and synergistic effects caused by the combined action of several substances are not considered.
The TGD details different sets of toxicity tests that are required on the basis of the quantity of substance produced per manufacturer per year. All substances that are produced at more than 10 kg/manufacturer per year must be tested (see Table 4-5). Tests of new substances are to be carried out in accordance with EU test guidelines (Annex V to Directive 67/548) or, if EU guidelines are not available, OECD guidelines.
STRATEGIES FOR SCREENING CHEMICALS FOR EFFECTS ON SPECIFIC SYSTEMS AND END POINTS
Because of the potential widespread impact of introducing into the environment anthropogenic materials that disrupt the endocrine system, EPA is developing strategies to screen chemicals for endocrine-disrupting activity, and OECD has developed a general framework for such evaluations. The National Research Council (NRC) undertook a study to clarify how environmental agents may be affecting human development and made recommendations for improvements to qualitative and quantitative risk assessment. The EPA and OECD endocrine-disruptor screening approaches and work of the NRC are provided as examples of testing approaches aimed at addressing specific systems and end points.
Environmental Protection Agency Endocrine-Disruptor Testing Strategy
In 1996, EPA formed the Endocrine Disruptor Screening and Testing Advisory Committee (EDSTAC) to advise on the development of a program for screening and testing chemicals for endocrine-disrupting activity.1 After considering that committee’s advice, EPA (63 Fed. Reg.
TABLE 4-5 European Union Testing Strategy
|
10-100 kg/manufacturer per year Acute toxicity—oral or inhalation |
1-10 tons/manufacturer per year Acute toxicity—oral route and a second depending on likely route of exposure Skin irritation Eye irritation Skin sensitization Repeated-dose toxicity—28 day Mutagenicity—bacteriologic and nonbacteriologic tests Reproductive-toxicity screen Pharmacokinetic assessment—based on data derived from above tests |
10-1,000 tons/manufacturer per year (50-5,000 tons cumulative) Level 1 Base Testing Set Acute toxicity—oral route and a second depending on likely route of exposure Skin irritation Eye irritation Skin sensitization Repeated-dose toxicity—28 day Mutagenicity—bacteriologic and nonbacteriologic tests Reproductive-toxicity screen Pharmacokinetic assessment—based on data derived from above tests Fertility study—in one species for one generation May be requiredon basis of level 1 testing results Fertility—second-generation study Teratogenicity—in second species Prenatal-development toxicity Subchronic/chronic toxicity Mutagenicity—additional testing |
|
100 kg-1 ton/manufacturer per year Acute toxicity—oral route and a second dependingon likely route of exposure Skin irritation Eye irritation Skin sensitization Mutagenicity—bacteriologic test Reproductive-toxicity screen Pharmacokinetic assessment—based on data derived from above tests |
|
>1,000+ tons/manufacturer per year (>5,000 tons cumulative) Level 2 Base Testing Set Acute toxicity—oral route and a second depending on likely route of exposure Skin irritation Eye irritation Skin sensitization Repeated-dose toxicity—28 days Mutagenicity—bacteriologic and nonbacteriologic tests Reproductive-toxicity screen Fertility study—in one species for one generation Chronic toxicity Carcinogenicity Pharmacokinetic assessment—based on data derived from base testing set plus additional studies May be required on basis of level 1 testing results Fertility—multigeneration study Prenatal-development toxicity Acute toxicity—in second species Repeated-dose toxicity—28 days—in second species |
42852 [1998]; 63 Fed. Reg. 71542 [1998]) developed its Endocrine Disruptor Screening Program (EDSP), which recommended screening of chemicals that have the potential to disrupt androgen, estrogen, and thyroid hormone systems. Both potential human and ecologic effects were to be addressed, and nonpesticide chemicals, contaminants, and mixtures were to be included in addition to pesticides. EPA developed a tiered approach for the EDSP. Its core elements are priority-setting—tier 1 screening to identify agents with the potential to alter the estrogen, androgen, or thyroid hormone systems and tier 2 testing to determine whether agents identified in tier 1 cause developmental toxicity through any of the three systems and, if so, their dose-response relationships.
Figure 4-1 shows an overview of the EDSP. The EDSTAC estimated that 87,000 chemical agents in use were candidates for screening for endocrine-disrupting activity. That would overwhelm the resources available for screening, so EPA proposed a “compartment-based approach” in which exposure and effects would be used for setting priorities for initial screening. The initial sort was intended to direct chemicals to one of four categories:
-
Category 1. A “hold” category comprising polymers with molecular weight greater than 1,000 daltons considered unlikely to cross
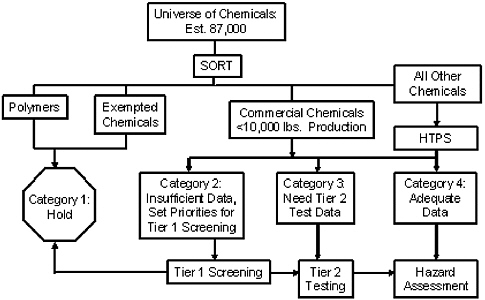
FIGURE 4-1 Endocrine-disruptor screening program overview. Source: EPA 2000.
-
cell membranes—estimated to amount to 25,000 of the 87,000 chemical agents.
-
Category 2. Chemicals with insufficient data to undergo tier I high-throughput screening that is designed to detect potential hormonal or biologic activity (binding to estrogen, androgen, or thyroid hormone receptors to elicit a biologic effect, such as transcription activation).
-
Category 3. Chemicals with existing data sufficient to bypass tier 1 screening and proceed directly to tier 2 testing.
-
Category 4. Chemicals with existing data sufficient to bypass both tier 1 and tier 2 testing for direct hazard assessment.
The EDSTAC also recommended that a scheme be developed for setting testing priorities among common mixtures. The EDSTAC recognized the practical impossibility of screening all possible chemical combinations but noted that some widely used mixtures, such as pesticide formulations and gasoline formulations, are potential candidates for testing.
Table 4-6 shows the tests included in each tier. The tier 1 screening battery combines in vitro and in vivo assays meant to detect chemicals that affect hormone systems regardless of mode of action; it is intended to minimize false negatives and false positives, and it includes several
TABLE 4-6 Endocrine-Disruptor Screening Testsa
|
Tier 1 Screening Tests |
|
In vitro tests |
|
Estrogen-receptor binding and transcriptional activation Androgen-receptor binding and transcriptional activation Steroidogenesis assay using minced testis |
|
In vivo tests |
|
Rodent 3-day uterotrophic assay—subcutaneous administration Rodent 20-day pubertal female assay with thyroid end points Rodent 5- to 7-day Hershberger Frog metamorphosis Fish gonadal recrudescence |
taxonomic groups and an array of end points to inform a weight-of-evidence evaluation. The in vivo assays are designed as an integral battery that complements the in vitro assays; they cover a wider array of mechanisms of action and incorporate pharmacokinetic determinants of mechanism of action.
Tier 2 testing is designed to determine the likelihood, nature, and dose-response relationship of the disruption of hormone systems in humans, fish, and wildlife. The tests include a wide range of doses administered through a relevant route of exposure throughout critical life stages and processes. The design of the tests also takes into consideration the possibility that the effects of chemical agents may be latent and thus not observed until later in life; this would be analogous to the effects of diethylstilbestrol. Thus, the tests were designed to encompass two generations and to allow determination of effects on fertility, mating, fetal development, neonatal growth and development, and transition from juvenile to sexual maturity.
Organisation for Economic Co-operation and Development Endocrine-Disruptor Testing Strategy
OECD has also developed a conceptual framework for testing endocrine disruptors. OECD emphasizes that the framework represents a toolbox rather than a testing scheme, with levels of tests and assessments that correspond to different levels of biologic complexity (see Table 4-7). That framework considers all testing data, including in vitro data, structure-activity relationships, and data that may become available from new technologies, such as genomics and proteomics. The lower levels of the testing framework are consistent with the hazard-identification phase of the UN Globally Harmonised System, a system for classifying and communicating chemical hazards (UN 2003). The testing and data development in the higher levels support consideration of dose-response relationships. The OECD endocrine-disruptor testing framework indicates that a chemical can enter at any step on the basis of available data or data requirements and leave the testing framework when available data are sufficient for an assessment.
Developmental-Toxicity Testing
Given the potentially devastating effects of human developmental defects, the NRC convened the Committee on Developmental Toxicity to
TABLE 4-7 OECD Conceptual Framework for Testing Endocrine Disruptors
evaluate the impact of environmental agents on human development. The committee proposed a multidisciplinary, multilevel, interactive approach to developmental-toxicity testing (NRC 2000, Chapter 8). The basic premise of the approach is that understanding the mechanistic basis of extrapolation between test animals (or in vitro assays) and humans will give risk assessment greater validity. The approach is different from a tiered-testing approach because testing may be initiated at any level and there is no unidirectional triggering of higher-level testing by results obtained at a lower level.
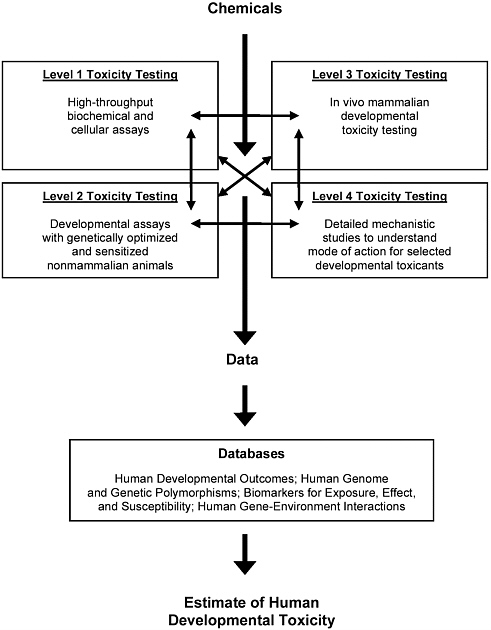
Figure 4-2 illustrates the possible testing scheme proposed by that committee. Testing in model systems is described as taking place on four levels. Level 1 includes molecular, biochemical, and cell-based assays that have high throughput and are expected to provide structure-activity information, relative potency of various chemicals tested in the same assay, insight into mixtures, and estimates of potency across chemical classes and assays. The proposed assays are designed to test chemical-induced alterations in conserved signaling and metabolic pathways. The committee envisioned that the application of the high-throughput screens could address 100,000 chemicals within 1 year. Level 2 proposes the use of nonmammalian animals to evaluate their response during developmental exposure. The fruit fly, nematodes, and zebrafish are mentioned as candidate organisms. Those model organisms could be partially “humanized” in their metabolism (that is, genetically
modified to express human metabolizing proteins) and sensitized to heighten the response of known important developmental pathways. The premise of testing in those models is that for some organs and structures, human and model organism differences are not great. It was estimated that 1,000-10,000 of those assays could be conducted each year. Level 3 involves testing in mammals, predominantly mice and rats, with the assumption that hundreds of assays could be conducted each year. The information expected to be gained in those animal tests is relative in vivo potency, the activity of chemical mixtures, mechanistic information, and dose-response relationships. Level 4 proposes detailed mechanistic evaluations of prototype members of families of chemicals whose mechanisms of action can be elucidated. Genetically optimized rodents are proposed as the likely test platform for those studies. The committee felt that about 10 such detailed evaluations could be conducted each year.
The model system and animal results are correlated with information gained from the assessment of toxicity, susceptibility, and chemical exposures in human populations. In particular, development of a number of databases is proposed, including a database of human developmental outcomes, a database of the human genome and genomic polymorphisms, a database of human biomarkers, and a database of human gene-environment interactions. The proposal recognized the challenge of creating and managing large databases and of linking and interrelating the mass of information.
Another important concept mentioned is the combining of information obtained from the model system and animal tests with that accumulated through an improved human surveillance program. It is less clear how the large mass of information generated in the proposed multilevel testing scheme is translated in practice into risk assessments. The committee’s report is presented at a conceptual level and discusses the potential for new technical approaches without providing a detailed plan for change. The committee’s proposal does not specify how the current testing paradigms and risk-assessment approaches could be modified and expanded to incorporate the new science and lead to a new scheme for data generation, animal-to-human extrapolation, and risk assessment. However, the committee’s report provides a useful framework for thinking about how different types and levels of testing and assessment could lead to an improved overall approach to chemical-toxicity assessments.
COMMITTEE OBSERVATIONS ON TOXICITY-TESTING STRATEGIES
In 1984, an NRC committee providing advice to the National Toxicology Program on testing priorities noted that there are far more chemicals in the human environment than can be evaluated for potential toxicity with available resources and methods (NRC 1984). That committee bemoaned the fragmentary information available with which to set priorities. The same holds true today. For the roughly 700 new industrial chemicals introduced into commerce each year, EPA essentially relies on its own structure-activity models to assess potential hazards and on information on use and estimated production volume contained in PMNs to characterize potential exposure. TSCA authorizes EPA to review existing chemicals, but toxicity and exposure information on them is typically so incomplete that it does not support the review process. EPA can require testing if it determines that a chemical meets a specific set of criteria; however, in vitro and whole-animal tests are rarely required. Thus, the basis for establishing priorities and requiring testing for industrial chemicals in the United States has not progressed much over the last 20 years.
The HPV program, which was a response to the lack of data collected under TSCA, uses a simple criterion to determine whether a chemical is to be included in the HPV testing program: nonpolymeric organic chemicals produced in or imported into the United States at a volume of 1 million pounds or more during the 1990 reporting year.2 Similarly, the EU uses production volume to assign priorities and define testing requirements. Unlike the U.S. HPV program, the EU program requires a minimum of testing of all chemicals produced at volumes of more than 10 kg/manufacturer per year. The extent of testing required increases with production volume; substantially greater testing, including carcinogenicity and pharmacokinetic studies, is required for the highest-production chemicals. For moderate- and high-production-volume chemicals, additional tests can be required on the basis of findings in the initial test battery and other tests. That contrasts with the minimal dataset requested in the U.S. HPV program, although EPA has indicated that it intends to use the results of the HPV program to assess further testing needs. For both the U.S. and EU HPV programs, it is unclear how other
exposure considerations—such as use, environmental persistence, and possible chemical breakdown to more active forms—might modify test selection and extent of testing and whether they have been sufficiently considered. Those other exposure considerations may lead to a better understanding of human exposure potential than production data alone.
Exposure considerations also dictate the degree of testing of direct and indirect food additives and pesticides. Testing is required for virtually all pesticides and all food additives that had not been sanctioned before the 1958 Food Additives Amendments or were not generally recognized as safe. Exposure is qualitatively considered in determining pesticide testing and is based on use. Potential exposures of the general population, especially via food, drive the greatest extent of testing. For indirect and direct food additives, a quantitative assessment of exposure (that is, concentration of the additive in the total diet) provides the basis of test selection. For direct food additives, study selection is modified by information on chemical structure. Regardless of the extent to which the “bright lines” used for food additives provide adequate protection, the overall approach of combining structural alerts with quantitative exposure information is an aspect of test-strategy design to explore for environmental agents.
The testing strategies discussed above generally use traditional, standardized toxicology tests. Results of an initial series of mandated tests can trigger additional testing to provide broader coverage of end points, exposure routes, or life stages or a greater depth of understanding of the effects observed. The tests that are triggered are also standardized toxicology studies.
Information generated by the testing strategies provides a basis for hazard identification (that is, assessment of whether a chemical has the potential to cause specific adverse responses regardless of exposure context). The test results indicate the exposures that produce adverse effects and thus help to characterize the dose-response relationship at least in the range of the experimental data, typically at high doses. The information produced is often sufficient for decision-making. For example, test results may indicate that a food additive or pesticide can be safely used as proposed. Given the doses at which toxicity is seen and the range of possible human exposures, risks may be so low that there is no need to curtail exposure even if one considers possible end points and life stages missed by the testing strategy. In contrast, if the margin between toxic doses and relevant human exposures is not large enough, further research may be needed to refine the dose-response relationship at lower doses,
particularly if some stakeholders find that the chemical is worth trying to keep on the market.
Thus, findings of toxicity studies can provoke a series of voluntary studies—often sponsored by industry or regulatory, scientific, or public health agencies—that seek to develop a better qualitative and quantitative understanding of dose-response characteristics and therefore a better understanding of the relevance of the findings for humans. The tests can be nonstandard and tailored to answer specific questions, such as those concerning mechanism of action or species differences in metabolism. The selection of tests and lines of inquiry can be ad hoc without a general framework for inquiry. For example, a general framework has not been adopted for generating and testing hypotheses and alternatives regarding mechanism of toxic action. Much to the frustration of sponsors, results of such testing can be found unacceptable for regulatory applications and not usable for refining risk estimates, although in several notable cases results have been found to be definitive. The ad hoc and unsystematic approaches for generating data for more detailed risk assessment contrast sharply with the mandated strategies for pesticide and food-additive registration and approval.
The toxicity tests and strategies discussed here have evolved primarily as a means of characterizing potential human health hazards and dose-response relationships at least at high doses. Existing strategies only partially recognize the toxicity-testing problems associated with exposure to mixtures and the associated problems in assessing aggregate exposures and cumulative risks. Dioxins, organophosphorus insecticides, and environmental estrogens have been tested as mixtures, but most testing continues to focus on individual chemicals. Different testing approaches generally stem from legislative mandates or from differences in individual agencies or program offices. Different approaches can result in inconsistent testing strategies among agencies or categories of chemicals even if the ultimate regulatory goal is the same. The differences in strategies for testing industrial chemicals by the EU and EPA are an example. The nature and extent of toxicity testing ideally would be guided by the regulatory risk-management decisions to be made and the assessments needed to support them.
NRC and OECD multilevel, multidisciplinary frameworks for evaluating developmental toxicants and endocrine disruptors provide insights into improving the standard regulatory framework for data generation and risk assessment. The general frameworks address key areas of uncertainty in cross-species and high-to-low dose extrapolation and
suggest approaches for incorporating high-volume screening of chemicals for those end points. Although considerable resources and commitment would be needed to develop such programs, they hold the promise of shifting to an improved system of testing and decision-making.
REFERENCES
EC (European Commission). 2003. Technical Guidance Document on Risk Assessment in Support of Commission Directive 93/67/EEC on Risk Assessment for New Notified Substances; Commission Regulation (EC) No. 1488/94 on Risk Assessment for Existing Substances, and Directive 98/8/EC of the European Parliament and the Council Concerning the Placing of Biocidal Products on the Market, 2nd Ed. EUR 20418 EN/1. European Chemicals Bureau, Institute for Health and Consumer Protection, Ispra, Italy [online]. Available: http://ecb.jrc.it/index.php?CONTENU=/Technical-Guidance-Document/ [accessed March 21, 2005].
EPA (U.S. Environmental Protection Agency). 1996. Discussion of registration, field testing, and experimental use permits. III Toxicology requirements. P. 3(e) in Microbial Pesticide Test Guidelines, OPPTS 885.0001. Overview for Microbial Pest Control Agents. EPA 712-C-96-280. Office of Prevention, Pesticides, and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/oppbppd1/biopesticides/regtools/guidelines/oppts_885_0001.htm#(e)%20Discussion%20of%20registration [accessed March 21, 2005].
EPA (U.S. Environmental Protection Agency). 2000. P. 8 in Environmental Protection Agency Endocrine Disruptor Screening Program: Report to Congress [online]. Available: http://www.epa.gov/scipoly/oscpendo/docs/reporttocongress0800.pdf [accessed March 21, 2005].
EPA (U.S. Environmental Protection Agency). 2004. Microbial Pesticide Test Guidelines OPPTS 885, Group C - Toxicology Test Guidelines. Office of Prevention, Pesticides, and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/oppbppd1/biopesticides/regtools/guidelines/microbial_gdlns.htm [accessed March 21, 2005].
EPA (U.S. Environmental Protection Agency). 2005. Endocrine Disruptor Screening Program, Assay Development and Validation, Assay Status Table. Office of Science Coordination and Policy; Office of Prevention, Pesticides, and Toxic Substances, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/scipoly/oscpendo/assayvalidation/status.htm [accessed Sept. 20, 2005].
FDA (U.S. Food and Drug Administration). 1993. Draft toxicology principals for safety assessment of direct food additives and color additives used in
food. “Redbook II.” Center for Food Safety and Applied Nutrition, U.S. Food and Drug Administration, Washington, DC.
FDA (U.S. Food and Drug Administration). 1997. Toxicological Testing of Food Additives. Office of Premarket Approval, Center for Food Safety and Applied Nutrition, U.S. Food and Drug Administration [online]. Available: http://www.cfsan.fda.gov/~dms/opa-tg1.html [accessed March 21, 2005].
FDA (U.S. Food and Drug Administration). 2003. General guidelines for designing and conducting toxicity studies. Section IV. B.1. in Redbook 2000, Toxicological Principles for the Safety Assessment of Food Ingredients, Office of Food Additive Safety, U.S. Food and Drug Administration [online]. Available: http://www.cfsan.fda.gov/~redbook/red-ivb1.html [accessed March 21, 2005].
GAO (U.S. Government Accounting Office). 2005. Chemical Regulation: Options Exist to Improve EPA’s Ability to Assess Health Risks and Manage its Chemical Review Program. GAO-05-458. U.S. Government Accounting Office, Washington, DC. June 2005 [online]. Available: http://www.gao.gov/new.items/d05458.pdf [accessed Oct. 24, 2005].
Gauderman, W.J., R. McConnell, F. Gilliland, S. London, D. Thomas, E. Avol, H. Vora, K. Berhane, E.B. Rappaport, F. Lurmann, H.G. Margolis, and J. Peters. 2000. Association between air pollution and lung function growth in southern California children. Am. J. Respir. Crit. Care Med. 162(4 Pt 1):1383-1390.
NRC (National Research Council). 1984. Toxicity Testing: Strategies to Determine Needs and Priorities. Washington, DC: National Academy Press.
NRC (National Research Council). 1999. Hormonally Active Agents in the Environment. Washington, DC: National Academy Press.
NRC (National Research Council). 2000. Scientific Frontiers in Developmental Toxicology and Risk Assessment. Washington, DC: National Academy Press.
OECD (Organisation for Economic Cooperation and Development). 2004a. Manual for Investigation of HPV Chemicals. Organisation for Economic Cooperation and Development [online]. Available: http://www.oecd.org/document/7/0,2340,en_2649_201185_1947463_1_1_1_1,00.html. [accessed Sept. 20, 2005].
OECD (Organisation for Economic Cooperation and Development). 2004b. Conceptual Framework for the Testing and Assessment of Endocrine Disrupting Chemicals. Prepared by the Secretariat of the Test Guidelines Programme, Based on the Agreement Reached at the 6th Meeting of the EDTA Task Force, 24 and 25 June 2002, Tokyo, Japan [online]. Available: http://www.oecd.org/dataoecd/17/33/23652447.doc [accessed March 21, 2005].
Peters, J.M., E. Avol, W.J. Gauderman, W.S. Linn, W. Navidi, S.J. London, H. Margolis, E. Rappaport, H. Vora, H. Gong, Jr., and D.C. Thomas. 1999.
A study of twelve Southern California communities with differing levels and types of air pollution. II. Effects on pulmonary function. Am. J. Respir. Crit. Care Med. 159(3):768-775.
Schettler T. 2001. Toxic threats to neurologic development of children. Environ. Health Perspect. 109(Suppl 6):813-816.
UN (United Nations). 2003. Globally Harmonized System of Classification and Labeling of Chemicals. United Nations Economic Commission for Europe [online]. Available: http://www.unece.org/trans/danger/publi/ghs/ghs_rev00/00files_e.html [accessed Nov. 17, 2005].