2
Epidemiology and Consequences of Traumatic Brain Injury—An Invisible Disability
Traumatic brain injury (TBI)—a brain injury caused by a blow or jolt to the head or a penetrating head injury that disrupts the function of the brain—is a leading cause of death and disability in the United States, particularly among very young children, adolescents, young adults, and elderly persons (NIH, 1998; Thurman et al., 1999; Coronado et al., 2005). Yet for many people who sustain a TBI, the effects of the injury are not obviously evident. The injury may not even be evident on neuroimaging in some cases (Gordon et al., 1998).
Because the damage to the brain from a TBI is hidden from view and the consequences are often not obvious to the casual observer, epidemiologists and other researchers often portray TBI as a “hidden” or “silent” epidemic (Box 2-1). The long-term consequences of TBI may manifest themselves in the form of altered cognition, personality, and behavior and, to a lesser extent, sensory and motor impairments (NIH, 1998; Thurman et al., 1999; Flashman and McAllister, 2002). Yet many health care professionals, community service workers, and the public are unaware of these consequences (Reynolds, 1993; Harris Interactive, 2000).
As background for the committee’s assessment of the impact of the TBI Program administered by the Health Resources and Services Administration (HRSA), this chapter provides basic information on the epidemiology and consequences of TBI. The focus here is on a particular subset of the TBI population—namely, individuals (and their families) with a TBI with post-acute impairments who require the services of myriad public and private service systems. Those individuals and the complex systems of care that
|
BOX 2-1 Mark is an attorney who does accounting. When you see him in his suit behind his desk punching numbers into his calculator you can’t tell that he needs cueing for such things as bathing, dressing, and other tasks of daily living. Lindsey is beautiful, charming, and lost. Injured as a college sophomore, she has returned 2 years later to audit her first class. She pretends to follow along, but she has yet to pass a test. Joey looks tough with his well-developed muscles as he struts along wearing his dark glasses to hide his feelings about his wife working and his inability to understand so much of what is happening around him. He is unable to accept his new less important and unpaid job in the photocopying department and depressed that he can no longer support his wife and four children. Anne’s first job, post-injury, was that of ticket taker at the movie theater, but she did not fit in. In addition to her frequent errors, she couldn’t recognize faces so she failed to acknowledge her coworkers. They were offended and considered her aloof. She could no longer drive and often was late for her job because she got lost while walking to work. She cried and angered easily and inappropriately. She was fired after 2 weeks. |
serve them constitute the group of individuals whose needs Congress addressed when it initially authorized the HRSA TBI Program in 1996 (P.L. 104-166) and then reauthorized it in 2000 (P.L. 106-310, 2000).1
EPIDEMIOLOGY OF TBI
TBI Incidence and Prevalence Estimates
In the immediate aftermath of TBI, a person may have a skull fracture, intracranial lesion, decreased level of consciousness for a period of time, or limited memory for events immediately preceding or following the onset of the brain injury, and other neurological or neuropsychological abnormalities. Initially, temporary swelling and bruising of the brain can produce a number of impairments such as confusion and loss of consciousness. When the initial swelling and bruising abates, the affected areas of the brain may return to normal functioning. In severe cases, the swelling may cause pres-
|
1 |
See Chapter 1 for an overview of the HRSA TBI Program and its legislative history. |
sure on the area of the brain that controls consciousness and wakefulness—the brainstem—thus causing coma or death.
TBI is known to affect millions of Americans, but experts widely acknowledge that precise estimates of the incidence and prevalence of TBI in the United States are elusive (NIH, 1998; Langlois et al., 2004). Available estimates of the incidence and prevalence of TBI are undercounts because most TBI surveillance systems draw only from hospital and emergency department records;2 however, people who have sustained a TBI may not be immediately evaluated at an emergency department, may not be hospitalized, or may not receive medical attention at all (Sosin et al., 1996; NIH, 1998; Schootman and Fuortes, 2000).
Furthermore, little is known about persons who sustain a TBI and are seen in doctor’s offices or other nonhospital settings. In individuals who sustain severe body injuries, or painful soft tissue injuries to the neck and shoulders, TBI may not be diagnosed until cognitive problems become evident. Some persons with a TBI may not seek care because they believe a “bump on the head” does not require medical attention or because they face barriers to care such as lack of health insurance (Reynolds et al., 2001). And in some cases, individuals who have sustained a TBI are not even recognized. Individuals with TBI who go uncounted are most likely persons with mild TBI or concussions (NIH, 1998).
Estimates of the number of people living with TBI-related disabilities in the United States are similarly imprecise. The number of people with long-term TBI-related disabilities has not been documented, because available surveillance data fail to differentiate between persons with TBI who have good recoveries and persons with TBI who experience persistent post-TBI symptoms and disability (NCIPC, 2003).
The Centers for Disease Control and Prevention (CDC), with a clear caveat that its estimates of the number of people living with a TBI-related disability in the United States are low, reports that at least 1.4 million TBIs occur in the United States annually (Figure 2-1) (Langlois, 2004). Of the 1.4 million people who sustain TBIs, approximately 50,000 die from their injuries and an estimated 80,000 to 90,000 incur a long-term, often permanent disability (Thurman et al., 1999). At a minimum, 5.3 million Americans are estimated to be living with a TBI-related disability (Thurman et al., 1999).
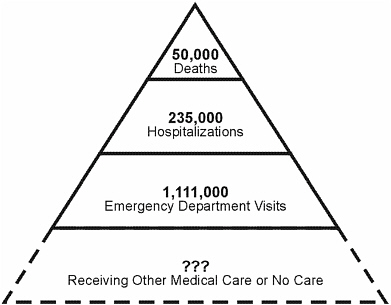
FIGURE 2-1 Average annual number of TBI-related emergency department visits, hospitalizations, and deaths, U.S., 1995–2001.
NOTE: Emergency department visits exclude persons who were hospitalized, died, transferred to another facility, or who had an unknown disposition. Hospitalizations exclude persons who died while being hospitalized.
SOURCE: Langlois et al., 2004.
Causes of TBI
The causes of TBI are varied. A TBI may result from a bullet entering brain tissue, forces due to extreme acceleration and deceleration produced by a motor vehicle crash or exposure to a bomb blast, or the sudden impact of falls, violent punches, construction accidents, or collisions on the playing field (NIH, 1998; Gondusky and Reiter, 2005; DVBIC, 2005).
During the period 1995–2001, the leading causes of TBI were falls (28 percent), motor vehicle accidents (20 percent), struck by/against3 (19 percent), and assaults (11 percent); these accounted for three-fourths of TBI-related emergency department visits, hospital stays, and deaths (Figure 2-2) (Langlois et al., 2004). Firearms are a major cause of TBI-related deaths (Thurman, 2001).
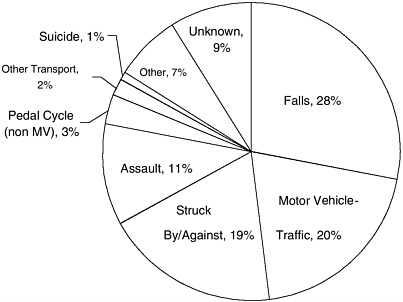
FIGURE 2-2 Average annual TBI-related emergency department visits, hospitalizations, and deaths, percent by external cause, U.S., 1995–2001.
SOURCE: Adapted from Langlois et al., 2004.
Risk Factors for TBI
Individuals who sustain a TBI are a heterogeneous group (NIH, 1998). Available TBI surveillance data related to TBI-related hospitalizations, emergency department visits, and deaths suggest that the population of individuals who sustain a TBI includes substantial numbers of the very young, the very elderly, as well as adolescents and young adults (Table 2-1).
Although many individuals who sustain a TBI were robust and healthy prior to their injury, others may have had one or more preexisting conditions that put them at risk (Corrigan, 1995; Guskiewicz et al., 2000; Coronado et al., 2005). The leading factors associated with risk for TBI-related hospitalization, emergency department visit, or death—age, gender, athletic activity, and military duty—are reviewed below.4
Age
The risk for TBI is age-related (Table 2-2). TBIs affect the very young (under age 5) at an alarming rate (1,120.7 TBIs per 100,000) that is 38 per-
TABLE 2-1 TBI-Related Emergency Department Visits, Hospitalizations, and Deaths, Number and Percent by Age Group, U.S., 1995–2001
|
Age Group |
Number |
Percent of Total |
Cumulative Percent |
|
0–4 |
216,000 |
15.5 |
15.5 |
|
5–9 |
133,000 |
9.5 |
25.0 |
|
10–14 |
125,000 |
9.0 |
34.0 |
|
15–19 |
160,000 |
11.5 |
45.4 |
|
20–24 |
103,000 |
7.4 |
52.8 |
|
25–34 |
184,000 |
13.2 |
66.0 |
|
35–44 |
166,000 |
11.9 |
77.9 |
|
45–54 |
101,000 |
7.2 |
85.1 |
|
55–64 |
53,000 |
3.8 |
88.9 |
|
65–74a |
50,000 |
3.6 |
92.5 |
|
Over 74 |
105,000 |
7.5 |
100.0 |
|
All ages |
1,396,000 |
100.0 |
|
|
aSample size is small so estimate may not be stable. bTotal percent does not add to 100 due to rounding. SOURCE: Adapted from Langlois et al., 2004. |
|||
cent greater than that for the age group with the next highest TBI rate, 15-to 19-year-olds (814.4 TBIs per 100,000).
As shown in Figure 2-3, accidental falls are the predominant cause of TBIs in young children. Motor vehicle traffic crashes are the predominant
TABLE 2-2 Rate of TBI-Related Hospitalizations, Emergency Department Visits, and Death, by Age, U.S., 1995–2001a
|
Age Group |
Rate per 100,000 |
|
0–4 |
1120.7 |
|
5–9 |
659.3 |
|
10–14 |
628.6 |
|
15–19 |
814.4 |
|
20–24 |
555.5 |
|
25–34 |
450.2 |
|
35–44 |
374.7 |
|
45–54 |
285.2 |
|
55–64 |
229.5 |
|
65–74b |
267.4 |
|
Over 74 |
659.1 |
|
All ages |
506.4 |
|
aAge-adjusted to the 2000 U.S. standard population. bSample size is small so estimate may not be stable. SOURCE: Adapted from Langlois, 2004. |
|

FIGURE 2-3 Average annual TBI-related rates for emergency department visits, hospitalizations, and deaths, by age group and external cause, U.S., 1995–2001.
SOURCE: Langlois et al., 2004.
cause of TBI among teenagers. The rate of TBI among adults tends to increase with age beginning at about age 65. Falls are by far the leading cause of TBI in this age group, accounting for more than half. Falls are the principal cause of TBI-related hospitalizations in older persons, especially among those with multiple, comorbid conditions (Coronado et al., 2005).
Gender
Males are far more likely than females to have a TBI (Figure 2-4). Fifteen- to 19-year-old males, for example, experience 174.5 TBI-related hospitalizations per 100,000 population compared with 81.1 TBI-related hospitalizations per 100,000 for similarly aged females.
Athletic Activity
The rate of sport-related TBI is not known. However, there is an extensive literature suggesting that TBI often goes undetected among high school, college, and professional athletes (Powell and Barber-Foss, 1999; Guskiewicz et al., 2000). Several studies also suggest that a history of concussion significantly increases the risk of sustaining another, more severe brain injury (Guskiewicz et al., 2000, 2003; Schulz et al., 2004).
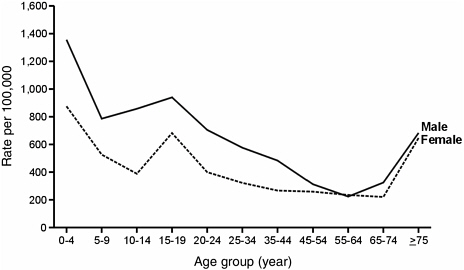
FIGURE 2-4 Average annual TBI-related rates for emergency department visits, hospitalizations, and deaths, by age group and sex, U.S., 1995–2001.
SOURCE: Langlois et al., 2004.
Military Duty
Military personnel in both noncombat and combat posts are at high risk for sustaining a TBI (Ommaya et al., 1996; DVBIC, 2005; Okie, 2005). One study, for example, found that 23 percent of noncombat, active-duty soldiers at Fort Bragg (n = 2,276) sustained a TBI during their military service (Ivins et al., 2003). The risk of TBI was highest among those soldiers with a prior TBI.
Furthermore, TBI has become a signature wound of the current war in Iraq (Box 2-2). Recent studies have found alarming rates of TBI among veterans injured by exposure to improvised explosive devices.
Native Americans
American Indians/Alaskan Natives have high rates of TBI-related hospitalizations compared with other races. Rutland-Brown and colleagues analyzed 1997–1999 hospital discharge data for persons in 13 states who were discharged alive after a TBI-related hospitalization (Rutland-Brown et al., 2005). The researchers found that among all adults, aged 20 to 44 years, American Indians/Alaskan Natives (78.5 per 100,000 persons) had the highest rate of TBI—almost 30 percent greater than that for whites (54.7 per 100,000 persons).
|
BOX 2-2 TBI has become a signature injury of the Iraq war. The high incidence of penetrating and closed-brain injuries is in part a result of U.S. soldiers’ increasing exposure to improvised explosive devices and improved military armor. Kevlar helmets, for example, often save lives but cannot prevent the internal bleeding, bruising, and tearing of brain tissue that result from exposure to blasts. The Defense and Veterans Brain Injury Center has studied the incidence of TBI at military health facilities. The center’s research and other recent studies find alarming rates of TBI among soldiers injured in Iraq. For example:
There is anecdotal evidence that soldiers who sustain TBIs may face unique barriers to both diagnosis and recovery. Some soldiers may return to their jobs and families without realizing the severity or consequences of their injury. Access to information and appropriate care appears to be a particular problem for veterans returning to rural areas. SOURCE: Peota, 2005; Okie, 2005; Gondusky and Reiter, 2005; DVBIC, 2005. |
Range in Severity
As discussed below, the terms commonly used for describing the severity of an acute TBI are mild, moderate, and severe; however, the severity of the initial brain injury is not sufficient to predict long-term consequences (Povlishock and Katz, 2005). It is important to recognize that some individuals in all severity groups experience a high rate of persistent symptoms.
The Glasgow Coma Scale is commonly used to determine the initial severity of TBI.5 A score of 13–15 is described as a mild TBI, 9–12 as a
|
BOX 2-3 Jane Woods was a 39-year-old senior economist working 55 hours a week for a think tank when she sustained a TBI. She is a graduate of Yale University, holds a Ph.D. in economics from Duke University, and is a former varsity athlete. Her “mild” TBI of 8 years ago has had a marked impact on her life. Today, she manages with effort to work 2 hours at a time in her volunteer job, and she can drive only 10 minutes at 10 mph before nausea forces her to stop. She continues to have auditory, visual, vestibular, and language deficits. Her summary: “Current criteria define my injury as mild; the changes in my life are radical.” |
moderate TBI, and ≤ 8 as a severe TBI.6 The variables typically considered in determining the initial severity of an acute TBI include decreased level of consciousness, duration of post-traumatic amnesia, skull fracture, bruises or blood clots in the brain, and neurological or neuropsychological dysfunction (Kay et al., 1993). In one study of hospital discharge data in 14 states, the CDC used the Glasgow Coma Scale to analyze severity of injury among persons hospitalized for TBI in 1997 (Langlois et al., 2003). The analysis found that approximately 10 percent of the hospitalized patients sustained severe TBIs; 10 percent, moderate; 75 percent, mild; and 6 percent, were of unknown severity.
The CDC further defines a mild TBI as an injury with any period of observed or self-reported transient confusion, disorientation or impaired consciousness; any amnesia with post-traumatic amnesia less than 24 hours; a loss of consciousness of 30 minutes or less; or observed signs of neurologic or neuropsychological dysfunction (NCIPC, 2003).
The majority (85 percent) of TBIs are considered “mild” (NIH, 1998; NCIPC, 2003; Bazarian et al., 2005). The signs and symptoms of mild TBI are often subtle and may be undetected by the patient, family members, and physicians in the period immediately following injury; the signs and symptoms may become apparent only when the individual attempts to resume his or her normal life activities.
The label “mild” TBI can also be misleading, because an estimated 10 to 20 percent of so-called “mild” TBI cases have significant persistent impairments (Box 2-3). Many individuals with mild TBI have significant
cognitive, physical, psychological, and social problems that lead to substantial disability and unemployment (Binder et al., 2005; Bazarian et al., 2005; Ruff, 2005); they are sometimes referred to as the “miserable minority.” Perhaps for this reason, the literature on identifying and treating the long-term consequences of “mild” TBI is growing (Kushner, 1998; Dikmen, 2001; NCIPC, 2003; Vanderploeg et al., 2003; Carroll et al., 2004; Peloso et al., 2004; Bazarian et al., 2005; Ruff, 2005).
CONSEQUENCES OF TBI FOR INDIVIDUALS, FAMILY MEMBERS, AND SOCIETY
The committee believes that for persons with TBI and the systems that serve them, it is far more important to identify and address the consequences of TBI than to characterize acute TBIs as mild, moderate, or severe. Despite the shortcomings in available research, a picture of the potential long-term impact of TBI has emerged. As discussed below, substantial proportions of persons with TBI report persistent limitations in activities of daily living; ability to return to work; social skills, relationships, and community participation. Family members trying to meet the needs of the injured person may experience declines in their own emotional and physical health status. There is also evidence that TBI has worrisome and significant costs for society at large—with particular burdens on an array of public and private systems including physical and behavioral health care delivery systems, schools, disability and welfare programs, and courts and correctional facilities.
Consequences of TBI for Individuals
For the majority of individuals with TBI, there are few, if any, long-term obvious effects (however, research is being conducted to better understand whether TBI creates vulnerability to future neurologic compromise). Yet some persons with TBI are likely to experience multiple signs and symptoms that appear in unique, unpredictable ways throughout their lifetime (Corrigan and Bogner, 2004). For these individuals, TBI brings a constellation of potential physical, cognitive, and behavioral consequences that may be temporary or permanent (Table 2-3) (NIH, 1998; NINDS, 2002). The consequences of TBI for individuals who suffer post-acute impairments are discussed below.7
TABLE 2-3 Constellation of Physical, Cognitive, and Behavior Changes After TBI
|
Physical Changes |
Cognitive Changes |
Behavioral Changes |
|
|
|
|
SOURCE: NIH, 1998. |
||
The direct consequences of TBI depend, in part, on which areas of the brain are injured (Figure 2-5). Injury to the frontal lobes of the brain, for example, can lead to an unusually broad range of symptoms, because this area of the brain is involved in many functions including fine motor skills, problem solving, spontaneity, memory, speech and language, initiation, judgment, impulse control, and social and sexual behavior (Thurman et al., 1999; Brown and Levin, 2001). Many TBIs involve the frontal lobes because they reside in a vulnerable location—directly behind the forehead and in a section of the skull with space for the brain to move around (NIDCD, 1998).
Recent research suggests that diffuse damage to brain tissue also significantly contributes to disturbances in concentration, attention, ability to handle multiple stimuli, and mood disorders (Smith et al., 2003; Buki and Povlishock, 2006). Diffuse injury occurs when a sudden jolt to the head causes the brain to move back and forth against the skull, leading to swelling and contusions in multiple areas of the brain. It is often produced by motor vehicle crashes and, sometimes, falls and assaults (Smith et al., 2003).
The sequence and nature of a TBI’s emerging symptoms depends on a range of critical factors including the site and severity of the injury; preexisting conditions, such as prior TBIs; behavioral, psychiatric, substance abuse, and learning problems; and a number of mediating variables including age, family income, health insurance, access to appropriate services; and social supports (Gordon et al., in press; Hibbard et al., 1998; Novack et al., 2001; Forducey et al., 2003; Dikmen et al., 2003; Mellick et al., 2003; Breed et al., 2004; Whiteneck et al., 2004a, 2004b). Diagnosing and under-

FIGURE 2-5 Architecture of the brain.
NOTE: This figure provides a superficial guide to major brain areas and is not meant to imply that specific functions mentioned are exclusively located in the areas shown; rather, related functions affect and are affected by other functions and brain regions in complex interactions to produce fully effective expression.
SOURCE: NINDS, 2005; Centre for Neuroskills, 2005.
standing TBI in infants, children, and adolescents can be particularly challenging because the effects of the injury may not become apparent until the child ages and fails to attain developmental milestones (Box 2-4).
Physical Health
The majority of individuals with TBI are mobile and able to care for themselves (Novack, 2000). Nevertheless, there are numerous physical
|
BOX 2-4 The complex physical, cognitive, and behavioral impairments associated with TBI have profound implications for children. In contrast to adults, pediatric TBI alters a developing brain. Furthermore, a child’s recovery occurs in the context of ongoing developmental processes. The TBI may not only affect previously learned skills, but also the capacity to gain new skills. As time passes and the child grows, the long-term impact of a child’s TBI may be misinterpreted or undetected altogether. Children injured early in life may appear to resume school activities successfully until they move on to middle school or high school and are expected to be increasingly competent and independent. Psychosocial disorders may not become obvious until the child moves into a more emotionally and socially challenging environment. SOURCES: NIH, 1998; Glang, 2004; Ylvisaker et al., 2005; Savage et al., 2005. |
health problems that are common after a TBI (Table 2-3) (Hibbard et al., 1998; NINDS, 2002; Breed, 2004).
Cognitive Abilities and Communication
Cognitive problems related to TBI typically have more impact on a patient’s recovery and outcome than physical limitations. Individuals with TBI with cognitive impairments often have difficulties concentrating, remembering, organizing their thoughts, making good decisions, solving everyday problems, and planning and foresight. They may be easily confused or forgetful. Language skills, both written and spoken, may also be impaired. Some people with TBI find it hard to learn new information or interpret the subtle cues and actions of others. As a result, they may act or speak inappropriately. Such deficits can impede one’s ability to communicate with others, to seek and obtain necessary services, and to maintain or develop all types of human relationships. Getting necessary care is also difficult because patients often forget appointments and struggle to articulate effectively and present their problems to providers.
Behavioral Competence
A substantial literature documents that TBI increases the risk of major depression, general anxiety disorder, obsessive-compulsive disorder, panic
disorder, post-traumatic stress disorder, anti-social behavior such as criminality and substance abuse, and suicide (Gordon et al., in press; Brown and Levin, 2001; Fann et al., 2004; Oquendo et al., 2004). The occurrence of these disorders appears to be independent of injury, age, severity of injury, and gender.
Individuals with TBI frequently report emotional problems related to impaired social relations, feelings of estrangement and isolation, difficulty accepting physical and lifestyle changes and difficulty accepting loss of control or competence in day-to-day decision making (Jordan et al., 2003). Individuals with TBI with preexisting behavioral and psychiatric problems may find that the brain injury exacerbates their condition and makes the management of day-to-day function all the more complex and difficult.
Depression is particularly prevalent post-TBI and is correlated with other problems including perceived impact of TBI, social support, cognitive symptoms, physical complaints and activity limitations, sexual dysfunction, and satisfaction with health and income (Granger et al., 1995; CDC, 1999; Hibbard et al., 2004).
It is important for health care professionals to recognize and treat psychological problems in individuals with TBI. Unfortunately, the potential for focusing on the psychological consequences of TBI can result in an inappropriate primary diagnosis of a psychiatric illness, ignoring the TBI diagnosis (McGuire et al., 1998; Torsney, 2004).
Daily Lives
It is difficult to capture the impact of TBI on an individual’s every day existence. Data collection and analysis are daunting challenges given the fragmented nature of TBI services and the inflexibility of their disparate data systems, lack of standardized definitions, and multiple public and private service systems.
Most of the available research draws from convenience samples, such as persons hospitalized for TBI, medical record reviews, and follow-up interviews with persons with TBI to document levels of impairment and need for assistance (Novack et al., 2001; Whiteneck et al., 2004b). Findings from such studies cannot be generalized to the overall TBI population and are limited by a variety of inconsistent outcome measures and other methodological details. A “better accounting” of the long-term consequences of TBI in a comprehensive sample of individuals with TBI is clearly needed (Dawson and Chipman, 1995). Advancing the research will be critical if federal, state, and private TBI programs are to meet the needs of individuals with TBI and their families.
Ability to Perform Activities of Daily Living (ADLs). An individual’s ability to perform ADLs is a well-established indicator of the person’s level
of disability and dependence on others. ADLs are basic, personal activities such as bathing, eating, dressing, mobility, transferring from bed to chair, and using the toilet (DHHS, 2003). ADLs also include higher level activities such as shopping, cooking, and managing personal finances.
The percentages of persons with TBI who are unable to perform specific ADLs are not known. The available studies report a wide range of estimates. Whiteneck and colleagues, for example, followed 1,591 Colorado adults who were hospitalized for TBI from 1996–1999. At 1 year post-TBI, 37 percent of the interviewees reported requiring assistance in either eating, bathing, dressing, toileting, walking, and/or using a wheelchair (Whiteneck et al., 2004b). In a study of 454 Canadian adults with TBI living in the community, Dawson and Chipman documented long-term rates of various indicators of disability. After an average 13 years post-injury, 15 percent of the sample needed assistance in personal care, and more than 30 percent reported needing help with basic activities such as shopping and managing personal finances (Dawson and Chipman, 1995).
Ability to Return to Work, School, and Productive Activity. The ability to be a productive member of society, whether in employment, school, or other settings, is associated with perceived quality of life after TBI (O’Neill et al., 1998; Steadman-Pare et al., 2001). There are no definitive estimates of employment and school attendance post-TBI, and little is known about how productive activity after TBI changes over time. The available research suggests, however, that psychosocial, cognitive, and physical impairments dramatically limit some individuals’ ability to return to productive activity, whether it is work, school, or other endeavors (Buffington and Malec, 1997). It also appears that successful employment after TBI is related to severity of injury, preinjury occupation or education, and age at injury (Wehman et al., 2005).
In the Colorado study, described above, return to work varied significantly by severity of injury and gender: 1 year after injury, less than half (47 percent) of the 207 individuals with severe injuries had returned to work, compared with 80.5 percent of the 1,273 persons with mild injuries (Whiteneck et al., 2004b). Regardless of severity, return to work was not necessarily with the same employer or in the same position as before injury.
Kreutzer and colleagues followed 186 previously employed adults who had been admitted to an acute care hospital within 24 hours of a mild, moderate, or severe brain injury (Kreutzer et al., 2003). Each subject received an individualized, comprehensive program of inpatient rehabilitation, including occupational therapy, physiatry and related medical services, psychology and neuropsychology, therapeutic recreation, social services, and speech and language therapy. The patients were interviewed annually over a 4-year, period and their medical records were reviewed. At 3 or 4 years after injury, just 42 percent of the subjects were employed.
Ability to Develop and Maintain Relationships. Sound physical health, cognition, and behavior are intrinsic to one’s social skills and ability to develop and maintain relationships. Impairments in these critical human abilities have profound implications for an individual’s sense of independence, perception of well-being, and overall quality of life (Jordan et al., 2003). Persons with brain injury are often susceptible to overstimulation and thus some traditional mental health group treatments may be overwhelming. Additionally, some housing options have a high degree of visual and acoustic stimuli, which may agitate the person with a brain injury and lead to a behavioral outburst (Torsney, 2004).
Consequences of TBI for Family Members
The emotional and physical health status of the family caregiver can be as compromised as that of the person with TBI (Thomsen, 1984; Hall et al., 1994). Many individuals with severe brain injury require ongoing care and supervision and are thus dependent on their families to meet their daily needs. Most families are not equipped to care for someone with the cognitive deficits and behavioral and emotional changes that are characteristic of TBI (Lezak, 1988). Families generally have little knowledge about the effects of brain injury, how to manage the associated problems, or how to care for individuals with brain injury.
The consequence is substantial stress for family caregivers who must radically change their lives to meet their loved ones’ long-term needs and financial burdens. In addition, dealing with the brain injured person’s “former self” and feeling trapped and isolated can contribute to high stress levels and compromised mental health. Family members experience not only the loss of income, but the loss of the relationship before the injury (Vandiver et al., 2003).
In general, family stress following brain injury revolves around three areas: isolation, quality of life, and health status (Lezak, 1988). With regard to isolation, studies have shown that friends and relatives may rally around the family in the early stages of recovery but tend to withdraw support and contact over time (Jordan et al., 2003). Clinical experience suggests that outpatient psychological care may be offered to people in the early stages of recovery, but family members may not be ready to receive this form of support at that time. Several years after injury, when problems are finally recognized, relatives may not know how to request services or may be ineligible for them because of insurance or other financial restraints (Gervasio and Kreutzer, 1997).
Consequences of TBI for Society
TBI appears to place a substantial burden on an array of social institutions and systems such as psychiatric facilities, courts and correctional facilities, schools, and disability and welfare programs. Definitive estimates of the prevalence of TBI among users of these institutions do not exist. Nevertheless, available research suggests that TBI definitely has an impact. Several studies of various convenience samples, for example, have found that half or more of individuals charged with a criminal offense or in prison have a history of TBI (Lewis et al., 1986; Sarapata et al., 1998; Slaughter et al., 2003).
Co-occurrence of TBI and substance abuse is well documented (Silver et al., 2001; Fann et al., 2002; Bombardier et al., 2002; Walker et al., 2003; Horner et al., 2005). A literature review of 11 studies of patients admitted to brain injury rehabilitation centers found that 50 to 66 percent of the patients had a history of alcohol or other drug abuse (Corrigan, 1995).
Murrey and colleagues reviewed the clinical and legal records of 3,133 adult inpatients in seven U.S. state-operated psychiatric hospitals (Murrey et al., 2004). The researchers found that 16.7 percent of the inpatients had a documented history of TBI, and 6.4 percent had dysfunction of thinking considered secondary to TBI. Silver and colleagues interviewed a probability sample of 5,034 adults in the New Haven metropolitan area for a National Institute of Mental Health epidemiologic study. They found that 43 percent of persons with a history of severe brain trauma had a diagnosed psychiatric disorder, twice the rate of persons (20 percent) with no TBI.
SUMMARY
This chapter has described the epidemiology and consequences of TBI for individuals, family members, and society. The immediate aftermath of a TBI may be a skull fracture, intracranial lesion, decreased level of consciousness or limited memory for events immediately preceding or following the injury, and other neurological or neuropsychological abnormalities. The immediate effects of a TBI are not necessarily predictive of longer term outcomes.
The repercussions of brain injury may be temporary or may result in profound disability. Individuals with TBI are likely to experience multiple signs and symptoms that appear in unique, unpredictable ways throughout their lifetime. The sequence and nature of a TBI’s emerging symptoms depends on the site and severity of the injury; preexisting conditions, such as prior TBIs; behavioral, psychiatric, substance abuse, and learning problems; and a number of mediating variables including age, family income, health insurance, access to appropriate services, and social supports.
Data on the epidemiology of TBI have limitations because they are drawn primarily from hospital and emergency department records, so they do not capture individuals with TBI who are seen in doctors’ offices or who do not seek medical care. In the United States, it is estimated that 80,000 to 90,000 individuals per year sustain a TBI with long-term, often lifelong implications. At a minimum, 5.3 million people in the United States live with a TBI-related disability.
Individuals with TBI are a heterogeneous group, including the very young, the very elderly, as well as adolescents and young adults. Although many individuals with TBI were robust and healthy prior to their injury, others may have had one or more preexisting conditions that put them at risk. Persons in the military are at heightened risk—TBI is the signature injury of the conflict in Iraq.
The majority of individuals with TBI are mobile and able to care for themselves. Nevertheless, various physical health problems are common after a TBI, including difficulties in balance and motor coordination, fatigue, headache, sleep disturbances, seizures, sensory impairments, slurred speech, spasticity and tremors, difficulties in urinary control, dizziness and vestibular dysfunction, and weakness.
Cognitive problems related to TBI typically have more impact on a patient’s recovery and outcome than physical limitations. Cognitively impaired persons with TBI are easily confused or forgetful; their language skills may also be impaired. Some people with TBI find it hard to learn new information or interpret the subtle cues and actions of others.
A substantial literature documents that TBI increases the risk of major depression, general anxiety disorder, panic disorder, post-traumatic stress disorder, antisocial behavior such as criminality and substance abuse, and suicide. Individuals with TBI with preexisting behavioral and psychiatric problems may find that the brain injury exacerbates their condition and makes the management of day-to-day function all the more complex and difficult.
It is difficult to capture the impact of TBI on an individual’s every day existence. Data collection and analysis are daunting challenges given the fragmented nature of TBI services and the inflexibility of their disparate data systems, lack of standardized definitions, and multiple public and private service systems. Nevertheless, substantial proportions of individuals with TBI report persistent limitations in activities of daily living, ability to return to work, social skills, relationships, and community participation.
The emotional and physical health status of the family caregiver can be as compromised as that of the individual with TBI. Most families are not equipped to care for someone with the cognitive deficits and behavioral and emotional changes that are characteristic of severe TBI. The consequence is substantial stress for family caregivers who must radically change their lives
to meet their loved ones’ long-term needs and financial burdens. There is also evidence that TBI has worrisome and significant costs for society at large, placing burdens on physical and behavioral health care delivery systems, schools, disability and welfare programs, and courts and correctional facilities.
REFERENCES
Bazarian J, McClung J, Shah M, Cheng Y, Flesher W, Kraus J. 2005. Mild traumatic brain injury in the United States, 1998–2000. Brain Injury. 19(2): 85–91.
Binder S, Corrigan JD, Langlois JA. 2005. The public health approach to traumatic brain injury: An overview of CDC’s research and programs. Journal of Head Trauma Rehabilitation. 20(3): 189–195.
Bombardier C, Rimmele C, Zintel H. 2002. The magnitude and correlates of alcohol and drug use before traumatic brain injury. Archives of Physical Medicine and Rehabilitation. 83: 1765–1773.
Breed ST, Flanagan SR, Watson KR. 2004. The relationship between age and the self-report of health symptoms in persons with traumatic brain injury. Archives of Physical Medicine and Rehabilitation. 85(Supplement 2): 61–67.
Brown SA, Levin HS. 2001. Clinical presentation of neurological sequelae of traumatic brain injury. In: Head Trauma: Basic, Preclinical, and Clinical Directions, Miller L, Hayes R, Newcomb J, eds. Canada: Wiley-Liss, Inc. Pp. 349–370.
Buffington A, Malec J. 1997. The vocational rehabilitation continuum: Maximizing outcomes through bridging the gap from hospital to community based services. Journal of Head Trauma Rehabilitation. 12: 1–13.
Buki A, Povlishock JT. 2006. All roads lead to disconnection?—Traumatic axonal injury revisited. Acta Neurochir (Wien). 148(2): 181–194.
Callahan CD. 2005. Traumatic brain injury: Rehabilitative treatment and case management, 2nd ed. Journal of Head Trauma Rehabilitation. 20(1): 110–113.
Carroll LJ, Cassidy JD, Holm L, Kraus J, Coronado VG. 2004. Methodological issues and research recommendations for mild traumatic brain injury: The WHO collaboration center task force on mild traumatic brain injury. Journal of Rehabilitation Medicine. Suppl. 43: 113–125.
CDC (Centers for Disease Control and Prevention). 1999. Report and Recommendations From the Traumatic Brain Injury and Public Health Meeting, April 12–14, 1999, Atlanta, GA. Washington, DC: CDC.
Centre for Neuroskills. 2005. Brain Map. [Online] Available: http://www.neuroskills.com/index.html?main=tbi/brain.shtml. [accessed 1/2/2006].
Coronado V, Thomas K, Sattin R, Johnson RL. 2005. The CDC traumatic brain injury surveillance system: Characteristics of persons aged 65 years and older hospitalized with a TBI. Journal of Head Trauma Rehabilitation. 20(3): 215–228.
Corrigan JD. 1995. Substance abuse as a mediating factor in outcome from traumatic brain injury. Archives of Physical Medicine and Rehabilitation. 76(4): 302–309.
Corrigan JD, Bogner J. 2004. Latent factors in measures of rehabilitation outcomes after traumatic brain injury. Journal of Head Trauma Rehabilitation. 19(6): 445–458.
Dawson D, Chipman M. 1995. The disablement experienced by traumatically brain-injured adults living in the community. Brain Injury. 9: 339–353.
DHHS (Department of Health and Human Services). 2003. Glossary of Disability, Aging and Long-Term Care Terms. [Online] Available: http://aspe.hhs.gov/daltcp/diction.shtml#ADLs [accessed 10/21/2005].
Dijkers MP. 2004. Quality of life after traumatic brain injury: A review of research approaches and findings. Archives of Physical Medicine and Rehabilitation. 85(Supplement 2): 21–35.
Dikmen S, Machamer J, Temkin N. 2001. Mild head injury: Facts and artifacts. Journal of Clinical and Experimental Neuropsychology. 23(6): 729–738.
Dikmen SS, Machamer JE, Powell JM, Temkin NR. 2003. Outcome 3 to 5 years after moderate to severe traumatic brain injury. Archives of Physical Medicine & Rehabilitation. 84: 1449–1457.
Durbin DR, Localio AR, MacKenzie EJ. 2001. Validation of the ICD/AIS MAP for pediatric use. Injury Prevention. 7(2): 96–99.
DVBIC (Defense and Veterans Brain Injury Center). 2005. Blast Injury. [Online] Available: http://www.dvbic.org/blastinjury.html [accessed 9/21/2005].
Elovic EP, Simone LK, Zafonte R. 2004. Outcome assessment for spasticity management in the patient with traumatic brain injury: The state of the art. Journal of Head Trauma Rehabilitation. 19(2): 155–77.
Fann J, Leonetti A, Jaffe K, Katon W, Cummings P, Thompson R. 2002. Psychiatric illness and subsequent traumatic brain injury: A case control study. Journal of Neurology, Neurosurgery, and Psychiatry. 72: 615–620.
Fann JR, Burington MS, Leonetti A, Jaffe K, Katon WJ, Thompson RS. 2004. Psychiatric illness following traumatic brain injury in an adult health maintenance organization. Archives of General Psychiatry. 61: 53–61.
Flashman L, McAllister T. 2002. Lack of awareness and its impact in traumatic brain injury. NeuroRehabilitation. 285–296.
Forducey PG, Ruwe WD, Dawson SJ, Scheideman-Miller C, McDonald NB, Hantla MR. 2003. Using telerehabilitation to promote TBI recovery and transfer of knowledge. NeuroRehabilitation. 18(2): 103–111.
Foster M, Tilse C. 2003. Referral to rehabilitation following traumatic brain injury: A model for understanding inequities in access. Social Science & Medicine. 56(10): 2201–2210.
Gervasio A, Kreutzer J. 1997. Kinship and family members’ psychological distress after brain injury: A large sample study. Journal of Head Trauma Rehabilitation. 12(3): 14–26.
Glang A, Tyler J, Pearson S, Todis B, Morvant M. 2004. Improving educational services for students with TBI through statewide consulting teams. NeuroRehabilitation. 19: 219–231.
Gondusky JS, Reiter MP. 2005. Protecting military convoys in Iraq: An examination of battle injuries sustained by a mechanized battalion during Operation Iraqi Freedom II. Military Medicine. 170(6): 546–549.
Gordon W, Brown M, Sliwinski M, Hibbard MR, Patti N, Weiss MJ, Kalinsky R, Sheerer M. 1998. The enigma of “hidden”traumatic brain injury. Journal of Health Trauma Rehabilitation. 13(6): 39–56.
Gordon W, Zafonte R, Cicerone K, Cantor J, Lombard L, Goldsmith R, Chanda T, Brown M. 2006. Traumatic brain injury rehabilitation: State of the science. American Journal of Physical Medicine and Rehabilitation. 85(4):343–382.
Granger C, Divan N, Fiedler R. 1995. Functional assessment scales. A study of persons after traumatic brain injury. American Journal of Physical Medicine and Rehabilitation. 74: 102–113.
Guskiewicz KM, Weaver NL, Padua DA, Garrett WE. 2000. Epidemiology of concussion in collegiate and high school football players. The American Journal of Sports Medicine. 28(5): 643–650.
Guskiewicz KM, Marshall SW, Cantu RC, Randolph C, Barr W, Onate JA, Kelly JP. 2003. Cumulative effects associated with recurrent concussion in collegiate football players: The NCAA Concussion Study. Journal of the American Medical Association. 290(19): 2549–2555.
Hall K, Karzmark P, Stevens M, Englander J, O’Hare P, Wright J. 1994. Family stressors in traumatic brain injury: A two-year follow-up. Archives of Physical Medicine Rehabilitation. 75: 876–884.
Harris Interactive I. 2000. Public Perceptions of Brain and Head Injuries. New York, NY.
Hibbard M, Uysal S, Sliwinski M, Gordon W. 1998. Undiagnosed health issues in individuals with traumatic brain injury living in the community. Journal of Head Trauma Rehabilitation. 13(4): 47–57.
Hibbard MR, Ashman TA, Spielman LA, Chun D, Charatz HJ, Melvin S. 2004. Relationship between depression and psychosocial functioning after traumatic brain injury. Archives of Physical Medicine & Rehabilitation. 85(Supplement 2): 43–53.
Horner MD, Ferguson PL, Selassie AW, Labbate LA, Kniele K, Corrigan JD. 2005. Patterns of alcohol use 1 year after traumatic brain injury: A population-based, epidemiological study. Journal of the International Neuropsychological Society. 11: 322–330.
Ivins B, Schwab K, Warden D, Harvey S, Hoilien M, Powell J, Johnson E, Salazar A. 2003. Traumatic brain injury in U.S. army paratroopers: prevalence and character. The Journal of Trauma Injury, Infection, and Critical Care. 55(4): 617–621.
Jordan C, Hunter S, Rycraft J, Vandiver V. 2003. Assessing families who are multistressed. In: Clinical Assessment for Social Workers. Jordan C, Franklin C, eds., 2nd edition. Chicago, IL: Lyceum Press. Pp. 313–350.
Kay T, Harrington D, Adams R. 1993. Definition of mild traumatic brain injury. Journal of Head Trauma Rehabilitation. 8(3): 86–87.
Kreutzer JS, Marwitz JH, Walker W, Sander A, Sherer M, Bogner J, Fraser R, Bushnik T. 2003. Moderating factors in return to work and job stability after traumatic brain injury. Journal of Head Trauma Rehabilitation. 18(2): 128–138.
Kushner D. 1998. Mild traumatic brain injury. Archives of Internal Medicine. 158: 1617–1624.
Langlois J, Rutland-Brown W, Thomas K. 2004. Traumatic Brain Injury in the United States: Emergency Department Visits, Hospitalizations, and Deaths. Atlanta, GA: National Center for Injury Prevention and Control.
Langlois JA, Kegler SR, Butler JA, Gotsch KE, Johnson RL, Reichard AA, Webb KW, Coronado VG, Selassie AW, Thurman DJ. 2003. Traumatic brain injury-related hospital discharges: Results from a 14-state surveillance system, 1997. MMWR Surveillance Summaries. 52(SS04): 1–18.
Langlois JA, Marr A, Mitchko J, Johnson RL. 2005. Tracking the silent epidemic and educating the public. Journal of Head Trauma Rehabilitation. 20(3): 196–204.
Lewis DO, Pincus JH, Feldman M, Jackson LJ, Bard B. 1986. Psychiatric, neurological, and psychoeducational characteristics of 15 death row inmates in the United States. American Journal of Psychiatry. 143(7): 838–845.
Lezak MD. 1988. Brain damage is a family affair. Journal of Clinical and Experimental Neuropsychology. 10(1): 111–123.
MacKenzie EJ. 1984. Injury severity scales: Overview and directions for future research. American Journal of Emergency Medicine. 2(6): 537–549.
MacKenzie EJ, Steinwachs DM, Shankar B. 1989. Classifying trauma severity based on hospital discharge diagnoses. Validation of an IDD-9CM to AIS-85 conversion table. Medical Care. 27(4): 412–422.
McGuire LM, Burright RG, Williams R, Donovick PJ. 1998. Prevalence of traumatic brain injury in psychiatric and non-psychiatric subjects. Brain Injury. 12(3): 207–214.
Mellick D, Gerhart KA, Whiteneck GG. 2003. Understanding outcomes based on the post-acute hospitalization pathways followed by persons with traumatic brain injury. Brain Injury. 17(1): 55.
Murrey GJ, Starzinski DT, LeBlanc AJ. 2004. Base rates of traumatic brain injury history in adults admitted to state psychiatric hospitals: A 3-year study. Rehabilitation Psychology. 49(3): 259–261.
NCIPC (National Center for Injury Prevention and Control). 2003. Report to Congress on Mild Traumatic Brain Injury in the United States: Steps to Prevent a Serious Public Health Problem. Atlanta, GA: CDC.
NIDCD (National Institute on Deafness and Other Communication Disorders). 1998. Traumatic Brain Injury: Cognitive and Communication Disorders. [Online] Available: http://www.nidcd.nih.gov/health/voice/tbrain.asp [accessed 10/17/2005].
NIH (National Institutes of Health). 1998. NIH Consensus Statement: Rehabilitation of Persons With TBI. Bethesda, MD: NIH. 16.
NINDS (National Institute of Neurological Disorders and Stroke). 2002. Traumatic Brain Injury: Hope Through Research. Bethesda, MD: NIH.
——. 2005. Know Your Brain: Brain Basics. [Online] Available: http://www.neuroskills.com/index.html?main=tbi/brain.shtml [accessed 10/17/2005].
Novack T. 2000. What to Expect After TBI. Presentation at the meeting of the Recovery After TBI Conference.
Novack T, Bush B, Meythaler J, Canupp K. 2001. Outcome after traumatic brain injury: Pathway analysis of contributions from premorbid, injury severity, and recovery variables. Archives of Physcal Medicine & Rehabilitation. 82: 300–305.
Okie S. 2005. Traumatic brain injury in the war zone. New England Journal of Medicine. 352(20): 2043–2047.
Ommaya AK, Dannenberg AL, Salazar AM. 1996. Causation, incidence, and costs of traumatic brain injury in the U.S. military medical system. Journal of Trauma-Injury, Infection, and Critical Care. 40(2): 211–217.
O’Neill J, Hibbard MR, Brown M. 1998. The effect of employment on quality of life and community integration after traumatic brain injury. Journal of Head Trauma Rehabilitation. 13(4): 68–79.
Oquendo MA, Friedman JH, Grunebaum MF, Burke A, Silver JM, Mann JJ. 2004. Suicidal behavior and mild traumatic brain injury in major depression. The Journal of Nervous and Mental Disease. 192(6): 430–434.
Ownsworth T, McKenna K. 2004. Investigation of factors related to employment outcome following traumatic brain injury: A critical review and conceptual model. Disability & Rehabilitation. 26(13): 765–783.
Peloso PM, Carroll L, Cassidy J, Borg J, von Holst H, Holm L, Yates D. 2004. Critical evaluation of the existing guidelines on mild traumatic brain injury. Journal of Rehabilitation Medicine. (Supplement 43): 106–112.
Peota C. 2005. Invisible wounds. Minnesota Medicine. 88(1): 13–14.
P.L. 106-310. Children’s Health Act of 2000.
Povlishock JT, Katz DI. 2005. Update of neuropathology and neurological recovery after traumatic brain injury. Journal of Head Trauma Rehabilitation. 20(1): 76–94.
Powell JW, Barber-Foss KD. 1999. Traumatic Brain Injury in High School Athletes. Journal of the American Medical Association. 282(10): 958–963.
Reynolds WE. 1993. Traumatic brain injury: A public policy analysis from a state perspective. American Rehabilitation. 19(2): 12–20.
Reynolds WE, Page SJ, Johnston MV. 2001. Coordinated and adequately funded state streams for rehabilitation of newly injured persons with TBI. Journal of Head Trauma Rehabilitation. 16(1): 34–46.
Ruff R. 2005. Two decades of advances in understanding mild traumatic brain injury. Journal of Head Trauma Rehabilitation. 20(1): 15–18.
Rutland-Brown W, Wallace LJD, Faul MD, Langlois JA. 2005. Traumatic Brain Injury Hospitalizations Among American Indians/Alaska Natives. Journal of Head Trauma Rehabilitation. 20(3): 205–214.
Sarapata M, Herrmann D, Johnson T, Aycock R. 1998. The role of head injury in cognitive functioning, emotional adjustment and criminal behaviour. Brain Injury. 12(10): 821–842.
Savage RC, DePompei R, Tyler J, Lash M. 2005. Paediatric traumatic brain injury: A review of pertinent issues. Pediatric Rehabilitation. 8(2): 92–103.
Schootman M, Fuortes LJ. 2000. Ambulatory care for traumatic brain injuries in the U.S., 1995–1997. Brain Injury. 14(4): 373–381.
Schulz MR, Marshall SW, Mueller FO, Yang J, Weaver NL, Kalsbeek WD, Bowling JM. 2004. Incidence and risk factors for concussion in high school athletes, North Carolina, 1996–1999. American Journal of Epidemiology. 160(10): 937–944.
Sherer M. 2005. Cognitive and behavioral rehabilitation from neurobiology to clinical practice. Journal of Head Trauma Rehabilitation. 20(3): 282–283.
Silver J, Kramer R, Greenwald S, Weissman M. 2001. The association between head injuries and psychiatric disorders: Findings from the New Haven NIMH Epidemiologic Catchment Area Study. Brain Injury. 15(11): 935–945.
Slaughter B, Fann J, Ehde D. 2003. Traumatic brain injury in a county jail population: Prevalence, neuropsychological functioning and psychiatric disorders. Brain Injury. 17(9): 731–741.
Smith DH, Meaney DF, Shull WH. 2003. Diffuse axonal injury in head trauma. Journal of Head Trauma Rehabilitation. 18(4): 307–316.
Sosin D, Sniezek J, Thurman D. 1996. Brain Injury. 10: 47–54.
State grants for projects regarding brain injury (2003). GPO Access. [Online] Available: http://www.access.gpo.gov/uscode/title42/chapter6a_subchapterx_parte_.html [2/9/2006].
Steadman-Pare D, Colantonio A, Ratcliff G, Chase S, Vernich L. 2001. Factors associated with perceived quality of life many years after traumatic brain injury. 26(4): 330–342.
Taylor HG. 2004. Research on outcomes of pediatric traumatic brain injury: Current advances and future directions. Developmental Neuropsychology. 25(1-2): 199–225.
Thomsen IV. 1984. Late outcomes of very severe blunt head traumas: A 10–15 year second follow-up. Journal of Neurosurgical Psychiatry. 47: 260–268.
Thurman D. 2001. The epidemiology and economics of head trauma. In: Head Trauma: Basic, Preclinical, and Clinical Directions. Miller LP, Hayes RL, Newcomb JK, eds. New York: John Wiley & Sons, Inc. Pp. 327–348.
Thurman DJ, Alverson C, Guerrero J, Sniezek J. 1999. Traumatic brain injury in the United States: A public health perspective. Journal of Head Trauma and Rehabilitation. 14(6): 602–615.
Torsney K. 2004. The need to explore the prevalence and treatment of acquired brain injury among persons with serious and persistent mental illnesses. Psychiatric Rehabilitation. 28(1): 75–77.
van Baalen B, Odding E, Maas AI, Ribbers GM, Bergen MP, Stam HJ. 2003. Traumatic brain injury: Classification of initial severity and determination of functional outcome. Disability & Rehabilitation. 25(1): 9–18.
Vanderploeg RD, Curtiss G, Duchnick JJ, Luis CA. 2003. Demographic, medical, and psychiatric factors in work and marital status after mild head injury. Journal of Head Trauma Rehabilitation. 18(2): 148–163.
Vandiver V, Johnson J, Christogero-Snider C. 2003. Supporting employment for adults with acquired brain injury. Journal of Head Trauma Rehabilitation. 18(5): 457.
Walker R, Hiller M, Staton M, Leukefeld CG. 2003. Head injury among drug abusers: an indicator of co-occurring problems. Journal of Psychoactive Drugs. 35(3): 343–353.
Wehman P, Targett P, West M, Kregel J. 2005. Productive work and employment for persons with traumatic brain injury. Journal of Head Trauma Rehabilitation. 20(2): 115–127.
Whiteneck GG, Gerhart KA, Cusick CP. 2004a. Identifying environmental factors that influence the outcomes of people with traumatic brain injury. Journal of Head Trauma Rehabilitation. 19(3): 191–204.
Whiteneck G, Brooks CA, Mellick D, Harrison-Felix C, Terrill MS, Noble K. 2004b. Population-based estimates of outcomes after hospitalization for traumatic brain injury in Colorado. Archives of Physical Medicine & Rehabilitation. 85(Supplement 2): 73–81.
Ylvisaker M, Adelson D, Braga L. 2005. Rehabilitation and ongoing support after pediatric TBI: Twenty years of progress. Journal of Head Trauma Rehabilitation. 20(1): 95–109.

























