8
Enhancing the Emergency Care Research Base
Emergency care is a broad field of inquiry involving many disciplines and cross-cutting themes. Unlike many other areas of medical research, which tend to be defined by organ systems or types of conditions, emergency care is uniquely defined by the urgency and location of treatment. The emergency care research field has spawned multiple branches, generally defined by specialty or research discipline, that have developed distinct but overlapping identities. The field also extends into disciplines well outside the traditional scope of medical research. Each branch includes basic science, clinical research, and health services research activities.
The fact that emergency care research defies easy description has been proven to be one of the principal challenges facing the field as it seeks its niche in the medical research and funding establishment. Figure 8-1 is an attempt to depict the scope of the field and necessarily is an oversimplification; the lines demarcating the three branches tend to suggest stronger distinctions than actually exist.
The first branch, emergency medicine research, is defined by time and place. It addresses principally conditions and interventions common to prehospital emergency medical services (EMS) and hospital emergency department (ED) settings, and its focus is on the acute management of patients. The research is conducted by emergency physicians, often in collaboration with specialists in other fields, such as pediatrics or cardiology. Emergency care research also extends significantly into prevention.
Trauma research is a parallel field of study that is also defined by time and place. It deals principally with the acute management of patients with traumatic injuries. Like emergency medicine research, it is concerned with
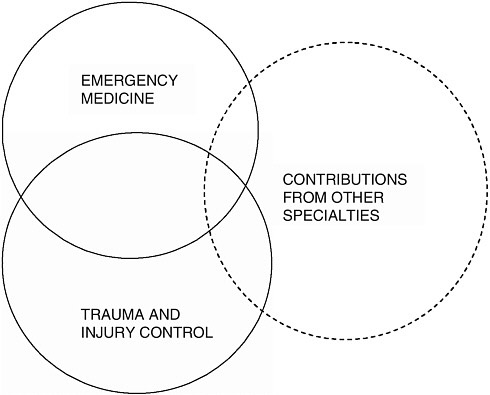
FIGURE 8-1 The scope of emergency care research.
the care of these patients in the prehospital and hospital settings; however, it reaches further into the inpatient setting, particularly the intensive care unit (ICU) and surgical departments, and deals with critical care and the operative management of trauma patients. In addition to trauma surgeons, the research involves specialists in critical care and anesthesiology, as well as collaborators in organ and disease specialties such as neurology and orthopedics. A significant focus of trauma research is service delivery and the effectiveness of trauma care systems.
The injury control field can be thought of as an arm of trauma research that has developed a distinct or rather several distinct areas of focus. It is concerned principally with the prevention of injury, but also overlaps significantly with the acute management of injury and has an additional focus on long-term rehabilitation following traumatic injury. It is one of the most interdisciplinary fields in all of medicine, involving the collaboration
of trauma surgeons, numerous medical specialties, engineers, behavioral scientists, and epidemiologists, to name but a few.
The third branch represents many other specialties—disease-, organ system–, and population-based—that lack a direct link to the emergency care setting but either independently or through collaboration with emergency medicine researchers make research contributions that impact emergency care. A significant amount of the research effort in both emergency and trauma care involves translation of findings from these fields into practice in emergency care settings. There has also been substantial research in emergency care that has flowed back to the specialties.
Finally, nursing research is a growing field that spans all three branches. Its principal focus is the clinical management of patients in all settings.
This chapter describes the development and current status of emergency medicine and trauma and injury research, the branches most germane to the present study, with reference to the other specialties as appropriate. The focus is on hospital-based, adult emergency care research; pediatric and prehospital EMS research are addressed in the two companion reports in the Future of Emergency Care series. The chapter also examines barriers to emergency care research and presents the committee’s recommendations for enhancing the emergency care research enterprise.
EMERGENCY MEDICINE RESEARCH
Emergency care research is vital to the health of Americans. It addresses the care of patients in their most vulnerable moments—when injury or sudden illness strikes. While most Americans have a need for emergency care only rarely, they count on it to be there when needed. Nearly 114 million visits were made to EDs in 2003, and traumatic injury is the leading cause of death among nonelderly adults. In contrast to the vast majority of patient encounters in medicine, the quality and speed of the care that is provided in the relatively brief emergency care encounter can mean the difference between life and death or a prolonged period of disability.
Although emergency medicine and trauma surgery are relatively young specialties, researchers have made important contributions to both basic science and clinical practice that have dramatically improved emergency care and resulted in significant advances in general medicine. Examples include assessment and management of cardiac arrest, including the development and refinement of guidelines for cardiopulmonary resuscitation (CPR) and the pharmacology of resuscitation; understanding and treatment of hemorrhagic shock; electrocardiogram (EKG) analysis of ventricular fibrillation; toxicology and detoxification; injury prevention and control; and uses of diagnostic methods and treatment protocols.
Emergency Care Research Infrastructure and Funding
Because emergency medicine and trauma surgery are young fields, they are not strongly represented in the political infrastructure of the National Institutes of Health (NIH), its various institutes, and its study sections. As a result, scant resources are allocated to advance the science of emergency care, and few training grants are offered to develop researchers who want to focus their work in the field.
A conference held in 1994 highlighted the need to strengthen the academic structure and funding for emergency care research (Josiah Macy, Jr. Foundation, 1995). The report resulting from that conference recommended that academic departments in emergency medicine be increased in number and enhanced, and that the specialty develop a research agenda and a strategic plan for its implementation. In response, the specialty took a number of actions to enhance academic departments and develop the capacity and funding of research in emergency care. In 2003, the American College of Emergency Physicians’ (ACEP) Research Committee reported on progress in emergency medicine research (Pollack et al., 2003). Their findings include the following:
-
Academic departments in emergency medicine more than doubled between 1991 and 2001, growing from 18 to 48 percent of medical schools. These increases occurred disproportionately among higher-ranked medical schools. At the time of the study, 63 percent of medical schools had either an academic department or a residency program in the field, and 44 percent had both.
-
Postresidency fellowships in the field increased from 18 in 1988 to 74 in 2002, although only 12 percent of available fellowships had a primary focus on research.
-
By 1999, 54 investigators had been named as principal investigators (PIs) on grants from NIH, the Centers for Disease Control and Prevention (CDC), the Agency for Healthcare Research and Quality (AHRQ), and others. In 2001 there were 40 active grants with emergency-trained PIs, but there are no data on the number of applications rejected.
-
The Emergency Medicine Foundation, a small specialty-supported foundation administered by ACEP, has provided development awards to 89 investigators at 53 academic institutions. However, more than 50 percent of those awards have gone to 12 individuals at 7 institutions.
A 2005 report of the ACEP Research Committee noted that emergency medicine residency programs had grown rapidly, from 1 such program in 1970 to 81 in 1990 to 132 in 2005. Currently there are 3,909 emergency medicine residents. The number of federally funded emergency medicine investigators has also increased rapidly but remains low—only 87 in 2005
(ACEP Research Committee, 2005). Just 0.05 percent of NIH training grants awarded to medical schools goes to departments of emergency medicine—an average of only $51.66 per graduating resident. In contrast, other medical specialties have much higher levels of support; for example, internal medicine receives approximately $5,000 per graduating resident per year (see Table 8-1).
While the pace and quality of emergency care–related research have improved steadily over the last two decades, further progress is limited by several factors. These include (1) a limited number of adequately trained laboratory, clinical, and health services investigators; (2) poorly defined professional research tracks (Stern, 2001; Lewis, 2004); (3) limited interdisciplinary collaboration and multi-institutional research networks; and (4) funding streams that are poorly geared to the nature of emergency care investigations (ACEP Research Committee, 2005).
Research Training Support
Research training grants and fellowships related to emergency care are funded by a number of sources, including institutions, foundations, and federal agencies. Postgraduate fellowships can be categorized into those that
TABLE 8-1 NIH Funding to Medical School Departments for Training Grants in 2003
|
Field |
No. of Awards |
Dollar Amounts |
Percentage of Total |
Active Residents/ Fellows |
NIH Training Grant Dollars per Resident |
|
Overall |
1,281 |
370,186,331 |
100.00 |
|
|
|
Internal Medicine |
354 |
107,209,870 |
29.00 |
21,351 |
5,021.30 |
|
Pathology |
78 |
28,289,147 |
7.64 |
2,257 |
12,533.96 |
|
Psychiatry |
78 |
18,176,767 |
4.91 |
4,522 |
4,019.63 |
|
Pediatrics |
81 |
17,547,387 |
4.74 |
7,773 |
2,257.48 |
|
Surgery |
41 |
8,302,760 |
2.24 |
7,623 |
1,089.17 |
|
Neurology |
24 |
5,654,160 |
1.53 |
1,339 |
4,222.67 |
|
Ophthalmology |
16 |
3,346,324 |
0.90 |
1,260 |
2,655.81 |
|
Anesthesiology |
10 |
2,640,197 |
0.71 |
4,719 |
559.48 |
|
Obstetrics/Gynecology |
13 |
2,324,220 |
0.63 |
4,681 |
496.52 |
|
Dermatology |
13 |
2,183,009 |
0.59 |
994 |
2,196.19 |
|
Otolaryngology |
11 |
1,989,202 |
0.54 |
1,071 |
1,857.33 |
|
Urology |
9 |
1,138,828 |
0.31 |
1,038 |
1,097.14 |
|
Neurosurgery |
2 |
599,544 |
0.16 |
775 |
773.61 |
|
Orthopedics |
4 |
390,055 |
0.11 |
3,024 |
128.99 |
|
Emergency Medicine |
1 |
198,012 |
0.05 |
3,909 |
50.66 |
|
Family Medicine |
0 |
0 |
0.00 |
9,529 |
0.00 |
|
SOURCE: ACEP Research Committee, 2005. |
|||||
are primarily clinical but include a research component (e.g., EMS, pediatric emergency medicine, toxicology) and those that are dedicated to research training. The former category is often funded by institutional resources. Frequently, patient care provides the financial support for the fellowship, limiting the amount of “protected time” trainees have to develop their research careers. It is generally accepted, however, that unless a research training program includes 2 years of dedicated research training (e.g., greater than 80 percent research time), it is unlikely to result in long-term success in today’s research climate (NIH, 2003). Thus postgraduate fellowship programs that are supported by clinical activity are unlikely to be an effective means of improving the nation’s research capacity in emergency care.
A substantial number of institutions offer dedicated postgraduate research fellowships, which may be funded using institutional resources, may be contingent on the individual applicant’s securing extramural funding, or may be funded by extramural support to the institution through an Institutional (T32) Grant. Currently, there is only one emergency care–related institutional training program supported by the T32 mechanism, and its focus is pediatric emergency medicine. No Institutional or Career Development (K12) Grant has ever been awarded directly to an academic department of emergency medicine (ACEP Research Committee, 2005), although some departments may have submitted grant applications under the name of the academic medical center hospital rather than the medical school.
The primary foundation-based supporters of emergency care research training are the Emergency Medicine Foundation (EMF), affiliated with ACEP, and the Society for Academic Emergency Medicine (SAEM). Both entities fund individual research fellowships for trainees who have completed residency training in emergency medicine, or in the case of SAEM, in pediatrics with the intent to pursue pediatric emergency medicine fellowship training. Currently, the EMF fellowship grants supply only a single year of training, although 2-year fellowships may be added. The SAEM individual research training grants provide 2 years of training. SAEM also funds an institutional training grant, through which 2 years of support is provided to the institution with the intent that the institution will then recruit an appropriate trainee. This funding mechanism was explicitly modeled after the T32 mechanism.
A handful of emergency care research trainees have secured individual NIH F32 National Research Service Award (NRSA) fellowship funding. Further, a notable number of emergency care researchers have obtained support for career development and educational activities through the K08 and K23 mechanisms (ACEP Research Committee, 2005).
As detailed in the 2005 ACEP report (ACEP Research Committee, 2005), a substantial proportion of all emergency medicine trainees intend to pursue an academic career, yet paradoxically, the support devoted to
emergency care research and research training is very low, especially compared with other medical specialties. While existing foundation support has modestly increased the number of well-trained emergency care investigators, substantial growth in the total available research training support will be required to expand the emergency care research capability nationwide.
Many have noted a concerning lack of young investigators, both in industry-sponsored clinical trials and among the ranks of federally supported clinical investigators. Sung and colleagues (2003, p. 1282) reported that “8 percent of principal investigators conducting industry-sponsored clinical trials are younger than forty years,” and “less than 4 percent of competing research grants awarded by the National Institutes of Health (NIH) in 2001 were awarded to investigators aged thirty-five years or younger” (see also Zisson, 2001; Goldman and Marshall, 2002). By contrast, investigators in emergency care specialties, including emergency medicine, pediatric emergency medicine, and EMS, are characterized by their relative youth. Physician-scientists in these fields are generally recently trained, and with the receipt of additional clinical research training may be well positioned to initiate productive, long-term clinical research careers. In its 2005 report, ACEP called for the development of 100 new investigators within 10 years through the NIH Mentored Career Development Award Program (K12) at an estimated cost of $50 million over 10 years. Sung and colleagues (2003, p. 1283) recommended that, as part of a strategy to increase the number of well-trained clinical investigators, academic health centers and research sponsors, including federal sponsors, “increase opportunities for training in all areas of clinical research, including health services and outcomes research, clinical trials, and research synthesis, and develop a mechanism for collecting longitudinal data on training program outcomes.”
Similarly, many have noted the lack of a sufficient pool of well-trained laboratory and patient-oriented investigators in emergency care. Nevertheless, emergency medicine investigators have made important contributions in laboratory investigations of shock, ischemia–reperfusion, cellular injury, early biomarkers for cardiac ischemia, cerebral resuscitation, and neuro-protection. For many years, medical training in the specialties of emergency medicine, pediatric emergency medicine, and trauma surgery were heavily focused on the development of clinical skills, with little formal training in research methodology. As noted by Stern (2001), formal fellowship training is now a well-recognized requirement for those embarking on a successful long-term research-based academic career. To address the shortage of training for new investigators in emergency medicine, the committee recommends that academic medical centers support emergency and trauma care research by providing research time and adequate facilities for promising emergency care and trauma investigators, and by strongly considering the establishment of autonomous departments of emergency medicine (8.1).
Research Funding
A 1994 review of nonmilitary research articles published in three emergency medicine journals revealed that the majority of articles did not list a source of funding. This is in contrast to other specialties, in which the majority of published research was funded. The literature review also found that funded studies published in the emergency medicine literature were less likely to be federally supported and more likely to be supported by industrial sources relative to studies published in the literature of other specialties (Wright and Wrenn, 1994). Although these results may be dated, federal funding, and in particular NIH funding, remains difficult for emergency medicine researchers to obtain (Morris and Manning, 2004). The limited amount of funding available for emergency care research extends across a wide range of institutes, programs, and sponsors, although NIH remains the key sponsor.
As noted earlier, because of the cross-cutting nature of emergency care, the field overlaps with many other medical disciplines. This makes it difficult to establish a unique funding home for emergency care within NIH and other research sponsors that tend to have a traditional orientation based on diseases or body parts. On the other hand, the cross-cutting nature of emergency care exposes it to many opportunities for collaboration with other research specialties and disciplines, and collaborating with established researchers in other fields may be a good way for emergency care investigators to obtain or expand their research funding.
National Institutes of Health NIH includes 20 institutes, seven centers, and four program offices contained within the Office of the Director (OD). NIH is the largest single source of support for biomedical research in the world, with a budget of over $27 billion in 2004 (IOM, 2004). All institutes but only some of the centers (e.g., the Center on Scientific Review [CSR]) provide research funding, while several other centers provide general support. All institutes and four of the centers receive individual congressional appropriations. The NIH institutes are organized into five categories, some by disease (e.g., the National Institute of Neurological Disorders and Stroke [NINDS]), some by organ system (e.g., the National Heart, Lung and Blood Institute [NHLBI]), some by stage of life (e.g., the National Institute of Child Health and Human Development [NICHD]), some by scientific discipline (e.g., the National Human Genome Research Institute [NHGRI]), and some by profession or technology (e.g., the National Institute of Nursing Research [NINR] and the National Institute of Biomedical Imaging and Bioengineering [NIBIB]) (IOM, 2003). None of the current institutes or centers is defined either by the site of care or the timing or urgency of care—defining characteristics of emergency care research. Perhaps for this
reason, NIH does not have an institute or center focused specifically on emergency services. Thus, many important emergency care clinical questions extend beyond the domains of single NIH institutes or centers. While both a 2003 Institute of Medicine (IOM) report (IOM, 2003) and the NIH Roadmap Initiative (Zerhouni, 2003) emphasized the importance of stimulating and funding trans-NIH research, and emergency care research questions naturally span the domains of multiple institutes and centers, the lack of attention to emergency care has not been effectively addressed. In fact, the term “emergency care” does not appear in the NIH Roadmap.
Other federal agencies Many other federal agencies provide small amounts of research funding in emergency care. AHRQ, for example, like NIH, does not have a dedicated funding stream for research on emergency services. However, it does have a long track record of funding grants in emergency care, such as a study on the effects of cost sharing on use of the ED, evaluation of technologies for identifying acute cardiac ischemia in EDs, and measurement of ED crowding (AHRQ, 2004).
The Health Resources and Services Administration (HRSA), through its Emergency Medical Services for Children (EMS-C) Program, sponsors the Pediatric Emergency Care Applied Research Network (PECARN), the first federally funded multi-institutional network for research in pediatric emergency medicine. The EMS-C Program also sponsors the National EMS Data Analysis Resource Center (NEDARC), which was established in 1995 to help states collect and analyze data on pediatric EMS systems and to populate the pediatric trauma registry. The HRSA Trauma-EMS Systems Program and the Office of Rural Health Policy also support research efforts in emergency care.
CDC’s National Center for Injury Prevention and Control (NCIPC) sponsors investigations in injury prevention and control and recently developed an Acute Care Research Agenda for the Future. NCIPC/CDC and the Consumer Product Safety Commission cosponsor the National Electronic Injury Surveillance System (NEISS), a longitudinal database with information from 100 hospital EDs on consumer product–related injuries, and since 2000 on all injuries. NCIPC also sponsors the Data Elements for Emergency Department Systems (DEEDS) project, a national effort to develop uniform specifications for data entered in ED patient records.
The Office of EMS in the National Highway Traffic Safety Administration (NHTSA) plays a lead role in coordinating activities related to EMS system development and research. Together with HRSA, NHTSA sponsored the development of the National Emergency Medical Services Research Agenda (NHTSA, 2001). The Office currently funds two key research initiatives: the Emergency Medical Services Outcomes Project, a study to develop metrics for use in EMS-related outcomes research, and the Emer-
gency Medical Services Cost Analysis Project, a study to develop metrics for assessing the costs and benefits of EMS. NHTSA and HRSA cosponsor the National EMS Information System (NEMSIS), a national database on EMS systems and outcomes that is operated by the National Association of State EMS Directors. NHTSA’s Office of Human-Centered Research sponsors the Crash Injury Research and Engineering Network (CIREN), a network of level I trauma centers that collect and share detailed research data on automobile crashes, injuries, and outcomes.
Although not research funding per se, funds are being provided by NHTSA’s Office of EMS for the National EMS Scope of Practice Model project, a joint initiative of the National Association of State EMS Directors and the National Council of State EMS Training Coordinators. The Longitudinal Emergency Medical Technician Attribute and Demographics Study (LEADS) is a NHTSA-funded project of the National Registry of Emergency Medical Technicians. An annual LEADS survey collects information on the EMS workforce.
The Centers for Medicare and Medicaid Services (CMS), the Department of Homeland Security, and the Department of Veterans’ Affairs also provide small amounts of funding related to emergency care research.
Private Funders
SAEM and EMF both provide investigator training grants, as described earlier. EMF awarded 18 grants in 2004–2005 totaling almost $500,000 (Pollack and Cairns, 1999; ACEP, 2005). The Robert Wood Johnson Foundation funded the Urgent Matters project, which provided grants to 10 hospitals and their communities for evaluating approaches to reducing crowding and improving patient flow. A small number of emergency medicine researchers received research training through The Robert Wood Johnson Clinical Scholars program. The National Emergency Medicine Association also provides research grants in trauma and emergency care.
Future Directions in Emergency Care Research
Pressing gaps remain in our understanding of emergency care in all three research areas: basic science, clinical research, and health services research. There have been several recent attempts to identify research priorities and key opportunities in emergency care (Aghababian et al., 1996; Maio et al., 1999; Seidel et al., 1999; Becker et al., 2002). The EMS Research Agenda for the Future project (Sayre et al., 2005) identified priority issues for targeted research efforts, including asthma, acute cardiac ischemia, circulatory shock, major injury, pain, acute stroke, and traumatic brain injury, as well as education and system design issues. Critical research questions identified
by these groups cut across basic science, clinical research, and health services research. Some fertile topics for research in each area are described below.
Basic Science
Because emergency medicine is defined by time and place, rather than body part or disease process, research in the field is often mischaracterized as being strictly translational in nature. But emergency medicine requires both basic discoveries and translation of those discoveries to the clinical setting. Basic research projects involving emergency medicine investigators focus on the following:
-
Characterization of the molecular events that cause delayed neuronal death after brain ischemia and other studies on neuronal injury (multiple NINDS grants).
-
The pathophysiology of carbon monoxide poisoning and mechanisms for the benefit of hyperbaric oxygen therapy (multiple NIH grants).
-
Understanding of the events that occur following ischemia–reperfusion injury from cardiac arrest, using animal models, cardiomyocyte cell culture models, and methods for inducing hypothermia for treatment of patients following cardiac arrest (NIH).
-
The pathophysiology of acute lung injury and acute respiratory failure (NHLBI).
-
Means of minimizing the risk of secondary ischemic brain injury during limited resuscitation from hemorrhagic shock and traumatic brain injury (Department of Defense [DoD]).
-
Identification of effective neuroprotective agents to limit tissue loss and enhance recovery following acute traumatic brain injury or stroke (NINDS, CDC).
-
Pathophysiology and treatment of traumatic spinal cord injury (NIH).
-
Hypothermia and gene expression following cardiac arrest (NIH).
-
Pathophysiological processes that contribute to the destruction of articular cartilage in a variety of disorders, including an evaluation of immunoprobes for lubricin from human synovial fluid (NIH).
-
Understanding of the human genomic and proteomic response to injury and injury recovery.
Clinical Research
Because of the wide range of patients, diseases, and interventions seen by physicians in emergency practice, these practitioners have a unique window on the state of treatment options available, including their shortcomings.
Thus emergency physicians have both the motivation and opportunity for focused efforts aimed at translating research into better modes of treatment. As a result, clinical research represents the most active area of emergency care research. Examples include the following:
-
The efficacy, safety, and dosages of medications for infants, children, adolescents, adults, and the elderly.
-
Definition of an effective and practical diagnostic and risk-stratification strategy for patients with possible pulmonary embolism (Kline and Wells, 2003; Brown et al., 2005; Courtney and Kline, 2005; Kline et al., 2004, 2005).
-
Development of evidenced-based protocols for common pediatric conditions (e.g., fever).
-
Evaluation of the pharmacokinetics and efficacy of promising clinical therapies for treatment of acute traumatic brain injury (Wright et al., 2005).
-
Development and testing of new therapies and strategies for resuscitation of the multiply injured trauma patient (Bickell et al., 1992; Coimbra et al., 1997; Angle et al., 1998; Sloan et al., 1999a; Cooper, 2004).
-
Evidence-based criteria for determining which patients with community-acquired pneumonia require hospitalization (current national guidelines are based largely on a risk stratification model created from data that did not include manipulation of the decision to admit (Fine et al., 1997).
-
Definition and testing of strategies for determining which patients with possible acute coronary syndromes require hospitalization and for those who do, definition of the appropriate level of care.
-
Identification and testing of new strategies for the prevention of secondary brain injury after both traumatic and ischemic insults (The Hypothermia after Cardiac Arrest Study Group, 1921; Stern et al., 2000; Neumar, 2000; Bernard et al., 2002; Nolan et al., 2003; Abella et al., 2004).
-
Use of blood substitutes by paramedics.
-
Evaluation of simplified methods of CPR instruction (Kellermann et al., 1989; Eisenberg et al., 1995; Todd et al., 1998, 1999).
-
Assessment of the potentially deleterious effects of hyperventilation on successful resuscitation following cardiac arrest (Auf der Heide et al., 2004).
Health Services Research
Emergency medicine by definition requires timely and efficient approaches to the delivery of services. The impact of the organization and mode of delivery has long been recognized as having a major impact on the
quality of care and outcomes—first codified in Crowley’s “golden hour” and Pantridge’s cardiac care in the field, and reinforced through military and civilian experience. But the organization and delivery of services is perhaps the weakest link in the emergency care evidence base. Even accepted doctrine, such as the value of paramedics in the field, has recently been overturned. This, then, represents a formative and essential area for research. Some of the key research questions in service delivery include the following:
-
The impact of bottlenecks in different hospital units (e.g., ICU, telemetry) on ED crowding and patient flow.
-
The effectiveness of queuing theory in smoothing patient volume to alleviate crowding, boarding, and diversion.
-
The effect of timeliness of out-of-hospital response, stratified by etiology and/or severity of injury.
-
Identification of which components of trauma systems impact outcomes and cost-effectiveness.
-
The causes of and solutions for missed diagnoses in the ED.
-
Validation of the use of prehospital 12-lead electrocardiography to direct patients with acute ST-elevation myocardial infarction (STEMI) to interventional cardiac centers.
-
The impact of medical direction in EMS systems.
-
Use of prehospital electrocardiography to identify and directly transport patients to a cardiac catheterization laboratory for percutaneous coronary intervention.
-
The impact of prearrival information from dispatchers about the condition of patients with respect to both arrival at the hospital and longterm outcome.
-
Evaluation of safe alternatives to endotracheal intubation for securing the airway in prehospital and ED settings.
-
The feasibility and cost-effectiveness of implementing point-of-care HIV testing of high-risk patients in the ED.
-
Development of a practical testing technology for evaluating mild traumatic brain injury and other causes of cognitive impairment in prehospital, sports, and ED settings.
-
Use of computers to screen ED patients for a variety of health risk behaviors, including intimate partner violence, depression, substance abuse, and suicide.
This and the above lists of basic science and translational research are not meant to be all-inclusive or even representative of current research challenges or priorities, but merely to suggest the breadth of important research questions in need of attention.
Multicenter Research Collaborations
Many of the important successes in emergency care research have been based on the establishment of large-scale multicenter research collaborations. Such collaborations enable researchers to assemble sufficiently large datasets to establish robust research findings.
There are a number of examples of successful studies by multicenter research collaborations. The National Emergency X-Radiography Utilization Study (NEXUS), for example, has investigated the use of cervical spine radiography in patients suffering blunt trauma (Hoffman et al., 1998, 2000). The Multicenter Airway Research Collaboration (MARC)/Emergency Medicine Network (EMNet) is studying respiratory disease management strategies in the ED (EMNet, 2005). The EMERGEncy ID Net collaboration provides important information on the characteristics and management of infectious diseases in the ED (Cydulka et al., 2003; Kim et al., 2004), as well as sentinel detection of emerging infectious diseases (Talan et al., 1998, 1999, 2003; Moran et al., 2000). The Resuscitation Outcomes Consortium network, sponsored by NIH and DoD, is addressing prehospital-based trauma and cardiac arrest resuscitation in North America. A number of emergency medicine departments are participating in or heading programs related to this endeavor. The Inflammation and the Host-Response to Injury study is a National Institute of General Medical Sciences (NIGMS) Glue Grant1 that has joined clinical level I trauma centers, genomic centers, and proteomic high-throughput centers in a multi-institutional, multidisciplinary attempt to explore the genomic, proteomic, and phenotypic host response to the stress of severe injury (Calvano et al., 2005). Finally, PECARN, sponsored by HRSA’s EMS-C program, focuses on prevention and management of acute illnesses and injuries in children through four research nodes (PECARN, 2003, 2005). The publication records of these collaborative efforts, as well as the impact of those publications on clinical care, illustrate the power of such research collaborations to address pressing clinical questions, as well as the ability of the emergency care research community to organize and conduct large-scale clinical research endeavors.
TRAUMA AND INJURY CONTROL RESEARCH
It is difficult to characterize the field of trauma and injury control research. For one thing, the field has expanded dramatically in scope from its early focus on treatment of injuries. It has become increasingly interdisciplinary and now includes investigators from a broad range of fields, such as
engineering, epidemiology, behavioral sciences, biomechanics, criminology, molecular biology, and human factors research.
While trauma and injury are, by and large, identical or at least overlapping, the terms do suggest some differences in focus, type of investigator, and setting. Historically, trauma research was focused clinically on treatment of injury and was strongly influenced by advances in trauma treatment gleaned from battlefield experiences. Injury research can be viewed as a newer endeavor, and one that has branched out in new directions. But even here the distinctions are nuanced rather than clear cut. The modern fields of trauma and injury research began to take shape in the 1960s as a result of the increasing number of highway deaths. The National Academy of Sciences/National Research Council (NAS/NRC) report Accidental Death and Disability (NAS and NRC, 1966) was followed by a burst of regulatory activity, including passage of the Highway Safety Act; establishment of NHTSA, the Occupational Safety and Health Administration, and the Consumer Product Safety Commission; and the founding of the American Trauma Society. These events collectively signaled a new national commitment to reducing death and disability due to injury.
The new field of injury science was based on the recognition that patterns of injury could be determined with the epidemiological tools of public health. William Haddon, a public health physician, set forth a scientific paradigm for analyzing injury based on the interaction between human and environmental factors (Haddon, 1968). The 1985 NRC/IOM report Injury in America (NRC and IOM, 1985) presented the idea of injury prevention and control as a separate discipline. It proposed the establishment of an injury center at CDC, which led to passage of the Injury Control Act of 1990. As a result of this legislation, the Division of Injury Epidemiology and Control was elevated to NCIPC. NCIPC’s focus on nonoccupational injuries was designed to complement a new center at NIH focused on occupational injuries—the National Institute on Occupational Safety and Health (NIOSH). While establishing a science base in injury prevention and surveillance, the development of these centers represented a divergence from trauma research, which remained more focused on treatment and service delivery.
The Trauma Field
The U.S. military experience during the Korean and Vietnam wars provided evidence that an organized health system with medical capabilities could improve chances of survival from trauma (GAO, 1991). Physicians returning from the war tried to apply the advances made and lessons learned during the war to civilian life. Through the availability of helicopters, the time required to evacuate the wounded from the battlefield was cut dra-
matically, resulting in decreased mortality rates. Other advances during the period include the availability of whole blood, well-organized medical teams, well-equipped forward hospitals, and more effective management of medical resources.
Along with advances made during the Korean and Vietnam wars, several medical and technology advances during this period coincided with the development of trauma centers. In 1956, Drs. Elan and Safar developed mouth-to-mouth resuscitation. In 1959, researchers at Johns Hopkins developed the first portable defibrillator and perfected CPR; the first out-of-hospital defibrillation occurred in 1969. These advances provided a means to stabilize victims, thereby making it possible for more critically ill patients to arrive at the hospital for care. During this period, prehospital EMS became more sophisticated, while at the same time trauma centers and systems were developed and formalized.
The American College of Surgeons Committee on Trauma (ACS COT) has also made significant contributions and played a major leadership role in the development of trauma centers. In 1976 ACS COT first published Optimal Hospital Resources for Care of the Seriously Injured (ACS COT, 1976). This document, updated most recently in 1999, identified the key characteristics for the categorization of hospitals as trauma centers. In 1987 ACS COT initiated an external review process for trauma centers. ACS COT also recently published Consultation for Trauma Systems, which provides guidelines for evaluating trauma system development (ACS, 1987).
In 1981 a seminal article, “Regionalization of Trauma Care,” outlined the key elements of the modern trauma system (Trunkey, 1981). The Major Trauma Outcome Study was undertaken in 1982 to improve scoring systems for trauma centers, establish national outcome data, and provide objective evaluations of quality assurance and outcomes for trauma care; by 1989, data on 170,000 patients from more than 150 institutions had been recorded (IOM, 1999). In addition, in 1986 ACEP published Guidelines for Trauma Care Systems, addressing prehospital care (ACEP, 1986).
A significant body of research has focused on the effectiveness of trauma systems. In 1998 the Skamania Conference was convened to review the medical evidence on trauma systems.2 The conference called for renewed federal funding for trauma system development and the drafting of a visionary document on the subject.
Trauma research has also focused on injury scales/scoring systems, leading to a succession of refinements to the precision and usefulness of these scales (e.g., the Abbreviated Injury Scale, the Injury Severity Score, the Anatomic Profile, the New Injury Severity Score, the Glasgow Coma Scale,
and the Revised Trauma Score). These scales are important for standardizing the measurement of injury and have multiple applications, including triage, diagnosis, and research.
Injury Control
The injury control field is focused less on treatment and more on surveillance and prevention. It links researchers in public health, medicine, and engineering and includes many disciplines. Systematic collection of injury data through such databases as NEISS, the National Trauma Data Bank, the Fatality Analysis Reporting System (FARS), and state trauma registries is critical to gathering sufficient observations to permit meaningful research. Most states have trauma registries, but the data elements are variable and generally not linked with one another. The American College of Surgeons (ACS) established the National Trauma Data Bank (NTDB) as a voluntary repository of trauma records. In 2005 the NTDB contained almost 1.5 million records from 565 trauma centers in 45 states, U.S. territories, and the District of Columbia. It represents 70 percent of level I and 53 percent of level II trauma centers (ACS, 2005).
Injury research has led to a wide range of prevention successes. By far the most important of these successes have occurred in prevention and control of motor vehicle crash injuries. Others include childproof containers; mandated use of smoke alarms; laws requiring motorcycle and bicycle helmets, sports pads, and mouth guards; and safe refrigerator disposal to prevent suffocation. Successes related to motor vehicles range from seat-belts, airbags, and child safety restraint systems to graduated driver’s license programs and improvements in highway design. An important current initiative is CIREN, whose mission is “to improve the prevention, treatment and rehabilitation of motor vehicle crash injuries to reduce deaths, disabilities, and human and economic costs” (NHTSA and CIREN Center Staffs, 2003, p. 1). CIREN researchers have had a significant impact in improving safety research, automobile safety, and emergency medical care.
Much of this research is concerned with the field of injury biomechanics—the study of physical and physiological responses to both penetrating and nonpenetrating impacts. Examples of the disciplines involved in this branch of study are robotics, physical therapy, orthopedics, physical and sports medicine, prosthetics, orthotics, and tissue engineering.
One area that has been highly underresearched relative to the magnitude of its impact is primary and secondary prevention of falls. Falls are a significant problem among toddlers and the elderly and are now the most common cause of traumatic brain injuries among the latter. With the aging of the population, this problem will only grow in importance over time (Wadman et al., 2003).
The injury field has focused largely on unintentional injuries but has begun to address intentional injuries—for example, those caused by firearms and suicide. Whereas the trauma field has long been interested in gunshot injuries from a treatment perspective, the injury field has looked at such injuries from the prevention perspective. Although widely accepted today, this new area of focus led to substantial debate about priorities in the field. Prevention of suicide and violence extends into the realms of behavioral science, sociology, and even economics. Indeed, the scope of injury research now encompasses many disciplines—epidemiology, behavioral sciences, biomedical science, biomechanics, criminology, sociology, engineering, law, molecular biology, and others.
Research Infrastructure and Funding
The majority of support for trauma and injury research comes from NIH and CDC, with limited support being provided by NHTSA, HRSA, AHRQ, DoD, and others.
National Institutes of Health
By far the most important source of funding for trauma research is NIH. The 1966 report Accidental Death and Disability recommended that NIH establish an Institute for Trauma. While this recommendation was never implemented, trauma research at NIH has grown. NIH convened a task force to study the needs and gaps in trauma research and produced the Report of the Task Force on Trauma Research (NIH, 1994). This report recommended doubling funding for trauma research centers, but sufficient funding was never appropriated to carry out this recommendation. Relevant areas of trauma and injury research funding are spread across multiple NIH institutes and centers—for example, NIGMS Research Centers in Trauma, Burn, and Perioperative Injury; the National Heart Attack Alert Program within NHLBI; and NINDS, which includes a Program on Trauma, Regeneration and Pain focused almost exclusively on neurotrauma. Finally, NICHD has a National Center for Medical Rehabilitation Research, which includes both injury-related and non-injury-related rehabilitation.
Total NIH support for research on trauma and injury is very limited in relation to the importance of both in terms of mortality, disability, and costs. The costs of trauma approach 10 percent of health care spending, and injury is the number one killer of nonelderly adults (IOM, 1999). Traumatic injury has surpassed heart disease as the most expensive category of medical treatment, resulting in $71.6 billion dollars in expenditures per year (AHRQ, 2006). In 1998, injury was the third-leading cause of death
in terms of years of potential life lost (YPLL), yet NIH injury research was collectively funded at a level of less than $200 million. In terms of YPLL, trauma received only $.10, compared with $3.51 for HIV/AIDS and $1.65 for cancer (IOM, 1999).
Just as important, with research spread across programs, there is little opportunity for coordination or the development of comprehensive research centers. The 1999 IOM report Reducing the Burden of Injury: Advancing Prevention and Treatment stated that “NIH lacks a focal point and a mechanism for coordinating disparate injury research projects and programs” (IOM, 1999, p. 229). The report recommended expanding the program within NIGMS and elevating the Trauma and Burn Program to a division. In addition, there is scant support for the development of investigators in the field.
Centers for Disease Control and Prevention
As noted earlier, the NRC/IOM report Injury in America: A Continuing Public Health Problem (NRC and IOM, 1985) led to the Injury Control Act of 1990, which elevated CDC’s Division of Injury Epidemiology and Control to NCIPC. NCIPC includes three divisions: Unintentional Injury Prevention, Violence Prevention, and Injury and Disability Outcomes. It operates much like an NIH center, with a focus on extramural research grants, plus cooperative agreements with states. NCIPC has nurtured biomechanics research and funded comprehensive Injury Control Research Centers. Some have argued that relative to NIH, CDC has funding limitations and lacks the infrastructure to pursue a strong basic and clinical research agenda (IOM, 1999). NCIPC supports no investigator training grants, but NIOSH does have a small number of pre- and postdoctoral training grants in occupational injury prevention. The IOM recommended that the center develop interdisciplinary training in epidemiology, biostatistics, biomechanics, and behavioral sciences through collaborations with NHTSA, HRSA, NIOSH, and others (IOM, 1999).
Other Agencies
With the exception of the NHTSA-supported CIREN program, described above, there is only limited support for trauma and injury research in other agencies. Most research in system design is sponsored by NCIPC and AHRQ, but health services research is not well funded in general, and trauma and injury represent a very small component of that research. HRSA’s EMS and Trauma Systems Program supported the development and evaluation of trauma systems until it was defunded for fiscal year 2006.
Current and Future Research Directions
Current directions in trauma and injury research have been the subject of several recent reports. The NIH and DoD Working Group on Trauma Research Program was convened in 2003 and developed a report that identified and summarized current trauma research priorities (Hoyt et al., 2004). It identified priorities in three key areas: basic science, clinical trials, and clinical research. In basic sciences, it identified cellular injury (immune response following injury), bleeding and thrombosis, central nervous system injuries, and multiple organ failure. Areas in need of clinical trials to establish efficacy included airway management, fluid resuscitation, therapies for controlling bleeding, adjuvants to control postinjury immune response, and body temperature management. The three top areas for clinical research were physiological monitoring, automated clinical data collection, and development of large-scale longitudinal datasets for research. The report cited the continuing lack of an organized infrastructure as an impediment to progress in resolving a number of key issues and addressed ways to build this infrastructure for resuscitation research, including development of a consistent informed consent process (for multicenter trials), formation of an animal model consortium, increased use of multicenter trials, centralized tissue banks, and standardized data collection and analysis.
In 2002, NCIPC developed a research agenda that addressed the following broad injury categories: injuries at home, sports, transportation, domestic violence, suicide, youth violence, acute care, disability, and rehabilitation. The agenda also identified four cross-cutting research priorities: translating research into programs and policies, improving parenting and controlling alcohol abuse, identifying the costs and consequences of injury, and building the research infrastructure.
In 2005, CDC updated the acute care chapter of the 2002 agenda. This revision identified seven research priorities:
-
Better translation of findings into patient care through guidelines
-
Evidence-based protocols
-
How trauma systems improve care
-
How mass casualty impacts acute care
-
Clinical prevention
-
Psychosocial impact of injury
-
Development of short- and long-term outcome measures
The report also called for enhancing research capacity through four actions: the development of acute care injury research networks; the conduct of research by mining current and future databases; the development of new investigators though training grants; and reductions in institutional barriers to research, such as Emergency Medical Treatment and Active Labor Act
(EMTALA) regulations. Finally, the report noted the need for more research on morbidity and disability outcomes (National Center for Injury Prevention and Control, 2005).
BARRIERS TO EMERGENCY CARE RESEARCH
There are unique logistical problems associated with conducting emergency care research, such as lack of a coordinating funding structure; the difficulty of establishing informed consent in emergency care situations; and the challenge of linking medical records to reconstruct an episode of care across prehospital, ED, and inpatient settings.
Organization and Funding of Emergency Care Research
Taken as a whole, the emergency care research enterprise has accomplished a great deal. Many of these accomplishments have been made with bootstrap funding and by poorly supported researchers in a disconnected fashion. But the field has reached a level of maturity that requires a new approach. There are well-defined areas of critical inquiry that require a coordinated and well-funded approach. In addition, there is a crucial need for an integrated research effort across disease lines that breaks down departments and requires multidisciplinary approaches to achieve effective translational research. This effort must include a wide range of disciplinary strengths—from epidemiology, pathophysiology, and toxicology to surgery, psychology, and biomechanics—to integrate the wide range of interrelated medical and sociological issues faced by the modern ED. It should be clear that the current uncoordinated approach to organizing and funding emergency and trauma care is ineffective. Therefore, the committee recommends that the Secretary of the Department of Health and Human Services conduct a study to examine the gaps and opportunities in emergency and trauma care research, and recommend a strategy for the optimal organization and funding of the research effort (8.2). This study should include consideration of training of new investigators, development of multicenter research networks, funding of General Clinical Research Centers that specifically include an emergency and trauma care component, involvement of emergency and trauma care researchers in the grant review and research advisory processes, and improved research coordination through a dedicated center or institute (8.2a). Congress and federal agencies involved in emergency and trauma care research (including the Department of Transportation, the Department of Health and Human Services, the Department of Homeland Security, and the Department of Defense) should implement the study’s recommendations (8.2b). This study should encompass the broad range of emergency care research, including emergency medicine, trauma, and injury and basic,
science, clinical research and health services research, and should consider ways to enhance the coordination of emergency care research across topics, disciplines, and agencies.
The inclusion of emergency care researchers on advisory and review committees has special merit in the committee’s view. NIH, for example, uses a wide variety of advisory committees: (1) initial review groups (IRGs, also known as study sections) and special emphasis panels (SEPs), (2) national advisory councils, (3) boards of scientific counselors, and (4) program advisory committees. The IRGs and SEPs perform the first level of peer review, scoring grant applications on technical and scientific merit. The national advisory councils perform a second level of peer review, providing advice to the institute or center both on the funding of individual applications and on more general issues related to the mission and goals of the institute or center. The combined review by the IRGs/SEPs and the national advisory councils is commonly termed the “dual review system” (IOM, 2003). The boards of scientific counselors perform retrospective reviews of intramural research programs and are not discussed further here. The program advisory committees provide input on research programs, future research directions, and the development of extramural research initiatives (IOM, 2003). The vast majority of members of advisory committees are appointed by either the NIH director or the directors of the individual institutes or centers.
Emergency care providers often have a unique perspective on the evaluation and management of specific syndromes and diseases, as they routinely manage the most acute and extreme manifestations of those conditions and must often act decisively with only preliminary clinical information. Thus, emergency care providers can provide important complementary perspectives during the framing of clinical research questions to be addressed by interdisciplinary clinical research teams and during the evaluation of research applications and proposals. These perspectives can be particularly valuable for judging proposals that require the timely recruitment of research subjects in acute care situations and for addressing the logistical challenges of conducting well-controlled clinical research in EDs, trauma centers, and other acute care environments.
General Clinical Research Centers (GCRCs) play a critical role in supporting the clinical research enterprise and serving as a fertile ground for the development and training of young clinical investigators. There are currently 87 GCRCs supported by the National Center for Research Resources, which include both inpatient facilities and ambulatory research clinics associated with academic health centers. These facilities are potentially valuable in providing mentorship to new clinical investigators and junior faculty and in facilitating the enrollment of subjects into clinical research studies. However, GCRCs rarely if ever support clinical research conducted in the ED, much less in out-of-hospital settings. Thus, emergency care investigators have
not had access to an important national resource. One reason is that most GCRCs are funded to conduct scheduled clinical research protocols. They are not well staffed, if staffed at all, to conduct emergency and trauma care research on a full-time basis. While it would be neither feasible nor perhaps prudent to staff all GCRCs in this way, a subset of GCRCs, particularly those based in hospitals with a major ED and level I trauma center, might be encouraged to compete for supplemental awards to support time-critical clinical trials on resuscitation and trauma care research.3
Protection of Human Research Subjects
Federal rules govern the protection of human research subjects, and these rules are enforced by institutional review boards (IRBs). Additional rules to protect the privacy of human subjects are defined in the Privacy Rule of the Health Insurance Portability and Accountability Act (HIPAA). The Office for Human Research Protections (OHRP) within the Department of Health and Human Services is the agency assigned to enforce protections for human subjects. The rules attempt to balance the value of important research against the potential harm to patients resulting from that research. Some have argued that the current rules overly restrict critically important research, particularly in emergency and trauma care (Newgard et al., 2005).
Informed consent requirements represent an important tool for evaluating new and promising therapies in an ethical and publicly transparent manner; however, complying with the requirements can be overly burdensome for emergency care researchers. Patients treated in the emergency care setting frequently have suffered acute, debilitating illnesses or injuries (e.g., cardiac arrest, traumatic brain injury) that affect their capacity to make informed decisions. Thus, potential research subjects often cannot participate in the informed consent process before participating in an interventional clinical trial, even when the investigational therapy offers the prospect of direct benefit to the individual subject. It is also difficult to secure informed consent because care must often be administered immediately. Currently, federal regulations (21 Code of Federal Regulations §50.24) allow a narrow exception to the general requirement for prospective, written informed consent for participation in research studies in the setting of an acute, debilitating illness or injury for which there is no accepted effective therapy (Biros et al., 1995, 1998, 1999; Baren et al., 1999; Sloan et al., 1999b; Lewis et al., 2001). Under this exception, however, it remains difficult to comply with the rules in many situations (NHTSA, 2001). As noted by Mann and colleagues
(2005, p. 1078), “…the logistical application of these ethical standards across institutions or among different research studies remains complex and variable.” Furthermore, state regulations occasionally preempt the federal exception for emergency care research. Active guidance from OHRP to states and individual IRBs could eliminate some of the current obstacles that discourage innovation in treatment approaches of potential benefit to critically ill or injured patients. The committee therefore recommends that states ease their restrictions on informed consent to match federal law (8.3).
Patient Confidentiality Protection
Under new rules established in 2000, all entities participating in federally funded research must obtain a federalwide assurance (FWA) from OHRP. The FWA is a document that ensures the intent of the research organization to comply with applicable federal laws and standards for the protection of human research subjects. The FWA program was intended to streamline the previous, more cumbersome system of single- and multiple-project assurances. But many patients seen in the emergency care setting, either those initially treated by EMS or those treated in community EDs, produce important health care utilization and outcome data that are stored at nonacademic community-based medical facilities. These facilities are unlikely to participate in federally supported research in general and therefore generally do not have an FWA in place. Newgard and colleagues (2005) examined the difficulties associated with effecting FWA agreements with community hospitals to obtain patient-level outcome data from a low-risk EMS study. The study involved an attempt to validate a triage rule for children seriously injured during automobile crashes through a retrospective chart review of cases at 27 pediatric receiving hospitals in Los Angeles County. The researchers were unable to achieve participation from all 27 hospitals, which they attributed to the complexity and risk of the FWA requirement. All 27 hospitals had agreed to participate in an interventional randomized controlled trial of airway management in children several years earlier, before the FWA requirement was in place (Gausche, 2000). To have robust and generalizable results, it is important to include outcome information from the full range of receiving facilities to which the EMS system delivers patients. The NIH Roadmap itself cites the need to remove barriers to collaborative clinical research between community-based providers and academic researchers (Zerhouni, 2003).
In addition, there is limited guidance regarding FWAs in EMS research. In the Field Administration of Stroke Therapy–Magnesium (FAST–MAG) trial, a $16 million NIH grant, investigators had to seek help from OHRP. It was finally decided that hospitals had to either have an FWA, apply to have an FWA, or use an academic medical center as a “parent FWA” and sign
a written agreement with the parent for their IRB to ensure protection of human subjects. Further, all of the 41 EMS agencies in Los Angeles County had to sign an agreement with the Los Angeles EMS Agency to allow the agency to serve as their FWA and oversee protection of human subjects. While for the most part successful, this effort has taken 2 years. To make it possible to conduct important emergency care research on representative populations in the community, the committee recommends that Congress modify Federalwide Assurance Program regulations to allow the acquisition of limited, linked, patient outcomes data without the existence of a Federalwide Assurance Program (8.4). One approach that has been suggested is to allow an experienced academic medical center IRB to serve as a regional IRB for community hospitals within a certain area, at least for minimum-risk research (Christian et al., 2002; Newgard et al., 2005).
SUMMARY OF RECOMMENDATIONS
8.1: Academic medical centers should support emergency and trauma care research by providing research time and adequate facilities for promising emergency care and trauma investigators, and by strongly considering the establishment of autonomous departments of emergency medicine.
8.2: The Secretary of the Department of Health and Human Services should conduct a study to examine the gaps and opportunities in emergency and trauma care research, and recommend a strategy for the optimal organization and funding of the research effort.
8.2a: This study should include consideration of training of new investigators, development of multicenter research networks, funding of General Clinical Research Centers that specifically include an emergency and trauma care component, involvement of emergency and trauma care researchers in the grant review and research advisory processes, and improved research coordination through a dedicated center or institute.
8.2b: Congress and federal agencies involved in emergency and trauma care research (including the Department of Transportation, the Department of Health and Human Services, the Department of Homeland Security, and the Department of Defense) should implement the study’s recommendations.
8.3: States should ease their restrictions on informed consent to match federal law.
8.4: Congress should modify Federalwide Assurance Program regulations to allow the acquisition of limited, linked, patient outcomes data without the existence of a Federalwide Assurance Program.
REFERENCES
Abella BS, Zhao D, Alvarado J, Hamann K, Vanden Hoek TL, Becker LB. 2004. Intra-arrest cooling improves outcomes in a murine cardiac arrest model. Circulation 109(22): 2786–2791.
ACEP (American College of Emergency Physicians). 1986. Guidelines for Trauma Care Systems. Dallas, TX: ACEP.
ACEP. 2005. Emergency Medicine Foundation. [Online]. Available: http://www.acep.org/webportal/Education/EMF/ [accessed August 1, 2005].
ACEP Research Committee. 2005. Report on Emergency Medicine Research. Dallas, TX: ACEP.
ACS (American College of Surgeons). 1987. Consultation for Trauma Systems. Chicago, IL: ACS.
ACS. 2005. National Trauma Data Bank Report 2005, Dataset Version 5.0. Chicago, IL: ACS.
ACS COT (ACS, Committee on Trauma). 1976. Optimal hospital resources for care of the seriously injured. Bulletin of the American College of Surgeons 61:15–22.
Aghababian RV, Barsan WG, Bickell WH, Biros MH, Brown CG, Cairns CB, Callaham ML, Carden DL, Cordell WH, Dart RC, Dronen SH, Garrison HG, Goldfrank LR, Hedges JR, Kelen GD, Kellermann AL, Lewis LM, Lewis RG, Ling LJ, Marx JA, McCabe JB, Sanders AB, Schriger DL, Sklar DP. 1996. Research directions in emergency medicine. American Journal of Emergency Medicine 14(7):681–683.
AHRQ (Agency for Healthcare Research and Quality). 2004. Funding Opportunities. [Online]. Available: http://www.ahrq.gov/fund/funding.htm [accessed January 22, 2004].
AHRQ. 2006. Costs of Treating Trauma Disorders Now Comparable to Medical Expenses for Heart Disease. [Online]. Available: http://www.ahrq.gov/news/nn/nn012506.htm [accessed May 16, 2006].
Angle N, Hoyt DB, Coimbra R, Liu F, Herdon-Remelius C, Loomis W, Junger WG. 1998. Hypertonic saline resuscitation diminishes lung injury by suppressing neutrophil activation after hemorrhagic shock. Shock 9(3):164–170.
Auf der Heide TP, Sigurdsson G, Pirrallo RG, Yannopoulos D, McKnite S, von Briesen C, Sparks CW, Conrad CJ, Provo TA, Lurie KG. 2004. Hyperventilation-induced hypotension during cardiopulmonary resuscitation. Circulation 109(16):1960–1965.
Baren JM, Anicetti JP, Ledesma S, Biros MH, Mahabee-Gittens M, Lewis RJ. 1999. An approach to community consultation prior to initiating an emergency research study incorporating a waiver of informed consent. Academic Emergency Medicine 6(12):1210–1215.
Becker LB, Weisfeldt ML, Weil MH, Budinger T, Carrico J, Kern K, Nichol G, Shechter I, Traystman R, Webb C, Wiedemann H, Wise R, Sopko G. 2002. The pulse initiative: Scientific priorities and strategic planning for resuscitation research and life saving therapies. Circulation 105(21):2562–2570.
Bernard SA, Gray TW, Buist MD, Jones BM, Silvester W, Gutteridge G, Smith K. 2002. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. New England Journal of Medicine 346(8):557–563.
Bickell WH, Bruttig SP, Millnamow GA, O’Benar J, Wade CE. 1992. Use of hypertonic saline/ dextran versus lactated Ringer’s solution as a resuscitation fluid after uncontrolled aortic hemorrhage in anesthetized swine. Annals of Emergency Medicine 21(9):1077–1085.
Biros MH, Lewis RJ, Olson CM, Runge JW, Cummins RO, Fost N. 1995. Informed consent in emergency research. Consensus statement from the coalition conference of acute resuscitation and critical care researchers. Journal of the American Medical Association 273(16):1283–1287.
Biros MH, Runge JW, Lewis RJ, Doherty C. 1998. Emergency medicine and the development of the Food and Drug Administration’s final rule on informed consent and waiver of informed consent in emergency research circumstances. Academic Emergency Medicine 5(4):359–368.
Biros MH, Fish SS, Lewis RJ. 1999. Implementing the Food and Drug Administration’s final rule for waiver of informed consent in certain emergency research circumstances. Academic Emergency Medicine 6(12):1272–1282.
Brown MD, Vance SJ, Kline JA. 2005. An emergency department guideline for the diagnosis of pulmonary embolism: An outcome study. Academic Emergency Medicine 12(1):20–25.
Calvano SE, Xiao W, Richards DR, Felciano RM, Baker HV, Cho RJ, Chen RO, Brownstein BH, Cobb JP, Tschoeke SK, Miller-Graziano C, Moldawer LL, Mindrinos MN, Davis RW, Tompkins RG, Lowry SF. 2005. A network-based analysis of systemic inflammation in humans. Nature 437(7061):1032–1037.
Christian MC, Goldberg JL, Killen J, Abrams JS, McCabe MS, Mauer JK, Wittes RE. 2002. A central institutional review board for multi-institutional trials. New England Journal of Medicine 346(18):1405–1408.
Coimbra R, Hoyt DB, Junger WG, Angle N, Wolf P, Loomis W, Evers MF. 1997. Hypertonic saline resuscitation decreases susceptibility to sepsis after hemorrhagic shock. The Journal of Trauma 42(4):602–660; discussion 606–607.
Cooper RJ. 2004. Emergency department triage: Why we need a research agenda. Annals of Emergency Medicine 44(5):524–526.
Courtney DM, Kline JA. 2005. Prospective use of a clinical decision rule to identify pulmonary embolism as likely cause of outpatient cardiac arrest. Resuscitation 65(1):57–64.
Cydulka RK, Rowe BH, Clark S, Emerman CL, Camargo CA Jr, MARC Investigators. 2003. Emergency department management of acute exacerbations of chronic obstructive pulmonary disease in the elderly: The multicenter airway research collaboration. Journal of the American Geriatrics Society 51(7):908–916.
Eisenberg M, Damon S, Mandel L, Tewodros A, Meischke H, Beaupied E, Bennett J, Guildner C, Ewell C, Gordon M. 1995. CPR instruction by videotape: Results of a community project. Annals of Emergency Medicine 25(2):198–202.
EMNet. 2005. Emergency Medicine Network. [Online]. Available: http://www.emnet–usa.org [accessed April 16, 2005].
Fine MJ, Auble TE, Yealy DM, Hanusa BH, Weissfeld LA, Singer DE, Coley CM, Marrie TJ, Kapoor WN. 1997. A prediction rule to identify low-risk patients with community-acquired pneumonia. New England Journal of Medicine 336(4):243–250.
GAO (U.S. Government Accountability Office). 1991. Trauma Care: Lifesaving System Threatened by Unreimbursed Costs and Other Factors. Washington, DC: GAO.
Gausche M. 2000. Effect of out-of-hospital pediatric endotracheal intubation on survival and neurologic outcome: A controlled clinical trial. Journal of the American Medical Association 283(6):783–790.
Goldman E, Marshall E. 2002. Research funding. NIH grantees: Where have all the young ones gone? Science 298(5591):40–41.
Haddon W Jr. 1968. The changing approach to the epidemiology, prevention, and amelioration of trauma: The transition to approaches etiologically rather than descriptively based. American Journal of Public Health & the Nation’s Health 58(8):1431–1438.
Hoffman JR, Wolfson AB, Todd K, Mower WR. 1998. Selective cervical spine radiography in blunt trauma: Methodology of the national emergency x-radiography utilization study (NEXUS). Annals of Emergency Medicine 32(4):461–469.
Hoffman JR, Mower WR, Wolfson AB, Todd KH, Zucker MI. 2000. Validity of a set of clinical criteria to rule out injury to the cervical spine in patients with blunt trauma. National emergency x-radiography utilization study group. New England Journal of Medicine 343(2):94–99.
Hoyt DB, Holcomb J, Abraham E, Atkins J, Sopko G, Working Group on Trauma Research. 2004. Working Group on Trauma Research Program Summary Report: National Heart Lung Blood Institute (NHLBI), National Institute of General Medical Sciences (NIGMS), and National Institute of Neurological Disorders and Stroke (NINDS) of the National Institutes of Health (NIH), and the Department of Defense (DoD). Journal of Trauma-Injury Infection & Critical Care 57(2):410–415.
The Hypothermia After Cardiac Arrest Study Group. 1921. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. New England Journal of Medicine 346(8):549–556.
IOM (Institute of Medicine). 1999. Reducing the Burden of Injury. Washington, DC: National Academy Press.
IOM. 2003. Enhancing the Vitality of the National Institutes of Health: Organizational Change to Meet New Challenges. Washington, DC: The National Academies Press.
IOM. 2004. NIH Extramural Center Programs: Criteria for Initiation and Evaluation. Washington, DC: The National Academies Press.
Josiah Macy, Jr. Foundation. 1995. The Role of Emergency Medicine in the Future of American Medical Care. New York: Josiah Macy, Jr. Foundation.
Kellermann AL, Hackman BB, Somes G. 1989. Dispatcher-assisted cardiopulmonary resuscitation. Validation of efficacy. Circulation 80(5):1231–1239.
Kim S, Emerman CL, Cydulka RK, Rowe BH, Clark S, Camargo CA, MARC Investigators. 2004. Prospective multicenter study of relapse following emergency department treatment of COPD exacerbation. Chest 125(2):473–481.
Kline JA, Wells PS. 2003. Methodology for a rapid protocol to rule out pulmonary embolism in the emergency department. Annals of Emergency Medicine 42(2):266–275.
Kline JA, Webb WB, Jones AE, Hernandez-Nino J. 2004. Impact of a rapid rule-out protocol for pulmonary embolism on the rate of screening, missed cases, and pulmonary vascular imaging in an urban U.S. emergency department. Annals of Emergency Medicine 44(5):490–502.
Kline JA, Novobilski AJ, Kabrhel C, Richman PB, Courtney DM. 2005. Derivation and validation of a bayesian network to predict pretest probability of venous thromboembolism. Annals of Emergency Medicine 45(3):282–290.
Lewis RJ. 2004. Academic emergency medicine and the “tragedy of the commons” defined. Academic Emergency Medicine 11(5):423–427.
Lewis RJ, Berry DA, Cryer H III, Fost N, Krome R, Washington GR, Houghton J, Blue JW, Bechhofer R, Cook T, Fisher M. 2001. Monitoring a clinical trial conducted under the Food and Drug Administration regulations allowing a waiver of prospective informed consent: The diaspirin cross-linked hemoglobin traumatic hemorrhagic shock efficacy trial. Annals of Emergency Medicine 38(4):397–404.
Maio RF, Garrison HG, Spaite DW, Desmond JS, Gregor MA, Cayten CG, Chew JL Jr, Hill EM, Joyce SM, MacKenzie EJ, Miller DR, O’Malley PJ, Stiell IG. 1999. Emergency Medical Services Outcomes Project I (EMSOP I): Prioritizing conditions for outcomes research. Annals of Emergency Medicine 33(4):423–432.
Mann NC, Schmidt TA, Richardson LD. 2005. Confronting the ethical conduct of resuscitation research: A consensus opinion. Academic Emergency Medicine 12(11):1078–1081.
Moran GJ, Talan DA, Mower W, Newdow M, Ong S, Nakase JY, Pinner RW, Childs JE. 2000. Appropriateness of rabies postexposure prophylaxis treatment for animal exposures. Emergency ID Net Study Group. Journal of the American Medical Association 284(8):1001–1007.
Morris D, Manning J. 2004. Research in Academic Emergency Medicine. [Online]. Available: www.saem.org/publicat/chap6.htm [accessed November 3, 2004].
NAS, NRC (National Academy of Sciences, National Research Council). 1966. Accidental Death and Disability: The Neglected Disease of Modern Society. Washington, DC: National Academy Press.
National Center for Injury Prevention and Control. 2005. CDC Acute Injury Care Research Agenda: Guiding Research for the Future. Atlanta, GA: Centers for Disease Control.
Neumar RW. 2000. Molecular mechanisms of ischemic neuronal injury. Annals of Emergency Medicine 36(5):483–506.
Newgard CD, Hui SH, Stamps-White P, Lewis RJ. 2005. Institutional variability in a minimal risk, population-based study: Recognizing policy barriers to health services research. Health Services Research 40(4):1247–1258.
NHTSA (National Highway Traffic Safety Administration). 2001. National EMS Research Agenda. Washington, DC: U.S. Department of Transportation.
NHTSA and CIREN Center Staffs. 2003. NHTSA Crash Injury Research and Engineering Network (CIREN) Program Report, 2002. Washington DC: NHTSA.
NIH (National Institutes of Health). 1994. Report of the Task Force on Trauma Research. Bethesda, MD: NIH.
NIH. 2003. Ruth L. Kirschstein National Research Service Awards for Individual Postdoctoral Fellow (F32). [Online]. Available: http://grants1.nih.gov/grants/guide/pa–files/PA–03–067.html [accessed April 17, 2005].
Nolan JP, Morley PT, Vanden Hoek TL, Hickey RW, Kloeck WG, Billi J, Bottiger BW, Morley PT, Nolan JP, Okada K, Reyes C, Shuster M, Steen PA, Weil MH, Wenzel V, Hickey RW, Carli P, Vanden Hoek TL, Atkins D, International Liaison Committee on Resuscitation. 2003. Therapeutic hypothermia after cardiac arrest: An advisory statement by the advanced life support task force of the International Liaison Committee on Resuscitation. Circulation 108(1):118–121.
NRC, IOM (National Research Council, Institute of Medicine). 1985. Injury in America: A Continuing Public Health Problem. Washington, DC: National Academy Press.
PECARN (Pediatric Emergency Care Applied Research Network). 2003. The Pediatric Emergency Care Applied Research Network (PECARN): Rationale, development, and first steps. Academic Emergency Medicine 10(6):661–668.
PECARN. 2005. About PECARN. [Online]. Available: http://www.pecarn.org/about_pecarn. htm [accessed April 16, 2005].
Pollack CV Jr, Cairns CB. 1999. The emergency medicine foundation: 25 years of advancing education and research. Annals of Emergency Medicine 33(4):448–450.
Pollack C, Hollander J, O’Neil B, Neumar R, Summers R, Camargo C, Younger J, Callaway C, Gallagher E, Kellermann A, Krause G, Schafermeyer R, Sloan E, Stern S. 2003. Status report: Development of emergency medicine research since the Macy report. Annals of Emergency Medicine 42(1):66–80.
Sayre MR, White LJ, Brown LH, McHenry SD, National EMS Research Strategic Plan Writing Team. 2005. The national EMS research strategic plan. Prehospital Emergency Care 9(3):255–266.
Seidel J, Henderson D, Tittle S, Jaffe D, Spaite D, Dean J, Gausche M, Lewis R, Cooper A, Zaritsky A, Espisito T, Maederis D. 1999. Priorities for research in emergency medical services for children: Results of a consensus conference. Annals of Emergency Medicine 33(2):206–210.
Sloan EP, Koenigsberg M, Gens D, Cipolle M, Runge J, Mallory MN, Rodman G Jr. 1999a. Diaspirin cross-linked hemoglobin (DCLHB) in the treatment of severe traumatic hemorrhagic shock: A randomized controlled efficacy trial. Journal of the American Medical Association 282(19):1857–1864.
Sloan EP, Koenigsberg M, Houghton J, Gens D, Cipolle M, Runge J, Mallory MN, Rodman G Jr, DCLHB Traumatic Hemorrhagic Shock Study Group. 1999b. The informed consent process and the use of the exception to informed consent in the clinical trial of diaspirin cross-linked hemoglobin (DCLHB) in severe traumatic hemorrhagic shock. Academic Emergency Medicine 6(12):1203–1209.
Stern SA. 2001. Fellowship Training: A Necessity in Today’s Academic World. [Online]. Available: http://www.saem.org/newsltr/2001/july–august/stern.htm [accessed April 17, 2005].
Stern SA, Zink BJ, Mertz M, Wang X, Dronen SC. 2000. Effect of initially limited resuscitation in a combined model of fluid-percussion brain injury and severe uncontrolled hemorrhagic shock. Journal of Neurosurgery 93(2):305–314.
Sung NS, Crowley WF Jr, Genel M, Salber P, Sandy L, Sherwood LM, Johnson SB, Catanese V, Tilson H, Getz K, Larson EL, Scheinberg D, Reece EA, Slavkin H, Dobs A, Grebb J, Martinez RA, Korn A, Rimoin D. 2003. Central challenges facing the national clinical research enterprise. Journal of the American Medical Association 289(10):1278–1287.
Talan D, Moran G, Mower W, Newdow M, Ong S, Slutsker L, Jarvis W, Conn L, Pinner R. 1998. EMERGEncy ID NET: An emergency department-based emerging infections sentinel network. Annals of Emergency Medicine 32(6):703–711.
Talan DA, Citron DM, Abrahamian FM, Moran GJ, Goldstein EJ, Emergency Medicine Animal Bite Infection Study Group. 1999. Bacteriologic analysis of infected dog and cat bites. New England Journal of Medicine 340(2):85–92.
Talan DA, Abrahamian FM, Moran GJ, Citron DM, Tan JO, Goldstein EJ, Emergency Medicine Human Bite Infection Study Group. 2003. Clinical presentation and bacteriologic analysis of infected human bites in patients presenting to emergency departments. Clinical Infectious Diseases 37(11):1481–1489.
Todd KH, Braslow A, Brennan RT, Lowery DW, Cox RJ, Lipscomb LE, Kellermann AL. 1998. Randomized, controlled trial of video self-instruction versus traditional CPR training. Annals of Emergency Medicine 31(3):364–369.
Todd KH, Heron SL, Thompson M, Dennis R, O’Connor J, Kellermann AL. 1999. Simple CPR: A randomized, controlled trial of video self-instructional cardiopulmonary resuscitation training in an African American church congregation. Annals of Emergency Medicine 34(6):730–737.
Trunkey DD. 1981. Regionalization of trauma care. Topics in Emergency Medicine 3(2): 91–96.
Wadman MC, Muelleman RL, Coto JA, Kellermann AL. 2003. The pyramid of injury: Using ecodes to accurately describe the burden of injury. Annals of Emergency Medicine 42(4):468–478.
Wright DW, Ritchie JC, Mullins RE, Kellermann AL, Denson DD. 2005. Steady-state serum concentrations of progesterone following continuous intravenous infusion in patients with acute moderate to severe traumatic brain injury. Journal of Clinical Pharmacology 45(6):640–648.
Wright S, Wrenn K. 1994. Funding in the emergency medicine literature: 1985 to 1992. Annals of Emergency Medicine 23(5):1077–1081.
Zerhouni E. 2003. Medicine. The NIH roadmap. Science 302(5642):63–72.
Zisson S. 2001. Anticipating a clinical investigator shortfall. CenterWatch 8(1).






























