10
Stomach Cancer and Asbestos
NATURE OF THIS CANCER TYPE
Despite a major decline in the incidence of stomach cancer (ICD-9 151; ICD-O-3 C16.0-16.9; see Figure 9.1) worldwide, and particularly in industrialized countries over the last century, it remains the second most common fatal cancer worldwide. An estimated 22,280 new cases and 11,430 deaths are projected to occur in the United States in 2006 (Jemal et al. 2006). The incidence of stomach cancer is about twice as high among men as among women, and is higher among nonwhites than among whites. In the United States, the incidence per 100,000 people varies markedly by race and ethnicity: the incidence in Asian and Pacific islanders is 23.0 and 12.8 in males and females, respectively; in blacks, 19.9 and 9.9; in Hispanics, 18.1 and 10.0; in American Indians and Alaska natives, 14.4 and 8.3; and in white non-Hispanics, 10.0 and 4.3. The age-standardized death rate from stomach cancer has declined on average by 2.5-3.0% per year since 1975.
The known risk factors for stomach cancer depend on the location of the tumor. Obesity is the principal known risk factor for cancers that develop in the upper portion (cardia) of the stomach in the United States. In contrast, severe nutritional deficiency is responsible for the high endemic incidence of that type of stomach cancer in some regions of China. Chronic infection with the bacterium Helicobacter pylori (H. pylori) in combination with tobacco-smoking is the main cause of cancer of the body (fundus) of the stomach and of the gastric antrum. Chronic infection with H. pylori causes chronic gastritis and loss of the acid-producing potential of the stomach.
About 90-95% of stomach cancers are adenocarcinomas, although other, less common tumors do occur. Most adenocarcinomas are thought to develop slowly—over many years. Premalignant lesions and early-stage cancers generally begin in the mucosa or inner lining of the stomach. The cells become progressively abnormal as they accumulate genetic damage. With malignant transformation, the cancers develop the capacity to invade the submucosa and muscle wall of the stomach, to extend from there into the subserosa and the outermost serosa that wrap the stomach, or to metastasize to other organs, such as the liver, lungs, and bones. Some types of lymphoma, a cancer of the immune system, also occur in the wall of the stomach; they account for about 4% of stomach cancers. Included among the other tumors are the slow-growing (indolent) lymphoma of mucosa-associated lymphoid tissue (MALT) and carcinoid, a hormone-producing tumor of the stomach and other organs.
In the United States, the median survival of persons with stomach cancer is less than 1 year after diagnosis, although the 5-year survival rate has increased slightly from 15.1% for patients diagnosed in 1974-1976 to 23.2% for those diagnosed in 1995-2001.
EPIDEMIOLOGIC EVIDENCE CONSIDERED
The committee deemed 36 papers on occupational cohorts and five population-based case-control studies to be informative for assessing the association of asbestos and stomach cancer. In that several citations on the cohorts reported separate findings for subgroups (by sex or by separate factory workforces), a total of 42 cohorts were included in this review.
Cohort Studies
The cohorts that presented usable information on the risk of stomach cancer were indicated in Table 6.1. Their histories and design properties are described in Table B.1, and the details of their results concerning cancer at this site are abstracted in Table D.4. The results of the cohort and case-control studies are summarized in Table 10.1, and Figures 10.1 and 10.2 are plots of RRs for overall exposures and for exposure-response gradients from the cohort studies reviewed.
Most of the occupational cohort studies were conducted in predominantly white populations in North America, Europe, and Australia. Two studies in China (Pang et al. 1997, Zhu and Wang 1993) were also included in the review. The reported associations between stomach cancer and occupational asbestos exposure in cohort studies were based on somewhat larger numbers of cases than those related to esophageal cancer, but statistical power in individual studies was generally low
TABLE 10.1 Summary of Epidemiologic Findings Regarding Cancer of the Stomach
|
Study Type |
Figure |
Comparison |
Study Populations Included |
No. Study Populations |
Summary RR (95% CI) |
Between-Study SD |
|
Cohort |
Any vs none |
All |
42 |
1.17 (1.07-1.28) |
|
|
|
|
High vs nonea |
Lower boundb |
12c |
1.31 (0.97-1.76) |
|
|
|
|
|
|
Upper boundb |
13 |
1.33 (0.98-1.79) |
— |
|
Case- control |
Any vs none |
All |
5 |
1.11 (0.76-1.64) |
0.32 |
|
|
|
Any vs none |
EAM = 1 |
3 |
0.91 (0.45-1.84) |
0.48 |
|
|
|
|
|
EAM = 2 |
2 |
1.43 (0.70-2.93) |
0.42 |
|
|
High vs nonea |
EAM = 1 |
5 |
1.42 (0.92-2.20) |
0.00 |
|
|
NOTE: CI = Confidence interval; EAM = exposure-assessment method; high quality, EAM = 1; lower quality, EAM = 2; RR = relative risk; SD = standard deviation. aIncluded studies that reported dose-response relationship (RR on an exposure gradient). bFor studies that reported dose-response relationship on multiple gradient metrics, the smallest “high vs none” RR was used to compute the lower bound, and the largest “high vs none” RR was used in computing the upper bound. cThis calculation omitted de Klerk et al. (1989), which reported RR = 0, but gave no information on the expected number of cases. |
||||||
because stomach cancer is a rare outcome in whites. Mortality was the outcome in most of the cohort studies reviewed. Insofar as stomach cancer generally has a high case-fatality rate, mortality is a relatively accurate measure of disease incidence.
All the cohort studies lacked data on stomach-cancer subsites and on potential confounding factors, such as H. pylori infection or diet. Confounding by factors related to socioeconomic status, which may be associated with these and other stomach-cancer risk factors, may have occurred in the comparisons of overall risks between occupational cohorts and the general population (such as standardized mortality ratios). However, the extent of potential confounding in the comparisons can only be speculated. Internal comparisons of exposure-response gradients within cohorts were unlikely to have been confounded appreciably, because important potential
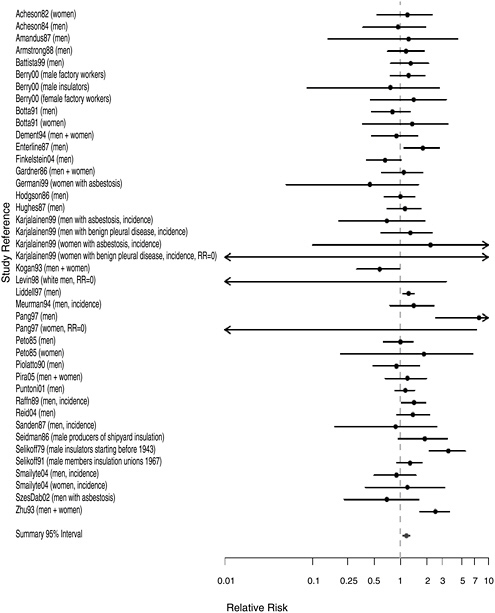
FIGURE 10.1 Cohort studies: RR of stomach cancer in people with “any” exposure to asbestos compared with people who report none.
confounders were probably, at most, weakly correlated with cumulative asbestos exposure.
The majority of overall cohort RR estimates exceeded the null value (1.0), indicating excesses, although estimates varied considerably in strength. The largest overall cohort RRs were seen among the earliest New
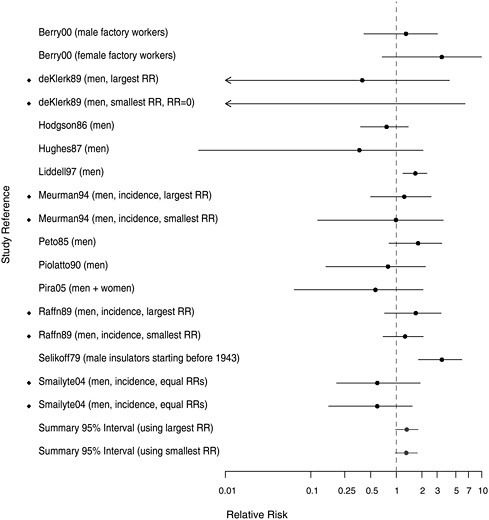
FIGURE 10.2 Cohort studies: RRs of stomach cancer among people in most extreme exposure category compared to those with none (◆ = more than one exposure gradient reported in citation, so the plot contains both highest and lowest estimates of risk for most extreme category over all gradients).
York-New Jersey insulation workers whose RR was 3.52 (Selikoff et al. 1979) and among two sets of workers in Chinese asbestos factories whose RRs were 4.4 and 2.4, respectively (Pang et al. 1997, Zhu and Wang 1993). The majority of studies reported small to modest overall cohort wide excesses (RR < 1.5). The combined RR was 1.17 (95% CI 1.07-1.28) (Table 10.1).
With respect to dose-response gradients, the summary estimates were stable whether based on the smallest (RR = 1.31, 95% CI 0.97-1.76) or
largest (RR = 1.33, 95% CI 0.97-1.79) estimates associated with the extreme exposure categories within the cohorts. Several studies indicated reasonably prominent exposure-response gradients. Among the more noteworthy findings were RR values exceeding 3.0 for the highest exposure categories of Quebec miners (Liddell et al. 1997) and of UK female asbestos-factory workers (Berry et al. 2000) and for the earliest US insulation workers (Selikoff et al. 1979).
Case-Control Studies
The five case-control studies of stomach cancer retained for thorough evaluation after exclusion of studies that did not assess exposure to asbestos or did not meet other exclusion criteria are listed in Table 6.5 according to quality of their exposure assessment. The details of the design aspects of those studies are presented in Table C.1 and their detailed results are ab-
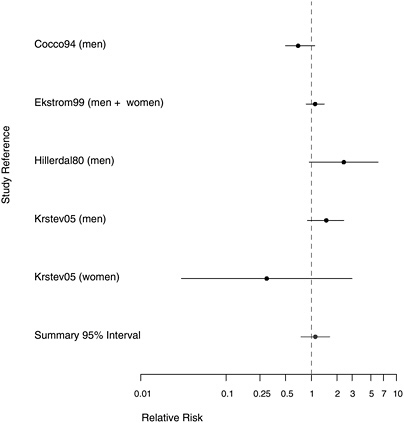
FIGURE 10.3 Case-control studies: RR of stomach cancer in people with “any” exposure to asbestos compared with people with none.
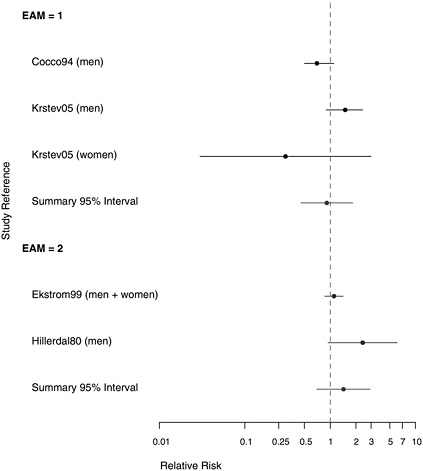
FIGURE 10.4 Case-control studies: RR of stomach cancer in people with “any” exposure to asbestos compared with people with none, stratified on quality of exposure assessment (top, EAM = 1: higher-quality exposure assessment; bottom, EAM = 2: lower-quality exposure assessment).
stracted in Table E.4. The findings of the studies are summarized in Table 10.1 and in the plots presented in Figures 10.3-10.5. Four of the case-control studies gathered data on potential confounders and adjusted risk estimates for asbestos exposure accordingly.
The findings for the case-control study populations with results reported for any asbestos exposure were mixed (Figure 10.3). Two found evidence of an increased stomach-cancer risk, two reported mostly null associations, and one suggested a deficit of stomach-cancer risk among women exposed to asbestos. The combined RR across these five was 1.11 (95% CI 0.76-1.64). That small overall RR increase was based largely on the two studies
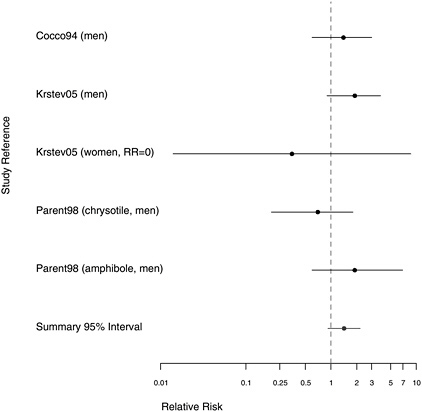
FIGURE 10.5 Case-control studies: RRs of stomach cancer among people in with extreme exposure compared with those with none.
with lower-quality exposure assessment (see Figure 10.4), whose aggregate RR estimate was 1.43 (95% CI 0.70-2.93), whereas the combined RR estimate for the three findings with better exposure assessments was 0.91 (95% CI 0.45-1.84). The strongest association (RR=2.40) was reported by a study in which asbestos exposure was inferred indirectly from the presence of pleural plaques rather than being based on quantitative estimates of cumulative exposure or duration (Hillerdal 1980).
The summary odds ratio (OR) increased when only extreme exposure was considered (OR = 1.42, 95% CI 0.92-2.20), but evidence suggesting a dose-response relationship was also inconsistent (Figure 10.5). The largest study (Cocco et al. 2004) gave a suggestion of greater risk with longer exposure, but the other two citations (Krstev et al. 2005, Parent et al. 1998)
both had one study group showing somewhat greater risk with more exposure and another group showing the reverse.
EVIDENCE INTERGRATION AND CONCLUSION
Evidence Considered
The committee’s final review covered 34 occupational-cohort studies, including a total of 42 cohorts, and five population-based case-control studies that provided data on stomach-cancer risk. The studies were conducted primarily among whites in North America, Europe, and Australia. Some of the studies reviewed information on exposure that enabled dose-response evaluation. In addition, the committee considered studies in which animals were exposed chronically to asbestos either by inhalation (Hesterberg et al. 1993, 1994; McConnell 2005; McConnell et al. 1994a,b, 1999) or by oral feeding (HHS 1983, 1985, 1988, 1990a,b,c); none reported increased incidence of stomach tumors.
Consistency
The occupational-cohort studies generally, although not uniformly, suggested a slightly increased risk of stomach cancer relative to the general population. There were also several instances in which reasonably strong dose-response gradients were observed in the occupational cohort studies. In contrast, the results from case-control studies were inconsistent, with no clear pattern emerging from the estimated risks of stomach cancer associated with asbestos exposure.
Strength of Association
Considering only the cohort studies, the committee noted that observed risk increases were weak to modest. There was also evidence from several studies supportive of relatively strong dose-response relationships. However, the few weak suggestions of dose-response gradients seen among the cohorts did little to augment the overall strength of evidence of a causal association. The case-control studies were far less informative than the cohort studies in their characterization of either overall associations or dose-response relationships between asbestos and stomach cancer.
Coherence
Asbestos bodies have been identified in human stomach tissue and at other sites in the gastrointestinal tract (Auerbach et al. 1980, Kobayashi et al. 1987). It is plausible that asbestos fibers could accumulate at sites of mucosal injury
and ulceration in the stomach, but the extent to which this occurs in exposed persons has not been investigated. As is the case for finding asbestos bodies at other sites, contamination during collection or processing of tissue samples is a possibility, as discussed in Chapter 4. The potential role of asbestos fibers as a cofactor with established risk factors (such as H. pylori) has not been investigated experimentally or epidemiologically.
Stomach cancer has received far less attention than mesothelioma or lung cancer in toxicologic research on asbestos. The very limited available literature on the topic provides no experimental evidence that asbestos fibers act as a direct or indirect carcinogen in the stomach.
Conclusion
Several considerations led the committee to its conclusion regarding causality for this site. The occupational-cohort studies were the most informative source of evidence. They revealed a generally consistent pattern of fairly modest risk increases. There was also some evidence, albeit inconsistent, of dose-dependence. Considerations of biological plausibility did not strengthen the case for causation. In particular, the extent of asbestos-fiber deposition and retention in human stomach mucosa is not known. Moreover, the limited available evidence from experimental research does not indicate that asbestos is carcinogenic to the stomach.
The committee concluded that evidence is suggestive but not sufficient to infer a causal relationship between asbestos exposure and stomach cancer.
REFERENCES
Acheson ED, Gardner MJ, Pippard EC, Grime LP. 1982. Mortality of two groups of women who manufactured gas masks from chrysotile and crocidolite asbestos: A 40-year follow-up. British Journal of Industrial Medicine 39(4): 344-348.
Acheson ED, Gardner MJ, Winter PD, Bennett C. 1984. Cancer in a factory using amosite asbestos. International Journal of Epidemiology 13(1): 3-10.
Amandus HE, Wheeler R, Jankovic J, Tucker J. 1987. The morbidity and mortality of vermiculite miners and millers exposed to tremolite-actinolite: Part I. Exposure estimates. American Journal of Industrial Medicine 11(1): 1-14.
Armstrong BK, de Klerk NH, Musk AW, Hobbs MS. 1988. Mortality in miners and millers of crocidolite in Western Australia. British Journal of Industrial Medicine 45(1): 5-13.
Auerbach O, Conston AS, Garfinkel L, Parks VR, Kaslow HD, Hammond EC. 1980. Presence of asbestos bodies in organs other than the lung. Chest 77(2): 133-137.
Battista G, Belli S, Comba P, Fiumalbi C, Grignoli M, Loi F, Orsi D, Paredes I. 1999. Mortality due to asbestos-related causes among railway carriage construction and repair workers. Occupation Medicine (London) 49(8): 536-539.
Berry G, Newhouse ML, Wagner JC. 2000. Mortality from all cancers of asbestos factory workers in east London 1933-80. Occupational and Environmental Medicine 57(11): 782-785.
Botta M, Magnani C, Terracini B, Bertolone GP, Castagneto B, Cocito V, DeGiovanni D, Paglieri P. 1991. Mortality from respiratory and digestive cancers among asbestos cement workers in Italy. Cancer Detection and Prevention 15(6): 445-447.
Cocco P, Palli D, Buiatti E, Cipriani F, DeCarli A, Manca P, Ward MH, Blot WJ, Fraumeni JF Jr. 1994. Occupational exposures as risk factors for gastric cancer in Italy. Cancer Causes and Control 5(3): 241-248.
de Klerk NH, Armstrong BK, Musk AW, Hobbs MS. 1989. Cancer mortality in relation to measures of occupational exposure to crocidolite at Wittenoom Gorge in Western Australia. British Journal of Industrial Medicine 46(8): 529-536.
Dement JM, Brown DP, Okun A. 1994. Follow-up study of chrysotile asbestos textile workers: Cohort mortality and case-control analyses. American Journal of Industrial Medicine 26(4): 431-447.
Ekstrom AM, Eriksson M, Hansson LE, Lindgren A, Signorello LB, Nyren O, Hardell L. 1999. Occupational exposures and risk of gastric cancer in a population-based case-control study. Cancer Research 59(23): 5932-5937.
Enterline PE, Hartley J, Henderson V. 1987. Asbestos and cancer: A cohort followed up to death. British Journal of Industrial Medicine 44(6): 396-401.
Finkelstein MM, Verma DK. 2004. A cohort study of mortality among Ontario pipe trades workers. Occupational and Environmental Medicine 61(9): 736-742.
Gardner MJ, Winter PD, Pannett B, Powell CA. 1986. Follow up study of workers manufacturing chrysotile asbestos cement products. British Journal of Industrial Medicine 43(11): 726-732.
Germani D, Belli S, Bruno C, Grignoli M, Dstat MN, Pirastu R, Comba P. 1999. Cohort mortality study of women compensated for asbestosis in Italy. American Journal Industrial Medicine 36(1): 129-134.
Hesterberg TW, Miller WC, McConnell EE, Chevalier J, Hadley JG, Bernstein DM, Thevenaz P, Anderson R. 1993. Chronic inhalation toxicity of size-separated glass fibers in Fischer 344 rats. Fundamental and Applied Toxicology 20(4): 464-476.
Hesterberg TW, Miller WC, Mast R, McConnell EE, Bernstein DM, Anderson R. 1994. Relationship between lung biopersistence and biological effects of man-made vitreous fibers after chronic inhalation in rats. Environmental Health Perspectives 102 (Supplement 5): 133-137.
HHS (US Department of Health and Human Services). 1983. Lifetime Carcinogenesis Studies of Amosite Asbestos (CAS NO. 12172-73-5) in Syrian Golden Hamsters (Feed Studies). NTP TR 249. Research Triangle Park, NC: National Toxicology Program.
HHS. 1985. Toxicology and Carcinogenesis Studies of Chrysotile Asbestos (CAS No. 12001-29-5) in F344/N Rats (Feed Studies). NTP TR 295. Research Triangle Park, NC: National Toxicology Program.
HHS. 1988. Toxicology and Carcinogenesis Studies of Crocidolite Asbestos (CAS No. 12001-28-4) in F344/N Rats (Feed Studies). NTP TR 280. Research Triangle Park, NC: National Toxicology Program.
HHS. 1990a. Lifetime Carcinogenesis Studies of Chrysotile Asbestos (CAS No. 12001-29-5) in Syrian Golden Hamsters (Feed Studies). NTP TR 246. Research Triangle Park, North Carolina: National Toxicology Program.
HHS. 1990b. Toxicology and Carcinogenesis Studies of Amosite Asbestos (CAS No. 12172-73-5) in F344/N Rats (Feed Studies). NTP TR 279. Research Triangle Park, NC: National Toxicology Program.
HHS. 1990c. Toxicology and Carcinogenesis Studies of Tremolite (CAS No. 14567-73-8) in F344/N Rats (Feed Studies). NTP TR 277. Research Triangle Park, NC: National Toxicology Program.
Hillerdal G. 1980. Gastrointestinal carcinoma and occurrence of pleural plaques on pulmonary X-ray. Journal of Occupational Medicine 22(12): 806-809.
Hodgson JT, Jones RD. 1986. Mortality of asbestos workers in England and Wales 1971-81. British Journal of Industrial Medicine 43(3): 158-164.
Hughes JM, Weill H, Hammad YY. 1987. Mortality of workers employed in two asbestos cement manufacturing plants. British Journal of Industrial Medicine 44(3): 161-174.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C, Thun M. 2006. Cancer statistics, 2006. CA: A Cancer Journal for Clinicians 56: 106-130.
Karjalainen A, Pukkala E, Kauppinen T, Partanen T. 1999. Incidence of cancer among Finnish patients with asbestos-related pulmonary or pleural fibrosis. Cancer Causes and Control 10(1): 51-57.
Kobayashi H, Ming ZW, Watanabe H, Ohnishi Y. 1987. A quantitative study on the distribution of asbestos bodies in extrapulmonary organs. ACTA Pathologica Japonica 37(3): 375-383.
Kogan PM, Yatsenko AS, Tregubov ES, Gurvich EB, Kuzina LE. 1993. Evaluation of carcinogenic risk in friction product workers. Medicina del Lavoro 84(4): 290-296.
Krstev S, Dosemeci M, Lissowska J, Chow WH, Zatonski W, Ward MH. 2005. Occupation and risk of stomach cancer in Poland. Occupational and Environmental Medicine 62(5): 318-324.
Levin J, McLarty J, Hurst GA, Smith A, Frank AL. 1998. Tyler asbestos workers: Mortality experience in a cohort exposed to amosite. Occupational and Environmental Medicine 55(3): 155-160.
Liddell FD, McDonald AD, McDonald JC. 1997. The 1891-1920 birth cohort of Quebec chrysotile miners and millers: Development from 1904 and mortality to 1992. Annals of Occupational Hygiene 41(1): 13-36.
McConnell EE. 2005 (October 27). Personal Communication to Mary Paxton for the Committee on Asbestos: Selected Health Effects. Available in IOM Public Access Files.
McConnell E, Kamstrup O, Musselman R, Hesterberg T, Chevalier J, Miller W, Thevenaz P. 1994a. Chronic inhalation study of size-separated rock and slag wool insulation fibers in Fischer 344/N rats. Inhalation Toxicology 6(6): 571-614.
McConnell E, Mast R, Hesterberg T, Chevalier J, Kotin P, Bernstein D, Thevenaz P, Glass L, Anderson R. 1994b. Chronic inhalation toxicity of a kaolin-based refactory cermaic fiber in Syrian golden hamsters. Inhalation Toxicology 6(6): 503-532.
McConnell EE, Axten C, Hesterberg TW, Chevalier J, Miller WC, Everitt J, Oberdorster G, Chase GR, Thevenaz P, Kotin P. 1999. Studies on the inhalation toxicology of two fiberglasses and amosite asbestos in the Syrian golden hamster: Part II. Results of chronic exposure. Inhalation Toxicology 11(9): 785-835.
Meurman LO, Pukkala E, Hakama M. 1994. Incidence of cancer among anthophyllite asbestos miners in Finland. Occupational and Environmental Medicine 51(6): 421-425.
Pang ZC, Zhang Z, Wang Y, Zhang H. 1997. Mortality from a Chinese asbestos plant: Over-all cancer mortality. American Journal of Industrial Medicine 32(5): 442-444.
Parent ME, Siemiatycki J, Fritschi L. 1998. Occupational exposures and gastric cancer. Epidemiology 9(1): 48-55.
Peto J, Doll R, Hermon C, Binns W, Clayton R, Goffe T. 1985. Relationship of mortality to measures of environmental asbestos pollution in an asbestos textile factory. Annals of Occupational Hygiene 29(3): 305-355.
Piolatto G, Negri E, La Vecchia C, Pira E, Decarli A, Peto J. 1990. An update of cancer mortality among chrysotile asbestos miners in Balangero, northern Italy. British Journal of Industrial Medicine 47(12): 810-814.
Pira E, Pelucchi C, Buffoni L, Palmas A, Turbiglio M, Negri E, Piolatto PG, La Vecchia C. 2005. Cancer mortality in a cohort of asbestos textile workers. British Journal of Cancer 92(3): 580-586.
Puntoni R, Merlo F, Borsa L, Reggiardo G, Garrone E, Ceppi M. 2001. A historical cohort mortality study among shipyard workers in Genoa, Italy. American Journal of Industrial Medicine 40(4): 363-370.
Raffn E, Lynge E, Juel K, Korsgaard B. 1989. Incidence of cancer and mortality among employees in the asbestos cement industry in Denmark. British Journal of Industrial Medicine 46(2): 90-96.
Reid A, Ambrosini G, de Klerk N, Fritschi L, Musk B. 2004. Aerodigestive and gastrointestinal tract cancers and exposure to crocidolite (blue asbestos): Incidence and mortality among former crocidolite workers. International Journal of Cancer 111(5): 757-761.
Sanden A, Jarvholm B. 1987. Cancer morbidity in Swedish shipyard workers 1978-1983. International Archives of Occupational and Environmental Health 59(5): 455-462.
Seidman H, Selikoff IJ, Gelb SK. 1986. Mortality experience of amosite asbestos factory workers: Dose-response relationships 5 to 40 years after onset of short-term work exposure. American Journal of Industrial Medicine 10(5-6): 479-514.
Selikoff IJ, Seidman H. 1991. Asbestos-associated deaths among insulation workers in the United States and Canada, 1967-1987. Annals of the New York Academy of Sciences 643: 1-14.
Selikoff IJ, Hammond EC, Seidman H. 1979. Mortality experience of insulation workers in the United States and Canada, 1943-1976. Annals of the New York Academy of Sciences 330: 91-116.
Smailyte G, Kurtinaitis J, Andersen A. 2004. Cancer mortality and morbidity among Lithuanian asbestos-cement producing workers. Scandinavian Journal of Work, Environment and Health 30(1): 64-70.
Szeszenia-Dabrowska N, Urszula W, Szymczak W, Strzelecka A. 2002. Mortality study of workers compensated for asbestosis in Poland, 1970-1997. International Journal of Occupation Medicine and Environmental Health 15(3): 267-278.
Zhu H, Wang Z. 1993. Study of occupational lung cancer in asbestos factories in China. British Journal of Industrial Medicine 50(11): 1039-1042.













