4
What We Drink
Water is essential to life and good health. Drinking water provides necessary hydration and serves as the carrier for a variety of substances, both beneficial and harmful, that enter and may be metabolically transformed by the body. The availability and quality of fresh drinking water are controlled by earth and atmospheric processes that generate the water cycle (e.g., NRC, 2004a, 2004b, 2004c). Water that falls on the land surface and does not become part of glaciers or polar ice caps eventually partitions into water that infiltrates into the ground and runoff, which moves by surface or shallow subsurface flow to lakes and streams. The rates of water infiltration are controlled by surface soil textures. The infiltrated water, in turn, partitions into water held in shallow soils, from which it can be removed by plants, and water that percolates downward to the water table where it becomes “recharge” to groundwater. Groundwater moving through geological layers composed of permeable materials (aquifers) eventually migrates to locations at or near the land surface, where it either becomes available for loss to the atmosphere by evapotranspiration or discharges to either fresh surface water bodies or the ocean (e.g., Dingman, 2002). The rates of flow and residence time of groundwater are influenced by the characteristics of the geological layers (Brusseau and Tick, 2006).
At deeper levels of the earth’s crust, water occupies the interstices (void spaces) in porous rocks such as fractured lava flows and sandstones, siltstones, and shales (mudrocks). These layered formations may accumulate in depositional basins to thicknesses of tens of kilometers, and consequently the interstitial water contained in such rocks is subjected to in-
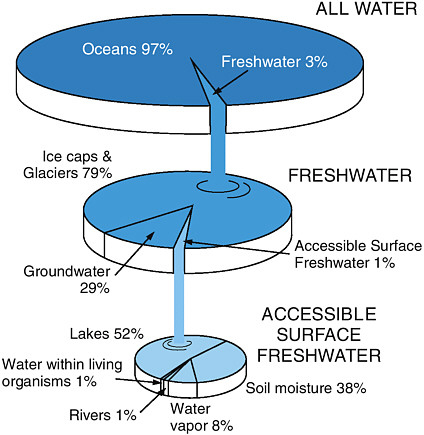
FIGURE 4.1 Distribution of the world’s water.
SOURCE: Courtesy “Earth Update” CD-ROM, Rice University and the Houston Museum of Natural Science; used with permission.
creased pressures and temperatures due to burial and compaction. The increased pressure and temperature result in enrichment of aqueous solutions within the rocks by soluble chemical species, especially salts. Such interstitial brines may be several times saltier than the world’s oceans, and thus fresh groundwater is typically limited to near-surface reservoirs.
Within the earth’s hydrosphere, fresh water comprises only 3% of the total water in the earth system. Because most fresh water is held in glaciers and polar ice caps, only ~30% of fresh water reserves are available as surface water or groundwater for human use (Dingman, 2002; see Figure 4.1). In many arid areas of the world, and even in some more humid locations, groundwater extraction rates by humans exceed natural recharge rates, and the available water stored in aquifers is decreasing. Agriculture
is the main user of fresh water, amounting to approximately 70% worldwide and up to 90% in developing countries. Together, irrigation and drainage (especially drainage water reuse) are a major source of salts and toxic trace elements in arid and semiarid regions.
Water, in both quantity and quality, is inextricably linked to public health. The availability of safe water is a basic human necessity and often is the first resource to become critically short in a natural disaster (such as recently encountered along the U.S. Gulf Coast in the aftermath of Hurricane Katrina). The availability and sustainability of safe water from surface and underground sources, particularly in the context of climate changes and population pressure, is a critical area of research that is beyond the scope of this report. This chapter will be limited to a description of the constituents in drinking water as potential benefits to public health (e.g., fluoride) and as potential hazards to public health (e.g., microbial contamination or dissolved toxic elements). The chapter will focus on the threshold research areas, with particular attention to the aspects of water and health that are directly influenced by earth science and the geological framework.
Drinking water contains a variety of substances that result from interactions with geological materials or from other sources such as atmospheric deposition, land application of fertilizer and wastes, mine drainage, and discharge of waste to surface water bodies. These include metals, major and trace elements, natural and anthropogenic organic substances, and microorganisms. Some of these constituents are essential nutrients; many have unknown or only suspected health benefits; and others are clearly health hazards.
In groundwater, inorganic constituents are transported primarily in a dissolved or nanoparticulate form. These constituents are able to enter the drinking water distribution system unless the water is subjected to appropriate treatment processes. Natural organic matter is also a component of natural waters, with largely unknown direct health implications. These substances have a well-established ability to form complexes with metals and potentially enhance the dissolution of minerals and mobilize sparingly soluble metal ions (Hem, 1985). In addition, natural organic matter interacts with chlorine and other drinking water disinfectants to form a dilute mixture of disinfection byproducts that may be mutagenic and/or carcinogenic (Jolley et al., 1984; NRC, 1987; Gerba et al., 2006). Anthropogenic organic materials have a wide variety of potential source points and a wider range of potential health impacts. For virtually all anthropogenic organic compounds, the geological framework—including surface topography, soils, and the vadose zone—exerts a fundamental control on the transport properties from source point to receiver.
HEALTH BENEFITS OF WATERBORNE EARTH MATERIALS
Some beneficial elements, such as calcium, magnesium, and fluoride, either occur naturally in water at sufficiently high concentration to positively influence human health or can be added to water as supplements. In addition, some microbes result in remediation of waterborne contaminants (bioremediation).
Calcium and Magnesium (Hard Water)
Calcium (Ca) and magnesium (Mg) are two of the three most abundant cations (along with sodium) in natural waters. These cations result from dissolution of a variety of rock-forming minerals, including feldspars, carbonates, and sulfate evaporites such as gypsum. Water “hardness” is a measure of the combined calcium and magnesium concentrations in water (see Figure 4.2). Because high concentrations of Ca and Mg generate residues when used with soaps, and boiler scale when water is heated and evaporated, “softening” by ion exchange is recommended for
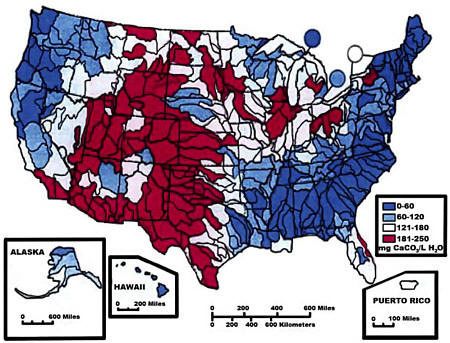
FIGURE 4.2 Water hardness across the United States, 1975 (milligrams per liter).
SOURCE: USGS web product; http://water.usgs.gov/owq/map1.jpeg.
water with a hardness exceeding 80 mg L−1. These practical considerations have led to widespread monitoring of water hardness in industrialized countries such as the United States.
There are documented beneficial health effects from the intake of calcium and magnesium, and some calcium and magnesium comes from daily water intake. Calcium is particularly important in the prevention of osteoporosis, with a recommended daily allowance for peri- and post-menopausal women of 1,200 mg (Wilkins and Birge, 2005). Whether calcium and magnesium in drinking water have a beneficial effect on the cardiovascular system is controversial—although many geographic studies have suggested an inverse association between water hardness and cardiovascular mortality, a recently published large case control study demonstrated no effect of calcium and magnesium intake from drinking water on the occurrence of myocardial infarction (Rosenlund et al., 2005).
Magnesium plays an important role as an activator of more than 300 enzymatic reactions, and the recommended daily magnesium intake for an adult is about 300–400 mg (SCF, 1993; NRC, 1997). Magnesium deficiency increases the risk to humans of developing various pathological conditions, such as vasoconstriction, hypertension, cardiac arrhythmia, atherosclerotic vascular disease, acute myocardial infarction, eclampsia in pregnant women, possibly type II diabetes mellitus, and osteoporosis (Rude, 1998; Innerarity, 2000; Saris et al., 2000). These relationships—reported in multiple clinical and epidemiological studies—have recently been supported by the results of experimental studies on animals (Sherer et al., 2001).
Fluoride
Fluoride, as it occurs in drinking water, has two beneficial effects. It helps prevent dental caries, particularly in children, and it contributes to bone mineralization and bone matrix integrity (ADA, 2005). The value of adding fluoride to water that has low natural fluoride levels has been recognized internationally, with community water fluoridation programs implemented in at least 60 countries. There have been periodic assessments of the benefits and risks of fluoridation. A 1991 report recommended the continued use of fluoride in the United States to prevent dental caries and advocated continued support for fluoridation of drinking water (HHS, 1991). The report also recommended the initiation of scientific assessments to determine the optimal exposure level of fluoride from all sources (i.e., combined exposure, not only from drinking water).
Excess fluoride consumption beyond recommended levels, however, may cause fluorosis (a condition that results in striations in tooth enamel) as well as skeletal deformities that can sometimes be severe, resulting in
functional impairments from diminished mobility. A recent analysis concluded that EPA’s drinking water standard of 4 mg L−1 is too high to protect against adverse health effects (NRC, 2006a). Because of this potential for both positive and detrimental health effects, there has been considerable research by earth scientists to determine the range of fluoride concentrations in natural waters and to elucidate the processes controlling hydrogeochemical cycling of fluorine (Edmunds and Smedley, 2005). Concentrations in surface waters are generally much lower than the range of 0.7–1.2 mg L−1 that promotes dental health (NRC, 2006a), and typically the natural abundance of fluoride in surface waters does not result in a net health benefit. Exceptions are lakes and rivers in volcanic areas where these water bodies may receive acidic geothermal fluids containing high concentrations of dissolved fluoride. The large ranges of fluoride concentrations in groundwater partially result from the variability of fluorine concentrations in geological materials and partially because dissolved calcium limits free fluoride by precipitating minerals that incorporate fluorine. Primary mineral sources in igneous rocks include biotites, amphiboles, apatite, and natural fluorite also occurs in hydrothermal veins. Soils may contain fluoride from the parent rock material as well as from anthropogenic inputs, particularly phosphate fertilizers and sewage sludge. Fluoride concentrations are highly variable on a local scale, due to the inherent heterogeneity of geological materials and the variations in flow paths and residence times that control water-rock interactions. Accordingly, geological and hydrogeological expertise is essential for both water supply programs in areas of potentially high fluoride concentrations and research efforts designed to relate health effects, such as skeletal fluorosis, to fluoride exposure.
Microbes—Natural Attenuation
Although waterborne pathogens are a globally important health problem (see below), beneficial microorganisms in ground and surface waters are also a fundamental part of the biogeochemical cycling of elements and nonpathogenic organisms, and these can be responsible for the rapid degradation of a range of organic contaminants. Contamination of shallow aquifers by petroleum products, including both gasoline and crude oil, is widespread both in this country and around the world. During past decades, leaking pipelines, transportation accidents, leaking tanks, and well-head leakage resulted in fears of a pending environmental catastrophe. As more data were collected during site investigations and cleanup, however, it was noted that in many cases the extent of the contaminant plume originating from the floating pool of hydrocarbon rarely extended farther than about 150 m from the source, no matter how big the source was or
how fast the groundwater was moving (Mace et al., 1997). While many processes contribute to the fate and transport of hydrocarbons in groundwater, the overwhelmingly most important process is the microbial degradation of petroleum compounds. In virtually all aquifers the rate of microbial degradation of the hydrocarbon contaminant by the native microbial consortium is fast enough that, combined with other attenuation mechanisms (dilution, sorption, and volatilization), the plumes were attenuated within ~150 m. This realization substantially changed the remediation strategy for hydrocarbon contamination of groundwater, and the term “natural attenuation” has now entered the lexicon of the environmental professional (NRC, 2000a). However, natural attenuation is not as effective for chlorinated organic solvents such as TCE and perchlorate, where reduced rates of natural degradation allow plumes to travel several kilometers (Brusseau and Tick, 2006).
HEALTH HAZARDS OF WATERBORNE EARTH MATERIALS
Health hazards from drinking water arise from natural or anthropogenic contamination of source waters used for potable use. In particular, contamination of groundwater is dependent on the earth’s materials that host the aquifer. The physical properties of the subsurface result in significant differences in the behavior of groundwater compared to that of surface waters. For example, residence times for groundwater range from a few years to hundreds of years or more. Dilution effects, either in water or the atmosphere, are much less significant for groundwater compared to surface water systems. In addition, the absence of light eliminates the possibility of photochemical reactions, a major route of transformation in lakes or streams. The net result is that once groundwater and the subsurface geological units are contaminated, they are very difficult to decontaminate, and therefore pollution prevention is critical for maintaining sustainable groundwater resources.
Risk assessment is an essential element of effective management of groundwater resources. There are two components to the risk of pollution from groundwater—groundwater vulnerability and contaminant load. Groundwater vulnerability is the intrinsic susceptibility of the specific aquifer in question to contamination (see Table 4.1). An aquifer that is close to the surface, overlain by sandy soil, and located in an area with high precipitation rates would clearly be more vulnerable to contamination than an aquifer in an area of low precipitation that is hundreds of meters below ground surface and overlain by clay soils or other relatively impervious material.
Factors involved in the contaminant load are the type of contaminant, the amount of contaminant released, the timescale of release, and the
TABLE 4.1 Factors Affecting Groundwater Vulnerability to Contamination
|
Factor |
Increases Vulnerability |
Decreases Vulnerability |
|
Depth to groundwater |
Shallow |
Deep |
|
Soil type |
Well drained (sandy) |
Poorly drained (high clay, organic matter content) |
|
Vadose zone physical properties |
Preferential flow channels |
Horizontal low-permeability layers |
|
Recharge |
High precipitation, high infiltration |
Low precipitation, low infiltration |
|
Subsurface attenuation processes |
Minimal attenuation |
Significant attenuation |
|
SOURCE: Brusseau and Tick (2006). |
||
mode of release. The pollution potential of a contaminant is controlled by its transport and fate behavior. Transport of contaminants from the source zone to groundwater necessitates travel through the soil and vadose zone, where attenuation processes such as sorption and biodegradation can act to reduce and limit such transport. For this reason, the soil and vadose zone are often referred to as a “living filter.” The degree to which contaminants will be attenuated is a function of the type of contaminant and the nature of the subsurface (NRC, 2000a). Generally, the greater the amount of contamination released, the greater the pollution potential, although the timescale and mode of release can also affect pollution potential. For example, releases from buried storage tanks may be more prone to cause groundwater contamination than releases from tanks stored above ground on concrete pads.
The greatest groundwater pollution risk is associated with locations where the aquifer has a high vulnerability and the contaminant loading is also high. There are several classes of contaminants within each of the major categories of chemical contaminants: organic, inorganic, and radioactive (see Table 4.2).
A variety of substances associated with human activity can contaminate surface and subsurface water supplies, many of which are virtually unaffected by the geological framework. Nitrate, for example, is an important contaminant in water (see Box 4.1)—principally derived from sewage or fertilizer contamination (NRC, 1995)—that does not significantly
TABLE 4.2 Examples of Organic, Inorganic, and Radioactive Groundwater Contaminants
|
Organic Contaminants |
|
Petroleum hydrocarbons (fuels)—benzene, toluene, xzylene, polycyclic aromatics, methyl tertiary butyl ether (MTBE) |
|
Chlorinated solvents—trichloroethene, tetrachloroethene, trichloroethane, carbon tetrachloride |
|
Pesticides—DDT (dichloro-diphenyl-trichloro-ethane), 2,4-D (2,4-dichlorophenoxyacetic acid), atrazine |
|
Polychlorinated biphenyls (PCBs)—insulating fluids, placticizers, pigments |
|
Coal tar/creosote—polycyclic aromatics |
|
Pharmaceuticals/food additives/cosmetics—drugs, surfactants, dyes |
|
Gaseous compounds—chlorofluorocarbons (CFCs), methane, sulfur gases |
|
Agricultural fumigants—methyl bromide |
|
Inorganic Contaminants |
|
Inorganic “salts”—sodium, calcium, nitrate, sulfate, fluoride, perchlorate |
|
Heavy/trace metals—lead, zinc, cadmium, mercury, arsenic |
|
Asbestiform minerals such as crocidolite, chrysotile, and erionite |
|
Radioactive Contaminants |
|
Occurring in solids—uranium, radium, strontium, cobalt, plutonium, cesium |
|
Occurring in gaseous form—radon |
|
SOURCE: Brusseau et al. (2006). |
sorb or react with geological materials. In contrast, arsenic and radium (plus radon) are examples of inorganic solutes that have natural earth material sources and significant potential to react with the geological system. These substances have received considerable attention from both the earth science and health science communities, but many uncertainties remain related to sources, exposures, and health effects.
Arsenic
Arsenic is a metalloid element found ubiquitously in nature, occurring in rocks and soil, coal, volcanic emissions, undersea hydrothermal vents (“black smokers”), hot springs, and extraterrestrial material. It is the twentiethmost abundant element in the earth’s crust, with an average concentration of 2 mg kg−1.
Worldwide, water contamination is the most common source of exposure to environmental arsenic. Currently, the regions of the world with the largest affected populations are Bangladesh and West Bengal in India. High arsenic levels in drinking water have been also reported in Argentina, Chile, China, Colombia, Hungary, Mexico, Peru, Taiwan, Thailand, and parts of the United States (NRC, 1999e, 2001b). In Bangladesh alone (see Box 1.1), it is estimated that more than 95% of the 120 million people
|
BOX 4.1 The Nitrogen Cycle and Nitrate The global nitrogen cycle is changing faster than any other major biogeochemical cycle (Townsend et al., 2003). Reactive nitrogen, that is, nitrogen in a chemical form other than N2, has important worldwide effects on air, water, soil, and human health. Human production of reactive nitrogen is estimated currently to be about 200 teragrams per year (Galloway et al., 2003), and this figure is increasing by about 15 Tg annually. Human addition of reactive nitrogen exceeds worldwide natural production (Fields, 2004). The major contributor is fixation of nitrogen by the Haber-Bosch process (100 Tg), most of which is used as fertilizer and released to the environment. Other human sources are nitrogen-fixing crops such as legumes (40 Tg), burning biomass (40 Tg), draining wetlands (10–20 Tg), and fossil fuel combustion in vehicles and for electrical generation (20 Tg). Airborne oxides of nitrogen in the troposphere (collectively called NOx) can produce ozone, which under certain conditions can cause or worsen asthma, cough, reactive airway disease, respiratory tract inflammation, and chronic obstructive pulmonary disease (e.g., Kierstein et al., 2006). At midlatitudes, nitrous oxide (N2O) behaves as a greenhouse gas, with each molecule being about 200 times as effective in absorbing outgoing infrared radiation as carbon dioxide. In the stratosphere, N2O catalyzes destruction of ozone. In addition, nitric acid has become an increasingly significant contributor to acid rain in industrial areas of the United States. Nitrate and nitrite are the two most common forms of reactive nitrogen in water. Both are highly soluble and quickly escape below the root zone into groundwater. Nitrate levels in groundwater in agricultural areas are |
living in this region drink tube well water and more than one-third of the tube well water contains arsenic above 50 µg L−1 (the guideline value recommended by the World Health Organization is 10 µg L−1; WHO, 2001).
A range of health effects have been associated with long-term chronic arsenic exposure, including cancer (skin, lung, bladder, and kidney), atherosclerosis, and peripheral vascular disease. Epidemiological data have also suggested a link with diabetes mellitus, hypertension, and anemia (Lerman et al., 1980). Many of these studies have been conducted in populations where the exposure to arsenic has been predominantly through contaminated drinking water (WHO, 2004; Smedley and Kinniburgh, 2005).
Although results of national-scale surveys offer a general guide to regions of the United States in which arsenic concentrations in drinking water may be linked to disease (e.g., Figure 4.3), they are not comprehen-
|
several times greater than in preagricultural groundwater. Increased nitrate loading to coastal areas provides nutrients that cause uncontrollable algae blooms, often rendering the water anoxic and unfit for fish and other oxygen-requiring aquatic organisms (NRC, 2000b). In humans, ingested nitrate is reduced to nitrite in the saliva. Nitrite is highly reactive, and infants are particularly susceptible. In infants, nitrite combines readily with fetal hemoglobin to inhibit oxygen transport, causing a condition called methemoglobinemia, or “blue baby syndrome,” which can lead to death. The international maximum contaminant level of 10 mg L−1 for nitrate in drinking water has been set to prevent this condition. The adverse effects of nitrate in infants are exacerbated under some conditions, such as gastrointestinal infection. In addition, nitrite can combine with commonly occurring secondary amines and amides to form a variety of carcinogenic N-nitroso compounds. Although many vegetables contain nitrate, the presence of vitamin C and other antioxidant compounds can inhibit the formation of N-nitroso compounds. Epidemiological data relating to cancer risk of nitrate are equivocal. A number of studies have evaluated the geographic distribution of cancer incidence or mortality rates with respect to the distribution of nitrate in water. Associations were observed for gastric and bladder cancers, with uneven findings. Case control and cohort studies, which consider exposure and health outcome on an individual basis, also are inconclusive. To resolve the question of nitrate’s carcinogenicity in human populations, it will be necessary to conduct studies in larger populations and include more precise estimates of exposure. |
sive and cannot be used to reconstruct doses to individuals. Studies that have examined data from wells over smaller regions in this country have often found large variations in arsenic concentrations that do not correspond to a simple spatial pattern (Ayotte et al., 1999; Peters and Blum, 2003; Root et al., 2005). Local-scale variations in concentration have been detected as a function of the geological formation providing water to wells and the depth of wells within a given formation. In some cases, large temporal variations in a given well have also been observed as a function of pumping conditions. From the earth science perspective, the development of improved understanding not only of the distribution of geological sources of arsenic, but also of the microbial, biogeochemical, and hydrogeological processes that control the mobilization of arsenic from geological materials is a major research challenge.
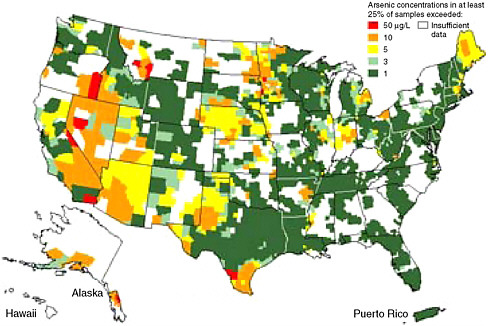
FIGURE 4.3 Arsenic concentrations found in at least 25% of groundwater samples in each county across the United States.
SOURCE: Ryker (2001).
Mercury
Mercury (Hg) in the environment is one of the most widely recognized and publicized pollutants. Natural phenomena, such as erosion of mineral deposits and volcanoes, as well as human activities such as metal smelting, coal-fired electricity generation, chemical synthesis and use, and waste disposal, all contribute to environmental mercury contamination.
Three main forms of mercury occur in the environment—elemental mercury (or quicksilver, Hg0); inorganic mercury (Hg1+, Hg2+); and organic methyl-, ethyl-, and phenylmercury. Each form has a different solubility, reactivity, and toxicity. Because of biomethylation and bioaccumulation, the effects of methylmercury (MeHg)—the most toxic of the organic forms—vastly exceed those of inorganic mercury as a result of the transport of MeHg across the blood-brain barrier as a complex with L-cysteine (Clarkson, 2002). Mercury is a potent neurotoxin which can cause developmental effects in the fetus as well as toxic effects on the liver and kidneys of adults and children. Over 60,000 babies born in the United States each year are at risk of neurodevelopmental effects from in utero exposure to methylmercury (NRC, 2000c). Sublethal effects of mer-
cury toxicity include diminished ability to learn, speak, feel, see, taste, and move. Children under the age of 15 are most vulnerable because their central nervous systems are still developing.
Although mercury toxicity can occur through skin contact, inhalation, or ingestion, the major route of human exposure to mercury released in the environment is through consumption of contaminated fish. Elevated concentrations of MeHg in fish occur even in the absence of direct anthropogenic discharges of mercury to the water bodies in which the fish live. Natural or geogenic sources of mercury also occur in lakes (Rasmussen, 1996). Elevated MeHg concentrations of fish in remote lakes are generally considered to be influenced by inputs of atmospheric inorganic mercury directly to the lakes and indirectly via their watersheds. Anthropogenic mercury emissions are probable contributors to mercury loading of lakes—these anthropogenic inputs originate as emissions from coal combustion, as waste incineration, and as emissions from other industrial and mining processes. Emissions from coal-fired power plants are a major source of mercury in the atmosphere and hence, by deposition, on land and in water. Volcanic emissions are a natural contributor of mercury to the environment, and recently regional and intercontinental dust movement has been suggested as a source of natural mercury contamination (Holmes and Miller, 2004).
Because of human activities, there has been significant redistribution of heavy metals, including mercury, from areas where they have little impact on human and animal health to areas where they can be detrimental to human health. Significant gaps remain in our quantitative understanding of the mercury cycle; although much of the process that forms MeHg is highly site specific, it is superimposed on a global cycle in which Hg(0) is the principal mercury species. Even basic questions, such as the effect on mercury concentrations in fish if atmospheric mercury deposition is reduced, or the extent of mercury toxicity where there is interaction with trace elements (e.g., selenium), have yet to be addressed. The complexity of the problem will require input from many disciplines, including public health and earth sciences.
Selenium and Molybdenum
Selenium and molybdenum frequently occur together in soils, and these trace elements can be concentrated by agricultural practices, for example, in the San Joaquin Valley in California (Ong et al., 1997). Agricultural irrigation of soils high in selenium and molybdenum results in solubilization, and ultimately bioaccumulation, of these trace elements. Kesterson Reservoir in California is a highly cited example where irrigation water that contained high selenium levels resulted in birth defects
and embryonic abnormalities in birds (Ohlendorf et al., 1990). The resulting restrictions on utilization of subsurface drainage have led to innovative technologies to reduce drainage volumes. These include reutilization of drainage water to irrigate salt-tolerant crops or halophytes, so that the volume of drainage water is reduced prior to salt concentration via solar evaporators (Oster and Grattan, 2002). The role of selenium ingested in food is described in more detail in the next chapter.
Radium and Radon
Radioactive contaminants occur naturally in groundwater originating from geological sources or are present in surface water as the result of contamination from a range of sources that include weapons testing, nuclear power plants, landfills, and medical applications. Radioactive concentrations may be several times higher in groundwater than in surface water (NRC, 1999c). Exposure routes for radium and radon include direct consumption of contaminated water and by inhalation (Appleton, 2005); development of lung cancer has been linked to household inhalation of radon (see Chapter 3).
Microbes
Microbial pathogens include bacteria, viruses, and protozoan parasites. Bacteria are common infectious agents implicated in many waterborne disease outbreaks—Vibrio cholerae, Helicobacter, Campylobacter, Salmonella, E. coli, and Shigella are bacteria known to cause infections and even death. Many bacterial diseases are due to failures of water treatment systems, resulting in consumption of untreated groundwater or surface water (e.g., Gerba and Pepper, 2006). One recent example involved inadequate treatment of the water supply in Walkerton, Ontario, which enabled high levels of a pathogenic strain of E. coli 0157:H7 to contaminate the entire drinking water system, resulting in seven deaths and hundreds of illnesses (McIlroy, 2001). Worldwide, waterborne diseases result in millions of deaths each year. Some 2.4 billion people in developing countries still have no access to basic sanitation, resulting in illnesses such as typhoid fever, dysentery, and cholera (WHO and UNICEF, 2000).
The role that viruses play in the waterborne transmission of human diseases is less well understood than that of many bacteria and protozoa, mainly due to the difficulties associated with detecting viruses in water. There are many routes of exposure, including consumption of drinking water polluted by viruses, consumption of shellfish harvested from con-
taminated water, consumption of food crops grown in soil irrigated with wastewater or fertilized with sludge, and contact with contaminated recreational water. Human enteric viruses are small (25–100 nm) and are encased in a resistant structure that protects them from environmental degradation and disinfection (Sobsey, 1989). Norwalk virus, the most widespread human calicivirus, causes outbreaks of waterborne and foodborne viral gastroenteritis. According to the Centers for Disease Control and Prevention (CDC), more than 96% of reported outbreaks of nonbacterial gastroenteritis characterized by nausea, vomiting, diarrhea, and an illness lasting one to three days are caused by Norwalk virus (Fankhauser et al., 1998), with an estimated 23 million cases per year in the United States (Mead et al., 1999).
Protozoan parasites are eukaryotic organisms that have been implicated as agents of waterborne disease. These include Giardia lamblia and Cryptosporidium parvum in particular, and the emerging pathogens Microsporidia and Cyclospora. Another emerging pathogen, Naegleria fowleri, is found in both soil and surface water environments. This protozoan parasite infects humans by entering via the nose and subsequently travels to the brain, where it multiplies in the central nervous system and ultimately results in death via primary amebic meningoencephalitis (Zhou et al., 2003). This parasite has recently been detected in 8% of drinking wells in southern Arizona (C.P. Gerba, University of Arizona, personal communication, 2006).
The transport of microorganisms through soils or the vadose zone is affected by a complex array of abiotic and biotic factors, including adhesion processes, filtration effects, soil characteristics, water flow rates, predation, the physiological state and intrinsic mobility of the cells, and the presence of biosolids. Viruses have a large potential for transport, although transport is limited when they adsorb to soil colloidal particles and biosolids. Virus sorption is controlled by the soil pH. The larger size of bacteria means that soil acts as a filter, limiting bacterial transport. Soil should also limit the transport of the even larger protozoa and helminthes. Soils and aquifers in fractured rock and porous media contain zones of preferential flow. Both water and small particles, including inorganic colloids and microorganisms, may be transported through these preferential flow zones and macrochannels very rapidly, and since particles are not subject to diffusion into the finer porosity matrix material, the result is extremely rapid transport. The transport properties of pathogens passing through heterogeneous porous and fractured media and colloid-facilitated transport of contaminants are a critical area of research that will demand expertise in hydrogeology, rock mechanics, geophysics, microbiology, and public health.
Pharmaceutical Substances
There are thousands of organic compounds that can potentially contaminate potable water sources, and many studies have examined the fate and transport of various classes of organic contaminants found in drinking water. Here the focus is on pharmaceuticals, including endocrine disrupting compounds (EDCs)—chemicals that modify the function of endocrine glands and their target organs (NRC, 1999b; Arnold et al., 2006; see Box 4.2)—as an example of an emerging organic contaminant category that occurs in groundwater and is a concern in many countries (Plant and Davis, 2003).
In 1999–2000 the U.S. Geological Survey (USGS) carried out a comprehensive reconnaissance in streams throughout the country that are potentially affected by human activities (Kolpin et al., 2002). Up to 95 trace chemicals frequently present in municipal wastewater were measured at 139 sites. Eighty percent of the waters tested by the USGS contained at least one of the 95 trace contaminants, and 82 of the 95 were present at one or more of the 139 sites (see Table 4.2).
Many studies of the potential for adverse effects from exposure to EDCs in humans and wildlife have been carried out in the United States and in Europe (e.g., NRC, 1999b; Arnold et al., 2006). EDCs in wastewater effluent have adversely affected fish and other wildlife, but there is con-
|
BOX 4.2 Endocrine Disrupting Compounds Endocrine disrupting compounds (EDCs) have a number of characteristics:
|
TABLE 4.2 Hormones and Hormone Mimics Observed in U.S. Surface Waters
siderable controversy as to whether human health has also been adversely affected by exposure to endocrine-active chemicals because of inconsistent and inconclusive results. Dose-response relationships are likely to vary for different chemicals and endocrine disrupting mechanisms, and such relationships may be species dependent. The exposure sets that do exist are primarily from chemical levels in various environmental media such as air, food, or water and may not reflect internal concentrations in blood or endocrine-regulated tissues. Exceptions to this are human breast milk and adipose tissue (e.g., Swan et al., 2005). Overall, more research is needed. It remains difficult to assess the risk to human and animal health from endocrine disruptors due to the necessity to extrapolate from low-dose exposures. An additional difficulty in assessing risk from endocrine disruptors is the possible synergistic effect from other environmental hazards. Because controversy will continue to surround EDCs and their potential short- and long-term risks to environmental and human health and welfare, EDCs remain an “emerging issue.”
OPPORTUNITIES FOR RESEARCH COLLABORATION
There is a rich array of opportunities for earth and public health scientists to collaborate on research that addresses health and drinking water quality. The earth science component of this research relates to improving the understanding of sources, transport, and transformations of potentially hazardous substances in water to ultimately determine the concentrations to which people are exposed through their drinking water. The health components of the research relate to quantifying and understanding the mechanisms of human responses to these exposures. The overall goal in all cases is to be able to predict potential health effects based on improved process-based understanding and, where appropriate, through modeling. Prediction of potential adverse health effects will provide the basis for development of effective prevention or mitigation measures related to either the water source or the human health response. High-priority collaborative research activities are to:
-
Determine the health effects associated with water quality changes induced by technologies and other strategies currently being implemented, or planned, for extending groundwater and surface water supplies to meet increasing demands for water by a growing world population. Of particular interest with respect to groundwater are changes in water quality induced by:
-
changes in rates and locations of groundwater extraction;
-
treatment of sewage effluent for potable reuse;
-
-
-
artificial recharge using stormwater and treated wastewater;
-
water “banking” via injection or aquifer storage and recovery;
-
extraction of brackish groundwater for desalination;
-
introduction of imported or recycled irrigation water; and
-
changes in land use, vegetation, and irrigation practices that alter rates of infiltration, drainage water runoff, and evapotranspiration.
-
All of these have the potential to alter the major and minor ion composition of groundwater. For example, fresh surface water stored in a brackish aquifer during aquifer storage and recovery will experience an increase in total dissolved solids due to mixing with ambient brackish water and dissolution of minerals from the aquifer matrix. Many of these may also introduce contaminants such as microbial pathogens, organic contaminants such as pesticides or solvents, and inorganic contaminants such as nitrates or metals. Of particular interest with respect to surface water are changes in water quality induced by urban and agricultural runoff, discharge of waste effluents from municipal or industrial sources (including the extractive mineral and energy industries), construction and operation of dams and reservoirs, drainage of wetlands, and channel modifications for purposes of flood control, navigation, or environmental improvement.
-
Identify and quantify the health risks posed by “emerging”contaminants, including newly discovered pathogens and pharmaceutical chemicals.The health effects of many naturally occurring substances at low concentrations and the health effects associated with interactions of multiple naturally occurring substances are poorly understood. Public health professionals and earth scientists will need to collaborate to identify emerging substances of potential concern and to improve understanding of the processes controlling the mobility of these substances in the environment, particularly in light of potential changes in concentrations induced by human activities that alter the land or the hydrological cycle. Of particular interest are “emerging” contaminants such as hormones, pharmaceuticals, personal care products, and newly identified microbial pathogens for which sources and transport processes are poorly understood. The synergistic and antagonistic interactions of mixtures of contaminants with naturally occurring substances in water also pose priority research questions. Examples of specific research priorities include an understanding of the:
-
fate and transport of prions from soil to groundwater and surface water and their relationship to disease incidence;
-
-
-
fate and transport of viruses through soil and vadose zones to groundwater and their relationship to disease incidence;
-
fate and transport of Naegleria fowleri from soil to water and disinfection strategies for contaminated wells;
-
fate and transport of endocrine disruptors through soil to groundwaters and the influence of long-term, low-level exposure on human health; and
-
fate, transport, and human health effects of perchlorate from soil and groundwater.
-




















