2
Potential Risks Associated with Hormone Treatment
Louise Brown, born on July 25, 1978, was the world’s first baby conceived via in vitro fertilization (IVF). And the egg that produced Louise Brown was created in her mother’s body in a completely natural way, without medications from the doctors who were helping Lesley Brown conceive.
Although Louise Brown’s birth is proof that it is possible to harvest an egg from a woman and fertilize it without the use of fertility drugs, this approach is inefficient. An egg must mature, must be retrieved and fertilized, then the resulting embryo must divide and grow in the laboratory, and finally it must implant in the uterus. At each stage, the process may be compromised, resulting in a small chance of success in each IVF cycle. For this reason, in the 1980s assisted reproduction specialists began treating the prospective mother—or the egg donor, if the eggs are coming from a woman other than the mother—with a series of hormone injections designed to increase the number of eggs that come to maturity in a given cycle. Thus, multiple eggs could be retrieved at one time with greatly improved chance of a successful outcome.
Today doctors have had two decades of experience with the use of hormone treatments to maximize the number of eggs that can be harvested from a woman, and they have become quite proficient in the production of oocytes. During that time they have also worked to improve the safety of the procedure and decrease the potential risks. Despite these improvements some risk will remain, because hormones have a powerful effect on the body—they could not increase egg production so dramatically if this were not true—and anything with a powerful effect on the body has the potential for harmful side effects as well.
Experience suggests that there are three main risks associated or potentially associated with the hormone treatment used in ovarian stimulation: ovarian hyperstimulation syndrome, cancer, and effects on future fertility. Each risk has its own characteristics and its own implications for egg donors.
OOCYTE PRODUCTION
To understand the potential risks associated with the hormone therapy used in egg donation, one must first understand the hormone therapy itself. And that, in turn, requires an understanding of how the body produces eggs, without outside intervention from administrated hormones.
When a baby girl is born, her ovaries contain roughly 2 million oocytes, each encased in a protective covering called a follicle (see Figure 2-1). At this point, all of the follicles are primordial follicles—follicles that have not taken the first steps down the developmental path that leads eventually to mature follicles. And each of these primordial follicles will sit dormant in a woman’s body for as long as 50 years or more, waiting for a signal—researchers are still not sure exactly what that signal is—that will cause that follicle to leave the primordial pool and begin slowly to mature.
Whatever that trigger is, throughout a woman’s life there is a steady exodus of these follicles from the developmental pool and down the developmental pathway that will lead eventually—if the follicle survives—toward maturity and ovulation. But only a very few survive, as noted at the workshop by Nicholas Cataldo, formerly an assistant professor of obstetrics and gynecology at Stanford University School of Medicine. At the time of a woman’s first menstrual period, she still has 400,000 or so of these primordial follicles, and by the time of menopause they are almost all gone—indeed, it is their absence that triggers menopause—which implies an average attrition of about 1,000 follicles a month. That attrition, Dr. Cataldo said, can occur anywhere along a follicle’s developmental path, from its first step as a primary developing follicle, through its secondary follicle stage, and on to its final stage as an antral follicle. It is the antral follicle that, under the influence of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), will grow into a large mature follicle with a mature oocyte in its fluid core.
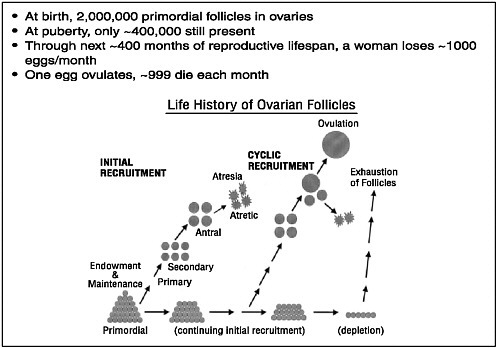
FIGURE 2-1 Follicle/egg number through the life span.
SOURCE: McGee and Hsueh, 2000.
Reprinted with permission from McGee, EA, Hsueh, AJWß. 2000. Initial and cyclic recruitment of ovarian follicles. Endocrine Reviews 21(2):200-214. Copyright 2000, The Endocrine Society.
Each month at the onset of the menstrual period, between 10 and 20 follicles will normally reach the antral stage, at which they are ready to grow into mature follicles under the influence of those hormones. Hundreds of other follicles that started down the developmental path at approximately the same time have regressed and have been reabsorbed by the body in the process known as atresia.
Once at the antral stage, the follicles require a certain level of FSH to survive and grow. In prepubertal girls and women taking birth control pills, without high enough levels of FSH to enable them to grow, the small antral follicles remain dominant. In a normally cycling woman, however, FSH levels begin to rise at the start of each menstrual cycle, and, under that influence, the antral follicles begin to grow. One of them will generally be slightly ahead of the others in its development, which gives it the advantage of being slightly more sensitive to FSH, which in turn causes it to grow faster than the other follicles and widen its lead.
This lead follicle is termed the “dominant follicle,” Cataldo said, because it acts as the dominant force in the ovary. “It basically makes the smaller ones die,” he said, and it does this by producing estradiol and possibly inhibin B as well, both of which are hormones that signal the pituitary gland to cut back on its production of FSH. With less FSH arriving in the ovaries, the smaller follicles do not have enough of the hormone to keep developing and avoid atresia. The dominant follicle, in contrast, is able to survive because of a mechanism that increases its sensitivity to FSH and allows it to keep growing with less FSH than the other follicles need. This process of selecting a single dominant follicle is the reason that women tend to ovulate only one egg per cycle, Cataldo explained. This limit is known to biologists as the ovulatory quota, and it is different from species to species. For example, pigs generally have litters of 6 to 12 piglets; and cows typically have 1 or, at most, 2 calves.
OVARIAN STIMULATION
Although only the dominant follicle survives to produce an oocyte in a normal monthly cycle, the other antral follicles can also survive and grow if there is enough circulating FSH. This is basic of the hormone therapy used to stimulate the ovaries and increase the number of oocytes that a woman can provide for assisted reproduction or for research.
The standard hormone therapy involves daily injections of gonadotropins—most often hormones with an action similar to FSH—beginning on about the third day of menstruation and lasting for about 10 days (see Figure 2-2), after having begun injections of a gonadotropin-releasing hormone (GnRH) to prevent premature oocyte release from the follicles. An alternative is to give the drug clomiphene to induce the body’s pituitary gland to release more FSH. With the right amount of hormones, all or almost all of the antral follicles will continue to grow. Occasionally, a GnRH agonist or a GnRH antagonist is added to the mix in order to prevent the body’s normal LH surge. Then, when ultrasound shows that the follicles have all reached the proper stage of maturation, with their oocytes ready for ovulation and fertilization, yet another hormone—human chorionic gonadotropin (hCG)—is given. This hormone would normally cause the follicles to ovulate and release their eggs in about 36 hours, but in practice the physician will retrieve the eggs from the follicles before that happens.
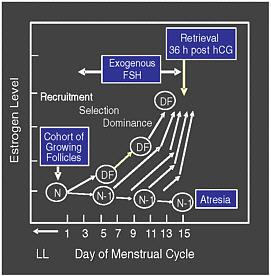
FIGURE 2-2 Follicular growth and selection.
SOURCE: Racowsky (2006). Reprinted with permission from the author.
Thus, as Marcelle Cedars, director of the Division of Reproductive Endocrinology and Infertility at the University of California, San Francisco, noted, hormone therapy acts to increase the number of available eggs in a way that is much different from how most people assume it works. “While we talk about stimulating donors, we really misspeak,” she said. “I have to explain this to patients quite frequently because the implication is that I can make more eggs be there. The reality is what I’m doing is rescuing those eggs from the antral stage forward that would otherwise undergo atresia.” And this, she noted, is an important point in understanding what the potential risks are for women who undergo this hormone treatment.
POTENTIAL RISK OF OVARIAN HYPERSTIMULATION SYNDROME
The most common side effect of the use of fertility drugs is what is called ovarian hyperstimulation syndrome (OHSS), Dr. Cedars said. “In essence,” she explained, “ovarian hyperstimulation is exaggeration of a desired response. We want to override a mechanism of getting a single egg.” A key factor in the development of OHSS is the administration of
the ovulation-inducing hCG; the syndrome will not occur in the absence of hCG.
Doctors diagnose OHSS by looking for a characteristic collection of symptoms. The ovaries are increased in size, and there may be gastrointestinal symptoms, such as nausea. The blood vessels become more permeable, and this leads to an accumulation of fluid that can collect in the abdomen and cause discomfort. If there is enough fluid buildup, the abdomen can become somewhat distended, which may lead to pressure on the diaphragm, which in turn can lead to shortness of breath and labored breathing. Blood volume may decrease, leading to an increased concentration of red blood cells.
Traditionally, cases of ovarian hyperstimulation syndrome were classified as mild, medium, or severe (see Box 2-1), Dr. Cedars said, but today the mild cases are not generally considered to be ovarian hyperstimulation syndrome at all, but rather a normal response to the hormone treatment. “It’s probably true that most women who undergo ovarian stimulation will have some mild symptoms of hyperstimulation,” she explained. Indeed, about 10 percent of women feel a lower abdominal or pelvic pain—called mittelschmerz—about halfway through a normal menstrual cycle, even though they have only a single follicle growing to its full size of about 20 millimeters. Since ovarian stimulation generally results in 10 or more follicles growing to a large size, it is not surprising that some women should feel pain as their ovarian capsule stretches to accommodate the multiple follicles that are growing. The mild cases generally have no serious complications, resolve themselves spontaneously, and are quite common in women after hormone treatment.
Moderate cases of ovarian hyperstimulation syndrome are characterized by more than minimal discomfort, a significant fluid buildup (ascites) in the abdomen, leading to a shortness of breath (dyspnea) because of the pressure on the diaphragm, and nausea and vomiting. The blood volume and concentration of red blood cells are normal, however, and the patients are usually handled on an outpatient basis.
Serious cases are relatively rare—about 100 to 200 cases for every 100,000 stimulation cycles—but they are the ones that are most worrisome to assisted reproduction specialists (Schenker and Ezra, 1994; Budev et al. 2005). They in turn are divided into three categories: A, B, and C (see Box 2-2).
|
BOX 2-1 Ovarian Hyperstimulation Classification
SOURCES: Schenker and Ezra, 1994; Delvigne, Rozenberg 2002; Budev et al. 2005. |
|
BOX 2-2 Severe Ovarian Hyperstimulation Classification
|
It is difficult to get good data on how often such serious complications occur, Dr. Cedars said, but there are some studies that offer an indication. A recent study from Finland, for example, found that kidney failure occurred in about 1.4 percent of patients who had severe ovarian hyperstimulation, or about 1 in 100,000 oocyte stimulation and retrieval cycles. Somewhere between 0.78 percent and 2.4 of patients with the severe form of the syndrome experience blood clots. This would translate into about 0.78 to 2.4 cases for every 100,000 stimulation cycles. Other
potential complications, such as adult respiratory distress, are so rare, Dr. Cedars said, that she could find no data to provide an estimate of the rate of occurrence.
It is also difficult to estimate a mortality rate, but, conservatively speaking, death appears to occur at a frequency between once every 450,000 and once every 50,000 egg donation cycles (among women with severe OHSS). The numbers are misleading, however, because they include patients who become pregnant with the eggs retrieved from their ovaries and later form blood clots during the pregnancy.
Generally speaking, Dr. Cedars warned, the data concerning the occurrence of ovarian hyperstimulation syndrome are not particularly good. For example, there are very few prospective studies—in which an investigator watches a large cohort of women from before the time they undergo ovarian stimulation until long enough afterward to know the final outcome. Three prospective studies that had reasonable sample sizes reported a risk for OHSS to be between 2.1 to 4.7 percent.
Without the control that comes from a prospective study, it is difficult to get a good measure of the rate of occurrence, and it is difficult to know what the risk factors were for each patient. And without knowing what the risk factors are, it is hard to identify groups of women who are particularly likely to suffer from ovarian hyperstimulation.
Finally, very few of the studies divide cases of the syndrome into early- and late-occurring classifications. Early cases of ovarian hyperstimulation occur within three to seven days of the injection of hCG given to start the ovulation process, and these early cases are caused by that hCG trigger. The late form of ovarian hyperstimulation, by contrast, occurs 12 to 17 days after the hCG trigger and happens in women who have become pregnant with their own fertilized eggs put back into the uterus after the egg retrieval process. In this case, the hyperstimulation syndrome is triggered by hCG from the placenta released early in the woman’s pregnancy.
The distinction between early and late is important, Dr. Cedars said, because the risk of severe complications appears to be about 4 to 12 times higher among women with the late-onset hyperstimulation syndrome. It is particularly important for the issue of donating eggs for research purposes, since these donors will not become pregnant immediately after donating their eggs and thus will not be affected by the late-onset type of hyperstimulation syndrome.
Doctors can use a number of strategies to help egg donors avoid hyperstimulation (see Box 2-3), Dr. Cedars said. The first is simply
to identify potential donors who are at high risk of developing ovarian hyperstimulation and tailor the stimulation to the individual patient’s physiology. One risk factor is simply youth: the younger a woman is, the more primordial follicles she has remaining in her primordial pool, and the more antral follicles she will have available for rescue. Thus, to avoid hyperstimulation in younger women, they should receive minimal or mild stimulation. By working from such information as a patient’s age, weight, and follicle count, Dr. Cedars said, a doctor can begin with an FSH dose based on those factors and then modify it as necessary. “We monitor during the course of the stimulation to further decrease the dose if too many follicles are developing or the estradiol levels are too high.”
A second group of women at high risk are those with ovulatory abnormalities, in particular, women with polycystic ovarian syndrome (PCOS). PCOS is an endocrine disorder that affects 5-10 percent of women and is characterized by irregular or missing ovulation, a higher than usual level of androgens, or male hormones, and multiple cysts in the ovaries. These cysts are follicles that have grown to the small antral stage but, because of abnormal hormone levels, never grew further and do not release an egg. Reproductive specialists have recognized for many years that stimulating these women’s ovaries, even in a very mild way, puts them at a high risk for hyperstimulation. And it now appears, Dr. Cedars said, that the at-risk group is larger than this. Even women who don’t have all the classic symptoms of PCOS—they may have regular ovulatory cycles, for example—but who have polycystic ovaries are still
|
BOX 2-3 Ovarian Hyperstimulation Syndrome Strategies for Prevention
|
at risk for hyperstimulation. Other risk factors include irregular menstrual periods and low body weight.
It is also makes sense to try different modifications of the hCG part of the treatment, she noted, since hCG acts as the trigger for the hyperstimulation. One approach, for example, is to decrease the hCG dose. A second would be to use recombinant LH in place of the hCG. LH has a similar effect to the hCG but has a shorter half-life, so it does not remain in the system as long and therefore might not be so likely to cause hyperstimulation. There is preliminary evidence to suggest that both of these approaches decrease the occurrence of ovarian hyperstimulation.
In summary, Dr. Cedars said, she thinks oocyte donation for research can occur safely. It is not possible to completely eliminate ovarian hyperstimulation, but it is possible to limit its incidence and severity. A strategy for doing that might include excluding women from donating their eggs who have irregular menstrual cycles, who have ovaries with a polycystic appearance, and perhaps even those with high levels of androgens, as well as modifying the hormone treatment regimen to minimize the factors that are known to make hyperstimulation more likely, such as a higher than normal egg follicle count.
POTENTIAL RISK OF CANCER
One of the most serious concerns about ovarian stimulation is that it may increase the chances that a woman will suffer certain types of cancer later in her life (see Figure 2-3). In particular, said Roberta Ness, chair of the Department of Epidemiology at the University of Pittsburgh, there are three types of cancer that would seem to have a plausible biological link to the hormone regimens used in ovarian stimulation: breast, ovarian, and endometrial cancers.
Breast cancer, she noted, is the most common form of cancer among women and the second most common cause of death for women. Ovarian and endometrial cancers are not as common but are still dreaded among women because of their fatality rates.
There are several reasons to be concerned that the hormones used in assisted reproduction might make these three cancers more likely, Dr. Ness said. First, all three of them seem to be affected by hormones. The cells of these three types of cancer all have estrogen receptors, for example, and women who have had children, women who have breast fed, and
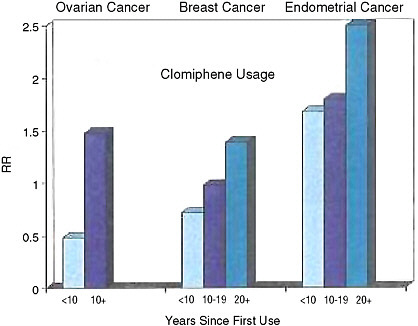
FIGURE 2-3 Clomiphene usage and risk of ovarian, breast, and endometrial cancers.
SOURCE: Louise Brinton, National Cancer Institute, personal communication, 2006.
women who had their ovaries removed are all at a lower risk for these cancers—which implies that hormones have some effect on them. Conversely, women who have a longer than average length of time between their menarche and their menopause are at a higher risk of developing these cancers.
Medical researchers think, Dr. Ness said, that breast tumors grow in response to a combined exposure to estrogen and progesterone. Endometrial cancer seems most affected by estrogen alone. And the risk of ovarian cancer may be increased by an increase in ovulation over a woman’s lifetime and by exposure to gonadotropins—i.e., mainly LH and FSH.
Given this, it seems reasonable to suppose that the hormones used in assisted reproduction may well have some effect on a woman’s risk of developing these three types of cancer, and the particular effect would depend on the details of the hormone’s actions. According to Dr. Ness, “Clomiphene should increase the risk of ovary cancer, perhaps even re-
duce the risk of breast cancer and perhaps the risk of uterine cancer dependent on its impact on the uterus as a selective estrogen receptor modulator (SERM).” The only way to know for sure, however, is to perform studies of women who have taken the hormones in the course of assisted reproduction therapy and compare their risk of cancer with controls who did not have the hormone therapy but who were similar in all other ways. However, that is not as easy as it sounds, Dr. Ness said, and one must be careful in interpreting the studies that have been done.
For example, infertility increases the risk of all three cancers, Dr. Ness said, so a study that compared women undergoing IVF with women in the general population might find the IVF group with a higher rate of cancer—but not because of the fertility drugs they had taken but rather because the infertility that led them to try IVF also made them more likely to develop these cancers. Women who receive assisted reproduction therapy may be more likely than others to get pregnant, and since pregnancy lessens the risk of the cancers, that can have an effect in the opposite direction, making it seem as though ovarian stimulation is less risky than it really is. Different drugs may work via different mechanisms, Dr. Ness pointed out, which means they would have different influences on cancer risk. Nor are all cancers the same. Different types of endometrial cancer, for example, may have different risk factors. All of these complications make it much more difficult to find the real relationship between ovarian stimulation and cancer risk.
With those caveats, Dr. Ness described what is known from epidemiological studies of the cancer risk for women who have had hormone treatment as part of an assisted reproduction program.
In the case of breast cancer, a systematic review of earlier breast cancer studies was published in 2005. Including more than 60,000 women who had undergone ovarian stimulation, it found that most of the 15 studies reviewed saw no significant association between this treatment and the risk of breast cancer.
For ovarian cancer, there have been two published meta-analyses that take data from a number of studies and do a combined analysis. One study in 2002 analyzed 8 case-control studies that included about 5,000 women who had taken fertility drugs and another 7,000 controls who had not. The bottom line, Dr. Ness said, was that the women who had taken fertility drugs had a rate of ovarian cancer basically identical to the rate for the women who had not taken fertility drugs. Thus, existing data do not support an increased risk for ovarian cancer among women who have taken fertility drugs. At this point in time, however, the state of knowl-
edge is not conclusive, and clarification of the exact relationship between ovarian cancer and treatment with fertility drugs will require additional long-term follow-up studies.
Analyzing the data in various ways—differentiating between women who took clomiphene and those who took gonadotropins, for example, and including women who had never been pregnant as the controls—still failed to show any increased cancer risk caused by fertility drugs, with one possible exception. When the cases of ovarian cancer were grouped into borderline cancers and invasive cancers, an increased risk of borderline cancers was seen among women who had taken fertility drugs. But these findings should be interpreted with caution, due to the possibility of surveillance bias. According to Dr. Ness, “Borderline cancers are ones that have a much, much better prognosis,” she said, “and they're ones that you would more readily find if you were looking for them.” Thus, since women who are undergoing assisted reproduction are more often clinically evaluated by their doctors, it is possible that their asymptomatic borderline tumors may be more commonly detected.
A second meta-analysis of assisted reproductive technologies and ovarian cancers looked not only at case-control studies but also cohort studies. The advantage of the cohort studies, which followed groups of women over a period of time after they took the fertility drugs, is that they make it possible to determine the relationship over time between the hormone exposure and the cancer risk.
For the case-control studies, there did appear to be a 50-percent increase in risk for ovarian cancer among the women who had undergone ovarian stimulation—but only when the control group was the general population. When only infertile controls were used, which is the more appropriate comparison, no increase in risk could be seen at all. And in the cohort studies, there was no increased risk at all. “All of that leads us to a conclusion,” Dr. Ness said, “that there's really not much going on.”
As for the relationship between fertility drugs and uterine cancer, there have been very few studies. “People didn't turn their attention to uterine cancer until fairly recently,” Dr. Ness explained. A review published in 2005 that analyzed the few existing studies on the subject found that women who had taken fertility drugs did have a higher rate of uterine cancer than the general population. The same study looked at the relationship between clomiphene use and uterine cancer and did control for infertility and other confounders, resulting in what Dr. Ness called “a pretty darn fair analysis.” The results indicated a possible increase in risk
with clomiphene use, but there were not enough subjects for the results to be statistically significant.
Looking more closely at the data, Dr. Ness said, there does seem to be some reason for concern. The numbers of women in the analyses are very small, so it is difficult to attain statistical significance for the results, but there are trends that deserve a closer look. With increased dosage, with a greater number of cycles of using the fertility drugs, and with more years since first use, the number of uterine cancer cases seemed to be going up. One study in particular focused just on women who had taken clomiphene and looked at the rates of various cancers. It found that as time elapsed since the treatment, there did seem to be an increase in risk for breast, ovarian, and endometrial cancers, with the highest risks for the endometrial cancers. This is of particular concern, Dr. Ness said, because it raises the possibility that many studies have missed the increased cancer risk because they haven’t followed their subjects for enough years.
Dr. Ness summarized what is known about fertility drugs and cancer risk this way: “There's no evidence that fertility drugs elevate the risk of breast cancer. There are a couple of little signals in maybe one study, but if we look overall at the literature, it is not terribly convincing. Infertility, not the assisted reproduction therapy, certainly increases the risk of ovarian cancer. There is no systematic evidence at this point that fertility drugs elevate the risk for invasive ovarian cancer. But for uterine cancer, where the data are too sparse to lead to any conclusion, I think that there's a greater concern. And the final concern is that these effects may not be evident until a longer period of time has elapsed between the exposure, the assisted reproduction therapy, and the cancer.”
POTENTIAL LONG-TERM FERTILITY EFFECTS
One of the major concerns that has been raised about the possible risks involved with hormone treatment is that the treatment may have some effect on a woman’s long-term fertility. “We've heard a lot about ovarian stimulation as a route to achieving more eggs,” Dr. Cataldo said. “The question exists whether this results in a depletion of the woman's egg supply. This is an important question, because if this were true, the retrieval of 20 eggs instead of the ovulation of 1 per cycle for 5 or 10 donation cycles might imply a considerable number of oocytes lost. And one might also worry that this could hasten the onset of age-related infer-
tility and even hasten the onset of menopause. If this were the case, repeated stimulations would be more problematic than one or two donation cycles.”
There is strong evidence against this idea, he said. The evidence comes from two sources: what is known about basic ovarian physiology and clinical experience.
As described above, a woman has some 400,000 primordial follicles when she first begins to menstruate, and she loses on average about 1,000 of those each month until she reaches menopause. Of those 1,000 per month, only 10 to 20 reach the stage of antral follicles; the rest die at various stages along the developmental path. Normally only one of the antral follicles completes its development and ovulates, with the rest dying and being absorbed by the body, but hormone treatment can rescue most of those.
The first thing to notice, Dr. Cataldo said, is that, according to the current understanding, ovarian stimulation does not cause a woman to lose any more eggs in a given month than she normally would, as all of the extra eggs made available by the hormone treatment are eggs that were slated for atresia anyway. But is it possible that the hormone treatment might somehow affect the rate at which the primordial follicles develop and so increase their rate of attrition throughout a woman’s life, causing her to exhaust her egg supply sooner than she otherwise would? Again, what is known about ovarian physiology suggests that this does not happen.
In particular, Dr. Cataldo said, throughout the entire process of follicular development, the hormones FSH and LH affect the follicle only during the last two weeks of its development. For the rest of the time, from the moment a follicle is pulled out of the primordial pool to the point at which it reaches the antral follicle stage, the development of the follicle is largely independent of those two hormones. And, since these are the hormones that are used in ovarian stimulation, it seems unlikely that the treatment would have any effect on follicular development prior to the antral stage.
Dr. Cataldo summarized: “The biology of follicle development predicts that there should be no reduction of follicle supply through repeat stimulation and hence no increase in infertility resulting from a decrease in egg supply.”
Furthermore, clinical experience backs up this conclusion. Dr. Cataldo described two clinical studies of women who had had repeated treatments of ovarian stimulation. The first were women from a Dutch
IVF program who had up to six treatments over a period of up to two to three years. “What you can see here is that the number of oocytes retrieved in successive cycles does not fall off, suggesting that we’re not pulling oocytes away from the next cycle each time we do a stimulation, and there doesn't seem to be any attrition in the response.”
A study of women from a Spanish IVF program reached a similar conclusion. These women donated eggs from one to nine times with a median of four months between cycles, so some of the women experienced hormone treatments stretching out over as much as three years. “Although the numbers are very small at the high numbers of cycles,” Dr. Cataldo said, “if you look at just the first 4 or 5 cycles, there appears to be absolutely no fall-off at all in terms of the ability to recruit roughly 15 or 16 oocytes per cycle from these donors in repeated use.”
So as far as either basic ovarian physiology or clinical experience indicates, there is no reason to think that repeated ovarian stimulation poses a risk to a woman’s long-term fertility. Still, Dr. Cataldo said, not everything is known on the subject, and there are several potentially important questions that have not yet been answered.
It would be helpful, for example, to have a longitudinal study that followed women who had ovarian stimulation all the way to menopause to find out what their reproductive future holds. Do women who have donated their eggs experience higher rates of infertility? And, if so, are there characteristics among these women at the time of their donation that are predictive of the later infertility? Are certain forms of infertility more common among woman who have donated than among the general population? And the ultimate milestone: Do women who have donated their eggs undergo menopause at an earlier age?
SUMMARY: WHAT WE KNOW ABOUT THE RISKS OF OVARIAN STIMULATION FOR OOCYTE PRODUCTION
To increase the number of eggs that can be retrieved from a donor, the usual strategy is to put the donor through a regimen of hormone shots that (1) keep most or all of the donor’s antral follicles continuing down the path to maturation instead of just one; (2) prevent the follicles from ovulating before the desired time; and (3) when it is time, prepare the follicles for the harvesting of the oocytes. The hormones used in this
regimen are known to have or suspected of having a variety of health effects, some minor and some potentially major.
The most prominent side effect of ovarian stimulation is ovarian hyperstimulation syndrome. Its symptoms include increased ovarian size; nausea and vomiting; increased permeability of the blood vessels, leading to an accumulation of fluid in the abdomen; breathing difficulties; hemoconcentration, or an increased concentration of red blood cells; kidney and liver problems; and, in the most severe cases, blood clots or kidney failure. The severe cases affect only a very small percentage of women who undergo in vitro fertilization—about 0.1 to 0.2 percent of all treatment cycles—and the Class C severe, or the most dangerous, are an even smaller percentage. Only about 1.4 in 100,000 cycles leads to kidney failure, for example.
The OHSS risks for egg donors are expected to be much lower than the OHSS risks calculated from women involved in IVF. OHSS occurs at two stages: early, 3 to 7 days after the hCG trigger is used to prepare the eggs for retrieval, and is a result of that trigger; and late, 12 to 17 days after the trigger, and is a result of the new pregnancy in a women who has successfully undergone IVF. The risk of severe complications is about 4 to 12 times higher in late-onset OHSS than in early-onset OHSS. Egg donors, because they will not be getting pregnant after donating their eggs, will not be affected by the late-onset OHSS and thus can be expected to have many fewer side effects than are seen in IVF patients.
Many observers have worried that the use of fertility drugs could lead to an increased risk of cancer—in particular, breast, ovarian, and uterine (including endometrial) cancers. One must be careful in interpreting epidemiological studies of women taking fertility drugs, because all of these cancers are more common in women with infertility, so merely comparing women taking fertility drugs with women in the general population inevitably shows an increased cancer risk. When the analysis is done correctly, accounting for the increased cancer risk due to infertility, the evidence does not support a relationship between fertility drugs and an increased prevalence of breast or ovarian cancer. More research is required to examine what the long-term impact fertility drugs may be on breast and ovarian cancer prevalence rates. For uterine cancer, the numbers are too small to achieve statistical significance, but it is at least possible that fertility drugs may indeed cause some increased risk of uterine cancer.


















