4
Class B Dealers and Animals
The previous chapters of this report have underscored factors that relate indirectly to determination of the desirability and necessity of animals from Class B dealers since use of such animals cannot be addressed without consideration of the broader context in which the Class B issue is embedded. The Committee was tasked to “determine the important biomedical research questions and common research topics in contemporary NIH-funded research where Class B dogs and cats are desirable/necessary.…” In addition, the statement of task given to the Committee requested data pertaining to “the frequency of these various research topics (i.e., number of grants where the potential exists or the source of the animal is identified as coming from a Class B source). Because there is no requirement to include such information in research records and reports, the Committee considered references to the use of “mongrel” dogs to infer that a number of studies with NIH funding used random source dogs. The Committee was unable to determine whether such dogs came from Class B dealers because mongrel dogs are available from various sources, including Class A dealers. The Committee therefore relied on its own expertise, input from scientific organizations, the testimony of individual investigators and an NIH representative, and the biomedical research literature both for evidence of general areas of research and for specific examples of physiological, anatomical, and genetic research in which random source animals have been used and may be desirable. These accounts, provided in Chapter 3, did not identify any unique or irreplaceable features that made it necessary to obtain random source animals from Class B dealers.
This chapter provides information about trends in the numbers of dogs and cats from Class B dealers used in research, sources of such animals from Class B dealers, challenges in AWA enforcement with respect to Class B dealers, and alternatives to the use of Class B dealers.
TRENDS IN THE NUMBER OF CLASS B DOGS AND CATS USED IN RESEARCH
According to the USDA, the use of dogs and cats in research has declined significantly over the last 30 years. However, data in Tables 1-1 and 4-1 indicate an increase in the use of all dogs and cats in research between 2006 and 2007. The reasons for this recent increase after more than two decades of decline are not understood. The USDA was unable to provide corollary data of year-by-year numbers of animals from Class B dealers, or if the increase was due to random source animals, purpose-bred animals, or animals used for NIH-funded research. Ten-year averages show a decrease in the use of dogs from 187,464 between 1978 and 1987 to 109,353 between 1988 and 1997, and 69,223 between 1998 and 2007. This represents a reduction of 63.1 percent. A parallel 59.4 percent reduction was observed in the 10-year averages of cats with a decrease from 58,526 between 1978 and 1987 to 34,828 between 1988 and 1997, and 23,737 between 1998 and 2007 (Table 4-1). The use of guinea pigs, hamsters, and rabbits has fluctuated over this same time period. Only the use of nonhuman primates increased (from 2001 to 2007 by 29%). It is estimated that rats and mice also increased although numbers are not reported for these species (Table 1-1).
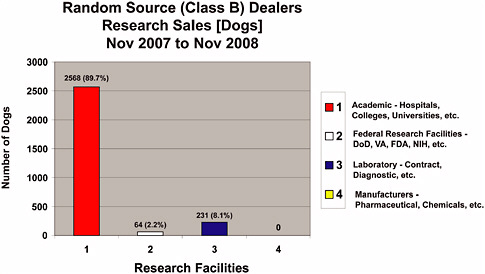
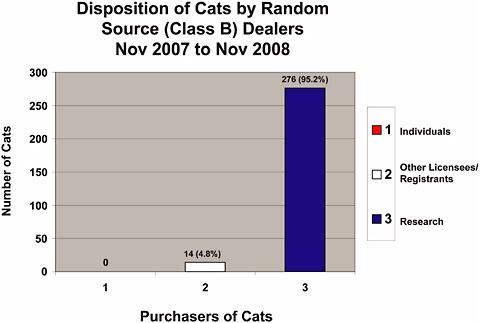
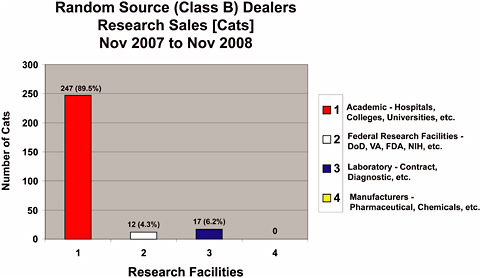
Statistics were not maintained by the USDA that discriminated between dogs and cats from Class A and Class B dealers for the last 30-year period; however, data were obtained from the USDA regarding animals from Class B dealers for November 2007 through November 2008 (Figures 4-1a-f). During Fiscal Year 2007 (FY 2008 was not available at the time this report was written), 72,037 dogs and 22,687 cats from all sources were used in research (Table 4-1). Combined dog and cat usage in research totaled 94,724 animals, roughly 9.2% of all species covered by the AWA that were used in research in 2007 (Table 1-1) and were reported to the USDA. For this reporting period, 2,863 Class B dogs (Figure 4-1b) and 276 Class B cats (Figure 4-1c) were sold for research representing only 4% of the dogs and 1.2% of the cats used in research. The combined total of dogs and cats from Class B dealers used in research represents only 3% of the total dogs and cats used in research and 0.3% of all animals reported to the USDA for research purposes.
Taking into account all animals used in research (assuming 90% of these are mice and rats), dogs and cats represent only 0.9% (Table 1-1)
TABLE 4-1 Total Numbers of Dogs and Cats Used in Teaching, Research, Experiments, and Tests, 1973–2007
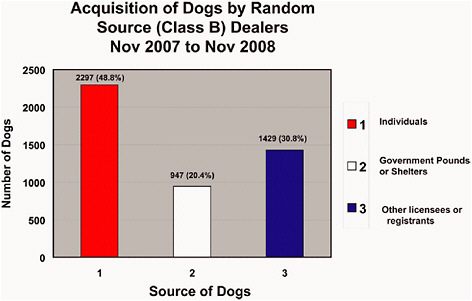
FIGURE 4-1a Acquisition of dogs by Class B dealers from eligible sources, November 2007–November 2008. Data source 4-1 a-f: USDA in response to Committee request, 2008.
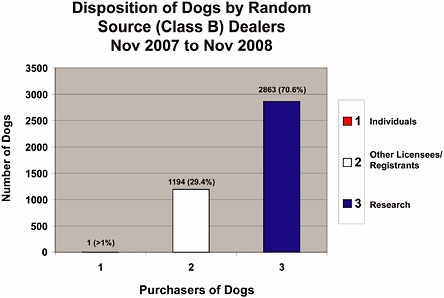
FIGURE 4-1b Disposition of dogs by Class B dealers, November 2007–November 2008. Disposition regulation (9 CFR 2.80) requires Class B dealers to maintain records for at least 1 year after an animal is disposed of, so the 12-month period represents the greatest amount of data USDA could access in response to a Committee request, 2008.
and animals from Class B dealers only 0.03% of all animals studied (USDA 2007). Thus, while random source dogs and cats from Class B dealers, shelters, and pounds may supply some of the animals needed in research, a reasonable conclusion is that Class A dealers provide most of the dogs and cats used in research.
Furthermore, over the past two decades a trend has emerged among research institutions to move away from the use of dogs from Class B dealers or to require justification for their use. For example, Duke University’s website states that “The Duke default for purchase of dogs for research is to use Class A dogs. The use of Class B dogs must be justified on a protocol-by-protocol basis [cost alone is insufficient justification]. The level of risk of using Class B dealers to the program or the research project is considered a level that generally outweighs the benefit.”1 The document lists the following reasons for this position: the animals’ questionable health status, poor condition, and aggressive temperament, and the institution’s inability to guarantee that an animal was not someone’s pet. Iowa State University, the University of Illinois at Chicago, the University of Arizona, Yale University, the University of Texas at Houston, the University of California at Los Angeles, and Massachusetts Institute of Technology are just a few of the growing number of institutions to adopt similar policies.2
THE ROLE OF CLASS B DEALERS IN PROVIDING RANDOM SOURCE ANIMALS
Changing state and local laws have made it more difficult for research institutions to directly obtain random source dogs and cats, particularly in states that do not allow access to pound and shelter animals. Class B dealers, who obtain animals from other states, pounds, shelters, private breeders, bunchers, and other Class B dealers, provide access to animals that research institutions would otherwise have difficulty obtaining.
The potential desirability of random source animals for research (e.g., because of their size, age, genetic diversity and naturally occurring infectious disease) was described in Chapter 3. But the relatively small number of dogs (fewer than 1,000; Figure 4-1a) and cats (fewer than 300; Figure 4-1b) obtained by Class B dealers from pounds and shelters calls into question the genetic diversity of the animals provided by these dealers and to what extent they truly represent the general population of these species. In fact, most dogs (but not cats) sold by Class B dealers are not random source animals,
and are therefore similar to animals available from dealers of purpose-bred animals.
In addition, Class B dealers primarily buy and sell generic dogs (mongrels and hounds) and cats and are seldom involved in or skilled with the selection of specific diseases or models, and indeed are likely to cull diseased animals. However, at least one of the currently licensed Class B dealers acquired animals with parasitism that were suitable for veterinary research.
TRENDS IN THE NUMBER OF CLASS B DEALERS
The declining trends in the use of dogs and cats for NIH-based research are evident in a concomitant and more rapid decline in the number of Class B dealers that sell animals for biomedical research. When demands for random source dogs and cats were highest, in the 1970s and 1980s, approximately 200 Class B dealers sold research dogs and cats. In the 1990s, that number fell to approximately 100, and it declined further when, in 2005, the number of USDA inspections of Class B dealers selling research dogs and cats rose to a minimum of 4 times per year, with an emphasis on traceback verification. There are now only 11 Class B dealers, one of which has been suspended for 5 years because of AWA violations and is not likely to resume operation (personal communication, Robert Willems, USDA, to Committee, January 2009); five others are under “intense scrutiny” (personal communication, Jerry DePoyster, USDA, to Committee, October 2008). One Class B dealer deals with non–random source hounds only, and another deals principally in animals with naturally acquired parasitism for (non-NIH) veterinary product research.
SOURCES OF DOGS AND CATS FOR CLASS B DEALERS
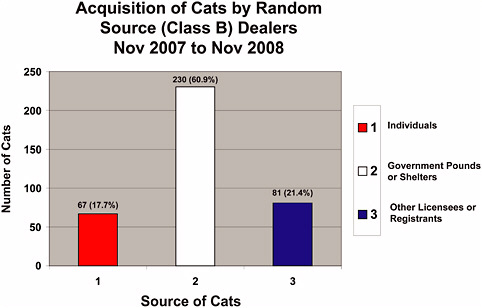
A review of 2008 acquisition data provided by the USDA revealed the following numbers specific to Class B dealers (Figure 4-1; see Chapter 1 for the AWR definition of eligible sources):
4,643 dogs acquired by Class B dealers in 2008:
49% from individuals (e.g., hobby breeders)
31% from other licensees or registrants (e.g., other Class B dealers)
20% from government pounds or shelters
378 cats acquired by Class B dealers in 2008:
61% from government pounds or shelters
21% from other licensees or registrants (e.g., other Class B dealers)
18% from individuals (e.g., hobby breeders)
Pounds and Shelters as Sources of Animals from Class B Dealers
Pounds and shelters serve as points of acquisition of animals for research either directly or through Class B dealers. Usually, these are (government-sponsored) contract pounds or private shelters, but because the latter rely on donations and public support they may have ceased providing animals for research.
Pound seizure became common after World War II when biomedical research experienced an upsurge and shelters and pounds were seen as a readily available source of surplus animals (Zawistowski 2008). However, many members of the public and animal advocates were upset about the practice, which they considered a betrayal of trust since the mission of shelters is to provide care, adoption services, and law enforcement; modern-day shelters also provide spay/neuter services, pet behavior counseling, veterinary care, and humane education.
In Iowa, a 2003 statute (145B; repealed in 2008) explained licensed pound seizure:
An institution so authorized by the Iowa Department of Public Health may request dogs from a pound. The pound may tender to such institution dogs in its custody seized or held by authority of the state, municipality, or other political subdivision. However, a dog shall not be tendered unless it has been held for redemption by its owner or for sale for a period of not less than three nor more than fifteen days. A dog lawfully licensed at the time of its seizure shall not be tendered unless its owner consents in writing. Unless a dog is sick or injured or lawfully licensed at the time of seizure, a pound shall not destroy a dog while a request of an authorized institution to that pound is pending. An institution obtaining dogs from a pound shall pay to the municipality or other political subdivision under whose authority each dog is held or was seized a reasonable fee not to exceed five dollars for each dog so obtained, and shall provide for the transportation of the dogs so obtained from the pound (Iowa 145B.4).
Thus the law allowed the acquisition of an animal by a research institution after only 3 days, a short time frame for the possible reunion of lost animals with their owners or for a new home to be found for relinquished animals. Once the minimum holding period expired, shelter operators seeking to find a home for an adoptable animal were in competition with research institutions or Class B dealers for those animals. A similar statute in Utah (UT26-26-4) states that “the authorized institution shall provide, at its own expense, for the transportation of such animals from the establishment to the institution and shall use them only in the conduct of scientific and educational activities and for no other purpose. The institution shall reimburse the establishment for animals received. The fee shall be, at a
minimum, $15 for cats and $20 for dogs. That fee shall be increased as determined by the department, based on fluctuations or changes in the Consumer Price Index.”
According to the American Anti-Vivisection Society, 16 states3 and the District of Columbia ban pound seizure, 3 states (Minnesota, Oklahoma, and Utah) mandate it, 9 states allow it,4 and the rest leave the decision to the municipality.5 These data may continue to change as states revisit these laws due to changing public attitudes and diminishing access to random source animals from pounds for research. In one instance, the state of California allows the release of unclaimed animals for research, but the individual counties have all enacted bans, in large part because of California Civil Code 1834.7, which requires any pound or shelter that provides living or dead animals to a biological supply company or research facility to post a publicly visible sign stating that “Animals turned in to this shelter may be used for research purposes or to supply blood, tissue, or other biological products.” Given a choice, the public chooses not to allow the resale of pound or shelter animals to research. A 1988 American Humane Association survey involving 26 shelters found that respondents overwhelmingly would not bring a lost animal to a shelter that released unclaimed animals to research (of the 2,438 responses gathered, 2,273 said no, 165 yes). They would also be much less likely to report a stray dog or relinquish their own pet to the shelter if they knew it could end up in a research laboratory (American Humane Shoptalk 1988) (see also Box 2-1).
Alternative Sources of Animals for Class B Dealers
As an alternative to shelters and pounds, the AWA allows Class B dealers to purchase animals from private individuals who have bred and raised the animals on their premises, such as hobby breeders, who are exempt under the AWA (see Chapter 1 for definitions). Hounds are the most common type of hobby breeder–derived dogs bought and sold for research. Class B dealers can also obtain random source animals from other USDA-licensed dealers, including other Class B dealers and auction houses. Auction houses and bunchers are required to have Class B licenses, although they are not considered random source Class B dealers. A review of sourcing data by the USDA indicates that auction houses have not recently been a source of dogs and cats for research purposes (personal communication, Robert Willems,
|
3 |
The 16 states are Connecticut, Delaware, Hawaii, Illinois, Maine, Maryland, Massachusetts, New Hampshire, New Jersey, New York, Pennsylvania, Rhode Island, South Carolina, Vermont, Virginia, and West Virginia. |
|
4 |
The 9 states are Arizona, California, Colorado, Iowa, Michigan, Ohio, South Dakota, Tennessee, and Wisconsin. |
|
5 |
www.aavs.org (accessed September 2009) |
USDA, to Committee, January 2009). Dog auctions are primarily involved with the pet trade; however, if auctions were to move into the research animal trade, the USDA would consider them random source Class B dealers and deal with them accordingly.
COST OF ANIMALS FROM CLASS B DEALERS
In an ideal world, cost would not be a factor in decisions about research, particularly those concerning animals. Realistically, however, resources are limited and researchers are constrained by financial concerns. Thus cost is a potential consideration for the continued use of animals from Class B dealers, which may cost less than animals from Class A dealers. Depending on circumstances, the financial incentives to use animals from Class B dealers may or may not be substantial.
According to information obtained from two Class B and three Class A dealers, the purchase price of a young, 20-25 kg (44-55 lbs) dog is $325-$350 for a random source animal and $600-$900 for a purpose-bred dog. However, oftentimes dogs and cats from Class B dealers are not free of disease and may require prolonged quarantine, socialization, treatment, or removal from the study altogether to avoid potential health threats to other animals or to people in the research facility. The hidden costs to address these conditions may substantially increase the actual final cost by hundreds of dollars per animal. Additionally, the price of USDA/APHIS oversight of Class B dealers (discussed below) represents a substantial cost to the U.S. government and ultimately the American public that is not borne by NIH, the research institution, or the research investigator.
In certain cases the difference in cost between dogs from Class A and Class B dealers may be prohibitive. For example, the cost of using unconditioned dogs from Class B dealers for an acute procedure would be substantially less than for dogs from Class A dealers; but much of the acute work in which an unconditioned dog from a Class B dealer would be an appropriate model, such as in a surgical training class, is beyond the scope of this report.
For studies that require older animals, the purchase price of animals from Class A dealers increases with the age of the animal; for example, one Class A vendor charges a base price of $730 for a 6-month old beagle and an additional $4.10/day after the animal’s first 195 days. Thus, a skeletally mature beagle from a Class A dealer for orthopedic research would cost over $1400/dog, a purchase price that some would consider prohibitive (Class A vendor beagle price list provided to the Committee, January 2009).
One of the largest components to the cost of the animal, regardless of the source, is that of transporting the animal from the vendor to the research location. Surface transportation of groups of animals may cost thousands
of dollars depending on the location of the recipient. Air transportation is even more expensive, done less frequently, and, with new regulations on airlines, may not be an attractive or even viable mode of animal shipping in the future. Obviously, the closer the source of the animals, the lower the shipping costs. In fact, based on the locations of the current Class B dealers and the institutions that use these animals, transportation costs appear to be a factor in the use of animals from Class B dealers: (as of October 2009) the 10 remaining licensed Class B dealers are in Illinois, Indiana, Kentucky, Michigan, Minnesota, and Pennsylvania.
AWA ENFORCEMENT
It is the responsibility of the USDA/APHIS Animal Care agency to ensure that Class B dealers abide by the AWA statutes and the AWR. As explained in Chapter 2, the specter of lost or stolen pets being illegally or inadvertently used in research has been a driving force in the increasingly rigorous revisions of the AWA and USDA/APHIS interpretation and execution of the law. However, in the more than 40 years since passage of the AWA, the USDA/APHIS has been unable to completely enforce the Act with respect to the activities of Class B dealers and has thus been unable to ease the concerns of the American public (Box 4-1). The reasons for this failure are multiple, as discussed below, and underscore the necessity of carefully crafting and enforcing laws if they are to have their intended effect.
USDA veterinary officers who testified before the Committee were appropriately circumspect about their personal opinions, but described long-standing problems with regulation of the Class B dealer system. All stated that AWR enforcement was feasible, with emphasis on tracebacks during inspections. When the Committee queried Jerry DePoyster, a Senior Veterinary Medical Officer with APHIS, he acknowledged the USDA/APHIS could not guarantee that a C.C. Baird-type incident would not be repeated, and reaffirmed the disproportionate effort and difficulties APHIS experiences in regulating Class B dealers. Robert Willems, APHIS Assistant Director for the Eastern Region, testified that Class B dealers are regulated more heavily than any other USDA licensee and that, when he was involved in west coast operations, the office invested over 800 hours and 1½ years investigating the violations of a single dealer. W. Ron DeHaven discussed with the Committee regulatory changes proposed while he served as a USDA Regional Director for Animal Care and Use, including a possible 2-year phase-out of Class B dealers discussed at a Public Responsibility in Medicine and Research (PRIM&R) conference “Animal Care and Use: Hot Zones, Grey Zones and Go Slow Zones” (Rudacille 1996).
According to information that Dr. Willems provided to the Committee, APHIS has responded to these incidents and public pressure by increasing
|
BOX 4-1 Problems with AWA Enforcement: A Case Study A notorious recent example is the case of the Class B dealer C.C. Baird.a For a number of years before 2003, USDA inspection reports indicated that Baird’s Martin Creek Kennels and Pat’s Pine Tree Farms operated within acceptable limits. Then a member of the organization Last Chance for Animals gained employment at Baird’s facilities and obtained over 70 hours of video surveillance of sick, dead, and dying animals with little or no protection from wet and cold, grossly unsanitary conditions, inadequate veterinary care, and multiple instances of cruelty and animal abuse. This documentation was given to the U.S. Attorney’s office, resulting in the largest multiagency (federal, state, and local) investigation of animal abuse in U.S. history. Included in the documentation was a conversation in which a buncher admitted to stealing animals that were probably people’s pets. A documentary film “Dealing Dogs” (HBO 2006) of these events was produced by Tom Simon and Sarah Tealer for Home Box Office. The Committee became familiar with the Baird case because of its importance to recent public perspectives (including congressional action) and decline in numbers of Class B dealers. Baird avoided imprisonment (but paid a large fine) by agreeing to testify to USDA and others in regards to multiple other ongoing Class B dealer investigations. It is important to note that despite uncovering extensive evidence of gross mismanagement and animal suffering by an undercover investigator from the animal protectionist community rather than USDA/APHIS, it still required over a year of administrative procedure and due process for the government to investigate, prosecute, and close this case, not to mention years of USDA inspection and approval of this dealer to remain in operation before the situation became public. The USDA increased its oversight of other Class B dealers by requiring more frequent inspections of dealer premises and by requiring USDA inspectors to regularly trace back the ownership of animals held by Class B dealers to verify that animals were legally obtained. These changes, together with a decrease in demand, contributed to the number of Class B dealers selling dogs and cats to research facilities from nearly 200 to 11. |
its regulatory oversight of Class B dealers by implementing in October of 2008 the new USDA internal SOP, Conducting Tracebacks from Random Source B Dealers. Whereas the regulations mandate annual inspections for research facilities and Class A dealers, Class B dealers now must undergo quarterly inspections with a major focus of these inspections on the acquisition of random and non-random source animals. This increased oversight is also now more feasible with the greatly reduced number of dealers. The legality of acquisition is evaluated by conducting tracebacks on a represen-
tative sampling of animals. For each dealer, tracebacks are performed on some animals at the facility at each inspection and on all animals present at the facility annually during one of the quarterly inspections. It must be made clear that due to the turnover of animals at a dealer facility, not all animals are traced back, only those on the premises at the time the inspection is conducted. In addition, during each quarter, approximately 25% of Class B dealers are now being subjected to 100% tracebacks of all acquisitions since the previous inspection and over the course of one year all Class B dealers will have undergone the process of 100% tracebacks. USDA inspectors are instructed to consider a traceback successful and complete when the origin of the animal has been traced to a legal source.
The USDA has expressed confidence that its increased scrutiny of Class B dealers is sufficient to address concerns about the Class B system and to keep pets out of the system. In 1998, Terry Medley, while serving as administrator of APHIS, stated in a letter to the House Committee on Agriculture, that the USDA was able to trace back original owners for more than 90% of the dogs brokered by Class B dealers (HSUS 2007; CBRA 2009), and Dr. DeHaven stated in testimony before this NRC Committee that in 2000 and 2001 the USDA was able to trace back original ownership for 95% of such dogs. This was well before the 2008 implementation of the new USDA SOP, Conducting Tracebacks from Random Source B Dealers, so it is likely that the current traceback figure is higher. But while such a success rate is admirable, it nonetheless suggests that the origins of 5-10% of animals in the Class B system are uncertain. Indeed, there remain loopholes in the system. For example, origination information is considered adequate if the sale of an animal is traced back to an auction. Thus, although there is no evidence that auction houses are currently used to sell animals to research institutions or Class B dealers, auction sales could serve as a mechanism to legitimize the sale of illegally acquired animals.
There is little evidence to prove that pets are stolen for research (HBO 2006) but the USDA could not offer assurances that pet theft does not occur, and agreed that such a crime is exceedingly difficult to prove, almost requiring an eyewitness. There are, however, descriptions of thefts provided by informants in prison (personal communication, Robert Willems, USDA, to Committee, January 2009) and documented accounts of lost pets that have ended up in research institutions through Class B dealers. For example, in June 2005, the University of Minnesota received from a Class B dealer a dog that was subsequently identified as a missing pet named Echo through microchip scanning by a veterinarian at the institution. Apparently Echo made his way to the university via a USDA-licensed Class B dealer from Michigan, who in turn reported buying Echo from another Class B dealer in Missouri (Fayetteville Free Weekly 2005).
The Committee requested FOIA access to USDA inspection reports over
the past three years of all licensed (as of October 2008) Class B dealers. Those reports revealed that one dealer purchased two cats from a private individual who during traceback investigation admitted that the animals were illegally acquired “strays.” Other citations involved incomplete acquisition documentation. Thus, increased traceback oversight is working at discovering violations, but these ongoing events illustrate that the law continues to be violated.
No system of laws and regulations can absolutely assure protection against theft of pets or misplacement of lost pets, but even single incidents, however few, are a breach of the public trust. The reasons for these deficiencies are multifactorial. The most significant factor is that the acquisition and resale of animals by dealers, bunchers, and individuals is profit-driven, and thus may foster corrupt practices and less attention to animal welfare issues. The system therefore requires rigorous enforcement, but APHIS is understaffed for the task, even with the reduction in numbers of dealers. According to testimony by Dr. Willems, some tracebacks are dead ends, with suspicion of violation, but lack of evidence. Even if staffing were substantially increased, prosecution of AWA abuses requires a step-wise approach to enforcement of the AWR, with documentation to create a “paper trail” of evidence involving citations with correction dates, requests for investigation, warnings, stipulations, formal complaints, and finally a hearing, all before violators can be legally prosecuted. These steps are mandated by federal law in the Administrative Procedures Act, which requires due process and places time constraints on APHIS authorities for action. Regarding the humane issues this Committee was also charged to examine, there is a strong concern that animals can only be removed if they are in need of immediate veterinary care, leaving the potential for animals that are severely stressed or in need of less intensive care to be left unattended indefinitely.
The Committee considered the question of whether animals are still being stolen for research, but was not able to answer conclusively based on the evidence provided. The Committee recognizes, however, that lost animals may find their way into the system inadvertently. Shelter lost-and-found systems range from use of computerized programs to random tours through the shelter by poorly trained staff and distraught owners. Other reasons lost pets are not reunited with their owners are poor breed identification, and lack of resources.
Implanted microchips are a tool used to identify animals and are currently provided by three different companies. Yet, according to testimony provided by USDA staff, inspectors do not check for microchips when performing tracebacks. In order to be effective, recent research has shown that microchip scans should be performed at least 3 times, because such equipment as computers, fluorescent lights, and stainless steel exam tables
can interfere with a scan (Lord et al. 2008). The failure to take these factors into account when scanning could lead to false negatives. The use of microchips for reuniting lost animals with their owners is further complicated by the fact that an effective universal scanner that can detect the various types of microchips is not always available.
All of these factors mean that lost animals may escape detection despite efforts by their owners to recover them. While the individual owner is understandably upset when a lost animal is euthanized at a shelter, the public is outraged (as evidenced by repeated calls to strengthen the legislation to protect pets) when the lost or stolen animal turns up at a research facility.
INCONSISTENCIES IN QUALITY AMONG CLASS B DEALERS
Although it is legal under AWA provisions to obtain dogs and cats from licensed Class B dealers, it is apparent that there are significant differences in standards among dealers in regards to facilities, animal care, and sanitation. There is no set standard among Class B dealers for veterinary care; the required veterinary care plan is left to the discretion of the individual veterinarian employed by the dealer, which lends itself further to disparities in care. Furthermore, there is no requirement to maintain medical records unless an animal is receiving veterinary care. There is also concern that the inspection reports may not always reveal the true conditions at the facility, as the Baird facility also passed its inspections despite its numerous violations (Box 4-1). Inspections are random and unannounced and it may take more than one attempt to actually inspect a facility, providing less scrupulous dealers an opportunity to hide violations and alter records. The Committee did not physically inspect facilities but became aware of such discrepancies after examining USDA/APHIS inspection reports for the last 3 years of all currently licensed Class B dealers. These reports revealed there are dealers that fully respected their obligations to the AWA, with virtually no citations accrued in the last 3 years. In contrast, other dealers were the source of repeated, usually minor and occasionally serious infractions of the law. The Committee recognizes that it is unfortunate that legitimate businesses are negatively impacted by less savory dealers.
ALTERNATIVES TO CLASS B ANIMALS
Chapter 3 discussed attributes, both positive and negative, of random source animals, attributes that pertain to random source animals whether they are obtained directly from pounds and shelters, obtained through Class B dealers, or obtained in other ways. Information provided to the Committee by Class B dealers and USDA officials indicates that a signifi-
cant number of dogs from Class B dealers are hounds from private owners (Figure 4-1a, “Individuals”) and that only 20% (947 of 4,672 total) came from pounds and shelters in 2008 (Figure 4-1a). In contrast, most cats (230 of only 378 total, or 61%) obtained by Class B dealers come from pounds and shelters (Figure 4-1d), many of which are probably stray and feral animals. These percentages indicate that Class A dealers can fulfill much of the demand for animals with similar characteristics. Justification for the research use of random source animals from Class B dealers is based on their cost, size, age, genetic diversity, infectious and naturally occurring diseases, and other conditions, but these attributes (other than cost) are generally not unique to animals from shelters and pounds.
Furthermore, it is important to emphasize that even if random source animals are considered desirable and necessary for NIH-based research, it is not clear that Class B dealers can ensure their availability from the diminishing number of cooperating pounds and shelters. And as sources of random source animals decline, the animals sold by Class B dealers are becoming increasingly similar in characteristics to those of Class A animals, except that they are of inferior quality to Class A animals for the reasons discussed in this report.
Class A Dealers
One mechanism that has been proposed to ensure continued access to genetically diverse, aged, or large breed dogs (attributes that are desirable in random source dogs) is to encourage these animals to be specifically bred and maintained by Class A dealers. Purpose-bred hounds available from Class A vendors are in fact somewhat genetically diverse. Although the colonies of such hounds have been closed for many years, the original stock was composed of various breeds (red bone, black & tan, blue tick, Tennessee walker, foxhounds, and brindle current) and the “descendants” do represent genetic diversity. In addition, a Class A colony of smaller mongrel dogs, originated from Class B dogs almost a quarter-century ago, is available and represents other mixed breed dogs (personal communication, Covance, to Committee, February 2009). Class A vendors also keep a few retired breeding animals or animals that are larger in size, but these are not always available. If the research community requires specific attributes—such as a particular age, physical conditioning, or physical attributes (e.g., larger size) not normally found in existing Class A colonies—Class A vendors may consider providing these models and the costs may not always be significantly higher than for animals from Class B dealers.
It would be impossible for Class A dealers of laboratory animals to maintain the diversity of dog and cat breeds (and thus the genetic diversity) that exists in the general dog and cat population, which could therefore be
an enormous potential resource of animal models of human disease. But Class B dealers do not provide such animals to the biomedical research community either; rather, they deal with various specific breeds of medium and large-sized dogs and random source cats.
Other Research Institutions
Some research institutions (e.g., the University of Florida, the University of North Carolina, and the University of California, Davis) maintain purpose-bred colonies of various breeds of dogs and cats. The Committee was unable to determine how many such colonies exist, because most are not supported directly by NIH, although their animals are used in NIH-related research. Indeed, these colonies are a relatively untapped resource for NIH for the acquisition of aged, genetically defined, or genetically diverse purpose-bred animals.
Random source dogs are used for age-related research, but a significant degree of aging research involves purpose-bred beagles (Cotman and Head 2008). Beagles are accessible from purpose-bred colonies maintained by a small number of research institutions (e.g., the Lovelace Foundation). Aged beagles from such colonies are particularly valuable models of human cognitive aging, and are actively used by the scientific community for these aging studies. These dogs manifest age-dependent decline in learning and memory and develop neurological disease with features similar to Alzheimer’s disease (AD) as well as age-related hippocampal and entorhinal neuronal loss, similar to that which occurs in humans. The aged Beagle model has also been used in immunotherapy studies for AD (Nippak et al. 2007; Siwak-Tapp et al. 2008; Vasilevko and Head 2009). In contrast to random source animals, which typically are of unknown age, defined-age purpose-bred dogs are also ideal for longitudinal studies and for studies evaluating the effects of long-term dietary variables or environmental enrichment.
To its credit, the Comparative Medicine Program of the NIH National Center for Research Resources has capitalized on the many genetic mutations among the pet dog and cat population by supporting the Referral Center for Animal Models of Human Genetic Diseases at the University of Pennsylvania School of Veterinary Medicine. This center has accrued a wealth of dog and cat models of human genetic diseases (e.g., metabolic diseases, bleeding disorders, immunologic disorders, dilatative cardiomyopathy and other cardiac disorders, osteogenesis imperfecta, mucopolysaccharidoses, and many others) through referrals about pets (e.g., from knowledgeable breeders, working dog organizations, and veterinarians). The center acquires, characterizes, and genetically analyzes submitted cases to validate homology to the human disease, and makes
animals available to the general research community by maintaining small nucleus breeding populations, collaborative interactions with colonies at other institutions, and germplasm. This laudable program directly addresses NIH needs for certain types of dog and cat models.
Another outstanding program, the Canine Comparative Oncology Program of the National Cancer Institute (NCI), takes full advantage of naturally occurring cancers in the general dog population. The aged and genetically diverse dog population is prone to many types of cancers that mimic the human disease—including non-Hodgkin lymphoma, osteosarcoma, melanoma, prostate carcinoma, pulmonary carcinoma, mammary carcinoma, soft tissue sarcomas, mast cell tumors, and others—and the rate of occurrence is sufficient to power preclinical trials.
The NCI program, established in 2003, includes a multicenter collaborative network of 18 veterinary teaching hospitals6 (the Comparative Oncology Trials Consortium; Figure 4-2) that fosters rigorously controlled preclinical trials of new cancer drugs intended for eventual use in humans. These preclinical trials provide guidance on the design of human studies, without the constraints of human phase I, phase II, and phase III trials, while also benefiting the client-owned animal patients. The program is linked to another NIH consortium, the Comparative Oncology and Genetics Consortium, which builds on the publication of the canine genome and maintains a biorepository of canine tumors, fosters collaborative opportunities between comparative oncologists, and initiates preclinical trials using pet dogs with cancers.
These NCI programs make use of naturally occurring cancers in dogs, which have significant similarities to the genomic profiles and biology of human neoplasms. For example, the comparable respiratory anatomy of large dogs and humans (discussed in Chapter 3) and the parallels in distribution of primary and metastatic lung cancers have allowed assessment of inhaled cytokine immunotherapy, which led the way for—and predicted the successful outcome of—early phase trials in humans. These programs benefit from the rich genetic and disease diversity of the general dog population, together with highly qualified veterinary and medical collaboration, client participation, and mutually beneficial advancement of human and animal health.
FIGURE 4-2 Map of current institution members of the Comparative Oncology Trials Consortium. Source: http://ccr.nci.nih.gov/resources/cop/COTC.asp
UNRESOLVED CLASS B COMPLIANCE ISSUES
The public harbors two major concerns about the use of Class B dogs and cats in research, and the Committee shares those concerns. The first is the perception of pet theft or displacement of lost pets by dealers who may profit through the sale of such animals to research. The second is the deplorable husbandry conditions that have been documented at some Class B dealers (AWI 2007).
With respect to the first concern, loopholes in the AWR permit pets to enter the research pipeline via Class B dealers who acquire and sell dogs and cats that originated from auctions, shelters, and pounds.
The second concern arises from the requirement that Class B dealers adhere to only the AWR, whereas institutions that receive NIH funding for research comply with PHS Policy, the Guide for the Care and Use of Laboratory Animals, and the U.S. Government Principles (defined in Chapter 1). Class B facilities are therefore not held to the same standards as NIH-funded research facilities. In addition, the USDA, which is responsible for enforce-
ment of the AWR, has different internal inspection manuals (see Chapter 1), allowing—and inconsistently applying—different standards of the AWR among dealers, research institutions, and exhibitors. It is difficult for the public to understand why there are different standards of care when the purpose of the AWR was to establish minimum standards.
Finally, the Committee recognizes that the USDA is severely hampered in its ability to implement the AWR standards. USDA has insufficient enforcement powers, including the ability to act more swiftly, assess sufficiently punitive fines, issue temporary injunctions, and impose immediate cease-and-desist orders for serious or repeat animal welfare citations. As explained above, because of insufficient staffing and the time-consuming multiple steps and documentation required for enforcement, only serious and repeated infractions are worth pursuing, allowing many “minor” infractions to persist unaddressed. It is of great concern to the Committee that animals can be removed from Class B dealer sites only if they are in need of immediate veterinary care, leaving the possibility that severely stressed animals or those in need of less intensive care may be left unattended.
These serious unresolved Class B compliance issues and humane concerns were major factors in the deliberations that led to the Committee’s final recommendations (Chapter 5).
REFERENCES
AWI (Animal Welfare Institute). 2007. The Animal Dealers: Evidence of Abuse in the Commercial Trade 1952-1997. M. E. Drayer, ed. Washington: AWI.
American Humane Shoptalk. 1988. April/May. 6(3).
CBRA (California Biomedical Research Association). 2009. Fact Sheet: The Pet Theft Myth. http://www.ca-biomed.org/pdf/mediakit/Petmyth.pdf
Cotman, C. W. and E. Head. 2008. The canine (dog) model of human aging and disease: dietary, environmental and immunotherapy approaches. Journal of Alzheimer’s Disease 15:685-707.
Fayetteville Free Weekly. 2005. Oct. 20-26. pp. 4-5. Fayetteville, AR.
HBO (Home Box Office). 2006. Dealing Dogs (video documentary).
HSUS (Humane Society of the United States). 2007. Class B Dealers of Random Dogs and Cats.
White Paper. July 2007. http://www.hsus.org/web-files/PDF/ARI/class-b-dealer-report-07-11-07-final-ii.pdf
Lord, L. K., M. L. Pennell, W. Ingwersen, R. A. Fisher, and J. D. Workman. 2008. In vitro sensitivity of commercial scanners to microchips of various frequencies. Journal of the American Veterinary Medical Association. 233(11):1723-1728.
Nippak, P. M. D., J. Mendelson, B. Muggenburg, and N. W. Milgram. 2007. Enhanced spatial ability in aged dogs following dietary and behavioural enrichment. Neurobiology Learning and Memory. 87:610-623.
Rudacille, D.1996. Public Responsibility in Medicine and Research (PRIM&R)—Animal Care and Use: Hot Zones, Grey Zones and Go Slow Zones. Center for Alternatives to Animal Testing Newsletter. (Summer):13(3).
Siwak-Tapp, C. T., E. Head, B. A. Muggenburg, N. W. Milgram, and C. W. Cotman. 2008. Region specific neuron loss in the aged canine hippocampus is reduced by enrichment. Neurobiology of Aging. 29:39-50.
USDA (U.S. Department of Agraculture). 2007. Animal Care Annual Report of Activities, Health Inspection Service APHIS 41–35–075. http://www.aphis.usda.gov/publications/animal_welfare/content/printable_version/2007_AC_Report.pdf
Vasilevko, V. and E. Head. 2009. Immunotherapy in a natural model of Aβ pathogenesis: the aging Beagle. CNS & Neurological Disorders Drug Targets. 8:98-113.
Zawistowski, S.2008. Companion Animals in Society. Clifton Park, NY: Thomson Delmar Learning. p. 81.