2
Clinical Research, Patient Care, and Learning That Is Real-Time and Continuous
INTRODUCTION
The United States is home to vital and dynamic biomedical research enterprises. Science-driven care, however, requires a focus beyond advancing understanding of disease mechanisms. The same level of passion, creativity, and rigor must be applied to the translation of these discoveries into effective patient care and improved health outcomes. Several recent reports call for improvements in the level, quality, and effectiveness of clinical research through a sharper focus on generating timely information that is relevant to care decisions faced by patients and providers and on the science of care delivery (Conway and Clancy, 2009; Dougherty and Conway, 2008; IOM, 2010, 2011).
Current clinical research capacity is underdeveloped, substantially fragmented, and limited in its ability to support such work, particularly learning in real-world settings (Califf, 2009; IOM, 2010, 2011). A more dynamic clinical research infrastructure that draws research closer to practice is needed to facilitate ongoing study and monitoring of the relative effectiveness of clinical interventions and care processes. Innovative research approaches (e.g., novel study designs, analytical tools, use of course-of-care data from electronic medical and personal health records) will also accelerate learning (IOM, 2011; Lauer and Collins, 2010).
The papers that follow explore leading opportunities to improve the efficiency, effectiveness, and volume of clinical research. They also describe strategies for fostering the development of the capacity and culture needed for real-time and continuous learning that anticipates research needs and
produces evidence that is timely, relevant, and applicable to real-world care. Patrick Conway, formerly of the Office of the Secretary of the Department of Health and Human Services (HHS), reviews the unique nature of comparative effectiveness research (CER) and its core aim of helping to inform decisions of patients, clinicians, and policy makers. Recent federal investments have expanded national capacity for CER, and ensuring the long-term success of this emerging enterprise will require a near-term focus on devising a sustainable funding strategy, developing a research agenda for a balanced portfolio of high-impact topics, selecting methods appropriate to information needs, and evaluating and reporting progress to the public. Elements essential to ensuring the availability and use of comparative information at the point of decision making are discussed as well. John Noseworthy and Sherine Gabriel of Mayo Clinic offer insights into what might be possible if health systems were better engaged in the research enterprise. To illustrate the potential, they review several examples of the use of clinical data captured as part of healthcare delivery to improve care quality and health outcomes. They describe key characteristics of knowledge-driven healthcare delivery systems of the future, including patient-centered care; real-time data and feedback; a culture of collaboration, innovation, and translation; health information technology (HIT); and delivery of high-value health care.
Without the willing participation of the public and patients as contributors to learning, however, capacity for research will remain limited. Diane Simmons and Kenneth Getz of the Center for Information and Study on Clinical Research Participation review current public and patient attitudes toward participation in clinical research and offer some suggestions for fostering a culture that better supports and encourages public appreciation of and participation in such research. Educational and outreach efforts by the clinical research community and other stakeholders are needed to enhance public awareness, enable participation, and sustain interest over time.
COMPARATIVE EFFECTIVENESS RESEARCH: PATIENT, CLINICIAN, AND POLICY NEEDS
Patrick Conway, M.D., M.Sc.
Office of the Secretary, Department of Health
and Human Services (formerly)
Cincinnati Children’s Hospital
National Investments in a Comparative Effectiveness Research Enterprise
The American Recovery and Reinvestment Act (ARRA) of 2009 dedicated $1.1 billion to CER and established the Federal Coordinating Council
(FCC) for CER. The FCC established a common federal government definition for CER, prioritization criteria for scientifically meritorious research (Box 2-1), and a strategic framework to guide investment decision making and priorities, and offered recommendations for initial priorities (Figure 2-1). The FCC definition of CER is as follows:
Comparative effectiveness research is the conduct and synthesis of research comparing the benefits and harms of different interventions and strategies to prevent, diagnose, treat and monitor health conditions in “real world” settings. The purpose of this research is to improve health outcomes by developing and disseminating evidence-based information to patients, clinicians, and other decision makers, responding to their expressed needs, about which interventions are most effective for which patients under specific circumstances. (FCC, 2009)
ARRA allocated $400 million to the National Institutes of Health (NIH), $300 million to the Agency for Healthcare Research and Quality (AHRQ), and $400 million to the Office of the Secretary for CER. The Office of the Secretary funding supported an array of data infrastructure projects, whose focus ranged from claims data to clinical data networks to patient registries. These investments also focused on selected priority populations, such as children and persons with multiple chronic conditions (Conway and Clancy, 2009). Another major priority for the Office of the Secretary
BOX 2-1
Federal Coordinating Council for Comparative Effectiveness Research
- Prioritization criteria for scientifically meritorious research
- Potential impact (based on prevalence of condition, burden of disease, variability in outcomes, costs, potential for increased patient benefit or decreased harm)
- Potential to evaluate comparative effectiveness in diverse populations and patient sub-groups and engage communities in research
- Uncertainty within the clinical and public health communities regarding management decisions and variability in practice
- Addresses need or gap unlikely to be addressed through other organizations
- Potential for multiplicative effect (e.g., lays foundation for future CER such as data infrastructure and methods development and training, or generates additional investment outside government)
SOURCE: FCC, 2009.
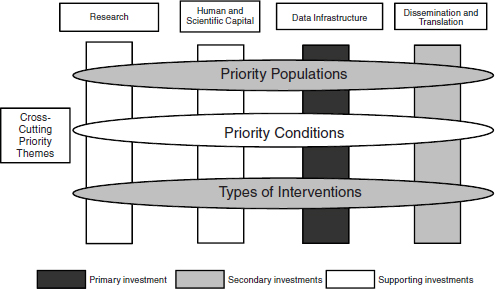
FIGURE 2-1 Federal Coordinating Council (FCC) strategic framework and recommended Office of the Secretary investment priorities for CER. This framework, including core activities (research, human and scientific capital, data infrastructure, and dissemination and translation) and cross-cutting priority themes (populations, conditions, and intervention types), was developed by the FCC to guide investment decisions and priorities.
SOURCE: FCC, 2009.
investment was dissemination of CER findings and their incorporation into practice. This effort will encompass both clinicians and patients, and will utilize networks and the delivery system. Finally, the Office of the Secretary investments targeted selected research topics—such as delivery systems; prevention; behavioral change; and priority populations, including racial and ethnic minorities and persons with disabilities—to complement the funding from AHRQ and NIH. Finally, the ARRA CER investment included funding for an ongoing inventory of CER and evaluation of the CER portfolio.
The Affordable Care Act (ACA) of 2010 included additional focus and funding for CER. Specifically, it established the Patient-Centered Outcomes Research Institute (PCORI). The duties of PCORI include identifying research priorities and establishing a research agenda. The Institute will have a 17-member Board of Governors and several expert advisory panels. Its budget will increase over time and likely exceed $600 million annually.
For the national CER program to have maximum impact on health and the value of healthcare delivery, the following five next steps have been proposed (VanLare et al., 2010):
- Develop an overall funding strategy, influenced by clinicians and patients and the program’s stakeholder advisory board.
- Establish an initial list of priority topics, and evaluate the current state of knowledge about each.
- With input from the advisory board and decision makers, select the research methods appropriate to fill the gaps in knowledge on a particular topic/question.
- Strive for a balanced portfolio of high-impact research topics.
- Evaluate progress, and report to the public.
CER Enterprise Implementation
Multiple additional considerations are involved in implementing a successful CER enterprise. First, the research methods must address the level of evidence necessary to influence decision makers. The level of evidence will vary based on the question and decision involved, but certain decisions may not require randomized controlled trials. Decision makers, including patients and clinicians, should be actively engaged in the planning and funding of research so CER meets their needs. The level of evidence needed to influence decisions should be considered an integral part of funding decisions and research designs. In addition, funding is needed to develop research methods and build understanding of how the methods used and the communication of findings will best meet decision makers’ needs (Chalkidou et al., 2009).
The FCC (2009) and IOM (2009) reports on CER were strongly influenced by public input, and ARRA includes funding for actively seeking input to guide CER (e.g., horizon scanning). Building on this work to involve stakeholders, PCORI should focus its efforts on maintaining and increasing clinician, patient, and other stakeholder input. This input is important because one of the unique aspects of CER is that it must be guided by the needs of routine practice and consumers, which involves a different paradigm from that which informs traditional, investigator-driven research. To garner input efficiently and effectively, technology and other means of obtaining “real-time” input from a broad sample of patients, clinicians, and other stakeholders are needed. Finally, a feedback loop from users and implementers of research back to the research enterprise is essential. Examples of such feedback loops exist in certain delivery systems and networks, but they need to become the rule as opposed to the exception.
The FCC report and subsequent ARRA funding (especially that for the Office of the Secretary) focused on dissemination and adoption. Without any investment in adoption, the CER enterprise will fail to translate comparative evidence into improved health outcomes (Dougherty and Conway, 2008). To better understand the factors underlying successful adoption
interventions will require research, supported by HIT as a tool for both driving and measuring adoption. Although challenging, successful adoption will provide significant rewards by allowing the healthcare system to deliver higher-quality care, better value, and better population health, fundamentally transforming U.S. health care.
Although research funders often do not systematically evaluate the impact of their research investments. Evaluation is critical not only for measuring impact but also for understanding how to improve future research investments. In contrast, the Office of the Secretary CER investment included evaluation of ARRA CER funding to focus on impact and guide future CER investments. Going forward, the CER research enterprise should be accountable to the public by evaluating investments and publicly reporting the evaluation results.
Concluding Observations
Overall, CER represents an opportunity to focus research on the needs of clinicians, patients, policy makers, and other decision makers. Unfortunately, all of these groups often are faced with health-related decisions for which insufficient evidence exists to guide decisions or the evidence is not adequately available at the point of decision making. Therefore, the evidence must be generated to meet decision makers’ needs and disseminated effectively to the point of decision making, in order to inform care and drive better outcomes and value.
HEALTH SYSTEMS AS RESEARCH PLATFORMS: ENHANCING SCIENCE, VALUE, AND INNOVATION
John Noseworthy, M.D., and Sherine Gabriel, M.D., M.Sc. Mayo Clinic
The IOM’s Learning Health System series of workshops has defined foundational elements for a healthcare system that enables both the implementation of best care practices and the real-time generation and application of new evidence. This paper focuses on one of these elements—health systems as research platforms. It considers how healthcare organizations can be structured to support a system that advances clinical research and produces and applies evidence that is timely, relevant, and applicable to real-world care. One example of such a system is Mayo Clinic, a health system with a long tradition of creating and sustaining research platforms built upon its rich clinical practice.
Traditionally, leading healthcare organizations have fostered the incorporation of discovery and clinical research into clinical practice. For more
than a hundred years, Mayo Clinic has applied a singular focus on excellent patient care through its primary value—“The needs of the patient come first”—and its mission statement—“Mayo Clinic will provide the best care to every patient every day through integrated clinical practice, education, and research.” Figure 2-2 shows a page from a patient ledger that contains the oldest medical records at Mayo Clinic. Written by Dr. William Worrall Mayo in 1866, it indicates that the patient record is “left open for further thought and research,” exemplifying the philosophy of continuous learning during the course of clinical care.
Mayo Clinic has applied its culture of patient-centeredness to incorporate advances from clinical research into practice as expeditiously as possible. Research has always been a fundamental component of Mayo’s core activities. The “conventional” approach of identifying key basic science and clinical research questions, publishing the results of these hypothesis-driven studies, and incorporating them into practice has helped Mayo Clinic grow its reputation as a knowledge-driven, patient-centered healthcare system. Three examples of this conventional approach are the Rochester Epidemiol-

FIGURE 2-2 Mayo Clinic patient ledger, 1866.
SOURCE: Reprinted courtesy of the Mayo Clinic.
ogy Project, the Total Joint Registry, and the recently completed Warfarin Project. A description of each of these efforts is followed by a discussion of two new transformational initiatives currently under way, designed to meet the healthcare needs of the future.
Rochester Epidemiology Project
The Rochester Epidemiology Project (REP) is a long-standing collaboration among healthcare providers in Olmsted County, Minnesota (Kurland and Molgaard, 1981; Kurland et al., 1970; Melton, 1996, 1997). Many decades ago, these providers formally agreed to share medical records collected during the course of care of Olmsted county residents in order to study the health and illnesses of people in this community. Inferences drawn from this unique population-based resource could then be used to inform and improve health and health care in the entire country. The REP is one of a few venues where population-based research can be conducted passively and on a routine basis. REP studies typically address disease incidence and prevalence, time trends, risk and protective factors, effectiveness of treatments, natural history and outcomes, genetic factors, quality of care, and cost of care through careful identification of cases and controls and of exposed and nonexposed individuals. The infrastructure for the REP has been NIH-funded since 1966, supports many individual NIH-funded research grants (approximately 40 during the past 5 years), and has yielded approximately 2,042 peer-reviewed research papers to date. From these publications have come such observations as the following: the occurrence of Guillain-Barré Syndrome is increased only slightly by swine flu vaccination (Beghi et al., 1985); silicone breast implantation carries a high risk of surgical complications, but is not associated with previously claimed significant autoimmune adverse sequellae (Beghi et al., 1985; Gabriel et al., 1994, 1997); routine immunizations do not increase the risk of autism (Barbaresi et al., 2005); and prophylactic bilateral oophorectomy is associated with both increased mortality and an increased risk of neurological disorders (Rocca et al., 2006, 2007, 2008).
Total Joint Registry
The Mayo Clinic Total Joint Registry1 is the most comprehensive joint replacement registry in the world. The database was established in 1969, has been carefully maintained since that time, and now contains data on 97,500 arthroplasties. Structured standardized information is gathered from
______________
1 See http://www.regulations.gov/search/Regs/home.html#documentDetail?R=090000648098c1d0 (accessed October 11, 2010).
patients before, during, and at scheduled intervals after their surgeries for the lifetime of the patient and the orthopedic implant. This registry has allowed for comparison of surgical techniques, implant types, patient demographics, comorbid conditions, and long-term outcomes. It has allowed physicians to determine which surgical practices and implant models are most effective over time and has informed decision making on these issues. The registry has been a valuable clinical and research tool, allowing orthopedic surgeons, for example, to compare planned hip replacement surgery with 35,000 such operations performed since 1966. Work based on this registry has enabled continuous improvement in the processes of care, favorably influencing lengths of stay, resource use, and the results of care (i.e., delivery of high-value care). The registry has served as a data source for more than 800 academic publications. It has enabled CER and has led to improvements in information systems that facilitate and enhance the continuity of care delivery after surgery.
Mayo Clinic Warfarin Project
The Mayo Clinic Warfarin Project is a third example of conventional, focused clinical research designed to improve the quality of care. This intervention was launched with the goal of reducing warfarin-related overanticoagulation, which is acknowledged to be a leading iatrogenic illness. In 2005, 18,700 inpatients were treated with warfarin at Mayo Clinic. It was determined that 3.5 percent experienced iatrogenic overanticoagulation, with a score greater than 5.0 on the blood coagulation International Normalized Ratio (INR) laboratory test, resulting from in-hospital warfarin administration, and no definitive, published, evidence-based guidelines for administering warfarin in the hospital existed. We recognized that there was considerable variability in risk for iatrogenic overanticoagulation across the five states where Mayo Clinic practices. Therefore, a prospective study was designed to develop a standardized warfarin protocol that would improve outcomes. The goal was to reduce the number of inpatients who had a single recording of an unsafe inpatient INR (greater than 5.0) from 3.5 percent to less than 1.5 percent. This project involved standardized rule-based algorithms supported by the prescriber using computerized provider order entry, with the hospital pharmacist determining the final dose from these algorithms. This work takes advantage of the principle of reflexivity; the prescribing system algorithm was improved dozens of times through the “plan, do, study, act cycle” based on surveillance, performance data, and user feedback. Since implementation of the standardized, rule-based algorithms, consistently fewer than 1.5 percent of Mayo Clinic warfarin inpatients have had an INR above 5.0, and there has been no increase in the proportion of patients with an INR below 1.7 after the third dose. Thus not only was the risk of potential hemorrhage reduced, but also the risk
of clotting was not commensurately increased. This change in practice has significantly reduced the risk of inpatient warfarin-related adverse events.
Transformational Initiatives
The High Value Healthcare Initiative
A learning healthcare system has as its central focus improving the value of care. For many years, Mayo Clinic has maintained an institution-wide emphasis on quality (best clinical outcomes, safety, and service), which has been applied both to the care of individual patients and through population-based strategies to manage chronic disease across communities of patients. Recently, Mayo Clinic has gone beyond its focus on the numerator (quality in terms of best outcomes, safety, and service) of the value equation (e.g., value = quality/cost) to address the denominator (i.e., the cost of care over time). Together with the Dartmouth Institute, Intermountain Healthcare, and the Geisinger Health System, Mayo is proposing a pilot project for the Innovation Center at Centers for Medicare & Medicaid Services (CMS) called the High Value Healthcare Project. This represents the first step in a broad-based initiative that will benchmark the costs of best practices over time, implement evidence-based best practice and patient shared decision-making models, and study new reimbursement models that better align payments with the outcomes of patient care. Together with similar initiatives under way around the country, the High Value Healthcare Initiative is motivating a fundamental transformation of health systems from their traditional focus on compliance with process to a more meaningful focus on results, outcomes, and value.
As famously reported by Balas and Boren (Balas and Boren, 2000), the lag time between the discovery of more efficacious forms of treatment and their incorporation into practice is unacceptably long—about 15–20 years. Moreover, a majority of patients at any given time receive care that is not supported by evidence-based research (Advisory Commission on Consumer Protection and Quality in the Health Care Industry, 1998). Stakeholders across the healthcare system—from patients to practitioners to payers—recognize the need for disruptive change to bring about fundamental improvements in health care. It is becoming increasingly apparent that such change requires new information systems to accelerate discovery, drive clinical research, identify best practices, and diffuse these practices rapidly across the profession.
The Mayo Clinic Enterprise Data Trust
The Mayo Clinic Enterprise Data Trust is an example of a centralized repository system created to manage, integrate, and share collective information resources with appropriate regulatory protections (Figure 2-3). This consolidated infrastructure can, for example, integrate biospecimen-related data generated by research core laboratories with phenotypic data extracted from clinical records through natural language processing techniques, analyze those data to develop improved biomarkers to guide therapy, and provide that information to physicians for use in caring for patients. Data and biospecimens collected during the course of care are fed back into the system to inform and improve the care of future patients. Systems such as this are needed to enable the evolution to a knowledge-driven healthcare delivery system.
Conclusions
The examples discussed above demonstrate how a patient-centered, knowledge-driven healthcare delivery system can serve as a research plat-
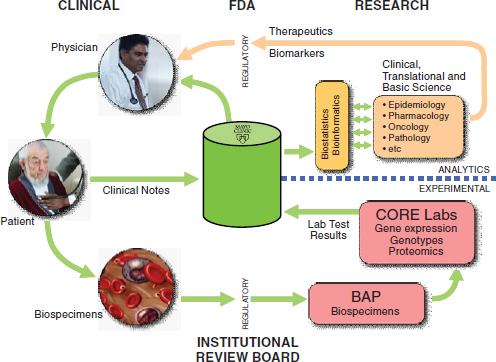
FIGURE 2-3 Schematic of Mayo Clinic Enterprise Data Trust.
SOURCE: Reprinted courtesy of the Mayo Clinic.
form both to improve outcomes for future patients and to identify the most compelling questions for future research. More important than these specific examples, however, are the underlying conceptual model and the set of fundamental principles upon which these (and other) examples are built. Although the specific examples may not be replicable elsewhere, the underlying model and key characteristics are widely translatable and transportable around the country and, indeed, around the world.
Figure 2-4 illustrates a model for a knowledge-driven healthcare delivery system of the future. As shown, knowledge generated from routine clinical settings is integrated with biological information garnered from biospecimen banks and other sources; aggregated and analyzed using sophisticated data warehousing and computational tools; and then used to improve patient outcomes through enhanced clinical practice, business processes, education, and research. The system rests on a foundation of data security and governance, metadata, and terminology standards.
The key characteristics of a knowledge-driven healthcare delivery system are summarized in Box 2-2. Patient-centeredness must be at the core of
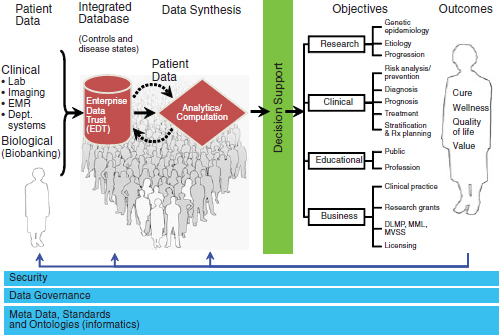
FIGURE 2-4 Model for a knowledge-driven healthcare delivery system. The model focuses on generating and synthesizing knowledge in daily clinical settings to advance research, education, clinical practice, and business operations to improve patient outcomes.
SOURCE: Reprinted courtesy of the Mayo Clinic.
BOX 2-2
Characteristics of the Knowledge-Driven Healthcare Delivery System of the Future
- Patient-centered care—a focus on quality (best results) and coordination of care
- Real-time data and feedback for providers at the point of care (horizon scanning)
- A culture of collaboration, innovation, and translation of scientific knowledge into improved health for patients and communities
- Health information technology systems—integration, standardization, interoperability
- Delivery of high-value health care in an information-enabled single practice
health care. Although patient-centered care is often assumed, it is but by no means universally applied. Providers must design and implement systems to be focused on quality and team-based, integrated, coordinated care centered on the patient. To enable the evolution to a knowledge-driven, learning healthcare delivery system, future HIT systems must have the capability to provide real-time data and real-time feedback to providers at the point of care/point of need. This capability will require scanning disparate internal and external data resources to rapidly find answers to scientific, clinical, and operational questions. HIT systems must also be integrated, standardized, and highly interoperable. Finally, the delivery of high-value care in the information-enabled practice of medicine requires a culture of collaboration, innovation, and translation of scientific knowledge into improved health for patients and communities. Although perfect and complete actualization of the conceptual model for each of the above characteristics may be a long way off, an intense focus on the development of integrated, patient-centered, and knowledge-driven healthcare delivery systems will lead to better health care and better health.
ENHANCING THE CULTURE OF PATIENT CONTRIBUTIONS TO LEARNING IN HEALTH CARE
Diane Simmons and Kenneth Getz, M.B.A.
Center for Information and Study on Clinical Research Participation
How can a durable relationship be built with the millions of past, present, and potential future clinical research study volunteers? A portfolio of strategic initiatives is needed to regain public trust in the clinical research enterprise and establish a culture that welcomes input from patients. Since its founding in 2004, the nonprofit Center for Information and Study on
Clinical Research Participation has developed an effective, multifaceted strategy of outreach and education to improve public understanding of the clinical research process and the important role of participation in advancing medical science. After a discussion of the current culture, this paper presents a number of examples of strategic initiatives for recruiting, retaining, and sustaining a community of study participants who will ultimately become the ambassadors for learning in health care.
Current Culture
During the past decade, public confidence and trust in the clinical research enterprise has eroded steadily (Center for Information and Study on Clinical Research Participation, 2006; HarrisInteractive, 2007; Kaiser Family Foundation, 2008). Distrust of clinical research professionals and of those organizations responsible for ensuring patient safety has increased dramatically. Public polls reveal startling statistics. For example, a 2007 poll among 1,726 U.S. adults found that 27 percent of respondents distrusted the Food and Drug Administration (FDA) “somewhat” or “very strongly.” That same poll found that only 31 percent of respondents believed that the FDA is effective at ensuring safety, down from 56 percent who held this belief in 2004 (HarrisInteractive, 2007). Equally alarming, nearly half (46 percent) of the 1,726 Americans polled said they distrusted Capitol Hill officials who govern regulatory oversight and drug development processes (HarrisInteractive, 2007).
Four of ten respondents (42 percent) distrusted pharmaceutical and biotechnology companies. The poll showed that a significantly higher percentage (39 percent) gave poor ratings to pharmaceutical and biotechnology companies for failing to serve consumers as compared with 1997, when 19 percent of Americans surveyed rated pharmaceutical and biotechnology companies on this item poorly (HarrisInteractive, 2007).
Nearly half (44 percent) of the 1,695 American adults polled in a January 2008 survey likewise reported having an unfavorable view of pharmaceutical and biotechnology companies. In that same survey, 27 percent of Americans said they did not trust these companies to offer reliable information about drug side effects and safety. And 45 percent said they did not trust research sponsors to inform the public quickly when safety concerns with a drug are uncovered (Kaiser Family Foundation, 2008).
The public displays similar levels of distrust in principal investigators and their study staff. A self-administered survey conducted among 717 U.S. adults in 2007 found high levels of public distrust in clinical research staff. This level of distrust was significantly higher among minority adults (HarrisInteractive, 2004). Nearly half (49 percent) of white respondents and 73 percent of minority respondents reported that it was “very likely”
or “somewhat likely” that they might be used as guinea pigs without their consent. One of four (25 percent) minority respondents and 22 percent of white respondents believed that their doctors would expose them to unnecessary risk in clinical trials (Braunstein et al., 2008).
The majority of respondents (72 percent) in a 2002 poll said they believed that physicians get involved in clinical research to help patients find new and better treatments (HarrisInteractive, 2002). Still, one of four said they believed that doctors and study staff are motivated to recruit volunteers primarily by money and selfish interests. In a 2005 survey, 25 percent of respondents said they believed physicians participate in clinical research to receive money from pharmaceutical and biotechnology companies, and another 20 percent said that physicians participate primarily for fame, glory, and publication rights (HarrisInteractive, 2005).
By extension, public distrust in clinical research professionals has tainted the public’s view of clinical research volunteers. In a 2002 poll, 8 of 10 Americans said they believed that study volunteers are taking a gamble with their health (HarrisInteractive, 2002). A 2006 survey conducted among 900 U.S. adults found that one of four believed people choose to participate in clinical research because they are “very sick without any other options” or they are “looking to make money.” A smaller percentage—19 percent—believed that people choose to participate in clinical trials to benefit the public’s health. In that same survey, 34 percent of respondents said they “do not admire” people who volunteer for clinical trials (Center for Information and Study on Clinical Research Participation, 2006).
As public appreciation for study volunteers has waned, public willingness to participate in clinical trials has also dropped. Research!America, for example, reported that in 2004, 55 percent of those polled said they would be willing to participate in a clinical trial, down from 63 percent in 2001 (Woolley and Propst, 2005). A later public poll, conducted in 2007, found that only 41 percent of white adults and 28 percent of minority adults would be “very likely” or “likely” to participate in clinical trials (Braunstein et al., 2008).
Investigative sites report that growing levels of public distrust have contributed to delays in bringing new treatments to market and to increased drug development costs. Since 2000, spending on patient recruitment promotional programs by investigative sites and research sponsors has grown by 12 to 14 percent annually, reaching more than $500 million in 2003 (Korieth, 2004). Enrollment rates dropped from 75 percent in 2000 to 59 percent in 2006, and retention rates fell from 69 percent to 48 percent during that same period (Kaitin, 2008).
In an attempt to understand how to improve patient recruitment and retention rates, the Center for Information and Study on Clinical Research Participation conducted focus groups among study volunteers to probe for
factors that most inform and educate clinical research participants. This research was conducted in 2009 at Piedmont Medical Group in North Carolina. In these focus groups, participants emphasized the essential role of study staff and healthcare professionals in volunteer recruitment and retention. They consistently described relationships with study staff as tough but fair, disciplined but supportive. At a time when recruitment and retention strategies and tactics tend to marginalize the role of study staff and trusted healthcare professionals, the results of these focus groups strongly suggest the need to engage these professionals more effectively as real assets in the clinical trial process.
The focus group participants revealed core motivations of all study volunteers regardless of age and socioeconomic status. These motivations can serve as a blueprint for patient and public education and outreach. Study volunteers
- want to feel that they are taking control of their medical condition and well-being,
- want to develop personal relationships with study staff,
- want to be treated as human beings, and
- want to know that their participation will make a difference.
Participating in medical research as a way to steer personal and public health reinforces the drive toward patient-centered health care. Yet the focus groups clarified that “patient-centered” does not mean that they want medical autonomy. Despite common expressions such as “take control,” study participants from the focus groups explained that they do not seek total independence in their efforts to improve their well-being. The prospect of being accountable to the research coordinator spurred and sustained volunteers’ interest in trials. In fact, the volunteer–study staff relationship forms and solidifies at several critical junctures. When research sites reach out to potential volunteers with the right messages and modes of communication at decisive moments and invite them to begin conversations, the relationship grows into a lasting commitment to the center and its staff.
The focus group participants’ comments show that money-focused recruitment campaigns and comparisons of research volunteers to “guinea pigs” or “lab rats” depersonalize the trial experience and keep the volunteers from feeling as though they are truly part of an extended research team. Typical perceptions of clinical research participation must shift before people can take part proudly and comfortably in a research community. The emphasis on monetary compensation in media and recruitment rhetoric impedes public and participant recognition that volunteers are part of a vital exchange in which they are compensated for sacrificing their time, effort, and even physical welfare.
To be effective, the education provided before and after trials must reflect what volunteers expect, need, and want to know about clinical research. The public’s primary source for education about the clinical research enterprise is the media, which tend to focus on sensationalistic accounts of human error, concealment, fraud, and corruption. More than two-thirds (69 percent) of Americans are exposed to information on clinical research studies through television, radio, print media, and Internet advertising. Only one of seven adult Americans is exposed to information about clinical research studies from a primary or specialty care physician (HarrisInteractive, 2004). Although the public has the greatest trust in information from healthcare providers, the medical and health professional communities are largely absent from efforts to educate the public and prospective volunteer communities. In a recent survey conducted among board-certified physicians in active community practices throughout the United States, fewer than half reported referring their patients to clinical trials, with an average referral rate for each physician of less than one patient per year (Getz and Faden, 2008).
Without broad understanding and context, recruitment advertising and promotional messages are met with, at best, passing curiosity from the public and prospective volunteers. Only 20 percent of those diagnosed with severe and life-threatening illnesses report considering clinical trials as a healthcare option (HarrisInteractive, 2004). Despite a wealth of online information available, less than 5 percent of the general public knows where to find information about relevant clinical trials (Getz, 2004). And the public is largely unaware of where clinical research is taking place. A 2005 public poll found that 62 percent of respondents were unable to name a single institution, company, or organization where medical and health research is conducted (Woolley and Propst, 2005). Research sponsors rarely, if ever, respond to media coverage, as government and corporate employees are usually instructed not to interact with journalists for fear of bringing more attention to a story or of appearing defensive and self-serving. As a result, the public is receiving a largely one-sided education in the clinical trials industry from the media.
Outreach Initiatives and Their Impact
If the public is to be engaged, the stage must be set with a national public education media campaign. The Center for Information and Study on Clinical Research Participation developed a public service campaign, with pro bono support from the international advertising agency Ogilvy HealthWorld, to educate and win over the public regarding the importance of participation in clinical research. During the campaign’s yearlong development process and extensive focus group testing, strong support was expressed for the easy-to-remember messaging and acknowledgment of or-
dinary people’s contributions to public health. During screenings before test audiences, the ads were lauded for their “humanity and emotional appeal.” Viewers declared, “These people are heroes in their own way,” “They’ve done something great for all of us,” and “I see the benefit of clinical research to society.”
This “Medical Heroes” public service campaign was market tested in 30 sites across 18 U.S. markets by Eli Lilly and Company. In the first wave of the market test, the control group was established as 12 markets that ran their typical recruitment ads; in 6 comparable markets, concurrent “Medical Heroes” ads were run, as well as recruitment ads, and these markets showed a 38 percent increase in patient recruitment rates relative to the control group. The test was repeated, and the results of the second wave showed that rates of response to recruitment ads more than doubled in the markets where the “Medical Heroes” campaign was run. The campaign met its ultimate goal of providing the public with increased awareness of research participation and an improved perception of clinical research volunteers.
Another Center for Information and Study on Clinical Research Participation initiative is a grassroots education and outreach process known as “AWARE for All-Clinical Research Education.” AWARE programs, held in major cities across the United States, bring together disease advocacy groups, hospitals and healthcare organizations, educational institutions, and community organizations to provide AWARE’s message directly to their constituents. In addition, distinguished local politicians and opinion leaders, physicians, healthcare providers, and clinical research professionals serve as keynote speakers and workshop leaders—all volunteering their time to help educate the public.
To date, more than 300,000 people have been impacted by the program. AWARE has put a human face on the people who volunteer for clinical trials while building public understanding of the risks and benefits of participating. The initiative is creating a movement at the local level, and there is a need to bring this form of outreach to many more communities. When asked whether they were more or less likely to participate in a clinical trial after attending AWARE, 75 percent of attendees responded “more likely.”
An additional example of an outreach initiative is post-trial communication with research volunteers. The Center for Information and Study on Clinical Research Participation and Pfizer collaborated to test a new process for routinely communicating clinical trial results to study volunteers after their participation has ended. Between June and December 2009, trial results for Celebrex®/Celecoxib and Sutent®/Sunitinib were translated into lay language by a team of consumer, science, and medical writers and published in print, web, and audio formats. These summaries were then tested
in focus groups among volunteers who had participated in the studies. The pilot study results demonstrate that a process for preparing and disseminating summaries of trial results to patients following their participation in clinical trials is feasible. Moreover, patients reacted very positively to the variety of formats and showed marked improvement in their comprehension of their clinical trial findings. Study personnel are also very receptive to disseminating summaries of trial results to their volunteers.
A final example of a public education and outreach initiative is the development of a traveling exhibit for science museums. Still in the preliminary planning stages, such an exhibit would provide inquiry-based, multimedia learning experiences focused on the how-to and importance of health research as presented by practicing scientists. It would use an innovative mix of video storytelling and digital support technologies to show people how real-world scientists conduct their research and create a continuum from basic to translational science to clinical trials that produce new treatments and solutions. This type of exhibit would highlight what it means to participate in a clinical trial and the impact of participation on science and drug discovery.
Conclusions
Despite low levels of trust and confidence today, there is no evidence to suggest that the public will abandon the clinical research enterprise outright. A foundation of general public support exists on which to rebuild public confidence and trust through education and outreach initiatives. Such initiatives need to focus on improving public awareness and appreciation of the study volunteer and the value of clinical research to the public health; repairing the credibility of research sponsors, study staff, and regulatory and human subject protection professionals; and engaging the public as partners in the development of new medical and health advances. Given how far public support has fallen, however, there is no time to waste in repairing and rebuilding trust and confidence.
To enhance the culture of patient contributions to learning in health care, a portfolio of strategic initiatives is needed, as shown in Figure 2-5. If general education about and awareness of the clinical research process are enhanced and if patients are enabled to participate because of the support network and tools provided to help them become active participants in clinical trials, recruitment and retention in trials will improve. With this solid foundation, volunteers will become a community of participants and ultimately the ambassadors of a process that advances medical science and improves the public health.
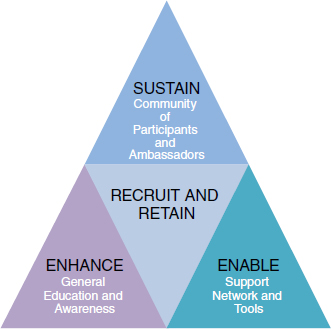
FIGURE 2-5 Model for enhancing the culture of patient contributions to learning in health care. A portfolio of strategic initiatives is needed that enhances the public’s general education about and awareness of the clinical research process and enables patients to become active participants in clinical trials. This foundation will lead to improvements in patient recruitment and retention and ultimately to the formation of a community of research volunteers.
SOURCE: Center for Information and Study on Clinical Research Participation model—permission for use in this publication authorized by Diane Simmons, President and CEO.
REFERENCES
Advisory Commission on Consumer Protection and Quality in the Health Care Industry. 1998. Quality first: Better health care for all Americans. http://www.hcqualitycommission.gov/final/ (accessed October 11, 2010).
Balas, E., and S. Boren. 2000. Managing clinical knowledge for health care improvement. In Yearbook of medical informatics. Bethesda, MD: National Library of Medicine. Pp. 65-70.
Barbaresi, W., S. Katusic, R. Colligan, A. Weaver, and S. Jacobsen. 2005. The incidence of autism in Olmsted County, Minnesota, 1976-1997: Results from a population-based study. Archives of Pediatrics & Adolescent Medicine 159(1):37-44.
Beghi, E., L. Kurland, D. Mulder, and W. Wiederholt. 1985. Guillain-Barre syndrome. Clinicoepidemiologic features and effect of influenza vaccine. Archives of Neurology 42(11):1053-1057.
Braunstein, J., N. Sherber, S. Schulman, E. Ding, and N. Powe. 2008. Race medical researcher distrust, perceived harm and willingness to participate in cardiovascular prevention trials. Medicine 87(1):1-9.
Califf, R. M. 2009. Clincal research sites—the underappreciated component of the clinical research system. Journal of the American Medical Association 302(18):2025-2027.
Chalkidou, K., D. Whicher, W. Kary, and S. Tunis. 2009. Comparative effectiveness research priorities: Identifying critical gaps in evidence for clinical and health policy decision making. International Journal of Technology Assessment in Health Care 25:241-248.
Center for Information and Study on Clinical Research Participation. 2006. Survey of 900 people’s perceptions of clinical research. http://www.ciscrp.org/professional/surveys/survey_dec2006_slide1.html (accessed October 11, 2010).
Conway, P., and C. Clancy. 2009. Comparative-effectiveness research—implications of the Federal Coordinating Council’s report. New England Journal of Medicine 361:328-330.
Dougherty, D., and P. Conway. 2008. The T3 roadmap to transform U.S. health care: The how of high quality care. Journal of the American Medical Association 299(19):2319-2321.
FCC (Federal Coordinating Council for Comparative Effectiveness Research). 2009. Report to the President and Congress. http://www.hhs.gov/recovery/programs/cer/cerannualrpt.pdf (accessed October 11, 2010).
Gabriel, S., W. O’Fallon, L. Kurland, C. Beard, J. Woods, and L. R. Melton. 1994. Risk of connective-tissue diseases and other disorders after breast implantation. New England Journal of Medicine 330(24):1697-1702.
Gabriel, S., J. Woods, W. O’Fallon, C. Beard, L. Kurland, and L. R. Melton, 3rd. 1997. Complications leading to surgery after breast implantation. New England Journal of Medicine 336(10):677-682.
Getz, K. 2004. Survey of 1,170 adults at the clinical trial congress in Philadelphia. Institute for International Research.
Getz, K., and L. Faden. 2008. Racial disparities among clinical research investigators. American Journal of Theraputics 15(1):3-11.
HarrisInteractive. 2002. There are many reasons why people are reluctant to participate in clinical trials. HealthCare News 2(7):1-4.
———. 2004. Views and attitudes of clinical research studies. HealthCare News 4(15):1-2.
———. 2005. New survey shows public perceptions of opportunities for participation in clinical trials has decreased slightly from last year. HealthCare News 5(6):1-13.
———. 2007. Lack of trust in both FDA and pharmaceutical companies makes drug safety a concern for many. HealthCare News 7(6):1-5.
IOM (Institute of Medicine). 2009. Initial national priorities for comparative effectiveness research. Washington, DC: The National Academies Press.
———. 2010. Redesigning the clinical effectiveness research paradigm: Innovation and practice-based approaches: Workshop summary. Washington, DC: The National Academies Press.
———. 2011. Learning what works: Infrastructure required for comparative effectiveness research: Workshop summary. Washington, DC: The National Academies Press.
Kaiser Family Foundation. 2008. Public opinions spotlight.
Kaitin, K. 2008. Growing protocol design complexity stresses investigators. Tufts.
Korieth, K. 2004. Taking patient recruitment in-house. CenterWatch Monthly Newsletter, 6-7.
Kurland L. T., L. R. Elveback, and F. T. Nobrega. 1970. Population studies in Rochester and Olmsted County, Minnesota, 1900–1968. In The Community as an Epidemiologic Laboratory: A Casebook of Community Studies. Edited by I. I. Kessler and M. L. Levin. Baltimore, MD: Johns Hopkins Press. Pp. 47-70.
Kurland, L., and C. Molgaard. 1981. The patient record in epidemiology. Scientific American 245(4):54-63.
Lauer, M. S., and F. S. Collins. 2010. Using science to improve the nation’s health system: NIH’s commitment to comparative effectiveness research. Journal of the American Medical Association 303(21):2182-2183.
Melton, L. 1996. History of the Rochester Epidemiology Project. Mayo Clinic Proceedings 71(3):266-274.
———. 1997. The threat to medical-records research. New England Journal of Medicine 337(20):1466-1470.
Rocca, W., J. Bower, D. Maraganore, et al. 2007. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology 69(11):1074-1083.
Rocca, W., B. Grossardt, M. de Andrade, G. Malkasian, and L. Melton, 3rd. 2006. Survival patterns after oophorectomy in premenopausal women: A population-based cohort study. The Lancet Oncology 7(10):821-828.
Rocca, W., B. Grossardt, and D. Maraganore. 2008. The long-term effects of oophorectomy on cognitive and motor aging are age dependent. Neurodegenerative Diseases 5(3-4):257-260.
VanLare, J., P. Conway, and H. Sox. 2010. Five next steps for a new national program for comparative-effectiveness research. New England Journal of Medicine 362(11):970-973.
Woolley, M., and S. Propst. 2005. Public attitudes and perceptions about health-related research. Journal of the American Medical Association 294(11):1380-1384.






















