Appendix A
Projecting the Burden of HIV/AIDS
In planning for a long-term response to the HIV/AIDS epidemic in Africa in the coming decade, the global community needs to ask how reliable projections for different variables can be. How well can we project the future course of the epidemic? How well can we predict the impact of treatment interventions? How well can we predict the impact of prevention efforts? How well can we predict the epidemic’s social, demographic, and financial burdens? How well can we predict the financial resources available to combat HIV/AIDS?
EPIDEMIOLOGICAL PROJECTIONS
The ability to predict the course and impact of the HIV/AIDS epidemic is reasonably good over the short term because the current situation evolves gradually, driven by biological or other relatively well-understood processes (e.g., those already infected progress to AIDS). Over the longer term, less predictable factors could alter the risk of HIV infection and the resources available to combat the epidemic. Eventually, game-changing technologies, such as a cure or vaccine, may exist, but the availability of resources will continue to impact populations’ access to them. One of the greatest uncertainties is the availability of treatment for those who need it, which will determine the numbers of deaths and impact the incidence of new infections.
In the absence of antiretroviral therapy (ART), the progression of HIV from infection to AIDS and death is a slow process with a median time to death of 11 years, with survival depending on age and other factors (Todd et al., 2007). Most of those progressing to AIDS and needing treatment over the next decade are already infected with the virus. Unlike many other aspects of this epidemic,
the need for treatment is predictable. The number of people on treatment over time is a function of the rates of diagnosis, treatment initiation, and survival on treatment. These rates are also predictable given assumptions about investment in treatment programs and the organization of those programs. The scale and success of treatment will determine the impact of treatment interventions. The social and economic burden of HIV/AIDS will depend upon the success of treatment programs, as well as the efforts undertaken to care for those affected. In contrast, the accumulation of new infections over time is more difficult to predict since it will depend on changing patterns of risk in the population, which in turn are influenced by demographic, cultural, and social phenomena, as well as the impact of preventive interventions.
Over the course of the epidemic, some patterns of risk behavior will likely change, making it difficult to link the current observed prevalence of HIV to measures of risk behavior with precision. Furthermore, the sensitive, private, and often stigmatized behaviors that place many at risk of acquiring HIV may not be accurately reported in surveys (Fenton et al., 2001; Slaymaker, 2004), making it difficult to relate incidence to behavior and predict how changes in behavior will influence incidence (Garnett et al., 2006). A straightforward assumption is that trends in HIV incidence will continue in the future. With incidence trends stable, future prevalence will be determined largely by the future expansion of ART, which has both direct and indirect effects on prevalence. The direct effect of ART expansion is to lengthen the lives of people with HIV, which consequently increases prevalence. ART also has several possible indirect effects on HIV prevalence, both beneficial and adverse, through its effects on transmission and therefore on incidence (see Table A-1).
In the aids20311 predictions, it is assumed that those on treatment have a reduced infectiousness (to 20 percent of the original transmission risk from an infected person to susceptible contacts) (aids2031 Consortium, 2011; Hecht et al., 2010). In formulating its baseline projections, the committee developed a model incorporating two of the biological effects from Table A-1—the reduction in transmission among those on effective ART and the increased exposure to risk caused by a longer HIV-infected lifespan. Since the degree to which the two behavioral effects in Table A-1 will manifest as treatment access increases is still unknown and will likely be responsive to national HIV/AIDS policies, the baseline model ignores these and the other indirect effects of ART on HIV transmission.2
Others have made projections of the impact of particular interventions over
TABLE A-1 Possible Effects of ART on HIV Transmission
|
Type of Effect |
Direction of Effect |
|
|
Beneficial (Slows transmission) |
Adverse (Speeds transmission) |
|
|
Biological |
Reduce infectiousness: ART lowers viral loads and may therefore lower the perepisode risk of transmission. |
Select for resistance: Imperfect adherence to ART selects for resistant strains of the virus, which can then be transmitted. |
|
|
|
Longer duration of infectivity: The greater longevity of HIV-infected people receiving ART has the unintended negative consequence of increasing the period in which the patient can transmit the virus. |
|
Behavioral |
Encourage prevention: ART may increase the uptake rates of prevention activities, particularly voluntary counseling and testing. |
Encourage disinhibition: People receiving ART, and both those who are and are not HIV-infected in the surrounding community, may engage in more risky behaviors than they would if ART were unavailable. |
|
SOURCE: Over et al., 2004. |
||
the next few decades. Granich and colleagues explore the impact of the so-called “test and treat” policy using a homogeneous population model in which the transmission coefficient within the force of infection is inversely proportional to prevalence (i.e., the model assumes that as prevalence increases, the risk of transmission decreases) (Granich et al., 2009a).3 This counterintuitive assumption allows prevalence to saturate, but generates some odd properties since increased treatment decreases incidence through this mechanism in addition to the calculated reduction in the transmission probability. Granich and colleagues also assume that diagnosis of HIV reduces risk behavior by 50 percent, a possibility supported only by studies of self-reported behavior change shortly after the start of treatment (Venkatesh et al., 2010). The authors do not consider the possibility that those undiagnosed would increase their risk behavior in response to the per-
ception that ART is free and easily accessible, thereby diminishing the perceived disadvantage of becoming infected. A further exploration of rapid testing and immediate treatment found that it could have a large impact on incidence but that heterogeneity in risk behavior prevented elimination of HIV transmission from occurring (Dodd et al., 2010).
Models of Johnson representing South Africa show that treatment improves survival and that prevention interventions can have an impact (Johnson, 2010). Models of Phillips and colleagues focus on patterns of treatment and the evolution of drug resistance within populations, predicting only marginally worse outcomes for syndromic management over a 20-year period (Phillips et al., 2008). However, the predicted risks of resistance are greatly increased if the viral load of patients on treatment is not monitored (Phillips et al., 2009).
The impact of prevention interventions in the Goals model4 used for aids2031 (Stover et al., 2003) is based on a review of trials of interventions with an estimate of efficacy for different specified risk groups in the populations. (See Box A-1 for a discussion of the use of the term “risk group.”) The implication of this approach is that only where efficacy has been observed in a trial can an intervention be assumed to reduce risk in the population (Bollinger, 2008).
PREDICTIONS OF FUTURE NUMBERS ON TREATMENT
To understand the impact of current and future treatment patterns, one must be clear as to what is meant by “coverage” of treatment in a given year, how this relates to the number of people who are newly recruited to treatment over the course of the year, and how these numbers in turn alter the expected pattern of mortality. Generally, coverage is defined as the fraction of those in need of treatment at a particular time who are receiving treatment. Thus, when the treatment guidelines of the World Health Organization (WHO) identified those with a CD4 count of less than 200 as in need of treatment, the coverage of treatment was the number of HIV-positive people treated divided by those whose CD4 count was below 200 who were still alive; the same calculation would apply for WHO’s 2010 threshold for needing treatment of a CD4 cell count below 350. This calculation includes those who do and do not know they are infected and relies on an estimate of decay in CD4 count, as it is independent of whether people know their CD4 count. A more restricted definition of coverage would be the fraction on treatment among those who know that they are infected and that they meet the treatment criteria. Coverage targets use the definition that coverage is the frac-
|
BOX A-1 Risk Groups The term “risk group” is used in describing and predicting the future course of an HIV/AIDS epidemic and in attempting to improve the allocation of resources across HIV/AIDS prevention alternatives. Epidemiological models of a sexually transmitted infection (STI) require defining subsets of the population who, at any one time, are more likely than others to engage in certain sexual behaviors that determine their risk of contracting and transmitting the infection. Within society, membership in such groups is highly dynamic. For example, children not engaging in sexual intercourse mature and initiate sexual activity, which places them in a risk group. Likewise, people marry, which can put them in a certain risk group, and if they divorce, they enter a different risk group. Some people begin engaging in transactional sex, thereby entering a risk group; when they cease this activity, they exit the risk group. However, both the concept of risk groups and the use of the term “risk group” raise ethical issues. It can be contended that the concept and the term connote prejudice toward the members of such putative groups, an attitude that could engender intolerance or even human rights abuses. Nonetheless, the committee was charged with presenting as clear and accurate a picture as possible of the future course of the HIV/AIDS epidemic and its consequences for Africa and the United States. The committee believes that the concept of risk groups is essential to this task and that this concept must be communicated as clearly and transparently as possible. At the same time, the committee wishes to avoid the possibility that its choice of words could be a source of intolerance or abuse of any person anywhere. To reconcile these objectives, the committee decided to use the term “risk group,” explaining that it could refer to people at particularly low or particularly high risk, and that effective interventions must recognize the right of individuals to choose their own sexual behavior while simultaneously facilitating the choice of safer behavior. |
tion of those in need of treatment that receive treatment; however, as people are put on treatment they survive longer, and the coverage of treatment accumulates.
In African health care facilities that deliver publicly funded ART services, recruitment of a new patient entails an implicit commitment by the facility and its funders to continue to provide that treatment under the same financial arrangements for the patient’s lifetime. To meet this commitment to enrolled patients implies that, when resources are scarce, the facility must give priority to existing enrolled patients over patients who have not yet begun treatment. This commitment limits the facilities’ freedom of choice when approached by people with HIV infection who have not yet begun treatment. Unlike a private-sector health care facility, which might decide to terminate its relationship with an existing patient to make room for a new patient, a public-sector facility that recognizes its
commitment to existing patients can enroll new patients only to the extent that it has the additional capacity to do so. This raises the possibility that among those who need ART by any definition and have not yet received a commitment, only a fraction will be started on treatment in a given year. This fraction, which is defined as the proportion of unmet need that is added to treatment rolls in a given year, is termed the “uptake rate.” (See Annex A-1 for a model of the relationship among initiation, coverage, and mortality.)
Future Numbers Needing Treatment with Different Assumptions About Changes in Incidence
The predicted number in need of treatment will depend in the long term on the pattern of incidence in the future. If incidence remains high, the cumulative number in need of treatment will rise. If incidence declines dramatically, however, it will still take some time before treatment need also declines. The slow response of the epidemic to even quite dramatic changes in either treatment or prevention policy is due to the long incubation period between HIV infection and the onset of symptoms (approximately 9 years from HIV acquisition to a CD4 count of 200, with variation depending on age, prior health condition, and other factors) (Aalen et al., 1997; Van Der Paal et al., 2007) and to the long survival of patients who receive ART. Most of the consequences of current policy changes will be indiscernible for years, making it difficult for citizens to appreciate the benefits of good policy decisions or to understand the costs of bad ones. Some of the decisions currently faced by policy makers relate to treatment guidelines and policies toward preventing mother-to-child transmission (PMTCT).
Treatment Guidelines
The WHO treatment guidelines reflect what is known from clinical trials and observational studies about survival on different ART regimens, the toxicities of drugs, and problems with adherence. Survival of those initiating ART is dependent on the CD4 cell count at initiation, with improved survival seen at higher counts. Over time, as drugs have become less toxic, it has become more acceptable to initiate treatment earlier, which led to the change in the WHO guidelines in 2010. That change in turn dramatically increased the fraction of those infected who need treatment and reduced coverage. The new guidelines also include a shift from recommending stavudine in first-line drug combinations to the less toxic but more expensive tenofovir. The guidelines represent what is believed to be best clinically for the individual, but following them will entail higher costs per patient and thus reduce the number of patients served within any given budget. (See the discussion of policy choices and associated trade-offs in Chapter 2.)
Impacts on Infants and Children
The WHO 2010 updated pediatric ART guidelines advise that all HIV-positive children less than 24 months of age be started on ART (WHO, 2010). The number of children infected with HIV is a function of the HIV prevalence in women of reproductive age; the fertility rate of those women; whether and for how long they breastfeed infants; whether PMTCT is used; and if it is, how efficacious the regimen is in preventing vertical transmission. The infection of children can be prevented directly by implementing PMTCT interventions or indirectly by preventing the spread of HIV through women.
Young people aged 15–24 account for an estimated 45 percent of new HIV infections worldwide. Globally, the number of children younger than 15 living with HIV increased from 1.6 million in 2001 to 2.1 million in 2008. Almost 90 percent live in sub-Saharan Africa (UNAIDS, 2008; UNAIDS and WHO, 2009).
Mother-to-child, or perinatal, transmission continues to account for a substantial, although decreasing, portion of these new HIV infections in many African countries (UNAIDS and WHO, 2009). It is estimated that 90 percent of children living with HIV acquired the virus during pregnancy, birth, or breastfeeding—all forms of HIV transmission that can be prevented (UNAIDS, 2008). In Swaziland, children were estimated to account for nearly one in five (19 percent) new HIV infections in 2008 (Mngadi et al., 2009; UNAIDS and WHO, 2009). Perinatally acquired infection accounted for 15 percent of new HIV infections in Uganda in 2008 (UNAIDS and WHO, 2009; Wabwire-Mangen et al., 2009).
HIV is the underlying reason for more than one-third of all deaths in children under the age of 5 (UNAIDS, 2008). Without ART, the progression of HIV infection in children is particularly aggressive, and many children die at a young age (UNAIDS, 2008). Among infants and children exposed to HIV, access to early testing, care, and treatment is insufficient. In 2009, in 54 reporting countries, only 15 percent of children born to HIV-positive mothers received an HIV test within the first 2 months of life (WHO, 2010). ART coverage among children less than 15 years of age in 2009 was only 28 percent (up from 22 percent in 2008); a mere 35 percent of infants in need received PMTCT prophylaxis in 2009 (up from 32 percent in 2008); and the percentage of pregnant women living with HIV receiving ART for PMTCT in 2009 was 53 percent (up from 45 percent in 2008) (WHO, 2010). Globally, however, coverage for PMTCT services did rise from 10 percent in 2004 to 45 percent in 2008; the drop in new HIV infections among children in 2008 suggests that these efforts are saving lives (UNAIDS and WHO, 2009) (see Figure A-1).
The causal links from PMTCT policy to treatment needs run in both directions. Not only does PMTCT expansion reduce the number of infants needing treatment, but the choice of an ambitious “test-and-treat” strategy for ART expansion would require initiating virtually all HIV-infected expectant mothers on lifetime ART regardless of CD4 count. This strategy would involve a significant
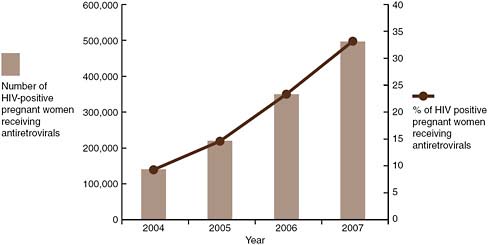
FIGURE A-1 Number and percentage of HIV-positive pregnant women receiving antiretroviral prophylaxis for PMTCT.
SOURCE: UNAIDS, 2008.
proportion of HIV-infected mothers starting therapy earlier than others and therefore remaining on treatment for a longer period of time.
AIDS orphans are defined as children who lose either a mother or father or both through HIV/AIDS-associated mortality. To predict the number of AIDS orphans, one must know the mortality rate of adults, their fertility history in the 18 years prior to death, and the survival of any children. The effort is complicated by vertical transmission of HIV leading to higher-than-background mortality in some children and the influence of HIV infection on reducing fertility. The use of ART makes it more likely that children will be born to HIV-infected mothers and less likely that they will acquire HIV infection vertically. However, ART also makes it less likely that the parent will die, and, of course, living parents decrease the societal burden of orphaned children. In general, ART should reduce the rates of orphanhood. However, a poor-quality program that delays mortality only for a short while could have the perverse effect of increasing numbers of orphans.
What Drives the Differences Among Projections?
Models include the details and assumptions required to address the questions they are designed to answer. Some models of future treatment need and patterns of survival on treatment start with an HIV-infected population and represent their declining CD4 count—either explicitly, as a function of time since infection, or using categories representing ranges of CD4 count through which individuals progress (Hallett et al., 2008; Kimmel et al., 2005; Phillips et al., 2008). Such models are useful in assessing the benefits of different treatment strategies and
provide a framework for exploring the cost-effectiveness of treatments. Other models represent transmission and include the influence of treatment or behavior change on the spread of infection (Baggaley et al., 2006; Blower et al., 2005; Granich et al., 2009a). Two approaches are possible in models exploring the impact of HIV prevention interventions. The first is to use changes in proximate risk behaviors that are the intermediate endpoints measured in trials. This is a relatively conservative approach adopted in the aids2031 models. A second approach is to assume that prevention interventions can generally have an impact reflecting an effect of “effort.” (See Annex A-2 for a more thorough discussion of what drives the differences among projections.)
ECONOMIC PROJECTIONS
The committee’s economic projections address the annual costs of treating a single patient, potential efficiencies, the cumulating costs of treatment, and the nature of treatment.
Annual Costs of Treating a Single Patient
The costs of treatment have been estimated in a number of different ways. One method is to seek prices for the various components of treatment (Hecht et al., 2009, 2010). Another method is to survey treatment programs and review the costs of their activities (OGAC, 2010c). A third method is to look at the overall costs and the number of people on treatment. In the estimation of treatment costs for aids2031, the costs include the unit costs of antiretroviral drugs, estimated as $167.65 for first-line and $1016.48 for second-line drugs based on average costs in 2007. Added to these figures are laboratory costs of $190.94 per patient per year and service delivery costs of $72.05 per patient per year for Africa; the service delivery costs are assumed to be greater in other regions. The result is an estimate of $430.64 per person per year treated (aids2031 Costs and Financing Working Group, 2010).
The above estimate is significantly below the actual costs based on the U.S. President’s Emergency Plan for AIDS Relief (PEPFAR’s) survey of facilities it supports, which resulted in an estimated total mean cost of $812 per patient per year (OGAC, 2010c). Of this total, PEPFAR bore an estimated $402 per patient per year on first-line treatment, with the rest covered by other donors and national resources. The purchase of antiretroviral drugs accounted for 39 percent of costs. Of those costs captured at the facility level, 20 percent were incurred above the facility level by supervising or sponsoring organizations, 5 36 percent were recurrent costs other than antiretroviral drugs, and only 5 percent were health system
|
5 |
Comparison with Figure A-2 below suggests, and the PEPFAR facility study authors confirm, that this facility-level study captured only a small portion of costs incurred above the facility level. |
investments (OGAC, 2010c). Included in these calculations are clinical staff salaries and benefits, laboratory and clinical supplies, nonantiretroviral drugs for opportunistic infections, building utilities, travel, contracted services, building renovation and construction, laboratory and clinical equipment, training, and a buffer stock of antiretroviral drugs.
These figures are somewhat puzzling when one compares the number of people treated and the overall costs reported by PEPFAR. Figure A-2 shows the average treatment costs per year per downstream PEPFAR patient computed from the Office of the Global AIDS Coordinator’s (OGAC’s) aggregate data (OGAC, 2010a) versus the total number of downstream patients. The best-fitting linear-in-logarithms prediction (shown as a diagonal line in the figure) suggests mild economies of scale for these national PEPFAR programs, with each 10 percent increase in scale associated with a 3 percent decline in average cost.
However, when one looks at the data, PEPFAR expenditure per patient is substantially greater than would be expected from PEPFAR’s own survey of facilities, suggesting that more than half of PEPFAR’s AIDS treatment expenditures are consumed before they reach the treatment site. Some of these additional costs above the facility level are justified as a contribution to the quality and efficiency of ART service delivery on the ground, while others are probably due to inadequate coordination, overly complex administrative procedures, and even fraud and abuse. The degree of efficiency in the delivery of HIV/AIDS services and the determinants of efficient, quality service delivery deserve active investiga-
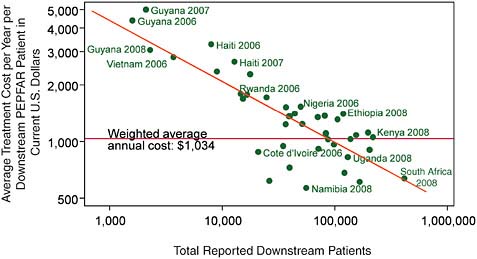
FIGURE A-2 Average unit costs of treatment reported by PEPFAR show mild economies of scale.
SOURCE: Committee analysis of OGAC data from OGAC 2010a,b.
tion. Only through an understanding of the variation in quality-adjusted efficiency across service facilities and contractors and the determinants of that variation can procurement policies be reformed to deliver more high-quality services for U.S. taxpayer dollars. PEPFAR is currently deepening its investigation of these issues, but without fully disclosing its methods or the data it is gathering at public expense. By shining a spotlight on inefficient or low-quality service providers, full public disclosure would enable improved efficiency in HIV/AIDS service delivery, thereby serving the interests of not only U.S. taxpayers but also the citizens of affected recipient countries.
The costs of treatment do not normally include the costs of counseling and testing—a necessary precursor to treatment in that it provides the diagnosis of infection and is the start of the pathway into care. However, counseling and testing could also be seen as prevention—directly if it influences the behaviors of those tested and indirectly if it improves the acceptability of the infection within the population, reducing stigma and risk. Evidence for the effect of counseling and testing on behavior is limited, with data suggesting that those diagnosed as HIV-positive reduce their risk behavior for a period of time; those diagnosed as HIV-negative do not change their behavior; and where there is couples testing, discordant couples significantly increase their condom use (Hallett et al., 2009a; Sweat et al., 2000). Further trial results powered to detect an effect of testing on HIV incidence are expected soon. Depending on these results, some portion of these testing costs should be added to the above costs of treatment, inflating the cost of treatment even more.6
Potential Efficiencies
Two options are available for expanding the number of people receiving ART: an increase in resources or an increase in efficiencies. The costs of antiretroviral drugs have fallen dramatically since the drugs were introduced in 1996 and currently constitute a minority of the costs of ART. To increase efficiency (i.e., decrease costs per patient), there may still be some scope for reduced drug costs, but the impact would be limited by other costs for which there may be more scope for reduction. There is a trend toward better-quality treatment with earlier initiation, viral load monitoring to ensure success, monitoring of adherence and other support services, more expensive drugs with a better profile, and second-line and salvage therapy. This trend results in increased costs per person treated, allowing fewer to receive treatment within a fixed budget. One option for achieving greater efficiencies is to make treatment simpler (UNAIDS, 2010), with
the implication in the short term (i.e., absent the development of more effective simple regimens) that the treatment would be less effective. Another option is to identify waste and inefficiencies in the organization of services.
Setting and accepting a target for the number of people receiving treatment that is in line with current treatment costs results in little incentive to reduce those costs. There is a negative correlation between the number of HIV-infected individuals in a country and the cost per person treated, suggesting modest economies of scale with respect to the magnitude of a national AIDS treatment program. However, further expansion will entail two sources of diseconomies as existing infrastructure and human resources are pushed to their limits. First, within any individual facility there is a point of minimum efficient scale beyond which further increases in patient load would entail increases in average cost per patient. Therefore, to reach more people, new facilities need to be developed, with appropriate staffing, support, and supplies. These facilities are likely to cover a smaller number of patients as they become more remote, so diseconomies are likely to develop related to the smaller-scale production. See Chapter 5 for a discussion of one promising policy response: increased delegation of tasks through task sharing with less specialized health care providers.
In looking at future numbers of HIV infections and numbers receiving treatment, the size of the population is important. Many African populations continue to grow, and population growth means that over time for a constant rate of infection, an increasing number of infections will be seen.
Cumulating Costs of Treatment
There are many models of the future course of the HIV/AIDS epidemic, which differ in their predictions according to the assumptions made. One model, produced for the aids2031 project, predicts financing needs for both treatment and prevention for Africa through 2031 under four different scenarios: current trends, rapid scale-up of treatment and prevention, “hard choices,” and “structural change” (an assumed change in behavior related to structural interventions [Gupta et al., 2008]).
According to this model, total required financing will be between $11 billion and $18 billion per year in 2020, depending on the scenario, as shown in Figure A-3. In this figure, current trends indicates that coverage of key interventions continues to expand to 2015 as it has in the past few years; rapid scale-up indicates that political will to achieve universal access is strong, and resource availability continues to grow rapidly; hard choices indicates that resources for HIV/AIDS programs are limited, so there is a focus on scaling up only the most cost-effective approaches for prevention; and structural change indicates a greater focus on structural change that can reduce vulnerability to HIV/AIDS and produce a more sustainable response (aids2031 Costs and Financing Working Group, 2010).
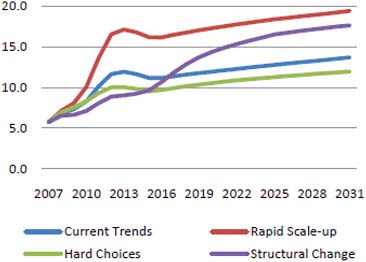
FIGURE A-3 One model predicts total financing needs of $11–18 billion in 2020, of which more than 25 percent would be for AIDS treatment.
SOURCE: aids2031 Costs and Financing Working Group, 2010.
HIV/AIDS prevention costs are difficult to estimate because they are so elastic and because their efficacy is so poorly understood (Marseille et al., 2007). However, HIV/AIDS treatment costs are relatively easy to project, as discussed above and in Chapter 2.
It should be noted that treatment costs accumulate over time. Depending on policy choices currently available, the cumulated cost of HIV/AIDS treatment between 2010 and 2050 is likely to be between $200 billion and $800 billion (Figure A-4), assuming that incidence declines as shown in Panel b of Figures 2-3 and 2-4 in Chapter 2.
Nature of Treatment
In considering the future of the HIV/AIDS epidemic and associated patterns of treatment, the characteristics of the management of patients can be considered.
Syndromic Initiation
If HIV-positive individuals are diagnosed through testing for antibodies and the facilities for CD4 counts are unavailable, the only criterion for determining whether a patient should start therapy is symptoms. If diagnosis of HIV infection is timely, this reliance on symptoms will automatically reduce the average CD4 count at which patients start treatment and their chances of survival. Likewise,
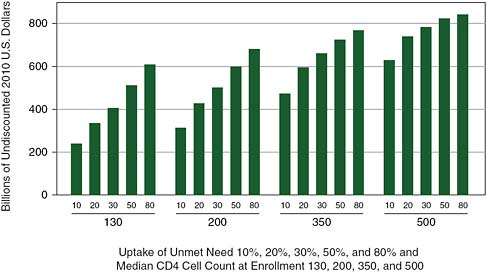
FIGURE A-4 Cumulated cost of HIV/AIDS treatment in Africa through 2050 by the proportion of unmet need enrolled each year and the median CD4 cell count at enrollment.
NOTE: Using the committee model, a bivariate sensitivity analysis comparing the influence of two parameters was used to show trade-offs, holding constant a set of epidemiological and cost assumptions as detailed in Annex A-1.
SOURCE: Committee projections using data from UNAIDS 2008.
where access to or uptake of counseling and testing is poor, people will be diagnosed as infected because of symptoms, and there will be a similar pattern of death. Using CD4 cell counts to determine the initiation of treatment is a simple and relatively inexpensive way of greatly improving the impact of treatment programs on years of life saved. However, questions arise of how frequently patients who are HIV-infected with a CD4 count above the initiation threshold should be monitored and how many tests should be done to confirm the accuracy of the CD4 measurement. As treatment coverage expanded, the initial goal was to find those in need of treatment and initiate them. However, as time progresses, those who did not initially need treatment will progress and become eligible. Either one can wait until people show symptoms, going back to the equivalent of syndromic initiation, or one can monitor their CD4 count over time.
Syndromic Management
A different set of questions arises about the management of those who have already been initiated on treatment. How frequently should they be seen? Should their CD4 count be monitored? What tests for toxicity of drugs are required?
And should measurements of viral load be made to identify successful viral suppression and failure? One approach is to manage syndromically; in a randomized controlled trial, such management was found to perform nearly as well as management with laboratory tests (Mugyenyi et al., 2010). However, failing to measure viral loads means that patients will have potentially high loads while they are exposed to antiretroviral drugs, providing selective pressure for the evolution of drug resistance (Gupta et al., 2009). The spread of resistant HIV would undermine the potential for successful treatment programs in the future (Blower et al., 2003).
Health Care Organization and Demands of Health Care Workers
The organization of health care can have an impact on the quality and accessibility of services, as well as their costs and feasibility. Who is legally and practically able to prescribe and administer antiretroviral drugs will depend upon the organization of health care in a country and the legal constraints on health care workers. A cascade model of care whereby patients with greater complications can be referred to secondary and tertiary services as required appears to be ideal, with less specialized health care workers maintaining patients on ART. Despite the potential for using workers with a range of skills, however, the demand for staff will be enormous if treatment is expanded to meet need. For example, in a
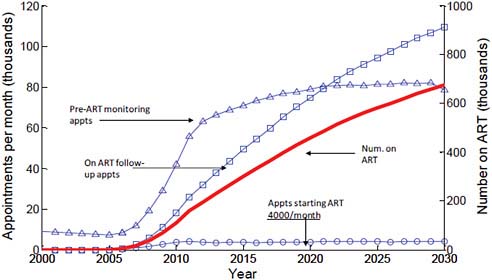
FIGURE A-5 Future treatment need and health system capacity in Zimbabwe.
NOTE: Universal access = 110,000 on treatment in 2010, 670,000 in 2030.
SOURCE: Hallett et al., in press.
modeling exercise focused on Zimbabwe, the number of appointments for monitoring patients over time can be predicted, revealing a large number of visits per health care worker per year. Chapter 5 examines the concept of task sharing, in which available personnel could redistribute the visits among a larger number of providers. Figure A-5 illustrates future treatment need and health system capacity in Zimbabwe.
REFERENCES
Aalen, O. O., V. T. Farewell, D. De Angelis, N. E. Day, and O. N. Gill. 1997. A Markov model for HIV disease progression including the effect of HIV diagnosis and treatment: Application to AIDS prediction in England and Wales. Statistics in Medicine 16(19):2191-2210.
Abbas, U. L., R. M. Anderson, and J. W. Mellors. 2006. Potential impact of antiretroviral therapy on HIV-1 transmission and AIDS mortality in resource-limited settings. Journal of Acquired Immune Deficiency Syndromes 41(5):632-641.
aids2031 Consortium. 2010. aids2031: About. http://www.aids2031.org/about (accessed October 21, 2010).
———. 2011. AIDS: Taking a long-term view. Upper Saddle River, NJ: Financial Times Science Press.
aids2031 Costs and Financing Working Group. 2010. Costs and choices: Financing the long-term fight against AIDS. Washington, DC: Results for Development Institute.
Baggaley, R. F., G. P. Garnett, and N. M. Ferguson. 2006. Modelling the impact of antiretroviral use in resource-poor settings. PLoS Medicine 3(4):e124.
Blower, S., E. Bodine, J. Kahn, and W. McFarland. 2005. The antiretroviral rollout and drug-resistant HIV in Africa: Insights from empirical data and theoretical models. AIDS 19(1):1-14.
Blower, S. M., A. N. Aschenbach, and J. O. Kahn. 2003. Predicting the transmission of drug-resistant HIV: Comparing theory with data. The Lancet Infectious Diseases 3(1):10-11.
Bollinger, L. A. 2008. How can we calculate the “E” in “CEA”? AIDS 22(Suppl. 1):S51-S57.
Bollinger, L., and J. Stover. 2010. Estimating global resource needs for HIV/AIDS 2007-2031. http://www.iom.edu/~/media/Files/Activity%20Files/Global/LongTermAIDS/2010-FEB-03/Bollinger.pdf (accessed October 21, 2010).
Dodd, P. J., G. P. Garnett, and T. B. Hallett. 2010. Examining the promise of HIV elimination by “test and treat” in hyperendemic settings. AIDS 24(5):729-735.
Fenton, K. A., A. M. Johnson, S. McManus, and B. Erens. 2001. Measuring sexual behaviour: Methodological challenges in survey research. Sexually Transmitted Infections 77(2):84-92.
Garnett, G. P., J. M. Garcia-Calleja, T. Rehle, and S. Gregson. 2006. Behavioural data as an adjunct to HIV surveillance data. Sexually Transmitted Infections 82(Suppl. 1):i57-i62.
Granich, R. M., C. F. Gilks, C. Dye, K. M. De Cock, and B. G. Williams. 2009a. Universal voluntary HIV testing with immediate antiretroviral therapy as a strategy for elimination of HIV transmission: A mathematical model. The Lancet 373(9657):48-57.
———. 2009b. Universal voluntary HIV testing with immediate antiretroviral therapy as a strategy for elimination of HIV transmission: A mathematical model. The Lancet 373(9657):48-57.
Gupta, G. R., J. O. Parkhurst, J. A. Ogden, P. Aggleton, and A. Mahal. 2008. Structural approaches to HIV prevention. The Lancet 372(9640):764-775.
Gupta, R. K., A. Hill, A. W. Sawyer, A. Cozzi-Lepri, V. von Wyl, S. Yerly, V. D. Lima, H. F. Gunthard, C. Gilks, and D. Pillay. 2009. Virological monitoring and resistance to first-line highly active antiretroviral therapy in adults infected with HIV-1 treated under WHO guidelines: A systematic review and meta-analysis. The Lancet Infectious Diseases 9(7):409-417.
Hallett, T. B., S. Gregson, S. Dube, and G. P. Garnett. 2008. The impact of monitoring HIV patients prior to treatment in resource-poor settings: Insights from mathematical modelling. PLoS Medicine 5(3):e53.
Hallett, T. B., S. Dube, I. Cremin, B. Lopman, A. Mahomva, G. Ncube, O. Mugurungi, S. Gregson, and G. P. Garnett. 2009a. The role of testing and counseling for HIV prevention and care in the era of scaling-up antiretroviral therapy. Epidemics 1:77-82.
Hallett, T. B., S. Gregson, E. Gonese, O. Mugurungi, and G. P. Garnett. 2009b. Assessing evidence for behaviour change affecting the course of HIV epidemics: A new mathematical modelling approach and application to data from Zimbabwe. Epidemics 1:108-117.
Hallett, T. B., S. Gregson, S. Dube, E. S. Mapfeka, O. Mugurungi, and G. P. Garnett. In press. Estimating the resources required in the roll-out of universal access to antiretroviral therapy in Zimbabwe. Sexually Transmitted Infections.
Hecht, R., L. Bollinger, J. Stover, W. McGreevey, F. Muhib, C. E. Madavo, and D. de Ferranti. 2009. Critical choices in financing the response to the global HIV/AIDS pandemic. Health Affairs 28(6):1591-1605.
Hecht, R., J. Stover, L. Bollinger, F. Muhib, K. Case, and D. de Ferranti. 2010. Financing of HIV/AIDS programme scale up in low-income and middle-income countries, 2009-31. The Lancet 376(9748):1254-1260.
Johnson, L. 2010. HIV/AIDS projections for South Africa. Presentation to the IOM committee on envisioning a strategy to prepare for the long-term burden of HIV/AIDS: African needs and U.S. interests, Washington, DC, February 3, 2010.
Johnson, L. F., L. Alkema, and R. E. Dorrington. 2010. A Bayesian approach to uncertainty analysis of sexually transmitted infection models. Sexually Transmitted Infections 86(3):169-174.
Kimmel, A. D., S. J. Goldie, R. P. Walensky, E. Losina, M. C. Weinstein, A. D. Paltiel, H. Zhang, and K. A. Freedberg. 2005. Optimal frequency of CD4 cell count and HIV RNA monitoring prior to initiation of antiretroviral therapy in HIV-infected patients. Antiviral Therapy 10(1):41-52.
Levine, R. 2008. Healthy foreign policy: Bringing coherence to the global health agenda. In The White House and the world: A global development agenda for the next U.S. president, edited by N. Birdsall. Washington, DC: Center for Global Development.
Marseille, E., L. Dandona, N. Marshall, P. Gaist, S. Bautista-Arredondo, B. Rollins, S. Bertozzi, J. Coovadia, J. Saba, D. Lioznov, J.-A. Du Plessis, E. Krupitsky, N. Stanley, M. Over, A. Peryshkina, S. G. P. Kumar, S. Muyingo, C. Pitter, M. Lundberg, and J. Kahn. 2007. HIV prevention costs and program scale: Data from the PANCEA project in five low and middle-income countries. BMC Health Services Research 7(1):108.
Mngadi, S., N. Fraser, H. Mkhatshwa, T. Lapidos, T. Khumalo, S. Tsela, N. Nhlabatsi, and H. Odido. 2009. Swaziland HIV prevention response and modes of transmission analysis. Mbabane: National Emergency Response Council on HIV/AIDS, UNAIDS.
Mugyenyi, P., A. S. Walker, J. Hakim, P. Munderi, D. M. Gibb, C. Kityo, A. Reid, H. Grosskurth, J. H. Darbyshire, F. Ssali, D. Bray, E. Katabira, A. G. Babiker, C. F. Gilks, G. Kabuye, D. Nsibambi, R. Kasirye, E. Zalwango, M. Nakazibwe, B. Kikaire, G. Nassuna, R. Massa, K. Fadhiru, M. Namyalo, A. Zalwango, L. Generous, P. Khauka, N. Rutikarayo, W. Nakahima, A. Mugisha, J. Todd, J. Levin, S. Muyingo, A. Ruberantwari, P. Kaleebu, D. Yirrell, N. Ndembi, F. Lyagoba, P. Hughes, M. Aber, A. M. Lara, S. Foster, J. Amurwon, B. N. Wakholi, J. Whitworth, K. Wangati, B. Amuron, D. Kajungu, J. Nakiyingi, W. Omony, D. Tumukunde, T. Otim, J. Kabanda, H. Musana, J. Akao, H. Kyomugisha, A. Byamukama, J. Sabiiti, J. Komugyena, P. Wavamunno, S. Mukiibi, A. Drasiku, R. Byaruhanga, O. Labeja, P. Katundu, S. Tugume, P. Awio, A. Namazzi, G. T. Bakeinyaga, H. Katabira, D. Abaine, J. Tukamushaba, W. Anywar, W. Ojiambo, E. Angweng, S. Murungi, W. Haguma, S. Atwiine, J. Kigozi, L. Namale, A. Mukose, G. Mulindwa, D. Atwiine, A. Muhwezi, E. Nimwesiga, G. Barungi, J. Takubwa, D. Mwebesa, G. Kagina, M. Mulindwa, F. Ahimbisibwe, P. Mwesigwa, S. Akuma, C. Zawedde, D. Nyiraguhirwa, C. Tumusiime, L. Bagaya, W. Namara, J. Karungi, R. Kankunda, R. Enzama,
A. Latif, V. Robertson, E. Chidziva, R. Bulaya-Tembo, G. Musoro, F. Taziwa, C. Chimbetete, L. Chakonza, A. Mawora, C. Muvirimi, G. Tinago, P. Svovanapasis, M. Simango, O. Chirema, J. Machingura, S. Mutsai, M. Phiri, T. Bafana, M. Chirara, L. Muchabaiwa, M. Muzambi, J. Mutowo, T. Chivhunga, E. Chigwedere, M. Pascoe, C. Warambwa, E. Zengeza, F. Mapinge, S. Makota, A. Jamu, N. Ngorima, H. Chirairo, S. Chitsungo, J. Chimanzi, C. Maweni, R. Warara, M. Matongo, S. Mudzingwa, M. Jangano, K. Moyo, L. Vere, N. Mdege, I. Machingura, A. Ronald, A. Kambungu, F. Lutwama, I. Mambule, A. Nanfuka, J. Walusimbi, E. Nabankema, R. Nalumenya, T. Namuli, R. Kulume, I. Namata, L. Nyachwo, A. Florence, A. Kusiima, E. Lubwama, R. Nairuba, F. Oketta, E. Buluma, R. Waita, H. Ojiambo, F. Sadik, J. Wanyama, P. Nabongo, J. Oyugi, F. Sematala, A. Muganzi, C. Twijukye, H. Byakwaga, R. Ochai, D. Muhweezi, A. Coutinho, B. Etukoit, C. Gilks, K. Boocock, C. Puddephatt, C. Grundy, J. Bohannon, D. Winogron, A. Burke, A. Babiker, H. Wilkes, M. Rauchenberger, S. Sheehan, C. Spencer-Drake, K. Taylor, M. Spyer, A. Ferrier, B. Naidoo, D. Dunn, R. Goodall, L. Peto, R. Nanfuka, C. Mufuka-Kapuya, D. Pillay, A. McCormick, I. Weller, S. Bahendeka, M. Bassett, A. C. Wapakhabulo, B. Gazzard, C. Mapuchere, O. Mugurungi, C. Burke, S. Jones, C. Newland, G. Pearce, S. Rahim, J. Rooney, M. Smith, W. Snowden, J. M. Steens, A. Breckenridge, A. McLaren, C. Hill, J. Matenga, A. Pozniak, D. Serwadda, T. Peto, A. Palfreeman, and M. Borok. 2010. Routine versus clinically driven laboratory monitoring of HIV antiretroviral therapy in Africa (DART): A randomised non-inferiority trial. The Lancet 375(9709):123-131.
OGAC (Office of the Global AIDS Coordinator). 2010a. Annual reports to Congress. http://www.pepfar.gov/progress/index.htm (accessed June 3, 2010).
———. 2010b. Operational plans. http://www.pepfar.gov/about/c19388.htm (accessed June 3, 2010).
———. 2010c. Report to Congress on costs of treatment in the President’s Emergency Plan for AIDS Relief (PEPFAR). Washington, DC: PEPFAR.
Over, M. 2009. AIDSCost computer program: Projecting future budgetary costs of AIDS treatment. Center for Global Development. www.cgdev.org/files/1422227_file_AIDSCost_manual_FINAL.pdf (accessed October 29, 2010).
Over, M., and P. Piot. 1996. Human immunodeficiency virus infection and other sexually transmitted diseases in developing countries: Public health importance and priorities for resource allocation. Journal of Infectious Diseases 174(Suppl. 2):S162-S175.
Over, M., P. Heywood, J. Gold, I. Gupta, S. Hira, and E. Marseille. 2004. HIV/AIDS treatment and prevention in India: Modeling the costs and consequences (health, nutrition, and population). Washington, DC: The World Bank.
Phillips, A. N., D. Pillay, A. H. Miners, D. E. Bennett, C. F. Gilks, and J. D. Lundgren. 2008. Outcomes from monitoring of patients on antiretroviral therapy in resource-limited settings with viral load, CD4 cell count, or clinical observation alone: A computer simulation model. The Lancet 371(9622):1443-1451.
Phillips, A. N., C. Gilks, and J. D. Lundgren. 2009. Cost-effectiveness of strategies for monitoring the response to antiretroviral therapy in resource-limited settings. Archives of Internal Medicine 169(9):904.
Slaymaker, E. 2004. A critique of international indicators of sexual risk behaviour. Sexually Transmitted Infections 80(Suppl. 2):ii13-ii21.
Stover, J., L. Bollinger, K. Cooper-Arnold, and The Futures Group International. 2003. Goals model for estimating the effects of resource allocation decisions on the achievement of the goals of the HIV/AIDS Strategic Plan. http://www.futuresinstitute.org/download/goals/Goals.pdf (accessed October 29, 2010).
Sweat, M., S. Gregorich, G. Sangiwa, C. Furlonge, D. Balmer, C. Kamenga, O. Grinstead, and T. Coates. 2000. Cost-effectiveness of voluntary HIV-1 counselling and testing in reducing sexual transmission of HIV-1 in Kenya and Tanzania. The Lancet 356(9224):113-121.
Todd, J., J. R. Glynn, M. Marston, T. Lutalo, S. Biraro, W. Mwita, V. Suriyanon, R. Rangsin, K. E. Nelson, P. Sonnenberg, D. Fitzgerald, E. Karita, and B. Zaba. 2007. Time from HIV seroconversion to death: A collaborative analysis of eight studies in six low and middle-income countries before highly active antiretroviral therapy. AIDS 21(Suppl. 6):S55-S63.
UNAIDS (The Joint United Nations Programme on HIV/AIDS). 2008. Report on the global AIDS epidemic. Geneva: UNAIDS.
———. 2010. Fact sheet: Treatment 2.0. Geneva: UNAIDS.
UNAIDS and WHO. 2009. AIDS epidemic update: December 2009. Geneva: UNAIDS.
UNAIDS/WHO/SACEMA Expert Group on Modelling the Impact and Cost of Male Circumcision for HIV Prevention. 2009. Male circumcision for HIV prevention in high HIV prevalence settings: What can mathematical modelling contribute to informed decision making? PLoS Medicine 6(9):e1000109.
Van Der Paal, L., L. A. Shafer, J. Todd, B. N. Mayanja, J. A. Whitworth, and H. Grosskurth. 2007. HIV-1 disease progression and mortality before the introduction of highly active antiretroviral therapy in rural Uganda. AIDS 21(Suppl. 6):S21-S29.
Velasco-Hernandez, J. X., H. B. Gershengorn, and S. M. Blower. 2002. Could widespread use of combination antiretroviral therapy eradicate HIV epidemics? The Lancet Infectious Diseases 2(8):487-493.
Venkatesh, K. K., G. de Bruyn, M. N. Lurie, L. Mohapi, P. Pronyk, M. Moshabela, E. Marinda, G. E. Gray, E. W. Triche, and N. A. Martinson. 2010. Decreased sexual risk behavior in the era of HAART among HIV-infected urban and rural South Africans attending primary care clinics. Journal of Acquired Immune Deficiency Syndromes 24(17):2687-96.
Wabwire-Mangen, F., M. Odiit, W. Kirungi, D. K. Kisitu, and J. O. Wanyama. 2009. Uganda HIV modes of transmission and prevention response analysis. Kampala: Uganda National AIDS Commission, UNAIDS.
Wagner, B., and S. Blower. 2009. Voluntary universal testing and treatment is unlikely to lead to HIV elimination: A modeling analysis. Nature Precedings. http://precedings.nature.com/documents/3917/version/1/files/npre20093917-1.pdf (accessed November 1, 2010).
White, R. G., K. K. Orroth, J. R. Glynn, E. E. Freeman, R. Bakker, J. D. Habbema, F. Terris-Prestholt, L. Kumaranayake, A. Buvé, and R. J. Hayes. 2008. Treating curable sexually transmitted infections to prevent HIV in Africa: Still an effective control strategy? Journal of Acquired Immune Deficiency Syndromes 47(3):346-353.
WHO (World Health Organization). 2010. Towards universal access: Scaling up priority HIV/AIDS interventions in the health sector. Progress report 2010. Geneva: WHO.
Williams, B. G., J. O. Lloyd-Smith, E. Gouws, C. Hankins, W. M. Getz, J. Hargrove, I. de Zoysa, C. Dye, and B. Auvert. 2006. The potential impact of male circumcision on HIV in sub-Saharan Africa. PLoS Medicine 3(7):e262.
ANNEX A-1
MODEL OF THE RELATIONSHIP AMONG TREATMENT INITIATION, COVERAGE, AND MORTALITY
A feature of a model with a user-selected target coverage rate is a pattern of rapid enrollment before the target rate is reached, followed by slow enrollment because the target has been reached. In such models, enrollment is largely unrelated to unmet need for treatment. An alternative approach is to specify the proportion of unmet need that the program absorbs every year, or the uptake rate, and allow this parameter to determine the growth of the coverage rate. The first approach—used, for example, in the aids2031 model —allows enrollment to rapidly attain some target coverage rate regardless of the real absorptive capacity on the ground. The second approach—used, for example, in standard economic models of investment—is sometimes referred to as the “partial adjustment” model and captures the reality that programs typically cannot adjust by more than a percentage of unmet need in any given year. Under the first approach, a country is assumed to care about enrollment only until its coverage target is reached, whereas in the second, it is assumed to offer an equal chance at enrollment to those with unmet need in perpetuity. While real policy could follow either pattern, the partial adjustment approach better captures the policy reality of insufficient resources to enroll everyone who needs treatment by any given definition.
Assuming a constant rate of people becoming newly in need of treatment, b, and the death rate of those in need, α, and those on treatment, μ, we can calculate the relationship between the fraction of those newly in need starting treatment immediately, ![]() ; the rate of those in need starting treatment, γ; and the patterns of treatment coverage and mortality. Where X is the number in need but not yet on treatment and Y is the number on treatment, the rate of change of these numbers is given by the ordinary differential equations
; the rate of those in need starting treatment, γ; and the patterns of treatment coverage and mortality. Where X is the number in need but not yet on treatment and Y is the number on treatment, the rate of change of these numbers is given by the ordinary differential equations
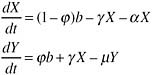
These equations have the equilibrium solution:
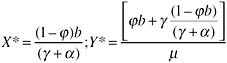
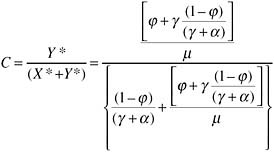
If the coverage of treatment is defined as the proportion in need, coverage at equilibrium is:
The number of deaths is simply αX + μY.
For given rates of HIV incidence and treatment initiation, an HIV epidemic will approach a long-run equilibrium coverage rate, which for high incidence and low treatment initiation will be substantially less than universal coverage.
ANNEX A-2
WHAT DRIVES THE DIFFERENCES AMONG PROJECTIONS
Various models include different levels of detail. The models of Blower assume a homogeneous population with respect to risk behavior, which allows a wider spread of HIV with a lower reproductive number than would be the case with a model including heterogeneity in risk (Blower et al., 2003, 2005). The model of Granich is also homogeneous with respect to risk, but it assumes that the incidence of infection is inversely proportional to prevalence, which allows prevalence to saturate and stabilize despite a higher basic reproductive number (Granich et al., 2009b). The models of Baggaley, Phillips, and Johnson include distributions of risk across the population (Baggaley et al., 2006; Johnson et al., 2010; Phillips et al., 2008). This heterogeneity in risk is observed in reported behavioral data, but the fine details are less certain. These models of Blower, Granich, Baggaley, Phillips, and Johnson have been used to explore a number of HIV/AIDS prevention interventions, including the impact of treatments for sexually transmitted infections (STIs) (Over and Piot, 1996; White et al., 2008), of adult male circumcision (UNAIDS/WHO/SACEMA Expert Group on Modelling the Impact and Cost of Male Circumcision for HIV Prevention, 2009; Williams et al., 2006), of counseling and testing (Hallett et al., 2009a), and of treatment (Abbas et al., 2006; Baggaley et al., 2006; Dodd et al., 2010; Velasco-Hernandez et al., 2002). The effect size of interventions will depend greatly on how resilient the spread of the virus is, which in turn depends on the importance assumed in the model for a high-risk group driving the epidemic. In addition, the frequency of unprotected sex and the pattern of mixing specific to different activity groups have an impact on model results. Other differences in models result from different definitions of coverage and the long-term impact of treatment. For example, the Goals model used by aids2031 (Bollinger and Stover, 2010; Hecht et al., 2010; Stover et al., 2003) and the AIDSCost model used by the committee (Over, 2009) both assume that after the first year, treatment failure occurs at a constant rate. This is a more pessimistic assumption than would be expected from the impact of treatment in developing countries.
Models with detailed patterns of risk behavior require many input parameters that can be estimated from local studies of behavior or from a fit between the observed epidemiology of HIV and the model outputs. Unfortunately, models that include a description of the range of behavioral interactions within populations will be overspecified, with multiple parameter sets being able to generate the observed epidemics. To an extent, this issue is being addressed in the development of models of HIV transmission (Hallett et al., 2009b; Johnson et al., 2010) in which multiple datasets are being combined for analysis. However, the detailed work required for predictions in one location does not allow for a complete
picture of the HIV/AIDS epidemic in Africa. In exercises across a wide range of countries, simpler methods have been applied, either using one “reasonable” parameter set (Hecht et al., 2010) or using estimated incidence at a recent time point as an input (Levine, 2008). The approach adopted by the committee in its model is to assume that interventions could achieve progressive reductions in incidence if sufficient incentives were provided (Over, 2009). This assumption allows the benefits of reduced incidence to be calculated without defining exactly how it is to be achieved.
To calculate the fraction of those infected needing treatment, a rate of progression to treatment need is applied. Of those needing treatment, a fraction receive it (this is the rate of treatment coverage per year). After 1 year, a fraction fail treatment and either die or are placed on second-line treatment, for which there is a further rate of treatment failure that is assumed to lead to death.
Estimating the future incidence of HIV based on incidence in a baseline year provides a basis for calculations that take account of future changes in coverage of treatment and prevention interventions. However, to represent the influence of a “core group” that has a high rate of HIV acquisition and transmission and therefore drives the epidemiology of infection, it is necessary to limit the fraction of the population susceptible to infection, k. A baseline incidence rate, IR0, is assigned to the year 2007—IR0= IR2007—which is an input variable that can be derived from the absolute incidence, AI0, in each country.
Since the absolute incidence is a function of the incidence rate and the fraction of the population to which it is applied, three different approaches are possible: to hold the incidence rate constant across countries, to hold a fraction of the population at risk constant across countries, or to vary both across countries. To reflect the very different prevalences across Africa, which presumably reflect the fraction of the population engaging in risk behaviors, country-specific fractions at risk, k, are estimated from a constant incidence rate:
where H0 is the number HIV-infected, and ![]() is the total population aged 15–64. IR0 is an external variable. A high value—for example, 0.2—would generate a low value of k relative to the prevalence in year zero and make HIV difficult to control compared with a high value of IR0—for example, 0.01—which would generate a high value of k.
is the total population aged 15–64. IR0 is an external variable. A high value—for example, 0.2—would generate a low value of k relative to the prevalence in year zero and make HIV difficult to control compared with a high value of IR0—for example, 0.01—which would generate a high value of k.
For future years, the above equation is rearranged to calculate the absolute number of new infections, which is a function of the incidence rate:
The prevalence at time t, Prevt , is the number infected, Ht, divided by the population size, ![]() . The proportion of those HIV-infected who are on treatment, θt, is assumed to influence the fraction of the population at risk through disinhibition, which is determined by the parameter Δ. If this value is negative, it is assumed that treatment acts to reduce the fraction of the population engaging in risk behaviors. The incidence rate is a function of changes in the prevalence of infection (i.e., the pool of infectious contacts within the population); prevention interventions that reduce susceptibility to infection and risk behaviors; and the proportion of those infectious with a reduced viraemia due to treatment relative to the baseline proportion treated:
. The proportion of those HIV-infected who are on treatment, θt, is assumed to influence the fraction of the population at risk through disinhibition, which is determined by the parameter Δ. If this value is negative, it is assumed that treatment acts to reduce the fraction of the population engaging in risk behaviors. The incidence rate is a function of changes in the prevalence of infection (i.e., the pool of infectious contacts within the population); prevention interventions that reduce susceptibility to infection and risk behaviors; and the proportion of those infectious with a reduced viraemia due to treatment relative to the baseline proportion treated:
where the impact of treatment on infection, τt, is given by τt= (1 – f .g.θt), where θt is the proportion of those infected treated, f is the fraction of infection that occurs after the initial primary viraemia, and g is the proportional reduction in transmissibility of those on treatment. The fraction of adult men circumcised is given by c, m is the proportion of adults that are men, and ε is the reduced susceptibility associated with circumcision. The parameter ωt represents prevention effort. Both the proportion circumcised and effort are externally input functions of time with a linear increase to a maximum value.
























