Summary
The field of corrosion science and engineering is on the threshold of important advances. To better comprehend corrosion and its effects, researchers have embraced emerging advances across many fields of science and technology to gain understanding of the mechanistic effects of these interactions on materials behavior and to relate this understanding to the underlying structure, composition, and dynamics. Accelerated progress in corrosion research is anticipated as a result of the stunning ongoing increase in the ability to tailor composition and structure from the nanoscale to the mesoscale; to experimentally probe materials at finer levels of spatial resolution as well as the dynamics of chemical reactions; and to model computationally intensive problems that unravel the nature of these reactions and the response of materials to the environment. Corrosion science today is presented with unprecedented opportunities to advance fundamental understanding of environmental reactions and effects from the atomic and molecular levels to the macro level. Advances in lifetime prediction and technological solutions, as enabled by the convergence of experimental and computational length- and timescales and powerful new modeling techniques, are allowing the development of rigorous, mechanistically based models from observations and physical laws. Advanced techniques for the manipulation of information and for analysis are critical to furthering our state of knowledge in corrosion science and engineering. Important in this regard are the development and handling of large data sets to extract knowledge and trends, to visualize emergent phenomena, and to comprehensively test predictions from models.
Materials design for corrosion resistance currently relies on expert knowledge and incremental improvements to well-tested compositions and structures. The use of increasingly sophisticated computational approaches to predict stability and properties and to probe reaction dynamics in complex environments can make materials design more effective and narrow the span of experimental investigation. Furthermore, progress in nanoscience, particularly the ability to synthesize and control precise nanostructures, creates new opportunities for corrosion scientists and engineers to explore the design of materials (including coatings and smart materials) and to establish the critical link between atomic- and molecular-level processes and macroscopic behaviors. Despite considerable progress in the integration of materials by design into engineering development of products, corrosion-related considerations are typically missing from such constructs. Similarly, condition monitoring and prediction of remaining service life (prognosis) do not at present incorporate corrosion factors. Great opportunities exist to use the framework of these materials design and engineering tools to stimulate corrosion research and development to achieve quantitative life prediction, to incorporate state-of-the-art sensing approaches into experimentation and materials architectures, and to introduce environmental degradation factors into these capabilities.
The Committee on Research Opportunities in Corrosion Science and Engineering defined corrosion as the environmentally induced degradation of materials, where “environment” is broadly construed but always includes some element of chemical reaction. There are many corrosion processes that operate over different temperature regimes, environmental conditions, and mechanical stress levels. The public perception of corrosion is generally limited to the degradation of metallic materials in aqueous environments, perhaps with some recognition that gases and condensed phases at high temperatures may also shorten the useful life of engineering materials. However, every material class is affected by corrosion in some way. Although the problem of corrosion is ubiquitous, research to reduce its magnitude has often received only modest attention.
Dramatic changes in societal factors now demand that prevention and mitigation of corrosion damage receive greater emphasis. Our quality of life is increasingly dependent on the application of diverse materials, including metals, polymers, ceramics, and semiconductor devices. This continuing trend is made more significant by the advanced requirements and designs that push past current experience and expose materials to ever-harsher chemical environments. Finally, and perhaps most importantly, increased awareness of the human impact on Earth’s environment is raising the public’s expectation not only for improved safety and high reliability, but also for green manufacturing and low environmental impact, including sustainability in consumer products, industrial and military equipment, and the infrastructure. When corrosion processes and their products act in direct opposition to these very desirable attributes, their effects must be mitigated. How-
ever, while corrosion generally has a societal cost, it also can be beneficial in that naturally degrading materials can provide a positive green effect once the useful lifetime of a device is reached. Environmentally degradable materials will reduce long-term waste storage issues, and the use of corrosion mechanisms may provide a more rapid path to a full life cycle for many products.
The most effective routes to corrosion mitigation rely on knowledge of the underlying mechanisms causing the corrosion. This alone would justify increased attention to the science base of corrosion research; however, understanding corrosion processes would also be beneficial for improving critical technologies such as batteries and fuel cells, semiconductors, and biodegradable materials.
There are many forms of corrosion, and while some are well understood at the macro level, complex interactions among the different forms are yet to be fully clarified. Further, at the detail level there is often relatively poor understanding of corrosion mechanisms, which makes it technically difficult to devise cost-effective engineering solutions to predict, avoid, and mitigate corrosion damage. These difficult problems have often been put on hold in favor of short-term, empirical fixes, but now appears to be an opportune time to readdress complex questions with new techniques. Advances in characterization (using, among other techniques, transmission1 and scanning2 electron microscopy, micro- and nanometer electrochemical probe methods, synchrotron beam lines and lasers, x-ray, and neutron spectroscopy and combinations of these methods for simultaneous information gathering) and computation and modeling (first principle, molecular dynamics, multiscale modeling, and informatics) have dramatically broadened the array of tools available. Furthermore, engineering practice has evolved, bringing organization to the science of new materials development—integrated computational materials science and engineering (ICMSE)—and order and predictability to the process of life-cycle management (prognosis3).
Lack of fundamental knowledge about corrosion and its application to practice is directly reflected in the high societal cost of corrosion. Although estimates of its aggregate cost vary widely, some studies suggest that corrosion consumes at least 2 to 4 percent of the U.S. gross national product. These estimates do not take into account the ancillary costs to the economy. Ancillary costs include loss of productivity due to deteriorated infrastructure, loss of full operational capability for industrial and military equipment and facilities, and added risk to public safety and
welfare.4 Tackling corrosion cost drivers represents a significant opportunity for the science and engineering community to make a valuable contribution to society.
Sometimes corrosion prediction and prevention do not receive the attention they require during engineering design of structures and systems. This might be because designers anticipate that corrosion problems arising in service can be readily mitigated over time with proper maintenance and repair to achieve desired longevity or because the in-service environment is unknown. The committee envisions a new paradigm in which this traditional reactive approach is transformed into a proactive engineering strategy with (1) appropriate tools used to accurately predict the onset and rate of deterioration of materials in actual or anticipated operating environments, (2) new materials and coatings expressly developed to provide superior corrosion resistance, and (3) design tools used to identify the best, most cost-effective options and to guide the selection of materials and corrosion-mitigation techniques. To support this vision corrosion research should focus on addressing significant gaps in corrosion knowledge so that the results can be applied directly to sectors that are significantly affected by corrosion damage. This means that corrosion science, which seeks to expand the frontiers of basic knowledge, should have its priorities guided by corrosion engineering needs.
Corrosion impacts our everyday life, affecting the health and welfare of the nation and leading to added energy and expense to combat it. Fortunately, a multitude of research and development (R&D) opportunities could lead to making substantial headway in reducing its impact. The committee believes that government-wide as well as society- and industry-wide recognition of the scope of the corrosion problem and a well-defined, coordinated, and reliably resourced program would have a high payoff for the nation. A robust R&D program should have a balanced portfolio of projects providing incremental advancements that result in high return on investment (ROI), as well as high-risk novel projects that could lead to transformative improvement, albeit with a longer time horizon and less attractive ROI in the short term. Because the committee chose to focus on general as well as federal government corrosion issues, it could not also include a separate strategy for each federal entity. The goal was to identify general research needs and organize these into a framework that could be used to facilitate a specific federal response.
This report identifies grand challenges for the corrosion research community, highlights research opportunities in corrosion science and engineering, and posits a national strategy for corrosion research. It is a logical and necessary complement to the recently published National Research Council report Assessment of Corrosion
Education,5 which emphasized that technical education must be supported by academic, industrial, and government research, as well as technology dissemination to accelerate the implementation of new and emerging technical advances. Although the present report focuses on the government role, this emphasis does not diminish the role of industry or academia.
Grand challenges in corrosion research might be identified from several perspectives. The committee chose to first explore technical challenges that pervade key national priorities such as energy, the environment, health, infrastructure/safety, and national security. These priorities account for an overwhelming portion of discretionary spending in the federal budget and are aligned with federal agencies with defined and related missions. Once identified, these technical challenges were organized by the committee into broad overriding themes, which the committee called the four corrosion grand challenges (CGCs):
CGC I: Development of cost-effective, environment-friendly, corrosion-resistant materials and coatings;
CGC II: High-fidelity modeling for the prediction of corrosion degradation in actual service environments;
CGC III: Accelerated corrosion testing under controlled laboratory conditions that quantitatively correlates with the long-term behavior observed in service environments; and
CGC IV: Accurate forecasting of remaining service time until major repair, replacement, or overhaul becomes necessary—i.e., corrosion prognosis.
The CGCs have been expressed as engineering and technology challenges, and these are deemed to be the drivers and guiding principles of the framework for prioritizing efforts. The committee believes that addressing these challenges will demand an integrated body of scientific and engineering research targeted at specific needs but coordinated to minimize duplication and take advantage of synergism. Figure S.1 is a hierarchical representation of the four CGCs, supported by a body of corrosion science and engineering research.
Between the CGCs and the underlying corrosion science in Figure S.1 is a layer depicting the need for dissemination of corrosion knowledge and research results to those facing the real problems of addressing the CGCs and the related corrosion issues. As in many other fields, there is a need for enhanced communication between researchers and engineers to make the existing knowledge as accessible as possible.
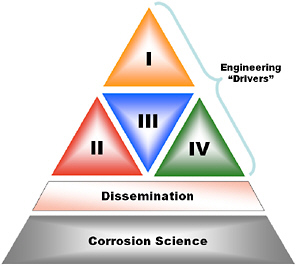
FIGURE S.1 Hierarchy of the four corrosion grand challenges identified by the committee.
The committee recognizes that each federal agency and department must establish its own priorities for addressing the challenges, depending on the relevance of the corrosion damage to their individual missions. The diverse responsibilities of the agencies might include funding such research or performing it as well as taking advantage of what is already known about mitigating corrosion to reduce the costs of such corrosion. Accordingly, the committee recommends that each agency and department use the underlying research opportunities as a framework for prioritization, each according to the benefits expected for its mission and allocating its resources appropriately. To facilitate the rapid dissemination of results, the committee encourages the involvement of industry in the planning and execution of the research and technology development.
Recommendation: Using as guidance the four corrosion grand challenges developed by the committee, each federal agency or department should identify the areas of corrosion research pertinent to its mission and draw up a road map for fulfilling its related responsibilities. In doing so, each should take a cross-organizational approach to planning and execution and should include input from industrial sectors that have experience in handling corrosion.
There are many research opportunities in the CGCs, each having significant gaps in basic understanding that need to be filled. Some of these gaps are listed below.
CGC I: Development of cost-effective, environment-friendly, corrosion-resistant materials and coatings.
-
Materials with inherently high corrosion resistance that also possess other important characteristics demanded by the applications of interest.
-
Design and modeling of new corrosion-resistant alloy chemistries and structures.
-
Durable, environmentally compatible, and cost-effective protective coatings that eliminate or significantly reduce corrosion.
-
Materials that biodegrade in a predictable and benign manner.
-
Determination of properties and design parameters/rules.
CGC II: High-fidelity modeling for the prediction of corrosion degradation in actual service environments.
-
Ability to predict effects of corrosion and the lifetimes of materials subjected to a wide range of service environments.
-
Computer modeling of material surfaces at the nanoscale, where corrosion initiates and propagates and where corrosion resistance must be imparted.
-
Increasing the fundamental understanding of new corrosion science and its utilization.
CGC III: Accelerated corrosion testing under controlled laboratory conditions that quantitatively correlates to long-term behavior observed in service environments.
-
Smart accelerated corrosion testing that accurately predicts performance under a range of exposures, ensuring durability, and early detection of unforeseen corrosion-related failure mechanisms.
-
Development of an “environmental corrosion intensity factor” that facilitates quantification of the acceleration provided by test conditions and enables prediction of performance, based on exposure time, in any combination of field environments.
-
Hypothesis-driven models to increase fundamental understanding of corrosion science, improve prediction of structural lifetimes, and optimize maintenance programs.
CGC IV: Accurate forecasting of remaining service time until major repair, replacement, or overhaul becomes necessary—i.e., corrosion prognosis.
-
Accurate and robust sensors that track and monitor corrosion damage and protection.
-
Automated defect-sensing devices for quality inspections.
-
Remaining life prediction “reasoners” based on measured corrosion deterioration and knowledge of a material’s capability in the particular environment.
-
Remaining life prediction for materials that have yet not shown any localized degradation.
-
Accurate assessment of alternative corrective actions.
Each of the four challenges is explored in the body of the report. In Chapter 3, the challenges are followed by descriptions of the underlying science and engineering research required to address them. The descriptions reveal that advances across many scientific disciplines will be needed to close current technological gaps, and they clarify the need for a balanced program of traditional single-investigator and multi-investigator efforts that will bring corrosion researchers into collaborative groups that include experts in characterization and computational modeling.
Recommendation: Funding agencies should design programs to stimulate single-investigator and collaborative team efforts and underwrite the costs of realistic test laboratories open to the corrosion community and its collaborators, including industry researchers. These programs should address the four corrosion grand challenges identified by the committee; provide a balance between single- and multi-investigator groups; develop collaborative interactions between corrosion, measurement, and computational experts; and be driven by both science and engineering applications.
A new development model, known as the technology “pull” paradigm, requires the engagement of multiple stakeholders, including researchers, applications engineers, design engineers, and material producers. Their efforts can reach fruition much faster if the technology needs are known in advance, allowing the research to focus on the critical issues. The other stakeholders are those who design the application for the new material and those who produce it in volume. This development model is undertaken by a consortium that engages interested, knowledgeable participants from conception to implementation.
Federal departments such as the DOD and the DOE currently support materials R&D in areas aligned with core mission requirements. Increasingly, these developments involve industry early in the planning process since materials developments are targeted for specific end applications.
Recommendation: Federal agencies should facilitate the formation of consortia of industry, university, and as appropriate, government laboratories chartered to address significant areas of opportunity in corrosion science and engineering. In consonance with best practices, industry should be involved at the earliest practical time in the structuring of these programs so that technology pull can realistically shorten the time between development and reduction
to practice. Also, early involvement by industries will facilitate their active participation as consortium members.
Research is critical but not in itself sufficient to tackle the CGCs. Advances in understanding will have little benefit and negligible ROI if they are not transferred to in-service practice. There are many ways to disseminate research results, but they are typically less than optimal in reaching medium and small firms with few engineers. Several options for enhancing dissemination are discussed in the body of the report. The committee feels strongly that each agency should include a plan for accelerating dissemination of the results achieved in addressing its part of the corrosion road map.
Recommendation: Each agency and department should assume responsibility not only for funding corrosion research but also for disseminating the results of the research.
Actions by the individual agencies are critical to this proposed national strategy, but the variety of materials applications and the broad commonality of the underlying science research suggest that collaboration across agency boundaries will add significant value. The Office of Science and Technology Policy (OSTP) should, accordingly, take the lead in optimizing the government effort. Such an integrated effort would constitute a much-needed national corrosion strategy.
Recommendation: The Office of Science and Technology Policy (OSTP) should acknowledge the adverse impact of corrosion on the nation and launch a multi-agency effort for high-risk, high-reward research to mitigate this impact. OSTP should set up a multiagency committee on the environmental degradation of materials. It should begin by documenting current federal expenditures on corrosion research and mitigation and then encouraging multiagency attention to issues of research, mitigation, and information dissemination. Collaboration among departments and agencies should be strengthened by collaboration with state governments, professional societies, industry consortia, and standards-making bodies.










