5
The Role of Smoking
Smoking is a natural candidate for explaining the divergence in life expectancy among high-income countries for two reasons. First, it is a major cause of mortality. In the United States, according to some estimates, smoking is the number one cause of preventable deaths, killing more than 440,000 Americans each year (American Heart Association, 2009; Centers for Disease Control and Prevention, 2004). Second, the number of smokers and historical smoking patterns vary from country to country, so smoking has the potential to explain differences among countries in mortality and life expectancy trends. This chapter describes the relationship between smoking and mortality, international trends in smoking, and various analyses of the effects of smoking-related mortality on life expectancy in different high-income countries. As will be seen, the evidence implies that smoking may indeed be a major factor explaining the divergent trends in life expectancy among high-income countries over the past several decades.
SMOKING AND MORTALITY
Although lung cancer is the best-known cause of death associated with smoking, a number of other smoking-related diseases increase the mortality rate among smokers, including various other types of cancer, coronary heart disease, chronic obstructive pulmonary disease (COPD), and cerebrovascular disease (Centers for Disease Control and Prevention, 2004). Thus determining the overall smoking-related mortality rate in a population can pose a challenge, as all of these diseases—even lung cancer to a relatively minor degree—appear in nonsmokers as well, making it
difficult to determine the percentage of deaths that can be attributed to smoking.
Prospective Cohort Studies
The most straightforward approach is to follow a large number of people over a number of years and compare the mortality rates of smokers and nonsmokers to determine the additional risk of death caused by smoking. Such prospective cohort studies, as they are called, have the advantage of allowing researchers to collect a great deal of information from participants and then use that information to determine the relationship between various medical and lifestyle factors—smoking history or high blood pressure, for example—and the various causes of death. Nonsmoking factors affecting mortality can be controlled in statistical analyses of the effects of smoking.
The main weakness of such studies in determining the risks of smoking is the difficulty of obtaining a precise measure of smoking behavior. Most prospective cohort studies ask participants about their smoking behavior when they first enter the study and assume that this behavior remains fixed throughout the course of the study (Preston et al., 2010b). In reality, people vary their smoking behavior over time, with some quitting and others starting or restarting and still others increasing or decreasing how much they smoke. Furthermore, 20 to 30 years can elapse between the time a person starts smoking and the time serious health effects appear (Lopez et al., 1994). The resulting errors in recording the smoking behavior of study participants will generally weaken whatever connections the study might find between smoking and various causes of death. Thus such studies will typically underestimate the risk of smoking.
The Cancer Prevention Studies (CPS-I and CPS-II) conducted by the American Cancer Society, which cover the years 1959–1965 and 1982–1986, respectively, are two of the largest such studies to date to examine the effects of smoking on mortality, and they have provided some of the most important information available on the subject. In the CPS-II, investigators asked 1.2 million volunteers to fill out questionnaires asking about their jobs, diet, alcohol and tobacco use, medical history, and family history of cancer as of 1982. Since then the study has kept track of deaths among the 1.2 million participants by monitoring the National Death Index (American Cancer Society, 2009). By 2006, 488,000 of the participants had died.
Working from the data accumulated by the CPS-II and comparing the numbers of smoking-related deaths among smokers with the number of deaths among nonsmokers, Mokdad and colleagues (2004) calculated that smoking had been responsible for 435,000 deaths in 2000. A second group used the CPS-II data to calculate how smoking affects life expectancy. Focusing on the difference in life expectancy between smokers who quit smoking
and those who never smoked, the researchers estimated that quitting smoking at age 35 added 6.9 to 8.5 years of life expectancy for men and 6.1 to 7.7 years of life expectancy for women compared with those who kept smoking (Taylor et al., 2002).
The reason why the number of deaths attributable to cigarette smoking has stayed as high as it has despite large reductions in the prevalence of smoking is that heavy smoking in the past has left a clear imprint on current mortality levels. One feature of that imprint is that the relative mortality levels of smokers have risen relative to nonsmokers, reflecting the heavy smoking histories of smokers today. Comparisons of the CPS-I and CPS-II data indicate that the age-adjusted death rate from lung cancer per 100,000 person-years increased among current smokers between the period covering the CPS-I (1959–1965) and that covering the CPS-II (1982–1986), from 187.1 to 341.3 in men and from 26.1 to 154.6 in women (Thun et al., 1997). The increased mortality risk appears to be partially attributable to differences between the two studies in smokers’ average age of initiation, number of cigarettes smoked per day by smokers, and duration of smoking. Among those who had never smoked, the age-adjusted death rate did not change substantially between studies for either males or females.
Indirect Measures of Mortality Due to Smoking
Prospective cohort studies make it possible to estimate directly the number of deaths caused by smoking or other factors, but they are expensive to carry out, require decades of commitment, and often are not available for populations one wishes to study. In particular, while the CPS-II offers reliable data on a large (but nonrepresentative) subgroup of the U.S. population, few other countries have comparable studies. Therefore, researchers have developed a variety of indirect ways to determine the number of deaths caused by smoking.
The major challenge researchers face when attempting to estimate smoking-related mortality in a population is obtaining reliable information about the smoking habits of the population. Peto and colleagues (1992) devised an innovative way of getting around the fact that good data on the smoking habits and histories of most populations one wishes to study are relatively scarce. The basic idea is that one can use the rate of lung cancer in a population to obtain a reasonably good estimate of the total smoking burden in that population. One starts with the amount of lung cancer mortality in the population, which is available wherever careful cause-of-death statistics are kept. The underlying assumption is that even if no one in a particular population of people smoked, there would still be a certain small number of lung cancer deaths, and it is possible to know what that small number would be by, for example, looking at the lung cancer
mortality rate among nonsmokers from the CPS-II. Then the number of lung cancer deaths in the population over and above the expected baseline is assumed to reflect the amount of smoking in that population—and in particular, the amount of damage caused by smoking, which is a function of the number of smokers and how much each has smoked over his or her lifetime. Although this method depends on several assumptions, they are reasonable ones, and this indirect measure of smoking behavior may even provide a better indication of smoking-inflicted damage in a population than measures based on asking people to report how much they have smoked over their lives (Preston et al., 2010b).
With this indirect measure of smoking, it is then possible to use mortality statistics in a population to estimate the mortality from various causes that can be attributed to smoking. This is what Peto and colleagues (1992) did to estimate the smoking-related deaths in various developed countries. In particular, they used relative risks of smokers versus nonsmokers for various diseases, calculated from the CPS-II. That is, they looked at how much more likely smokers were to die from such causes as coronary heart disease or cerebrovascular disease, based on the CPS-II cohort study, and then assumed that the same relative risks would hold for smokers in other countries. Then by applying these relative risks to the population of smokers in a given country—estimated from the lung cancer mortality rates—they could calculate what percentage of deaths from each disease was due to smoking. According to their estimates, 35 percent of the deaths attributable to smoking are from vascular diseases, 24 percent from lung cancer, and 16 percent from COPD. The authors estimate that among U.S. men aged 35 and older in 2000, 24 percent of all deaths were attributable to smoking; the comparable figure for women is 20 percent (Peto et al., 2006).
Preston and colleagues (2010a) developed an alternative approach for estimating smoking-related mortality from cause-of-death statistics. Instead of using relative risks derived from a cohort study, they performed a large-scale statistical analysis of deaths from lung cancer and from all other causes in 21 countries over the period 1950–2006. The idea behind this approach is that “lung cancer mortality is a reliable indicator of the damage from smoking and that such damage has left an identifiable imprint on other causes of death at the population level” (Preston et al., 2010a, p. 2).
There have been a number of other variations on the original Peto method, each with the goal of estimating the amount of smoking-related mortality in a population. Before using this sort of approach to analyze the effects of smoking on mortality in the various countries studied here, it is useful to examine historical smoking patterns in those countries, which are at the root of the patterns seen in smoking-related mortality. In doing so it is important to keep in mind that smoking trends lead mortality trends by 20 to 30 years (Lopez et al., 1994).
INTERNATIONAL SMOKING PATTERNS
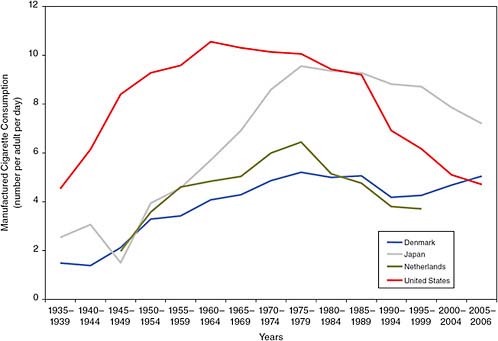
Fifty years ago, smoking was much more widespread in the United States than in Europe (see Figure 5-1). Compared with most Europeans, a greater proportion of Americans smoked, and they smoked more intensively (Pampel, 2010). There are probably many reasons for the higher level of smoking in the United States, including the fact that cigarettes were made freely available to many military and civilian groups during World War II; higher per capita income in the United States, which made cigarettes seem more affordable than in other countries; excellent growing conditions for tobacco in part of the United States; intense advertising campaigns in magazines, on billboards, and on the radio; the widespread smoking of popular movie stars on the silver screen; and the presence of a number of major tobacco companies (Brandt, 2007; Ravenholt, 1990). Over the next two decades, the prevalence of smoking grew steadily in most countries, with the United States continuing to have one of the highest levels. After 1964, when the Surgeon General’s office released its authoritative report on the adverse effects of cigarette smoking, the increase in smoking slowed, stopped, and eventually reversed in the United States. The peak of smoking among males occurred in cohorts born around 1915–1920 and among women in cohorts born around 1940–1944 (Preston and Wang, 2006). During the 1970s, smoking began to decline in most high-income countries, but the decline has been much greater in the United States than in most of Europe, particularly among men (Cutler and Glaeser, 2006). Today there are a number of European countries, as well as Japan, where the per capita consumption of cigarettes is greater than in the United States (see Figure 5-1).
Within each of the countries, the diffusion of smoking among the population has tended to follow a similar pattern. Smoking first catches on among people in higher socioeconomic brackets, then makes its way into the rest of the population. When smoking-related health issues begin to emerge, it is the people in the higher socioeconomic brackets who first begin to cut back on smoking so that in later stages, when smoking is in decline, it is much more common among those in the lower socioeconomic brackets (Pampel, 2010). The difference between men and women shows a similar pattern across countries. Smoking catches on first among men, and women are generally a couple of decades behind in terms of the numbers who smoke. Over time, smoking levels off among men while it continues to grow among women, and when smoking decreases, it tends to decrease more rapidly among men than among women, closing the gap between them (Pampel, 2010).
Focusing specifically on smoking among females, Pampel (2003) found that women in the United States and Northern Europe took up smoking in large numbers much earlier than women in Southern Europe and Japan. In particular, he identified two “clusters” of countries with similar patterns of
female smoking. In the first, consisting of the United States, Denmark, the Netherlands, and England and Wales, about a third of middle-aged women were smoking during the 1970s. In the second, consisting of France, Italy, Spain, and Japan, many fewer middle-aged women—from 5 to 17 percent—were smoking during the 1970s. As Pampel notes, if indeed smoking was a major factor in the diverging trends in life expectancy, the mortality trends from country to country should mirror these trends in smoking.
SMOKING AND LIFE EXPECTANCY IN HIGH-INCOME COUNTRIES
By using the method of Peto and colleagues (1992) or one of its variants to derive estimates of total smoking-related mortality from data on lung cancer mortality, it is possible to compare smoking-related deaths across countries and through time and thus to estimate the role of smoking in the divergence in life expectancy trends. This section describes two such studies that used different approaches and came up with very similar answers.
Preston, Glei, and Wilmoth (2010b)
Preston and colleagues (2010b) began with yearly cause-specific death counts from the World Health Organization’s (WHO’s) Mortality Database (World Health Organization, 2009) for 21 high-income countries from 1950 to the present. The distribution of deaths by cause of death, divided into 5-year age groups (50–54, 55–59, 60–64, 65–69, 70–74, 75–79, 80–84, and 85 and up) and into males and females, was combined with annual all-cause death counts and mortality rates from the Human Mortality Database (Human Mortality Database, n.d.) to determine the numbers of deaths due to lung cancer by country, year, gender, and age group. Then, instead of assuming that relative mortality risks for smokers based on the CPS-II (in the United States) could be extrapolated to other populations, as Peto and colleagues (1992) had done, the authors applied a regression analysis to investigate the macro-level statistical association between lung cancer mortality and mortality from all other causes of death. Using these results, they estimated how many of the deaths were due to smoking, on the assumption that the mortality due to smoking could be inferred from the number of lung cancer deaths over and above the baseline for lung cancer mortality.
The results of this analysis are shown in Table 5-1, which gives the estimated fraction of deaths for ages 50 and above that can be attributed to smoking for males and females in the 21 high-income countries during the years 1955, 1980, and 2003. One obvious result is that smoking was responsible for a much greater percentage of deaths among men than among women, although women were catching up in a number of countries. In
TABLE 5-1 Estimated Fraction of All Deaths at Ages 50 and Older Attributable to Smoking in 1955, 1980, 2003, by Gender and Country
|
Country |
Males |
Females |
||||
|
1955 |
1980 |
2003 |
1955 |
1980 |
2003 |
|
|
Australia |
0.07 |
0.22 |
0.17 |
0.00 |
0.04 |
0.10 |
|
Austria |
0.15 |
0.21 |
0.17 |
0.01 |
0.02 |
0.05 |
|
Belgium |
0.09 |
0.30 |
0.27* |
0.00 |
0.01 |
0.05* |
|
Canada |
0.07 |
0.22 |
0.24 |
0.01 |
0.06 |
0.19 |
|
Denmark |
0.07 |
0.22 |
0.20 |
0.01 |
0.06 |
0.16 |
|
Finland |
0.18 |
0.28 |
0.17 |
0.01 |
0.02 |
0.04 |
|
France |
0.05 |
0.17 |
0.19 |
0.00 |
0.00 |
0.02 |
|
Hungary |
0.07 |
0.22 |
0.30 |
0.01 |
0.05 |
0.13 |
|
Iceland |
0.03 |
0.06 |
0.16 |
0.00 |
0.11 |
0.18 |
|
Ireland |
0.04 |
0.17 |
0.19 |
0.02 |
0.07 |
0.14 |
|
Italy |
0.04 |
0.20 |
0.23 |
0.00 |
0.01 |
0.04 |
|
Japan |
0.01 |
0.11 |
0.20 |
0.00 |
0.03 |
0.09 |
|
Netherlands |
0.10 |
0.32 |
0.26 |
0.00 |
0.01 |
0.09 |
|
New Zealand |
0.08 |
0.21 |
0.17 |
0.00 |
0.06 |
0.12 |
|
Norway |
0.02 |
0.09 |
0.16 |
0.00 |
0.01 |
0.07 |
|
Portugal |
0.02 |
0.07 |
0.12 |
0.00 |
0.00 |
0.01 |
|
Spain |
0.04 |
0.14 |
0.22 |
0.00 |
0.00 |
0.00 |
|
Sweden |
0.03 |
0.10 |
0.09 |
0.00 |
0.02 |
0.06 |
|
Switzerland |
0.09 |
0.19 |
0.16 |
0.00 |
0.01 |
0.04 |
|
United Kingdom |
0.16 |
0.30 |
0.20 |
0.02 |
0.09 |
0.15 |
|
United States |
0.08 |
0.23 |
0.22 |
0.01 |
0.08 |
0.20 |
|
*Estimates based on data from 2004 for Belgium. SOURCE: Preston et al. (2010b, Table 4-2). Reproduced with permission. |
||||||
Iceland by 2003, smoking actually accounted for a larger percentage of deaths among women than among men. The United States was one of only a few other countries in which the percentages for women approached those for men. In general, although smoking accounted for a negligible percentage of deaths among women in every country in 1955, the numbers have grown steadily since that time, particularly in the past quarter century. The exceptions to that trend are France, Portugal, and Spain, where smoking-related deaths still account for just a tiny percentage of total deaths among women. The pattern among men has been different. By 1980 smoking-related deaths were a significant percentage of all deaths among men for almost every country, and thereafter the risk increased in 10 countries and decreased in 11, including the United States.
Next, Preston and colleagues (2010b) calculated what the life expectancy numbers would have been for different years if there had been no smoking. By comparing these estimated nonsmoking life expectancy rates with the real rates, they could see the effect of smoking on life expectancy
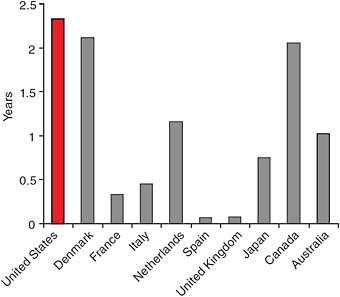
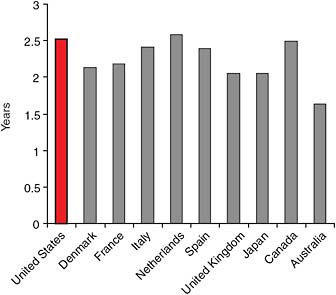
in the different countries. The results for life expectancy at age 50 in 2003 for the 10 countries considered in this report are shown in Table 5-2 and Figures 5-2 and 5-3. As can be seen from Table 5-2, smoking had a negative effect on life expectancy at 50 for both men and women in all the countries surveyed. The smallest effect—a loss of 0.08 years—was among women in Spain, and the largest—a loss of 2.58 years—was among Dutch men. Other than in the Netherlands, the greatest effect on male life expectancy at 50 was seen in the United States (a loss of 2.52 years), Canada (2.49 years) and Italy (2.41 years). Among women, smoking had the greatest effect on life expectancy in the United States (a loss of 2.33 years), Denmark (2.12 years), and Canada (2.06 years). Figures 5-2 and 5-3 show graphically that there is much less variability in the male than in the female gains from eliminating smoking, probably because smoking was much more widespread among males than females in developed countries.
The average effect of smoking on female life expectancy in the nine countries other than the United States was a loss of 1.07 years. Since the effect in the United States was a loss of 2.33 years, smoking cost U.S. women an average of 1.26 years in life expectancy relative to women in the other countries. By contrast, smoking cost U.S. men only 0.31 year in life expectancy relative to the average for men in the other nine countries.
The total gap in life expectancy at age 50 between the United States and the nine other countries is 1.61 years for women and 0.76 year for men (see Table 5-2). Thus, the difference in the estimated damage caused by smoking accounts for 78 percent (1.26/1.61) of the shortfall of U.S. female
TABLE 5-2 Life Expectancy at Age 50 in 2003 Before and After Removal of Deaths Attributable to Smoking
|
Country |
Males |
Females |
||||
|
With Smoking |
Without Smoking |
Difference |
With Smoking |
Without Smoking |
Difference |
|
|
Australia |
30.63 |
32.25 |
–1.63 |
34.59 |
35.61 |
–1.02 |
|
Canada |
29.82 |
32.31 |
–2.49 |
33.85 |
35.91 |
–2.06 |
|
Denmark |
27.77 |
29.89 |
–2.13 |
31.66 |
33.78 |
–2.12 |
|
France |
28.83 |
31.01 |
–2.18 |
34.59 |
34.92 |
–0.33 |
|
Italy |
29.46 |
31.88 |
–2.41 |
34.19 |
34.64 |
–0.45 |
|
Japan |
30.47 |
32.52 |
–2.05 |
36.66 |
37.41 |
–0.75 |
|
Netherlands |
28.34 |
30.92 |
–2.58 |
32.55 |
33.69 |
–1.15 |
|
Spain |
29.00 |
31.39 |
–2.39 |
34.44 |
34.52 |
–0.08 |
|
United Kingdom |
28.62 |
30.67 |
–2.05 |
32.21 |
33.87 |
–1.66 |
|
United States |
28.46 |
30.98 |
–2.52 |
32.25 |
34.58 |
–2.33 |
|
Non-U.S. average |
29.22 |
31.43 |
–2.21 |
33.86 |
34.93 |
–1.07 |
|
SOURCE: Adapted from Preston et al. (2010b, Table 4-4). Reproduced with permission. |
||||||
life expectancy relative to the mean of the nine other countries. For males, smoking accounts for about 41 percent (0.31/0.76) of the much smaller U.S. shortfall. Thus, smoking appears to be a very important factor in the subpar life expectancy of the United States, especially among women.
The numbers tell an interesting story about Japanese life expectancy. A number of observers have noted the anomaly that although the Japanese smoke heavily, the country is at the top of the life expectancy rankings (Stellman et al., 2001). As Table 5-2 shows, removing the effects of smoking-related mortality increases Japanese life expectancy in both men (by 2.05 years) and women (0.75 year) by values that are close to the average for the rest of the countries in the study. Thus, despite Japan’s high life expectancy, smoking has left a clear imprint on its mortality.
Preston and colleagues (2010b) also pinpoint how trends in life expectancy between 1950 and 2003 were affected by smoking during that period. Among men, smoking-related mortality had a negative effect on trends in life expectancy at age 50 in 9 of the 10 countries examined here. Only in the United Kingdom did declines in smoking-related mortality have a positive effect: an increase of 0.5 year in life expectancy at 50. In contrast, men in the United States lost an additional 0.8 year in life expectancy at 50 between 1950 and 2003 because of smoking, but changes in smoking cost men even more in Spain (1.3 years), Italy (1.2), France (1.1), the Netherlands (1.1), Denmark (0.9), and Japan (0.9) over this period. Part of the variation across countries reflects differences in the timing of the smoking epidemic: men in the United Kingdom and the United States adopted smoking earlier and have since exhibited declines, whereas the smoking epidemic hit other countries (e.g., Japan, Spain, Italy) much later (see Table 5-1). Thus over this period, the trends for men capture the latter portion of the smoking epidemic (including its waning) for some countries, such as the United Kingdom and the United States, but only the escalating portion for many others.
There was no corresponding drop in smoking among women during this time, and, not surprisingly, smoking had a negative effect on female life expectancy trends from 1950 to 2003 in all 10 countries. The largest decreases in life expectancy at 50 attributable to smoking occurred in Canada (a loss of 1.4 years), Denmark (1.5 years), and the United States (1.6 years). By contrast, there was very little effect on the life expectancy of women in Spain (a loss of 0.1 year), France (0.2 year), Italy (0.3 year), and Japan (0.3 year).
Between 1950 and 2003, the gain in U.S. women’s life expectancy trailed the mean gain in the other nine countries by 2.1 years. Preston and colleagues’ (2010b) results suggest that 42 percent of this shortfall (0.9/2.11) is attributable to the greater impact of smoking among U.S. women. Correspondingly, gains in life expectancy among U.S. men lagged behind those in the other nine countries by an average of 0.2 year, but differences in smoking-attributable mortality account for only 6 percent of the shortfall.
The effects of smoking on life expectancy trends in the United States are illustrated in Figure 5-4. The solid lines are the actual life expectancy trends, while the dotted lines represent what the trends would hypothetically look like if smoking-related mortality were removed. For men, the difference between the two trend lines widened steadily from 1950 to 1990, increasing from 0.7 year to 3.1 years, but then the gap began closing and had decreased to 2.5 years in 2005. An implication of the figure is that a rise in smoking-attributable mortality is responsible for the leveling off of male life expectancy in the 1960s and early 1970s. For women, on the other hand, the gap remained small until around 1975, when it began increasing rapidly, and by 2005 it had grown to 2.3 years—nearly as large as the gap for men in that year. The gap for women is expected to level off and then begin declining, in concert with lung cancer mortality.
Staetsky (2009)
Staetsky’s (2009) study differs from the work of Preston and colleagues (2010b) in that she relied on the original method developed by Peto and
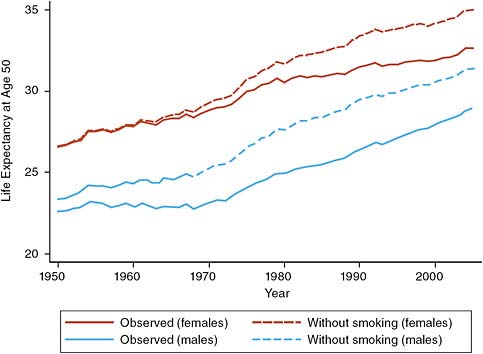
FIGURE 5-4 U.S. trends in observed and estimated life expectancy at age 50 without smoking, by gender.
SOURCE: Preston et al. (2010b, Figure 4-1). Reproduced with permission.
colleagues (1992) to estimate smoking-related mortality. She also looked at 8 high-income countries instead of the 21 that Preston and colleagues worked with, and she focused on women aged 65 and older instead of considering both men and women aged 50 and older as Preston’s group did. Her results are very much in line with those of Preston and colleagues—she, too, concludes that a large part of the divergence in female life expectancy trends among various high-income countries can be attributed to smoking patterns—but she offers some additional perspectives that are worth noting.
First, Staetsky observes that the eight countries she studied fall into two obvious clusters, each with similar mortality trends and smoking patterns among women. In the first cluster, consisting of the United States, Denmark, the Netherlands, and England and Wales, a large percentage of women—one-third or more—were smoking during the 1970s. Women in the second cluster, consisting of France, Italy, Spain, and Japan, had much lower rates of smoking. Mortality from lung cancer and other smoking-related diseases was relatively low for women in this second cluster of countries during the 1980s and 1990s, and their life expectancy rose steadily throughout this period. By contrast, women in the United States, Denmark, and England and Wales had much higher smoking-related mortality during this period, and their life expectancy slowed significantly relative to the other cluster of countries. The Netherlands was something of an anomaly, with lung cancer mortality trends falling somewhere between those of the two clusters. The reason, Staetsky concludes, is that women in the Netherlands took up smoking somewhat more slowly than women in the United States and Denmark, although more rapidly than women in the other cluster, so smoking affected their health to a degree falling somewhere between that in the two clusters.
Staetsky also addresses the issue of the apparent mismatch between the high levels of smoking in Europe today and the lower levels of smoking-related diseases relative to the United States. Statistics show that smoking currently is much more common in most European countries than in the United States among both men and women (British Heart Foundation, 2008). So how could smoking explain the fact that life expectancy has been increasing more slowly for U.S. women than for women in most European countries? The answer lies in the 20- to 30-year period between the time people start smoking and the time major health effects appear. In the period from the 1950s to the 1970s, U.S. women were smoking at a much higher rate than women in most European countries, particularly those in Southern Europe, and it is the smoking behavior during that period that affected life expectancy in the 1980s and 1990s. Now that smoking is more common among women in Europe than among those in the United States, the effects are likely to be seen in life expectancy trends over the next couple of decades.
DISCUSSION
While this chapter has focused on the United States in comparison with other countries, it should be noted that smoking has been implicated in the poor performance of other countries as well. In a paper commissioned for this study, Christensen and colleagues (2010a) conclude that smoking is the major factor explaining why Denmark’s life expectancy has fallen behind that of neighboring Sweden. Likewise, Juel (2008) used data from 1997-2001 to calculate that nearly all the difference in male life expectancy between Denmark and Sweden and about three-quarters of the difference in female life expectancy could be attributed to smoking- and alcohol-related deaths. Smoking-related deaths, estimated using the Peto/Lopez method, were the more important of the two.
In short, it appears that smoking is responsible for a good deal of the divergence in female life expectancy being examined here. During 1950-2003, gains in life expectancy at age 50 were 2.1 years lower among U.S. women compared with the average of the other nine countries examined in this study (5.7 vs. 7.8 years gained, respectively); Preston and colleagues (2010b) estimate that smoking accounts for 42 percent of this shortfall. In terms of the gap in life expectancy between the United States and other countries in 2003, Preston and colleagues find that differences in the estimated damage caused by smoking account for 78 percent of the 1.6-year gap for women and 41 percent of the 0.8-year gap for men. It appears that smoking has also caused significant reductions in life expectancy in Denmark and the Netherlands, two other countries with relatively poor life expectancy trends. On the other hand, smoking has also had a major negative effect on life expectancy in Canada, where life expectancy trends have been much more favorable. Thus although smoking clearly helps account for the lagging performance of the United States, it is only one of many factors affecting trends in life expectancy.