1
Introduction
People live in a world of growing interdependency and complexity. The old English word “connexity” is an appropriate description that helps to define the combination of connectivity and complexity that is our reality. Tick-borne diseases (TBDs), including Lyme disease, are certainly embedded in our world of “connexity.” This group of diseases defies simple cause and effect explanations and, while science has enabled us to uncover critical information on TBDs, we also realize that much more remains hidden.
TBDs represent some of the world’s most rapidly expanding arthropodborne diseases, yet there still are significant gaps in our understanding and an incomplete knowledge of them. While we can map the genome of Borrelia burgdorferi, the spirochete that causes Lyme disease, we still lack clarity in the natural history, epidemiology and true ecology of this pathogen as well as for other microbes involved with other TBDs. The state of the science is promising, but we lack a national, integrated research road map and an appreciation of the process of system thinking in considering these diseases and their human impact.
Rather than focusing on a more reductionist approach to science and research, we must fully understand parts in relationship with the whole and how they influence one another. The field of complex systems is relevant to the study of TBDs. This new field cuts across traditional disciplines of science, medicine, and the social sciences. It focuses on parts, wholes, and relationships. TBDs are problematic because causes and effects are not obviously related or are not closely associated in time and space. In addition, ecological knowledge has been grossly underused both to under-
stand emerging infectious diseases and to reduce the burden of disease and mitigate its expression.
Vector-borne diseases, including diseases transmitted by ticks, continue to be a public health concern in the United States and abroad. Ticks are arthropods that belong to two large groups: hard (ixodid) and soft (argasid) ticks. Soft ticks undergo no more than seven molts during their life cycle while hard ticks undergo three (see Figure 1-1). The life cycle duration varies
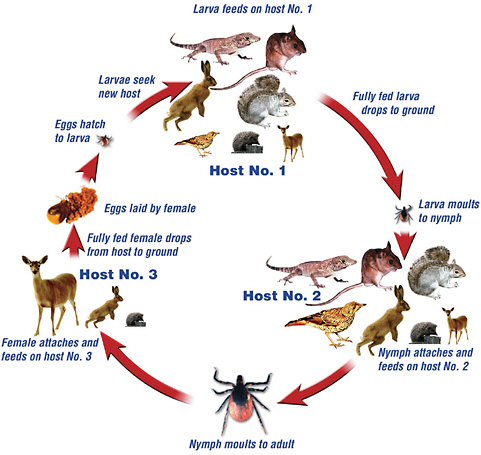
FIGURE 1-1 The life cycle of a three-host tick, such as Ixodes and Dermacentor sp., illustrating the common host for each stage. In this example, beginning prior to the first host, the eggs hatch to larvae and then feed on the first host. After the larval feeding is complete, the larvae drop from the host and molt to the nymph stage. At this stage, the nymph attaches and feeds again. It then drops off of the second host and molts to an adult. The adult tick attaches to a third host for a final meal. Following the final meal, the tick drops off and eggs laid by a female tick restart the process.
SOURCE: Reprinted with permission of Dr. Jeremy Gray.
as each of these life stages requires a blood meal from a vertebrate host. Ticks are highly adaptive to environmental change. In warmer climates, the life cycle duration may be less than a year. In colder climates, ticks can go months or years without feeding when the hosts are not available; the life cycle can encompass 3 years or longer. Numerous vertebrate species, such as rodents, deer, and rabbits, participate in zoonotic cycles that maintain infectious organisms in nature. Typically, a tick becomes infected with a virus, bacterium, or protozoan by feeding on an animal (reservoir host) that has the infection in its blood or by transovarial transmission of infected ova from an infected adult female tick. Ticks, in turn, transmit pathogens to reservoir animals in their salivary secretions while feeding.
This unusual lifestyle requires extraordinary evolutionary adaptation of the pathogen to the hosts, both vertebrate animals and ticks. Each of the tick-borne infections is initiated by the inoculation of the pathogen in saliva secreted by the feeding tick. Acquiring a blood meal occurs in a short feeding period only by soft ticks. Hard ticks feed for a period of days. Thus, ticks have evolved salivary components with pharmacologic activities of anticoagulation to maintain blood flow in the feeding site and modulation of inflammation and immunity to prevent deleterious host responses to the tick. The pathogen is transmitted via the tick saliva secreted during feeding (Kaufman and Nuttall, 1996), which has pharmacologically active substances (e.g., antihemostatic, vasodilatory, anti-inflammatory, and immunosuppressive) to aid in effective transmission. Infectious inocula can benefit from the local effects of the saliva, and through this transmission severe or fatal illness can result.
The local tick bite site lesion is useful for diagnosis of Lyme disease (erythema migrans) and R. parkeri infection (eschar). A multiple circular rash (Lyme disease); inflammation of the head and neck (tularemia); maculopapular, sometimes petechial, rash (Rocky Mountain spotted fever); and maculopapular, sometimes vesicular, rash (R. parkeri infection) occur frequently. Cutaneous lesions are observed less often in other tick-borne diseases in the United States.
Ticks have been recognized as a source of infections for humans for more than 100 years. In the United States, more than a dozen infectious diseases are transmitted by ticks (Table 1-1). The most common tick-borne diseases in the United States include Lyme disease, babesiosis, anaplasmosis, ehrlichiosis, relapsing fever, tularemia, Rocky Mountain spotted fever, and other rickettsioses. The geographic distributions of TBDs vary depending on the prevalence of the pathogen and vectors and the ecological system in which they are embedded. Additionally, tick-borne infectious diseases vary tremendously in their severity of illness. Severity of illness corresponds to visceral involvement (e.g., interstitial pneumonia and encephalitis) in Rocky Mountain spotted fever and human monocytotropic (or monocytic)
TABLE 1-1 Tick-Borne Infections in the United Statesa
|
Disease |
Agent |
Vector(s) |
Intracellular/Extracellular |
Chronic/Prolonged/Acuteb |
Life-threatening |
|
Lyme borreliosis |
Borrelia burgdorferi |
Ixodes scapularis, Ix. pacificus |
E |
Acute >> Chronic |
No |
|
Babesiosis |
Babesia microti |
Ix. scapularis |
I |
Prolonged |
Seldom |
|
Rocky Mountain spotted fever |
Rickettsia rickettsii |
Dermacentor variabilis, D. andersoni, Rhipicephalus sanguineus |
I |
Acute |
Yes |
|
Maculatum disease |
R. parkeri |
Amblyomma maculatum, A. americanum |
I |
Acute |
No |
|
Human monocytotropic ehrlichiosis |
Ehrlichia chaffeensis |
A. americanum, D. variabilis, Ix. pacificus |
I |
Acute |
Yes |
|
Ewingii ehrlichiosis |
E. ewingii |
A. americanum, D. variabilis |
I |
Acute |
No |
|
Human granulocytotropic anaplasmosis |
Anaplasma phagocytophilum |
Ix. scapularis, Ix. pacificus |
I |
Acute |
Yes |
|
Tick-borne relapsing fever |
Borrelia turicatae, B. hermsi |
Ornithodoros turicatae, O. hermsi |
E |
Prolonged |
Yes |
|
Tularemia |
Francisella tularensis |
D. andersoni, D. variabilis,A. americanum |
I/E |
Acute |
Yes |
|
Powassan/Deer tick virus encephalitis |
Powassan and deer tick viruses |
Ix. scapularis, D. andersoni |
I |
Acute |
No |
|
Colorado tick fever |
Colorado tick fever virus |
D. andersoni |
I |
Acute |
Rare |
|
Southern tick-associated rash illness (STARI) |
unknown |
A. americanum |
unknown |
unknown |
No |
|
a Worldwide, there are approximately 865 species of ticks (Keirans and Durden, 2005), of which four species, I. scapularis, I. pacificus, I. ricinus, and I. persulcatus, are the primary vectors for Lyme disease, babesiosis, and human anaplasmosis. Other tick-borne illnesses that may occur in the United States include Rickettsia sp. 364D strain carried by D. occidentalis on the Pacific coast, E.muris-like organisms in the upper midwestern states, Babesia duncani, and B. divergens-like infections. The most frequently imported travel-associated tick-borne illness is African tick bite fever caused by R. africae. Rickettsia massiliae, a potential human pathogen, has been identified in Rh. sanguineus ticks in the United States, but not yet in humans. Coxiella burnetii, the agent of Q fever, occurs in ticks, but transmission in the United States is associated with inhalation of aerosols from animal parturition. b E=extracellular; I=intracellular. |
|||||
ehrlichiosis. These illnesses, human granulocytotropic anaplasmosis, and tularemia can also manifest clinically as sepsis. Rocky Mountain spotted fever, however, is among the most virulent infectious diseases known, and human monocytotropic ehrlichiosis, human granulocytotropic (or granulocytic) anaplasmosis, tick-borne-relapsing fever, and tularemia are life-threatening diseases. Currently no documented cases of mortality are associated with Rickettsia parkeri, Ehrlichia ewingii, and southern tick-associated rash illness (STARI), and only very rare cases are associated with Borrelia burgdorferi (Kugeler et al., 2011). More than half of the tick-borne diseases in the United States are emerging infectious diseases—many of which have been recognized only in the past two decades. Given the growing list of tick-borne diseases, one would predict that there are others involving zoonotic cycles yet to be discovered.
Many TBDs such as ehrlichioses, anaplasmosis, Lyme disease, and Rocky Mountain spotted fever are on the rise as animal reservoirs and tick vectors have increased in number and range and humans have inhabited areas where reservoir and tick populations are prevalent (Ismail et al., 2010). From a public health standpoint, this disease trend is of growing concern. Also of growing concern is our incomplete knowledge and understanding of the complex interactions of ticks, hosts, pathogens, and habitats and the potential impact of climate change. Improvements in our knowledge of this group of emerging and re-emerging diseases and their dynamics will be needed to reduce the risk of infection and the burden of these diseases.
THE GENESIS OF THE WORKSHOP
The U.S. Senate and House included in the Appropriation Bill in September 2009 the following:
The Committee encourages the [NIH] Director, in collaboration with the Director of NIAID, to sponsor a scientific conference on Lyme and other tick-borne diseases. The Committee believes that the conference should represent the broad spectrum of scientific views on Lyme disease and should provide a forum for public participation and input from individuals with Lyme disease. (U.S. Congress, House, 2009)
In March 2010, the National Institutes of Health (NIH) and the National Institute of Allergy and Infectious Diseases (NIAID) contracted with the Institute of Medicine to form a committee to plan the workshop and summarize the viewpoints in a workshop report. NIAID charged the committee to hold a workshop that discussed the state of the science, but that
did not include a discussion of treatment guidelines. The committee was to provide opportunity for public input into the activity.
THE COMMITTEE APPROACH
The Committee on Lyme Disease and Other Tick-Borne Diseases: The State of the Science met on April 29, 2010, in a planning session that was open to the public. Federal agencies conducting research in the field were invited to make brief presentations to the committee. These presentations were followed by a public comment period during which interested members of the research, advocacy, and patient communities were invited to make brief remarks. Individuals also were encouraged to submit comments to the project’s e-mail address.
Following the initial meeting, the committee decided that it needed to provide additional opportunities for input from patients and groups not represented by the individuals who attended the planning meeting. The committee held four listening sessions on June 2, 15, 18, and 25, 2010, to hear from residents of the Southeastern United States (Georgia, North Carolina, South Carolina), the Southwest and West (Arizona and California), the Midwest (Iowa, Michigan, Minnesota, and Wisconsin), and members of the Native American population, respectively. The publicized target populations served as guidelines for those registering for the listening sessions, and the committee did not exclude anyone who wished to register but did not fit into one of the groups. A multiplicity of viewpoints with diverse ideas for workshop topics and speakers were expressed in the listening sessions, e-mail submissions, and planning meeting. Appendix C summarizes the public input into the agenda of the workshop.
The committee recognized the limitation of a 2-day workshop, which meant that not all proposed topics or speakers could be accommodated. The development of the agenda was driven primarily by topics that would cover the state of the science in tick-borne diseases: surveillance, burden of disease, diagnosis, diagnostics, at-risk populations, environmental and host interactions, pathogenesis, and prevention, as well as the human face of the disease. Per NIAID’s charge to the committee, discussion of treatment guidelines was excluded from the workshop. With any complex scientific discipline it is difficult to limit discussion on a disease without some references to treatment. The committee further excluded the topics of physicians’ discipline by state medical boards and insurance reimbursements from the workshop. Although these topics are of concern for many patients and clinicians, they fell outside the scope of the state of the science.
In addition to the speaker presentations, the committee commissioned 10 papers to gather further information on the state of the science of tick-borne diseases. The papers address a number of areas, including diagnostics,
emerging infections, tick-transmitted microbes, vaccines, environmental contribution to tick-borne diseases, atypical Lyme disease, global burden of disease, case definitions, and a patient perspective. The patient perspective paper addresses the human aspect of tick-borne diseases. All of the commissioned papers are included in Appendix A. The Committee also requested information from federal agencies that conduct research or have programs associated with tick-borne diseases in their current research programs on tick-borne diseases. The information is summarized in Appendix B. Finally, the committee recognized that even with a generous allotment of time for discussion, a number of comments would not be able to be expressed during the workshop. The additional comments sent to the committee by e-mail are summarized in Appendix E.
Upon reviewing the presentations and comments during the workshop and in the pre-workshop listening sessions, the committee acknowledged that the language and terminology used to describe various facets and manifestations of Lyme disease and coinfecting conditions are inconsistently applied. Rather than offering its own interpretation of terms and definitions used by the various presenters, the committee has transcribed the terms as they were used by the workshop participants. This does not imply that the committee believes that terms such as “post-Lyme disease,” “post-treatment Lyme disease,” “persistent Lyme disease,” and “chronic Lyme disease” are or are not interchangeable, differ in meaning or value, or have differing scientific validity. Similar confusion exists regarding terminology related to recurrent and relapsing Lyme disease with or without reinfection. As highlighted by many presenters, a standard lexicon that is consistently applied and understood would improve and advance research efforts regarding Lyme disease and other TBDs and likely improve patient care.
This workshop summary report is a reflection of what occurred during the workshop held on October 11–12, 2010. As part of the charge, the committee invited individuals with diverse viewpoints to present and participate at the workshop. The committee recognizes that not all viewpoints to fully discuss the nuances of the state of the science were represented at the meeting, nor with the number of topic areas could a point–counterpoint discussion occur. This workshop was designed to not reach concensus but to discuss a range of ideas. The committee did not weigh the scientific evidence on any topic, but it did ask the presenters to supply the references for their remarks. Where the references were available, they were included. Furthermore, the summary report should not be interpreted as a consensus of the Institute of Medicine, the committee, or its sponsors. The views presented, including the key knowledge gaps and research opportunities, are those of the individual speakers.
The reader will note that the workshop report is organized into eight additional chapters, which summarize the committee’s preliminary
introductions to the respective chapter and the corresponding presentations. The presentations from speakers and sessions may be presented in a different order than that reflected in the agenda. Chapter 2 provides a broad overview of tick-borne diseases from a systems perspective. Chapter 3 provides the societal and patient perspective of Lyme disease. Chapter 4 reflects the ecology, tick biology, and host interactions discussions. Chapter 5 provides a broad overview of the surveillance, spectrum, and burden of tick-borne diseases. It further includes a discussion of at-risk populations. Chapter 6 reviews the latest research on pathogenesis of four tick-borne diseases: Lyme disease, anaplasmosis, ehrlichioses, and rickettsial diseases. Chapter 7 reviews the state of the science on diagnostic tools for tick-borne diseases and the challenges for physicians in the treatment of these diseases. Chapter 8 provides a short overview of the prevention, including vaccines and non-pharmaceutical measures. Chapter 9 summarizes the viewpoints from participants on the research gaps, opportunities, and priorities for the field of tick-borne diseases.








