9
Medical Device Innovation and Public Policy in the European Economic Community
JOHN HUTTON
The general theme of this volume is “improving the translation of research findings into clinical practice. ” This is often taken to mean speeding up the process by which research findings can be developed into pharmaceutical products or medical devices—that is, turning inventions into innovations in the economic sense. In such circumstances public policy would be directed toward stimulating clinical research and smoothing the passage of new products into the marketplace, on the assumption that all advances in medical technology must be regarded as beneficial. This concept would be more readily accepted if the current diffusion and use of medical technology were considered appropriate universally.
In fact, the interpretation of improving the translation of research findings into clinical practice can be broadened in at least two directions. The relevant research findings could be taken to include studies of the effectiveness and cost of existing technologies, as well as the development of new ones, and the idea of improvement could be taken to mean better selection of socially desirable innovations. The broader interpretation opens the scope of public policy to include technology assessment, both in identifying deficiencies in existing technologies and in identifying the potential benefits and costs of new developments. This is the perspective from which the first two sections of this paper review the public policy in this area, the interested parties in policy formulation, and their reactions in different circumstances. The third section reviews the characteristics of European health care systems and the nature of the European medical devices market, and the fourth section examines the potential influences on policy in the European context, using illustrations from different countries. The final section deals with
developments in policy at the European Community level and how they may influence the actions of national governments.
PUBLIC POLICY AND INNOVATION: A MODEL
Social Objectives
Defined in the most general terms, the objective of most societies is to improve the quality of life of its members. Quality of life has many dimensions, but it is increasingly recognized that health is an important one because of its influence on an individual's ability to enjoy the other aspects of life. The links between health and quality of life have been developed to the point where the benefits of health programs are now generally assessed in terms of their impact on patients' quality of life. A variety of instruments and methods have been used (1,2). Figure 9.1 sets out the relationships among health, quality of life, and economic growth that are relevant to the discussion of the benefits of medical device innovation.
The left-hand column describes the assumed relationship by which more medical technology leads to better health care, which in turn leads to better health and improved quality of life. The right-hand column describes, in equally simplistic terms, a series of economic relationships. Increased research generates more innovation, which increases industrial output thus leading to higher incomes. The improvement in standard of living will have an impact on quality of life for society in general, although the benefits may be spread unevenly.
In fact, the economic and health elements are inextricably linked by cross-relationships at different levels. For example, higher national income levels can improve health through the provision of better housing, education, and nutrition and therefore can influence the health-related aspects of quality of life. Increased industrial output may improve the capability to provide health care, but it may also increase the demand for health services because of industrially related injury and illness. The links between research and industrial innovation are essential for the production of new medical devices that cannot be taken beyond the prototype stage within the laboratory or hospital research environment.
Accept for the moment that the relationships outlined in Figure 9.1 hold. Before we can say that efforts to increase medical device innovation are desirable, we must look at the alternatives. If resources are drawn into the health sector to develop more medical technology, this may simply switch resources from other sectors of the economy, resulting in transfers of benefit without necessarily producing a net gain (Figure 9.2 ). To produce a net gain in social value, medical device innovation must either improve the cost effectiveness of health care by providing increased changes in quality of life per dollar spent or produce equally effective technology at lower cost.
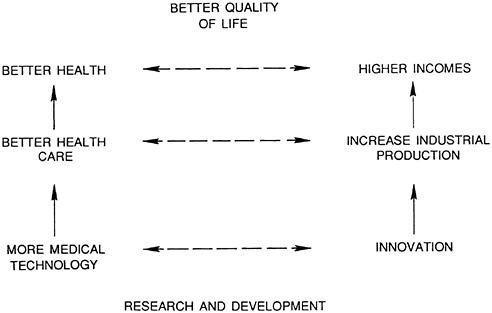
FIGURE 9.1 Social objectives
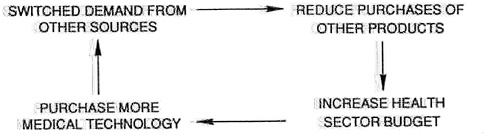
FIGURE 9.2 Transfers of benefit.
Also, it must be shown that innovation in the medical device industry is a more effective way of increasing quality of life and productive efficiency than innovation in other sectors of the economy.
Parties of Interest
The interest groups involved in medical device innovation can be grouped into three categories: manufacturers, health care providers, and government policy makers. Within each of these broad groups there may exist conflicts of interest between subgroups that make unambiguous identification of the objectives of the groups more difficult. For example, the attitude of established firms toward complicated and costly regulatory procedures may be
different from that of small companies trying to enter a market. Equally, the attitude of providers of health care toward new technology may be rather different from that of the health care financing bodies. Even within a hospital there will be a difference of attitude among managers, doctors, and boards of governors (3). At the government level overall objectives may be less easily determined because of the many policy concerns relating to health. For example, for reasons of macroeconomic policy, the government may wish to restrict public expenditure, including that on health care, while at the same time wishing to promote better quality of care and the development and diffusion of new technologies. Similarly, a desire to improve access to care by disadvantaged groups may not be compatible with policies to increase the cost effectiveness of service provision (4).
Because of the disparate objectives of subgroups within the main parties of interest, generalizations about policy responses are unwise. However, in the following section an attempt is made to characterize the different perspectives of public policy and the response to it by industry.
Policy Responses
Analysis of public policy responses to new technology generally takes place in the framework of a technology assessment model. In Figure 9.3 three key elements of technology assessment are represented: technical feasibility, social acceptability, and cost effectiveness.1 Technologies that exhibit all three characteristics, represented by the shaded area, are considered appropriate for implementation. Where technologies meet only two of these conditions, policy may be directed toward meeting the third. For example, in Figure 9.3, area (b), if a technology is socially acceptable and potentially cost effective, an effort to achieve technical feasibility could be supported by public policies aimed at increasing targeted research and development (R&D) expenditures.
The development of a drug therapy for acquired immune deficiency syndrome (AIDS) might fall into this category. On the other hand, in area (c) an existing technology that is considered cost effective may not be accepted by the section of the population that would benefit. In this circumstance investment in health education programs might be the appropriate policy response. A media campaign to promote cancer screening in groups most at risk would be one example. Technically feasible interventions that are publicly accepted or even in demand, area (a), pose problems for public health funding agencies if they are not cost effective. The overuse of expensive diagnostic testing procedures is an example. The policy response might be to fix reimbursement rates below the cost of using inappropriate technology in an attempt to delay diffusion of the more expensive methods. The reimbursement system needs to be quite complex in order to permit the use of the more expensive technology in cases where it is appropriate.
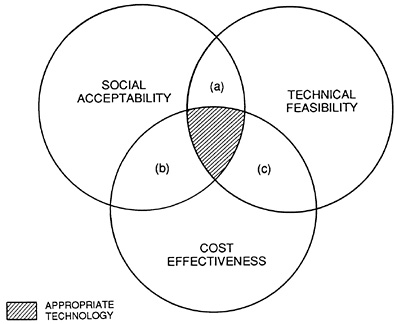
FIGURE 9.3 Technology assessment model.
Equally important are the responses of the manufacturers of the technology to the system of economic and bureaucratic incentives and disincentives. These can be analyzed in a similar way, but as Figure 9.4 shows, the industry is more likely to be concerned with the economic viability of technology, that is, marketability and profit, rather than its cost effectiveness and social value.
Thus, the third factor in Figure 9.4 is economic viability, and the co-incidence of the three factors indicates marketable technology. In area (b) a socially acceptable innovation that appears to be economically viable but is not yet technically feasible is likely to be encouraged by public and private organizations. Both government and industry will have an incentive to invest in further R&D, as is evident in the case of AIDS drugs. Where industry feels that a developed product is economically viable but not yet socially acceptable, area (c), the solution is more difficult. A public education campaign and promotion to health care providers who might influence public opinion may be one option. A recent example of this is the resistance from large sections of society to the application of advances in genetic engineering, even though they would probably not be the group affected, nor would they necessarily benefit from the new techniques. The third case involves a technically feasible and socially acceptable technology, area (a), for the use of which public health care funders are reluctant to reimburse and private demand is insufficient to produce economic viability. One possible response by the industry would be to undertake further development to reduce the
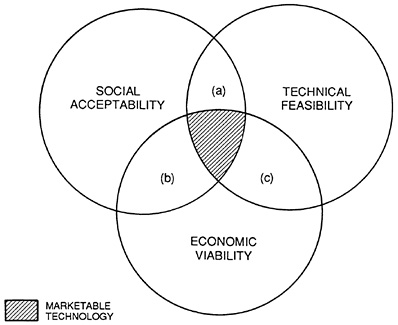
FIGURE 9.4 Diffusion model.
costs of the innovation. Alternatively, industry might lobby funding authorities for better reimbursement, with the support of patient groups and clinicians wishing to obtain the best, most modern medical care. The diffusion of whole-body computerized tomography (CT) scanning in the United Kingdom initially was funded extensively from private and charitable sources. The large amount of private funds ultimately drew in public funds, making the market viable. This was not the case for magnetic resonance imaging (MRI) in the United Kingdom, where tighter restrictions on public funds available for acquisition of the new technology severely affected its marketability and the apparently less dramatic nature of the innovation reduced public interest in raising charitable funds for its purchase.
Whereas the responses of government and industry to the existing situation will focus on increasing the marketability and cost effectiveness of technology, both parties will be pursuing long-term policies to alter the domains of Figure 9.4. R&D activities will expand the area of technical feasibility, and general economic growth will increase the economic viability of innovations through the consequent increase in purchasing power. These two elements clearly are closely linked. The pace of technological change has a strong influence on the rate of economic growth and richer societies adopt innovations more readily. Thus, the area of triple intersection in Figure 9.4 might increase over time, indicating marketable technology will increase not through convergence of the domains but through expansion of the domains.
THE EUROPEAN HEALTH CARE ENVIRONMENT
Europe has a wide variety of systems of health care organization and provision. In each country there is a mixture of public and private provision and public and private finance. This distinction is important because in many countries care is delivered in private hospitals but funded from public sources. The following framework is useful in classifying the different elements in the systems (6).
Public Health Care Systems
In these the whole system is owned, planned, managed, and financed by public authorities. Examples of such systems are the United Kingdom, Sweden, Denmark, Ireland, and Italy. The essential feature of the organization of these systems is that payment to individual health care institutions, such as hospitals and individual doctors, is not related directly to the number of patients treated.2 In the United Kingdom and Ireland a small proportion of funding comes from user charges, but the bulk comes from national taxation. Although the administration of the system is decentralized through regional and district structures, the national budgetary system gives the central government very strong control over total spending on health care.
In Denmark and Sweden national tax finance is supplemented by local taxation, and the service is managed by the counties. The budgetary system is similar for hospitals. They have local control over the use of funds, but the overall size of the budget is controlled centrally.
Social Security Health Care Systems
These systems are planned by public authorities, but funding comes from insurance agencies belonging to the social security system, from private insurance, and, directly, from patients. Health care is provided by institutions in public and private ownership. The health care systems of Germany, France, Belgium, the Netherlands, Spain, Portugal, and Greece come into this category.
Insurance contributions to the sick funds come from employees and employers and may be subsidized from general taxation. Membership of an insurance scheme is generally compulsory, although the scheme may be private rather than public. Reimbursement systems vary among countries. In France patients must pay for services and claim back the proportion of cost covered by the insurance. More generally, the sick funds are billed directly by the hospital or doctor providing the service. Because the income of service providers is linked directly to service provision, concern has been expressed over the tendency for expenditures to rise. As a consequence,
extra controls over the overall budgets of hospitals have been introduced in France and the Netherlands (8).
Private Health Care Systems
Where the hospital service is publicly owned and funded from taxation, as in the United Kingdom and Denmark, the private sector is relatively small, as very few people carry private health insurance. In countries where provision of services by the private sector is much higher, as in Spain, Germany, and France, the main source of funding is still the public sector, through the social security system. Although there is less government control over the operation of privately owned hospitals, the reimbursement system can be used to control service provision. Purely private medicine, in the sense of services provided by independent physicians in privately owned facilities and paid for directly by patients (out-of-pocket or through private insurance), is not a major feature of any European system.
THE EUROPEAN MEDICAL DEVICE MARKET
The European market for medical devices is characterized by a high level of intercountry trade; a strong overseas presence, particularly by United States companies; and a concentration of production in many submarkets with a small number of large multinational companies producing a large proportion of the output.
It was estimated in 1985 that Europe represented 25 percent of the world market for medical devices, which was worth around 30 billion dollars. The U.S. share was 53 percent (9). An analysis of the United Kingdom trade position for 1985 gives a good illustration of the complicated patterns of trade flows. The United Kingdom imported £470 million in medical devices and exported £560 million. Over 60 percent of the imports came from other European countries, whereas 42 percent of U.K. exports went to Europe. Direct trade with the United States represented 26 percent of U.K. imports and 17 percent of U.K. exports (9). This is only a partial indicator of U.S. involvement in the U.K. and European market, since much of the intra-European trade involves products manufactured in Europe by subsidiaries of U.S. companies.
The major European countries generally have a small number of major international medical device companies and a large number of small specialist companies. There is a considerable amount of merger activity. Smaller companies with innovative products often are absorbed by larger companies with the resources to market products on an international basis. Although the major companies have extensive research laboratories, many innovations come initially from universities and research institutes or nonspecialist companies. For example, the CT scanner was developed by EMI, a U.K. electrical
goods company with no previous medical products. The same firm developed the earliest clinical MRI system in the United Kingdom in collaboration with university researchers.
The largest European companies in the medical device market are Siemens of West Germany and Philips of the Netherlands, although both companies have significant production facilities in several other European countries. U.S. companies such as IGE of New York, Hewlett-Packard, and Baxter International also have a strong presence in Europe. The relative importance of individual country markets varies with the size of their overall economies but is also related to the characteristics of the health systems. For example, a recent survey of the imaging systems market in Europe found that West Germany had the largest share (42 percent), followed by France (20 percent), Italy (15 percent), and the United Kingdom (10 percent) (10).
FACTORS INFLUENCING MEDICAL DEVICE INNOVATION
Research and Development
Research activity is spread throughout the private and public sectors in European countries. A large amount of basic research is funded from public sources, by either direct government funding for national research organizations such as the Dutch Organization for Applied Scientific Research, or grants to other bodies. A great deal of research is undertaken by university institutes with government funding, often through academic commissioning bodies such as the U.K.'s Research Councils, as well as contracts with industrial companies (9,11). The proportion of public and private funding for research varies between sectors and between countries. In the United Kingdom, public funding has traditionally been high compared with the contribution of private industry. In other countries, such as West Germany, the industrial contribution has been much stronger, particularly in basic research, and the whole activity is seen much more as a partnership between government and industry to achieve longer-term economic objectives.
Generally, European countries see the funding of basic research as a government responsibility, with much of the health-related work being funded from the education budget, which supports universities and equivalent institutions, rather than from Ministry of Health budgets (9,11). Applied R&D of innovations is seen much more as the responsibility of industry. Although industry may receive some support from government, public sector organizations generally are not involved. In the development of the lithotripter in West Germany, the Dornier Company received research grants totaling 8.9 million German marks over an 8-year period from the Ministry for Research and Technology. Because of the expected cost effectiveness of the equipment in comparison with surgical techniques for the treatment of kidney stones,
the health insurance organizations promoted the rapid diffusion of the technology, aided by the political support given to a German innovation (12). The advantages of a strong domestic market, such as that enjoyed by the West German manufacturers, are illustrated by the much slower diffusion of the Dornier lithotripter in France, partly because of the potential development of a competing French product (13).
Regulation
Direct regulation of the marketing of medical devices exists in all European countries, but in no case does the system approach the strictness and detail of the U.S. system (14). Because the United States is still the world's most important market for medical devices, European manufacturers ensure that their products comply with U.S. standards, and, by doing so, they will also meet European requirements. The problem for manufacturers is that a separate process of approval has to be undertaken in each country, leading to an excessive administrative burden. This may well deter innovation by smaller companies without the resources to meet bureaucratic requirements (15).
Regulation may be product specific, as in France, or manufacturer specific, as in the United Kingdom (14). In the former system each product has to meet a range of safety and technical performance criteria. In the United Kingdom the system of Good Manufacturing Practice assessment has been established. Companies registering under the scheme must get approval of the organization and quality of their production processes. Products from approved companies can then be marketed to the U.K. Health Service (provided they meet conventional international electrical safety standards).
Although time consuming and sometimes expensive to meet, the regulatory systems for devices have not been a major impediment to the diffusion of new technology. This has partly been the result of the lack of requirements for demonstration of clinical effectiveness or economic efficiency in the approval process, which is essentially a technical exercise. A more important influence on innovation has been the regulation of technology in clinical use (16,17). This is achieved primarily through adjustment of the reimbursement system.
Reimbursement
Public health systems, such as the United Kingdom, Sweden, and Denmark, have controlled the diffusion of technology through the size of the overall budget given to each hospital. No formal barriers are raised against particular technologies, but budget constraints limit the freedom of individual units to acquire expensive items of equipment. In some cases positive moves have been taken to encourage the diffusion of some technologies and
their related equipment, through the provision of extra central government funds. For example, the promotion of the U.K. heart transplantation program and the expansion of renal dialysis services in Denmark and the United Kingdom followed this pattern (6,16,17).
Since 1986, public hospitals in France have been given fixed annual budgets to cover expenses for the treatment of social security members in their area. They are still able to charge fees for service for patients coming from outside their area. The system does not, however, apply to private hospitals. For certain expensive medical technologies specific approval is required from the Ministry of Health, which attempts to achieve a sensible diffusion of such technologies, geographically and between private and public sector hospitals. The Carte Sanitaire (or health map) is based on predetermined criteria relating to population and other characteristics of each region. The technologies regulated in this way include linear accelerators, CT and MRI scanners, dialysis machines, and lithotripters (8). Once approval has been given for acquisition of a technology, public hospitals can receive financial assistance for its purchase and are eligible for reimbursement for its use. Proposals have been put forward to extend these controls to private hospitals (10).
Similar controls have been instituted in other social security-funded systems, such as the Dutch Article 18 of the Hospital Provisions Act. It requires the government to produce a national plan for certain expensive technologies that cannot be used without a license. The technologies to which this provision applies are similar to those controlled under the French system. An additional provision allows the government and the Sick Funds Council to fund procedures on an experimental basis while they are being evaluated and before they come under Article 18. This was done initially with heart and liver transplantation (11).
More generally in social security systems, control over the diffusion and use of medical devices is exercised through the reimbursement rate. This has the advantage of applying to all users of the devices in both private and public sectors. Even in West Germany the continued rise in health care costs has led to restrictions on the approved applications of techniques such as MRI and more careful consideration of the level of reimbursement (18).
THE ROLE OF THE EUROPEAN COMMUNITY
Given the background of different systems of technical assessment of equipment for safety, design, and performance and the variable planning controls on the diffusion and use of devices in health care, what hope is there of any standardization? The most likely vehicle is the European Community, which has for many years been trying to coordinate the activities of its member states in this field (19). The Community has no official role in health care—this is regarded as a national government responsibility—
but it has a direct interest in industrial matters. Its interest is centered on opening access to markets and ensuring fair competition.
The development of the Community's role in industrial policy carries further implications for the medical device industry, which is dominated by multinational companies. The ability of national governments to control such companies is declining, but an active community anti-trust policy could have a stronger influence. Through its industrial policy role the Community has devoted resources to R&D in industries related to health care. Two of the most important justifications for funding one such program were “national interest” and “European cooperation” (20). To justify further R&D programs in the field of medical informatics, the need to improve “European competitiveness” features strongly (5,6). The European Commission is much more involved in funding applied research in industry because of its limited powers in the field of heath care, although it does have a medical and health-related research program (17). In the area of regulation, the European Community is encouraging the standardization of testing procedures for medical devices by member states in preparation for the “single market” in 1992. In spite of earlier delays and pessimism, significant progress is being made (14,19,21). Directives eventually will be introduced for different types of device, introducing stricter requirements, particularly with regard to clinical evidence on safety and performance.
CONCLUSIONS
Several trends that emerge from this overview of the European scene are significant from the U.S. perspective. All European governments are concerned about the cost and efficiency of their health care systems and are taking a more active interest in medical technologies as a potential cause of increased costs. As a consequence, there is increased emphasis on the cost effectiveness of innovations rather than just technical and clinical matters. How far this will influence the industry in terms of the quantity and nature of the devices that are selected for development remains to be seen.
The influence of the European Community is increasing, particularly through its encouragement of harmonization and standardization of regulations among member states. This will greatly ease the burden on manufacturers selling from within the Community, but its effect on those outside is less easy to predict. As the Community's standards are unlikely to be stricter than U.S. regulations, it seems plausible that U.S. companies not located in Europe would also benefit from a system requiring approval of products in only one member state in order to market throughout the Community.
In terms of competitive R&D the Community is keen to support the activities of European manufacturers through programs administered by the Commission. This compensates for the restrictions placed on national governments by Community competition laws. Although encouraging complete
freedom of competition within its boundaries, the Community may take on a more protective attitude toward the outside world. However, the truly global nature of the market, the dominance of multinational companies, and the dependence of many European companies on U.S. markets make it unlikely that the medical device market would be chosen for a confrontation with the United States on trade policy.
NOTES
1. This form of presentation was first used by the author at the Workshop on Technology Assessment and Industry at the 1989 Conference of the International Society for Technology Assessment in Health Care, held in London. It was subsequently used in a report produced for the European Commission to which the author contributed (5). He is grateful to Dr. Anne-Marie Warning of WHO, Copenhagen, for her contribution to the development of the approach.
2. The current reforms of the U.K. National Health Service (7) will change this, but the new system will not come into operation until April 1991. Under the new arrangements a proportion of hospital budgets will be dependent directly on the number of patients treated. Similar changes also are under consideration in Sweden.
REFERENCES
1. Torrance GL. Measurement of health state utilities for economic appraisal . Journal of Health Economics 1986 ; 6 : 1-30 . 2. Kind P. The Design and Construction of Quality of Life Measures . Discussion Paper No. 43. Centre for Health Economics, University of York , 1988 .
3. Greer AL. Adoption of medical technology: the hospital's three decision systems . International Journal of Technology Assessment in Health Care 1985 ; 1 : 669-680 .
4. Hutton J. The conflict between industrial policy and health policy in the UK medical equipment market . In Hutton S , Hutton J , Pinch T and Shiell A (eds). Dependency to Enterprise . London : Routledge , 1991 .
5. Commission of the European Communities . Research, Development and Technology in Medical and Bio-Informatics: Specification of Priority Tasks in Research, Development and Technology . Commission of the European Communities, Directorate General XIII , Brussels , 1989 .
6. Groot L. An overview of the systems and regulations concerning medical technology and the diffusion of six technologies . In Stocking B (ed). Expensive Health Technologies . Oxford : Oxford University Press 1988 ; op.cit. : 3-18 .
7. Department of Health . Working for patients . London : Her Majesty's Stationery Office , 1989 .
8. Lacronique JF. Technology in France . International Journal of Technology Assessment in Health Care 1988 ; 4 : 385-394 .
9. Advisory Council on Applied Research and Development . Medical Equipment . London : Her Majesty's Stationery Office , 1986 .
10. Editorial . West Germany leads EC imaging market . Clinica 1990 ; 383 : 10 .
11. Banta HD , Gelijns AC. Steering Committee on Future Health Scenarios . Anticipating and Assessing Health Care Technology . Volume I . Dordrecht : Martinus Nijhoff , 1987 .
12. Kirchberger S. The process of diffusion of the lithotripter in the Federal Republic of Germany . In Stocking B (ed). Expensive Health Technologies . Oxford : Oxford University Press , 1988 ; 54-59.
13. Dirieux P , Blum C , Jolly D. The Introduction of the First Lithotripter in France . In Stocking B (ed). Expensive Health Technologies . Oxford : Oxford University Press , 1988 ; 46-53 .
14. Sargentini A , Mariani L. Regulations governing medical devices . In Rapparini, R (ed). The Health Service Market in Europe . Brussels : Elsevier 1984 ; 46-83 .
15. Johnson P. Problems Facing Manufacture of Electromedical Devices . In Rapparini R (ed). op.cit. : 99-108 .
16. Stocking B (ed). Expensive Health Technologies . Oxford : Oxford University Press , 1988 .
17. Drummond MF (ed). Economic Appraisal of Health Technology in the European Community . Oxford : Oxford University Press , 1987 .
18. Krebs KH , John J. Federal Republic of Germany . In Drummond MF (ed). Economic Approval of Health Technology in the European Community . Oxford : Oxford University Press , 1987 ; 170-188 .
19. Rapparini R (ed). The Health Service Market in Europe . Brussels : Elsevier , 1984 .
20. Lavelle SM , Morucci JP , Dawids S. Committee in Bio-Medical Engineering Report on Technology Transfer and Assessment . In Drummond MF (ed). Economic Appraisal of Health Technology in the European Community . Oxford : Oxford University Press , 1987 ; 85-88.
21. Higson G. 1989—Year for big decisions in the active implantable medical devices directive . Clinica 1988 ; 383 : 12 .














