3
The Changing Economics of Pharmaceutical Research and Development
HENRY GRABOWSKI
These are interesting times for the research-oriented pharmaceutical industry. There have been several developments in recent years that are significantly changing the basic economics of pharmaceutical research and development (R&D). Since total investment and payback periods in pharmaceuticals now span three decades or more, it may be some time before all of the implications are apparent. However, some important changes in industry structure have begun to occur already.
This chapter provides an overview of several factors influencing the current and future environment for pharmaceutical R&D. The following section examines current trends in R&D opportunities, R&D costs, product life cycles, effective patent life, and prescription prices. The second section discusses some of the main results from a recently completed study of returns on past new drug introductions (1). This analysis provides a useful perspective for evaluating the impact of current industry trends. The final sections present conclusions and public policy implications.
CURRENT TRENDS
R&D Opportunities
The state of R&D opportunities in pharmaceuticals is no longer the source of concern that it once was. Industry R&D directors are very optimistic.
*Commentaries on the economics of pharmaceutical innovation can be found in Appendixes A and B.
TABLE 3.1 New Drug Products Achieving $100 Million Sales in the U.S. Market Within the First 6 Years of Market Life
|
Number Discovered |
||
|
Period |
Total |
Outside United States |
|
1970-1974 |
2 |
1 |
|
1975-1979 |
8 |
2 |
|
1980-1984 |
12 |
7 |
|
Total |
22 |
10 |
The combined research efforts of academia, government, and industry have produced major advances in biomedical science, many of which offer promising clinical applications. Increased knowledge of physiological processes at the molecular level enable researchers to develop more selective and potent pharmaceutical targets. New research tools, such as electron microscopy and X-ray crystallography, and new research techniques associated with biotechnology have helped enhance the search for significant new compounds. Because of these advancements, pharmaceutical industry R&D now can be categorized more as a “discovery by design” approach, as opposed to the random screening of compounds that was once prevalent.
We have seen many more economically and therapeutically important drugs emerge from the R&D pipeline over the past decade.Table 3.1 provides an analysis of the number of drugs introduced between 1970 and 1984 that achieved 100 million dollars in annual sales in the first 6 years of market life. Using this definition of commercially important drugs, Table 3.1 shows that there were 22 such compounds over this period. The time trend shown is especially striking. There were only 2 such products in the 1970-1974 period, 8 in 1975-1979, and 12 for 1980-1984.1
This table also shows that foreign discoveries account for 10 of the 22 commercially important drugs in the United States during this period. This reflects the multinational character of the pharmaceutical industry. Pharmaceuticals discovered abroad have been a prominent source of U.S. market introductions over the past two decades. Conversely, sales of U.S.-discovered products in foreign countries have been important to earning satisfactory returns on R&D by U.S. firms.
It is also worth noting that while the relevant 6-year period for evaluating the sales of 1985-1989 introductions is still less than half completed, there already are eight compounds achieving $100 million in annual sales
from this cohort.2 This period is therefore likely to easily exceed the preceding one in terms of commercially significant compounds.
Another interesting development concerns the therapeutic orientation of these big commercial successes. The leading therapeutic category for these compounds during the 1970s was antibiotics and non-steroidal anti-inflammatory drugs (NSAIDs). For the 1980s the leading category was cardiovascular agents. This class of drugs accounted for over half the major compounds introduced. This trend reflects the changing orientation of pharmaceutical R&D.
The introductions of major new products for cardiovascular and other clincial conditions have begun to have a significant impact on clinical practice. Significantly fewer prescriptions are being written for older therapies such as diuretics. They have lost much of their share of the market to several products that were not even present during the 1970s. Newer product categories, such as the ACE inhibitors, the calcium channel blockers, and the cholesterol reducers, now account for a significant and rapidly growing share of the cardiovascular drug market. It is also important to note that the rate of deaths per capita in the United States from cardiovascular causes has decreased significantly over the past decade (2). This reflects, at least in part, the availability of new and better medicines (2-4).
Another measure of innovative performance that has received attention in the literature involves the diffusion of new chemical entities (NCEs) across major world markets. Drugs that have been adopted in a majority of these markets have been categorized as internationalized or consensus NCEs. This measure has been employed by Coppinger and Haas, Barral, Thomas, and in my own research on international competitiveness (5-8). Less than one-quarter of the new products introduced worldwide achieve the status of consensus drugs in these studies. There is also evidence of a significant statistical correlation between the international acceptance of a new drug and its therapeutic and commercial significance (6).
Figure 3.1 indicates that the U.S. pharmaceutical industry had the largest share of consensus NCEs for the period 1970-1985. The U.S. industry's share of consensus drugs—43 percent—is approximately double its 24 percent share of worldwide introductions over this same period. Furthermore, the United States has had as many consensus NCEs as all of the countries in Europe together. These data indicate that the U.S. industry has been at the forefront of the pharmaceutical innovation process. Whether this will continue in the 1990s and beyond depends on a number of technological, economic, and public policy factors.
R&D Costs
There is strong evidence that the R&D process in the United States is becoming significantly longer and costlier.Figure 3.2 shows a plot of the average duration of the Investigational New Drug (IND) and New Drug
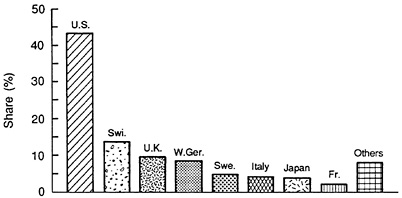
FIGURE 3.1 Consensus new drug approvals by nationality of originating firm, 1970-1985. (Source: Grabowski H. An analysis of U.S. international competitiveness in pharmaceuticals. Managerial and Decision Economics, 1989, Special Issue:27-33.)
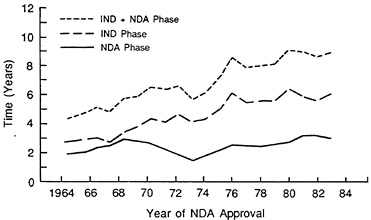
FIGURE 3.2 Duration of IND and NDA phases, self-originated NCEs of U.S. firms. (Source: Center for the Study of Drug Development.)
Application (NDA) phases for annual new drug approvals between 1964 and 1984. By the mid 1980s, the IND or clinical investigational phase averaged over 6 years and the NDA or regulatory review phase was about 2 1/2 years. If we add to this a pre-clinical phase of 3 or more years, we obtain a mean total R&D time of over 12 years. This total R&D time has been trending inevitably upward.3
The bar graph in Figure 3.3 shows annual industry R&D expenditures, expressed in constant dollars.4 The solid line shows the annual number of new drug introductions. This figure indicates that R&D expenditures have increased several fold, even after adjustment for economy-wide inflation. The increase in R&D investment is most dramatic in the past decade. At
the same time, the annual number of new drugs has changed only moderately. While the issue of R&D costs is best analyzed at the level of individual drugs, the two series in Figure 3.2 suggest that R&D investment costs per new drug introduction are increasing significantly in real terms.5
The Center for the Study of Drug Development at Tufts University recently has completed a microeconomic study of R&D costs. The principal investigators in this study are Joe DiMasi, Ron Hansen, Lou Lasagna, and myself (9). This analysis is designed to estimate the average R&D cost for NCEs discovered and developed by U.S.-owned firms (i.e., their self-originated NCEs). Data were obtained on a random sample of 93 drugs first tested in humans between 1970 and 1982. In this analysis the costs of drug candidates that fail in pre-clinical and clinical trials are incorporated into the average costs of the new drug introduction. R&D expenditures also are capitalized to the date of marketing introduction to reflect the time costs associated with an investment in pharmaceutical R&D.6 Our best estimate is that it takes an average of $231 million (in 1987 dollars) and 12 years to discover and develop a new drug. Of this total, $114 million is the out-of-pocket R&D costs and $117 million is the time cost associated with the 12-year average investment period. In addition, we find substantial variability around this mean cost estimate. Research is continuing with respect to how R &D costs vary by therapeutic category and other characteristics.
Our findings imply that average R&D costs per new drug introduction have been increasing significantly. An earlier analysis by Hansen (10) using the same general methodology found an average R&D cost of $54 million (in 1976 dollars). Hansen 's R&D cost estimate is $100.7 mil
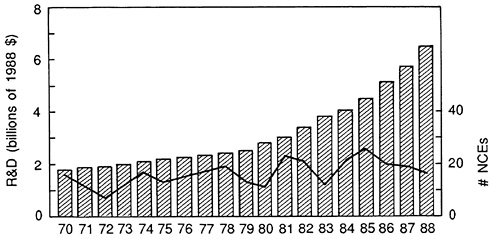
FIGURE 3.3 FDA approvals versus R&D spending. (Source: data on new drug introductions from the FDA and pharmaceutical R&D expenditures data from annual surveys of the Pharmaceutical Manufacturers Association.)
lion expressed in 1987 dollars. Hence, in real terms total capitalized costs are about 2.3 times larger in our study than in the earlier period analyzed by Hansen.7 What factors account for this increase in real R&D costs per new drug introduction? This is clearly an important issue for further research. Some key factors can be highlighted on the basis of our present knowledge. First, pharmaceutical R&D now entails significantly greater expenditures in the discovery phase. In addition, we found that the probability of success in the clinical phase has been increasing. These trends are consistent with a more science-based search process for new compounds. To date, however, the higher success rates at the clinical stage have not been sufficient to offset the greater costs expended elsewhere in the R&D process. A second factor associated with longer R&D times and higher costs per new drug introduction is the shift in research focus toward therapeutics to treat chronic clinical conditions such as cardiovascular disease and cancer. Chronic disease drugs require more long-term testing and greater overall resource investments prior to commercial introduction.8 A third factor accounting for higher R&D costs is the rapid escalation in the out-of-pocket costs of clinical trials and the greater capital equipment requirements associated with current R&D activities in the pharmaceutical industry. There are striking changes in this regard emerging from our analysis compared to the situation of a decade ago. Understanding the forces underlying this rapid increase in out-of-pocket costs is an important topic for future research.9
Product Life Cycles
Whereas R&D investment costs have been increasing, product life cycles have been getting shorter. This is the result of faster follow-on from competing new drugs and increased generic competition when patents expire. John Vernon and I have studied the life cycles of drugs introduced since the 1970s (1). It appears the sales volume of the average drug tends to peak somewhere around 10 to 12 years after market introduction. This research is ongoing, but there is definite information to suggest faster follow-on from competing drugs than in the past. The same factors that make the “discovery by design” research approach more effective, with higher probabilities of success, apparently also generate more intense competition from “fast followers.” 10
The biggest change in the commercial life cycle of a product in the 1980s, however, is due to increased competition from generics. Several years ago, when a patent expired, a manufacturer would lose part of the market share to generics, but at a fairly slow pace. A study by Statman (12) of the 1970s found that several years after patent expiration, the typical pioneering brand still maintained an 80 to 90 percent market share in terms of unit sales.
This situation changed dramatically in the wake of the 1984 Drug Price Competition and Patent Restoration Act and its related developments. One
major part of this act shortened and simplified the regulatory process for generic drugs by allowing the submission of an abbreviated new drug application (ANDA). This allowed generics an easier and faster entry into the market. John Vernon and I recently examined the experiences of 18 economically significant drug products whose initial generic competition occurred in the post-1983 period (13). For these drug compounds, the average product was subject to 25 generic competitors and lost approximately half its market share within 2 years. There was a tendency for the rate of sales erosion to accelerate with more recent patent expirations. Compared to the period before the 1984 act, today's commercial environment is much more competitive with regard to generics.
Effective Patent Life
Mindful that the 1984 act would result in greater generic competition and shorter times to recover R&D costs, legislators sought to grant brand manufacturers some relief by providing partial restoration of patent time lost during the clinical development and regulatory approval periods for new product introductions. Given this other objective of the 1984 act, what can be said about the effective patent life for current new drug introductions?
To date, most introductions have received partial benefits under the transition terms of the act.11 Figure 3.4 shows some initial results for the transition period from a study by Kaitin and Trimble (14). In particular, it shows the impact of the 1984 act on the average effective patent life for new drug introductions from 1982 to 1986. For the post-1984 12 period,
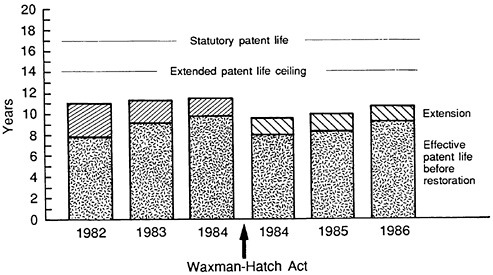
FIGURE 3.4 Effective U.S. patent life. (Source: Center for the Study of Drug Development.)
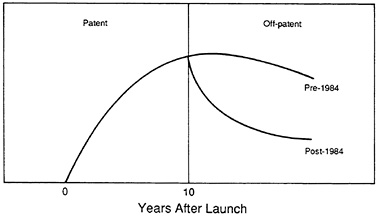
FIGURE 3.5 Waxman-Hatch Act: generic impact on sole-source product sales.
Figure 3.4 shows what the average effective patent life would have been without any restoration (the black portion) and the additional patent life provided by the act (the hatched portion). The total patent life for a new drug introduction in the post-1984 period averaged around 10 years. Of this total, the average additional benefit provided by the act in restored patent life is 1 1/2 years. These figures apply to the drugs in the transition phase, since most of these drugs were already patented and in clinical development when the act was passed.
We have also done some simulations at Duke on what the patent restoration periods will be when their benefits become fully effective. This involves restoring one-half of the time lost in the clinical development phase plus all of the time lost in the NDA approval process, subject to a cap of 5 years as well as other constraints. On average, recent introductions would get about 3 years of patent extension, based on our analysis. Some drugs will get the full 5 years and others will get none, depending on their status.
These analyses indicate that the average drug will probably have 10 to 12 years of effective patent life when the full patent restoration benefits from the 1984 act are implemented. The patent life may even be less if average development times continue to increase. In addition, when patents expire, there will be a rapid loss in sales and revenues of the pioneering product to generic imitators.
Figure 3.5 is a schematic diagram of the change that has occurred since the 1984 act was passed. Before 1984 loss of sales to generics was gradual; the erosion of sales in the post-1984 period has been much more rapid. This sharp decline in sales in the post-patent period is likely to intensify in the 1990s under the additional pressures of aggressive cost containment-driven generic substitution policies. In summary, manufacturers will most likely face an environment in which they must recoup their initial R&D
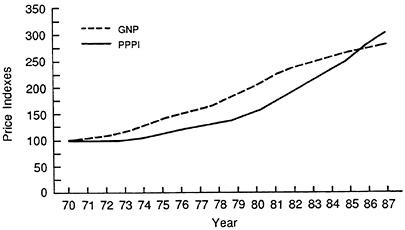
FIGURE 3.6 GNP versus PPPI. (Source: original data from U.S. Bureau of Labor Statistics and Economic Report of the President.)
expenditures and earn the majority of their positive return during the first 10 to 12 years of effective patent life.
Drug Prices
There has also been an important change in industry pricing behavior during the past decade. During the 1970s, U.S. prescription drug prices lagged inflation. At the end of the 1970s, however, prescription drug prices began to increase faster than inflation. This pattern persisted through the 1980s as illustrated in Figure 3.6, which shows a plot of the pharmaceutical producer price index (the PPPI) 13 versus the Gross National Product (GNP) price deflator. Both indices are valued at 100 in 1970 for comparative purposes. The GNP series advances faster during the 1970s, but the PPPI, with its faster growth in the 1980s, has a higher value than the GNP deflator by the end of this period.14
Increasing drug prices above the rate of general inflation has been one of the main strategic responses of the industry to higher R& D costs, shorter product life cycles, and increased generic competition. Analysis of the return on investment of new drug introductions, discussed in the next section, indicates that higher real prices in the 1980s had an important influence on industry's realized return on R&D investment.
RETURN TO PHARMACEUTICAL R&D
John Vernon and I recently completed a study investigating the economic returns of new drugs introduced during the 1970s (1). Our analysis employs a sample of 100 pharmaceuticals introduced between 1970 and 1979.
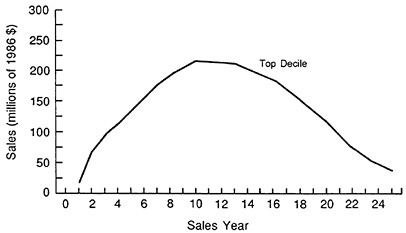
FIGURE 3.7 Sales profile of 1970-1979 compounds. (Source: Grabowski H, Vernon J. A new look at the returns and risks to pharmaceutical R&D. Management Science, 1990, 36:804-821 [using original data from IMS America Inc.].)
The average R&D cost per new drug introduction is based on Ron Hansen's original research work (10), but his figures are adjusted to take account of the time period of our introductions. Cash flows are estimated from historical sales data and are also projected forward in time using representative lifecycle profiles. We have assembled between one and two decades of data on sales and related variables for each compound in our sample so that we can estimate the returns with some degree of confidence.
Figure 3.7 shows the sales profiles for the top decile of compounds ranked by sales. This is an estimate of dollar sales on a worldwide basis. For this group of drugs, sales peak in year 10 and diminish gradually over an expected economic life of 25 years. Discounted cash flows are estimated from each drug's sales profile, utilizing a number of parameters pertaining to both U.S. and foreign operations.
Some of the key results of this study are as follows:
-
The economic breakeven point occurs 23 years after market introduction for the mean compound.15
-
A real annual return of 9 percent is earned by the mean compound.
-
Returns are significantly influenced by the higher drug prices of 1980s.
-
The distribution of returns is highly skewed.
The first two findings are illustrated in Figure 3.8. The cumulative present value curve (gross of R&D outlays) for the mean compound is negative in the early years of product life owing to the large upfront capital investment and to market launch expenditures. It becomes positive by the
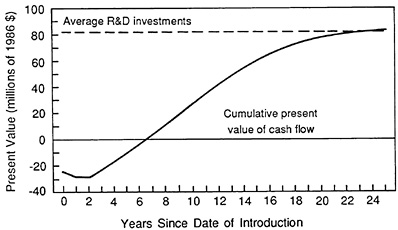
FIGURE 3.8 Present value of cash flow versus R&D investment. (Source: Grabowski H, Vernon J. A new look at the returns and risks to pharmaceutical R&D. Management Science, 1990, 36:804-821.)
sixth year of market life and achieves equality with the average after-tax R&D investment in year 23 of market life.
The after-tax present value of the mean compound, which is gross of R&D costs, equals $83.5 million by the end of the drug's projected market life. Our estimated after-tax present value of R&D costs for this period is $81 million. Since both series are capitalized at 9 percent, their approximate equality by the end of the projected market life implies that the mean compound earned a real annual return in the neighborhood of 9 percent. This is in line with our estimates of returns for investments of comparable riskiness over this period.16
Another major finding is that the higher real drug prices in the 1980s had an important effect on the returns to the 1970s introductions. A sensitivity analysis indicates that if no real price increases had occurred in the 1980s, the cumulative present value of after-tax cash flows for the average drug in Figure 3.8 would have been reduced by 16 percent. Under this scenario, the typical new drug introduction would not have been able to achieve breakeven in economic terms within its expected market life.
Finally, it is important to note that the revenues from new product introductions are highly skewed. This is illustrated in Figure 3.9. This figure shows the distributions of present values of after-tax cash flows by deciles for the sample of 1970s new product introductions. The top decile has an estimated after-tax present value that is several times the average after-tax R&D investment. At the same time, only the top three deciles have present values in excess of average R &D costs.17 These lower decile products may be important therapeutically and may also contribute economically in terms
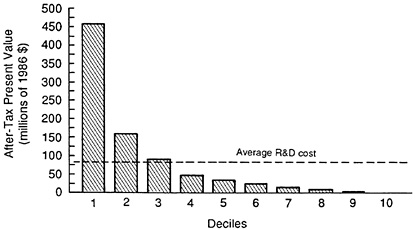
FIGURE 3.9 Present values by decile. (Source: Grabowski H, Vernon J. A new look at the returns and risks to pharmaceutical R&D. Management Science, 1990, 36:804-821.)
of incremental cash flows. However, these results indicate that from a financial perspective, a firm must occasionally have a drug or two in the top decile of sales if it is to cover the large fixed costs of pharmaceutical R&D (i.e., common discovery costs and the costs of the compounds that fail in the development phase).
IMPLICATIONS
If we turn now to a consideration of current and future returns to R&D, what insights can we garner from the above analyses? First, current new drug introductions will require, on average, substantially higher real R&D costs than previous introductions. Furthermore, these costs will have to be recovered over shorter product life cycles. New drugs introduced during the 1970s had an average patent life of 15 years and a gradual loss of sales over the assumed market life of 25 years. Current new drugs, whose patents expire in the 1990s or beyond, can expect an effective patent life of 10 to 12 years and a very rapid loss of sales in the post-patent period because of aggressive generic competition. Hence, a firm needs to realize most of its economic return from a new compound within the first 10 to 12 years of market launch. For pharmaceuticals launched during the 1970s, about one-third of their present value came from sales in the period between years 12 and 25. These revenues will have to be earned earlier in the life cycle by current new drug introductions, or imaginative strategies will have to be devised to extend the product life cycle.18
On a more optimistic note, the 1990s may see more commercially and therapeutically important innovations than in the past. Hopefully, the growing
level of industry R&D investment portends continued success. Preliminary analysis indicates that sales revenues for new drug introductions in the 1980s have been increasing steadily.
In sum, current economic trends will place a greater economic burden on the industry to achieve higher sales levels and more breakthrough products than in the past. This will be true given the present environment of expanding R&D costs, shorter product life cycles, and increased generic competition.
Two other likely implications of this new environment for future pharmaceutical competition are worth mentioning—greater earnings variability and increased concentration of industry sales and assets. Even if the firms in the pharmaceutical industry are successful in developing significant innovations that achieve a healthy return on investment, they still face greater risk and volatility. There are going to be companies, even within a general industry climate of relatively successful research, that will have their mainstay products come off patent, without anything immediately coming out of the R&D pipeline to replace the missing sales. Earnings and, consequently, stock values are likely to fluctuate. There have been many examples of this already. This type of unstable situation will be prevalent in the 1990s and will likely produce an environment of more mergers and acquisitions and thus greater industry consolidation.
PUBLIC POLICY CONSIDERATIONS
It is clear from the above analyses that R&D costs and returns on investment are highly sensitive to development and regulatory approval time. As we saw earlier, regulatory approval times currently average about 30 months for a new drug introduction. In some of our simulation analyses, we found that a 1-year decrease in regulatory approval times decreases breakeven lifetime by 3 to 4 years (15). This is due primarily to the fact that regulatory delays occur at the beginning of the product life cycle. If regulatory clearance times can be shortened, it would not only increase effective patent life, but firms would also realize their return on investment and subsequent profit at an earlier time. This would enable more drugs to cover their R&D costs and would therefore be a stimulus to further innovation. It is therefore worth examining current regulatory approval and clinical development procedures with an eye to accelerating what has become a very lengthy process. Making this process more efficient could have a high economic and social payoff.
Another factor influencing the returns to R&D is reimbursement policy. Many states now use formularies as criteria for Medicaid drug reimbursement. This process can result in significant time lags beyond those for Food and Drug Administration (FDA) regulatory approval before a drug is eligible for Medicaid reimbursement.Figure 3.10 shows the delays in formulary approval from a study of the Medicaid programs in six states (16). The
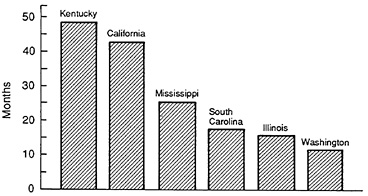
FIGURE 3.10 Medicaid formulary delays. (Source: Grabowski H. Medicaid patients ' access to new drugs. Health Affairs, 1988, 7:102-114.)
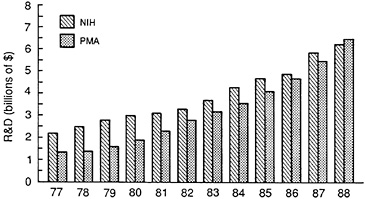
FIGURE 3.11 R&D expenditures: NIH versus PMA members. (Source: Compiled from original data from NIH and PMA.)
average delays range from 11.6 months in Washington state to over 40 months in Kentucky and California. These delays in giving approval result in lost revenue and lower expected returns to new drug introductions. Furthermore, there was no observed tendency for drugs of greater therapeutic significance to be more available to Medicaid patients in these states.19
The use of formularies in state Medicaid programs, and more generally in private sector managed health care settings, appears to be increasing over time (17). This could have a significant negative effect on the incentives for drug innovation in the 1990s. It is therefore appropriate to study whether formularies are really cost beneficial to society and, if so, whether reimbursement and coverage decisions can be made on a more timely basis.20
A third policy concern is the slowing rate of public sector funding for biomedical R&D relative to that of the private sector. Publicly funded biomedical research promotes much of the fundamental knowledge on which
the private sector search for effective new medicines is based. As discussed earlier, the rate of National Institutes of Health (NIH)-related biomedical R&D expenditures has been slowing. The drug industry has responded to these changes over the past decade by increasing its level of R&D investment. The relative changes over time are illustrated in Figure 3.11. In the late 1970s pharmaceutical industry R&D was about half that of the NIH. By 1988 the expenditures were approximately equal and proceeding on very different growth paths. The slower growth in public expenditures reflects the budget deficit problems of recent years. If left unchecked, this is likely to result in a downward pressure on both private and societal R&D opportunities in the 1990s. A vigorous public sector program in biomedical R&D is necessary to ensure that the search process for new medicines remains highly productive throughout this decade and that the United States remains at the forefront of this effort.
NOTES
1. The products that meet these sales criteria are as follows: 1970-1974, Keflex and Motrin; 1975-1979, Naprosyn, Tagamet, Mandol, Clinoril, Lopressor, Mefoxin, Ceclor, and Corgard; 1980-1984, Capoten, Carafate, Proventil, Tenormin, Xanax, Cardizem, Feldene, Halcion, Lopid, Procardia, Zantac, and Augmentin.
2. Using sales data through 1988 for this cohort, the eight drugs exceeding $100 million in annual sales are Mevacor, Rocephin, Seldane, Pepcid, Vasotec, Activase, Cipro, and Prozac.
3. The comparable figure on total R&D time in the early 1970s was between 9 and 10 years.
4. To express each year's R&D expenditures in dollars of constant purchasing power (i.e., to correct for inflationary changes), nominal values are divided by the GNP price deflator. The latter provides a representative price index for the goods and services produced for the U.S. economy.
5. The R&D expenditures series in this figure are based on Pharmaceutical Manufacturing Association (PMA) data. They include foreign and domestic R&D outlays of U.S. owned firms, but only the R&D expenditures in this country of foreign-owned firms. The NCE series counts all United States introductions, including those originating in United States and foreign countries. A complete economic analysis of these data would need to make adjustments in these series to make them consistent. In addition, it would need to take account of the lag in time between R&D expenditures and NCE introductions (9).
6. Time costs reflect the fact that a dollar obtained earlier in time is worth more than one obtained later in time, since it can be invested and earn a positive rate of return. In this analysis we used estimates of the opportunity cost of capital for the pharmaceutical industry to capitalize R&D expenditures.
7. There is also an indication of rising real costs from other studies of R&D costs in pharmaceuticals surveyed in our article (9).
8. In addition, drugs based on biotechnology appear to require greater investments than the more traditional chemically based approach, judging from industry
R&D outlays and product successes to date. However, this is a topic that needs further research.
9. The executive of one major pharmaceutical company recently analyzed data from his firm's records relevant to this issue (11). These data indicate that the amount of information and the number of patients required to support an NDA increased significantly in the 1980s. He also mentions the complexity and scope of research and the adoption of expensive new technologies as important factors leading to rising R&D costs.
10. There is also case evidence consistent with this hypothesis. Consider Tagamet, which was quickly followed by Zantac, which in turn was followed more recently by products such as Axid and Pepcid. One also has Capoten, followed quickly by Vasotec, with many other ACE inhibitors now in the pipeline. Contrast that with older products like the diuretics of an earlier era, where there was a much longer period of dominance by the pioneer brands.
11. The full patent term benefits apply to drugs that were not yet in the clinical development stage or not yet patented when the 1984 act was passed.
12. The effect on drugs introduced prior to the 1984 act is not of major significance to this discussion because of a one-time retrospective patent extension provision in the law that applies to a limited group of pre-1984 introductions.
13. The PPPI reflects the annual change in the prices of a market basket of established pharmaceutical products.
14. Neither the PPPI nor the GNP price deflator adequately adjusts for quality changes that occur over time. For sectors with rapid technological advances, such as pharmaceuticals or computers, the comparison of these government-derived indices can be somewhat misleading because these indices would tend to overstate the relative degree of price inflation in research-intensive sectors.
15. The economic breakeven point is computed on the basis of capitalized values, using a cost of capital of 9 percent.
16. We estimated that pharmaceuticals had an opportunity cost of capital of 9 percent on the basis of an analysis of financial returns for investments of comparable risks over an extended period (1).
17. Not all the compounds will have R&D costs equal to the average compound, as emphasized in the prior section. However, the available evidence indicates that revenues are much more skewed than R&D costs and the two variables are imperfectly correlated.
18. In order to extend the life cycle for products coming off patent, firms are devoting more effort to new delivery systems and also to over-the-counter status for some pharmaceuticals.
19. In particular, there was no relationship observed in our analysis between a drug's ranking of 1A, 1B, or 1C by the FDA and its availability to Medicaid patients in these six states.
20. A major issue in evaluating the cost effectiveness of formularies is to examine their system-wide effect on health care costs. In particular, formularies may lead to lower costs for the restricted pharmaceuticals, but higher costs elsewhere in the health care system. This result has, in fact, been observed in a number of case studies (18-20).
REFERENCES
1. Grabowski H , Vernon J. A new look at the returns and risks to pharmaceutical R&D . Management Science 1990 ; 36 : 804-821 .
2. Battelle Human Affairs Research Centers . The Value of Pharmaceuticals: A Study of Selected Conditions to Measure the Contribution of Pharmaceuticals to Health Status BHARC-01390/010 , Washington, D.C. , March 1990 .
3. Goldman L , Cook EF . The decline in ischemic heart disease mortality rates . Annals of Internal Medicine 1984 ; 101 : 825-883 .
4. Weinstein MC , Stason WB. Cost effectiveness of interventions to prevent or treat coronary heart disease . Annual Review of Public Health 1981 ; 2 : 363-395 .
5. Coppinger P , Hass A. International patterns in the availability of important new chemical entities introduced since 1970 . Paper presented at the 22nd Annual Drug Information Association Meetings , Washington, D.C. , June, 1986.
6. Barral PE. Ten years of results in pharmaceutical research throughout the world (1975-1984), Puteau Cedex France , 1985
7. Thomas LG. Spare the rod and spoil the industry: a ten nation study of competitive advantage in pharmaceuticals . Paper presented to the International Joseph Schumpeter Society Meetings , Charlottesville, Va. , June 1990 .
8. Grabowski H. An analysis of U.S. international competitiveness in pharmaceuticals . Managerial and Decision Economics 1989; Special Issue : 27-33 .
9. DiMasi J , Hansen R , Grabowski H , Lasagna L. The cost of innovation in the pharmaceutical industry . Forthcoming in the Journal of Health Economics, June 1991 .
10. Hansen, RW. The pharmaceutical development process: estimates of current development costs and times and the effects of regulatory changes . In RI Chien , ed. , Issues in Pharmaceutical Economics , Lexington, Mass . : Lexington Books, 1979 , 151-187 .
11. FDC Reports The Pink Sheet . Lilly estimates 3,300 person years go into development of successful new molecular entity; current antibiotic project is requiring 10,000 patients 1989, nos. 51 and 52:14 .
12. Statman M. The effect of patent expiration on the market position of drugs . In Helms R , ed., Drugs and Health , Washington, D.C. : American Enterprise Institute , 1982 .
13. Grabowski H , Vernon J. Brand Loyalty, Entry and Price Competition in Pharmaceuticals After the 1984 Drug Act . Duke University , Department of Economics , Discussion Paper Series, July 1990 .
14. Kaitin K , Trimble AG. Implementation of the drug price competition and patent term restoration act of 1984: a progress report . Journal of Clinical Research and Drug Development 1987 ; 1 : 263-275 .
15. Grabowski H , Vernon J. The Regulation of Pharmaceuticals: Balancing the Benefits and Risks . Washington D.C. : American Enterprise Institute, 1983 .
16. Grabowski H. Medicaid patients' access to new drugs. Health Affairs 1988 ; 7 : 102-114 .
17. Gold M , Joffe M , Kennedy TL , Tucker M. Pharmacy benefits in health maintenance organizations . Health Affairs 1989 ; 8 : 182-190 .
18. Smith D , McKeicher P. The elimination of selected drug products from the Michigan Formulary: a case study . Hospital Formulary 1984 ; 19 : 366.
19. Bloom B , Jacobs J. Cost effects of restricting cost-effective therapy . Medical Care 1985 ; 23 : 872.
20. Reeder E , Lingle E. An evaluation of the South Carolina medical open formulary system . Reston, Va. : National Pharmaceutical Council , 1988 .


















