Key Concepts: Chemical Sensitization
Michael D. Lebowitz
INTRODUCTION
Sensitization to chemicals can be defined as changes in the organism, usually the immunochemical system, by exposure to a chemical such that further chemical exposure leads to recognition by the organism. Such recognition will lead to a response that is marked by a greater reaction at lower doses than what would be observed in non-sensitized individuals. This is usually called hypersensitivity (Turner-Warwick, 1978). Inhulation of the antigen/allergen in an individual previously sensitized leads to an allergic reaction, such as rhinitis or conjunctivitis. If the skin is sensitized, as in allergic contact dermatitis, then contact will cause an oedematous response and/or a rash. Pulmonary (airway) sensitization manifests itself through bronchial constriction or obstruction (Davies and Blainey, 1983; Hetzel and Clark, 1983; Ramsdale et al., 1985). Some chemicals can produce different types of "allergy'. There are various known and hypothesized mechanism for sensitization. There are also host susceptibility factors, including genetic predisposition, which will play a role in sensitization and in disease manifestation (Turner-Warwick, 1978; Gregg, 1983). There must be some differentiation also between irritation and sensitization (Burge et al., 1979; Newman-Taylor and Davies, 1981). Irritants do not have the immunochemical recognition signal. Irritation in a non-sensitized individual usually leads to a slower, less serious response, at higher doses. This does not prevent irritants from producing or enhancing inflammation, which can be prolonged, and which can produce hypersensitivity reactions (e.g., bronchial hyperreactivity) when stimulated (discussed below). There are about 10-20% of almost any population which show greater "sensitivity" to irritants, which is usually organ (e.g., eye) specific, by responding at lower doses (Weber, 1984; EPA, 1986). (Acute infectious diseases may produce some of the same symptoms also, which would require other means of differentiation.) Chemicals have different potencies as sensitizers, not entirely dependent on the mechanism of sensitization. For instance, platinum salts have a very high potency (i.e., up to 95% properly exposed become sensitized) While formaldehyde has a low potency (i.e., 1-5% properly exposed become sensitized) (Brooks, 1982; Newman-Taylor and Davies, 1981.).
MECHANISMS OF SENSITIZATION
Classic Sensitization
The B cell IgE mediates various immediate hypersensitivity states, including typical allergy to biological aeroallergens. IgE mediation is involved heavily in the pathogenesis of asthma (Hetzel and Clark, 1983; Turner-Warwick, 1978). Several chemicals, such as TDI, TMA, platinum salts, form protein bound haptens that act as antigens (Weill and Turner-Warwick, 1981; Brooks, 1982; Chan-Yeung, 1990; Newman-Taylor and Tee, 1990). Basically, IgE forms antibodies to certain antigens/allergens that are inhaled, absorbed, and/or ingested. This is the recognition (and sensitization) phenomena. Specific IgE then persists in the sensitized; it is an outcome that can be assayed. There are IgE receptor sites on basophils and mast cells. When an IgE antibody-antigen binding occurs, the IgE can attach to these receptors. This usually leads to degranulation of the cells and subsequent release of histamine, chemotactic factors for platelets and other active cells (e.g., eosinophils, neutrophils), and other active mediators (e.g., PG's/LTB). This starts a chain reaction (or cascade) of immunochemical phenomena which leads to the hypersensitivity response, and can lead to inflammation. (Ibid.) Anti-IgG can lead to basophil activation as well, as can the cells and some of the chemotactic factors (e.g., PAF) directly (Marone, 1989). (The mechanisms are discussed in more detail in the Immunology and Animal Models section.) IgE is genetically linked, as are other immunoglobulins (Turner-Warwick, 1978; Gregg, 1983; Lebowitz et al., 1984; McGue et al., 1989). IgE also has an amplification-control cycle that probably involves some subsets of IgG and secretory IgA. IgG itself is important in sensitization and hypersensitivity (e.g., in TMA hypersensitivity). IgE mediated activity is associated also with T-lymphocyte activity in the lung, as found from bronchoalveolar lavage (BAL) studies (Gerblich et al., 1991). There can also be IgG blocking antibody that somehow prevents IgE mediated reactions. (Secretory IgA is the most abundant immunoglobulin the lung, but its role in amplification-control of IgE is poorly understood. Turner-Warwick, 1978). Sensitization can occur in the lower respiratory tract, usually called hypersensitivity pneumonitis (or extrinsic allergic alveolitis), such as isocyanate disease. The nature of the protein-hapten deserves attention; T-and B-cells appear to have specific roles (Ibid.). Mediators (such as lymphokines) and receptors, and of other factors require further elucidation. Secretory IgA, along with IgG, and T-cells (especially suppressor/cytotoxic CD8 cells) are usually involved in hypersensitivity pneumonitis (Schlueter, 1982; Patterson et al., 1990, Semenzato, 1991). Some of the other known IgE-mediated chemical hypersensitivities are platinum and nickel salts, cobalt, other isocyanates (in addition to TDI), other anhydrides (in addition to TMA, such as phthalic acid anhydride), and organic acids (such as plicatic acid from red cedar) which are essentially all low molecular weight compounds which form protein haptens (Brooks, 1982; Marone, 1989; Allegra et al., 1989; Nemery, 1990, Chan-Yeung, 1990). Based on skin reactions, chromium salts, and colophony flux probably have IgE mediated immediate hyper-sensitivity. Based on this, and sensitization through ingestion, there are other chemicals, such as formaldehyde and ethylene oxide, which may turn out to have some IgE mediated hypersensitivity (Rockel et al., 1989). The various metals/metal salts appear to have IgG involvement also (as shown by precipitin formation), as do isocyanates and anhydrides (Adams et al, 1988; Thurmond and Dean, 1988; Patterson et al., 1990). Some of the haptens formed will produce late phase reactions as well, related to influxes of eosinophils, PMN's, inflammatory mediators (op cit.). Non-atopics will produce IgE
antibodies if antigen is administered with certain adjuvants (Marsh et al., 1972), though these responses are short-lived. This implies an IgE suppressive mechanism, which appears to be T cell mediated, and this suppression is probably due to CD8+ T cells (Diaz-Sanchez and Kemeny, 1990). Certain antigens can sensitize non-atopics as well (e.g., platinum salts, castor bean dust), which implies that they have IgE-potentiating properties. Such potentiation appears related to increased CD4+ T cells (Ibid.). Certain common chemicals (e.g., ozone) or mixes (e.g., diesel exhaust, including with HCHO) appear to be appropriate adjuvants (Meggs). Sometimes, elevated IgE and eosinophilia are associated with smoking (Burrows et al., 1989), and with acute and persistent respiratory disease, as in rubber workers involved in a thermoinjection process, where the suggested sensitization has not been related to one of the component chemicals (Bascom et al., 1990). Lymphocyte factors related to immunoglobulin activity also activate human basophils (Marone, 1989). One has to consider the triggering mechanism in predisposed individuals, such as specific triggers of inflammation (e.g., HCHO). Asthmatics and those with BHR, or those with such predispositions may be such susceptibles. It is claimed that those with autoimmune disorders (e.g., Lupus/SLE, rheumatoid arthritis-RA) may be predisposed also (Thrasher et al., 1989; Ashford). SLE and RA are associated with circulating immune complexes, which may play a role (discussed elsewhere) (Turner-Warwick, 1978). One also has to consider factors that affect interleukins which have such an impact in the chain of events after presentation with the antigen, including activation of basophils (e.g., by IL-3). IL-1 is a basic mediator of intercellular activity within and between immune system functions, including having a role in tissue homeostasis; IL-1 inhibitory activity (IHA) is reduced in interstitial lung disease (Nagai et al., 1991), and IL-1 is related to cell-mediated immunity as well.
Inflammation
Whatever the mechanism, inflammation (in airways, skin, possibly other organs) appears to be a key trait in many sensitization conditions (Reed, 1988; Bonini et al., 1989; Hogg et al., 1991). (Although it is discussed elsewhere as well, it is worth mentioning some major components of inflammation.) Epithelial release of mediators, including histamine and chemotactic factors, appears to be a major component of inflammation and a mechanism of importance in sensitization and subsequent triggering by chemicals. The increased activity of mononuclear cells (including PAM's), polymorphonuclear cells (including neutrophils) and eosinophils are part of the basic mechanisms involving inflammation. The release of oxidants, such as hydrogen peroxide (H202), as with exposure to formaldehyde, and superoxide union from these cells is a key contributor to damage and responses, occurs more in sensitized organisms, and can occur with immune complexes (Adams et al., 1988; Rossi et al., 1989; Allegra et al., 1989; Cerasoli et al., 1991). The lack of sufficient, effective antioxidants could be a mechanism of sensitivity (as in those with genetic defects, such as those who have little or no ceruloplasmin). It is interesting to note that only scarce airway inflammatory cell activity has been reported in at least some asthmatics between attacks (Saetta et al., 1989). Airway inflammation is probably the main cause of bronchial hyperreactivity by itself or by contributing to bronchial obstruction (although obstruction can occur by other mechanisms). The increase of airway or skin permeability, permitting processes that lead to mast cell or basophil degranulation, leads to mediator release. Mucosal edema, an aspect of inflammation, call increase epithelial
permeability, alter smooth muscle dynamics, and stimulate neural pathways (Bucca and Rolla, 1989). The occurrence of immune complexes in situ is important also (as discussed above); basophils can be activated by C3a and C5a (Marone, 1989). Macrophages are major cells in acute and chronic inflammation, immune responses including the complement system and immune deficiencies, are involved in hematopoesis and coagulation, and promote proliferation of many other cells. They present antigen to T cells. They release plasminogen activator and classes of toxins such as neutral proteases. Exposure to lymphokines is part of macrophage activation. Derived factors can activate basophils directly (Turner-Warwick, 1978; Adams et al., 1988; Thurmond and Dean, 1988).
Other Mechanisms of Sensitization
There are several hypothesized mechanisms, each with preliminary experimental verification of its potential role. Epithelial cells in the mucosa of bronchi and intestine can express Ia antigens. Ia-bearing epithelium can present antigens to specific T-cells, to initiate the classic immune response (Allegra et al., 1989). "It is possible that epithelial cells, like all antigen-presenting cells, act as accessory cells in immune responses by producing IL-1, which is responsible for T-cell migration and proliferation." (Ibid.) Also, HLA-DR antigens from airway epithelial cells are seen in those with interstitial lung disease (Ibid.). HLA-DR is part of the MHC (major histocompatibility) locus, and MHC-peptide linkages have been shown to be specific for some compounds (op tit.). Further, there may be epithelial cell-surface receptors with potential protein (i.e, hapten) specificity, probably involving IgG binding and myosin, which would induce intracellular changes (op tit; DeMarzo et al., 1989). Mast cells have receptors for a variety of antigens and histamine-releasing factors, in addition to anti-IgE and-IgG (Marone, 1989). It is unknown whether the "antigen" receptor would accept specific chemical-protein haptens. IgG, which has such high affinity for binding proteins, may play a role in this function. In rhinitis, and probably in asthma, there are specific chemical sensitive mast cells in the epithelium, such as formaldehyde-sensitive mast cells in the nasal epithelium, which are related to increased numbers of mast cell progenitors in airway mucosa (Otsuka et al., 1985, 1986, 1987; Barret and Metcalfe 1987). These epithelia can produce cytokines which promote mast cell growth in the presence of lymphocytes (Ibid.; Denburg et al., 1987). Local cells synthesized close to progenitors influences which type of mast cell develops, such that "derangements in the airway epithelium may result in a generation of factors which can induce a selective expansion of a mast cell sub-population.'' (Allegra et al., 1989). These mast cells, as found in bronchial secretions, BAL's and bronchial epithelium of asthmatics, are leaky and have the characteristics of the specific chemical-sensitive mast cells (as described above) (Allegra et al., 1989). The increased numbers of these specific mast cells can be activated in the airway, by provocation and possibly by other triggers, and cause bronchoconstriction only in those with these characteristics (Ibid.). The same exposure-epithelial mast cell response would be likely in the intestine as well, and possibly other locations where there are mast cells with their superficial location in epithelium. Epithelial cells are capable of activating mast cells also thru the release of specific mediators (e.g.,15 HETE) (Marone, 1989).
The possibility of neuro-sensitizers has been mentioned as a possibility, involving the adrenergic, cholinergic and/or NANC (neither) system(s). Mast cell-nerve interactions are biologically significant as reviewed recently (Bienenstock et al., 1991). Endogenous biochemical compounds are important in the regulation of smooth muscle tone, which is
more evident in sensitized individuals (such as asthmatics) (Barnes et al., 1984; Randem et al., 1987). The autonomic system appears to be involved as well (Nadel and Barnes, 1984; Postma et a1.,1985). The adrenergic system may be more important in this regard (Barnes et al., 1984). Central parasympathetic efferent signals are important for the circadian rhythm of bronchial tone (Dreher and Koller, 1990). The endogenous factors may only differentiate sensitized individuals; this requires further exploration. Although there is no knowledge of the specificity of muscarinic receptors (in the cholinergic system) for specific haptens, changes in the patterns of muscarinic receptors in sensitized individuals requires further exploration (Paggiaro et al., 1989). Neuropepities are important in asthma (Barnes, 1991). Threshold doses of PGD2 and takykinins (neuropeptides in the NANC system) to produce acetycholine induced bronchospasm (a cholinergic phenomena) were modified in animals presensitized (with ovalbumin) (Omini et al., 1989), implying a different set and time-course of reactions in sensitized organisms. Psychological conditioning may cause most cell degranulation in the rat (Bienenstock et al, 1991). Other ANS involvement, including that related to the limbic system, and hypothalmic responses is discussed elsewhere (C.Miller, I.Bell).
Different Pathways
The hypersensitive also may use a different pathway that essentially does not exist in the general population (as stated also by Burrell, and partly discussed above), which may be genetically determined. This may relate to inflammation directly, or to BHR (which appears to be, at least as far as entrainment, an independent genetic trait). (Figure 1) Asthmatics have a similar distribution, but different immunoreactivity of myosin heavy chains in bronchial smooth muscle compared to normal subjects (De Marzo et al., 1989).
Irritant Mechanisms
Pulmonary alveolar macrophages are engaged in response to irritants. They can release chemotactic factors (including PAF, ECT, NCT) and other mediators after stimulation by irritants (op cit.). Lysosomal enzymes are released, specifically after phagocytosis and cell lysis. Other mononuclear cells may play a role also. Irritants also stimulate PMN's (neutrophils), which release proteases. Eosinophils release MBP endotoxin, and all release free radicals (see above). These cells are the hallmarks of inflammation and the biochemicals released increase it, even when the stimulant is an irritant. Irritants can also produce endothelial cell damage which results in arachadonic acid metabolism (and circulation of prostaglan-dins, leukotrienes). Increased permeability leads to exposure of mast cells or basophils, which can then be activated to release other mediators (Thurmond and Dean, 1988). However, the type and level of mediator activity is less under irritation stimuli. Stimulation of C-fibers lead to release of Substance P and other neuropeptides (NKA, NKB), including vasoactive intestinal peptide (VIP); these are part of the NANC (nonadrenergic noncholinergic) system. Such stimulation may lead to epithelial release of mediators, and mast cell degranulation (Marone, 1989). Stimulation of cholinergic mechanisms (the vagal afferent pathway) may be important, though there is no indication that muscarinic receptors would have a "memory" for specific irritants (see above). Thus, chemical irritant contact may produce symptoms of disease that mimic
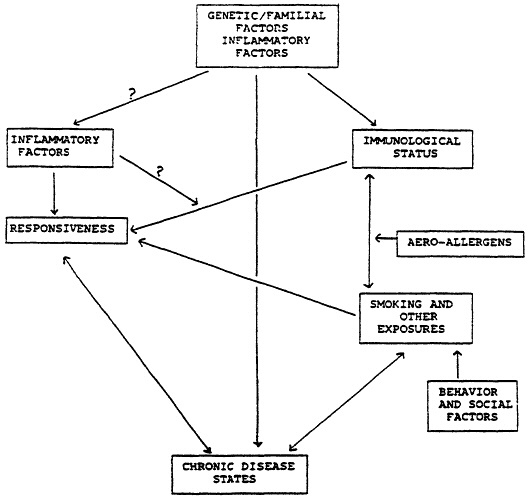
Figure 1 Hypothesized relationships.
immunological syndromes, but have a lack of antigen (Brooks, 1988; Thurmond and Dean, 1988). Protective responses, such as sneezing, coughing, and eye tearing, occur with stimulation of the epithelial irritant receptors. Irritants may even produce direct injury to epithelium. The "pseudoallery" or "reactive airway disease syndrome' usually requires exposure to relatively large amounts of the agent, usually occurs without a latency period after exposure (i.e., no "sensitization" period), and usually don't show reactivation of symptoms upon challenges with smaller amounts of the irritant or similar irritant (Ibid.). The irritant mechanisms last not much longer than due to the presence of the stimulus. Chronic inflammation can occur as well, leading to persistent symptoms, with high exposures to irritants; this may lead to reactive airway disease (Tarlo and Broder, 1989; Brooks, 1988). This form of chronic inflammation can result in increased susceptibility to other agents and to infection; implicated agents include chlorine, ozone, particles, SOx, NO2 (Gearhart and Schlesinger, 1986; Bylin et al., 1988; Wegman and Eisen, 1990). Examples of exposure
agents that may produce such disease include acids, chlorine, phosgene, paints, calcium oxide, sulfur dioxide (Tarlo and Broder, 1989). Irritant stimuli may lead to adaptation or tolerance as well; ozone, NO2, H2S, phosgene and calcium oxide are agents that lead to reduced/attenuated responses (Wegman and Eisen, 1990). Irritant responses must be differentiated also from odor responses (Cain et al., 1987). The mechanisms of such tolerance or adaptation are not known. Certain ''sensitizers" are pulmonary, dermal and sensory irritants as well, such as isocyanates, anhydrides, formaldehyde, ethylene diamine and paraphenylenediamine (Cain et al., 1987 ). Differentiation of responses is not always easy initially. After sensitization, stimulation can become non-specific as well, such that these and other irritants may produce similar responses. Even odors can produce non-specific responses in asthmatics (Shim and Williams, 1986).
FORMALDEHYDE
Formaldehyde is ubiquitous, especially in the indoor environment (Table 1). Formaldehyde is regarded as a upper respiratory irritant because it has a high solubility in water and is therefore captured by the wet mucus membrane. Retention of inhaled formaldehyde is shown to be almost 100% at the inhaled site in dogs (Egle, 1972). Malorney et al., (1965) showed that during an immediate following intravenous (IV) infusion of formaldehyde, the erythrocytes rapidly pick it up, leading to a very short-term increase in plama before the erythrocytes oxidize it to formic acid. This is partly (1/6th) secruted in urine; the rest is oxidized to carbon dioxide and water. Formaldehyde (HCHO) irritant effects. are presumed to be due to a nonspecific bronchial reflex response, which can increase flow resistance (Coffin and Stokinger, 1977). The effects of formaldehyde may be aggravated by interactions of other chemicals (Goldsmith and Friberg, 1977). The detection of the odor of formaldehyde in the air starts at about 0.05 ppm (0.06 rag/m3) (WHO/EURO, 1987) and is recognizable through most concentrations of exposure (Table 2). Exposure of a half hour to an hour is all that is needed to produce primary irritation; there is a dose response (Loomis, 1979: NRC 1981a). The lowest concentration causing pharyingitis is said to be 0.5 ppm (Stokinger and Coffin, 1968). Exposure to concentrations of formaldehyde gas of 0.1 ppm causes general irritation of the upper respiratory tract and other symptoms in children (Burdach and Wechselberg, 1980; NRC, 1981a). Concentrations from 1-5 ppm provoke coughing, constriction in the chest, and other symptoms (Loomis, 1979; NRC, 1981; Bardana, 1980; and Walker, 1964). Tolerance to eye and upper airway irritation may occur after I to 2 hours of exposure (NRC, 1981a; Blejer and Miller, 1966; Kerfoot and Mooney, 1975; Shipkovitz, 1968). However, even if tolerance develops, the irritation symptoms can return after I to 2 hour interruptions of exposure (Ibid; CPSC Ad Hoc Task Force, 1979). Ingestion usually leads injury to the larynx and trachea, gross effects on the GI tract, pneumonia, hardening of the lungs, hyperemia and edema of the lungs, and systemic damage. Increased mucolytic (decreased ciliary) activity has been shown related to formaldehyde exposure in animals ill various studies (Carson et al., 1966; Cralley, 1942; Dalhamn, 1956; Hoffmann and Wynder, 1977; Kensler and Battista, 1963; Kotin, 1966). Decreased ciliary activity reduces the host defense mechanism to chemicals and micro-organisms, and may lead to increased cell wall permeability. Acute respiratory infections have been associated with formaldehyde exposure in children (Burdach and Wechselberg, 1980; NRC, 1981a; Tuthill, 1984). Trinkler (1968) found inflammatory changes in bronchial tracts of subjects who used formalin solution as a histological fixative.
Orringer and Mattern (1976) and Rockel et al., (1989) saw an increase frequence of acute and chronic bronchitis in dialyzed patients. There was a reactive, eosinophilic component.
TABLE 1
|
Contribution of Various Atmospheric Environments to Average Exposure1 |
|
|
Source |
mg/d |
|
1. Air |
0.02 |
|
Ambient (10% Of time) |
|
|
Indoor |
|
|
Home (65% of time) |
|
|
Conventional |
0.5-2 |
|
Prefabricated (particle board) |
1-103 |
|
Workplace (25% of time) |
|
|
No occupational exposure2 |
0.2-0.8 |
|
With 1 mg/m3 occupational exposure |
4-5 |
|
ETS |
0.1-1 |
|
2. Smoking (20 cigarettes/d) |
|
|
|
1 |
|
1 Contributions of food and water is low, so they are ignored here. 2 Assuming the normal formaldehyde concentration is in conventional buildings 3 Currently unusual. Source: WHO/EURO 1987 Air Quality Guidelines for Europe (EURO Series #23) Copenhagen, 1987, pp 91-104. |
|
Potential Mechanisms of Formaldehyde Induced Respiratory Sensitivity
Formaldehyde possesses the capacity to bind and alter protein constituents, such as: amino acids, proteins, nucleic acids, nucleosides, and nucleoproteins (Morin et al, 1964; Semin et al. 1974.; Goh el al., 1979; Bardana, 1980). Therefore, it is quite possible that this reactive chemical combines with respiratory tract proteins to form an immunoreactive, hapten-protein complex Formaldehyde reacts directly with free amino groups, altering the
TABLE 2
|
Effects of Formaldehyde on Humans After Short-Term Exposure |
||
|
Concentration of formaldehyde mg/m3 |
|
|
|
Estimated median |
Reported range |
Effect |
|
0.1 |
0.06-1.2 |
Odor threshold in 50 % of people (including repeated exposure) |
|
0.5 |
0.1-1.9 |
Eye irritation threshold |
|
0.6 |
0.1-3.1 |
Throat irritation threshold |
|
3.1 |
2.5-3.7 |
Biting sensation in nose, eyes |
|
5.6 |
5-6.2 |
Tolerable for 30 rain (tearing) |
|
17.8 |
12-25 |
Strong flow of tears, lasting for 1 hr |
|
37.5 |
37-60 |
Danger to life, edema, inflammation, pneumonia |
|
125 |
60-125 |
Death |
|
Source: WHO/EURO 1987 Air Quality Guidelines for Europe (EURO Series #23) Copenhagen, 1987, pp 91-104. |
||
character of the proteins, which could make the chemically altered constituents antigenic in themselves or can result in the formation of macro-molecular formaldehyde protein complexes and thus produce hyper-sensitivity in some persons (Bardana, 1989; Loomis, 1979; Stokinger and Coffin, 1968). Supposedly, an IgE ISO PAST (Pharmacia, Sweden) was developed for formaldehyde and was positive for exposed dialysis patients (Rockel et al., 1989); others have evidently used formaldehyde conjugated to human serum albumen for immunoglobulin determinations (see below). Anti-N-like antibodies were found in dialysis patients using HCHO-treated cells (Sandler et al., 1979), which is evidence of type H auto-allergy (Consensus Workshop, 1984). Complement activation (via the alternate pathway) is another possible mechanism (Hakin et al, 1984; Rockel et al., 1989). C3a and C5a can activate basophils (Marone, 1989). More than one mechanism may be at work (Ibid.). Thrasher et al. (1990) report increased IL2 receptor cells and total B Cells (using monoclonal antibodies to LEU10) in those with long-term HCHO exposures (by report) and various presenting multiple organ system symptoms, compared to controls (with short-term exposure as chiropractic students. They also found increased titres (1:8 or greater) to B-cell
(IgG, IgM and IgE) isotypes to formaldehyde conjugated to human serum albumen; they indicate that these isotypes have proven positive in other investigations; increases in both IgG and IrE are hard to explain, and IgM increases haven't been found related to such immune disorders before (see above section, Turner-Warwick, 1978). Helper/Suppressor T-cell ratios don't appear different. They also reported increased titres (1:20) to auto antibodies (ASS, APC, ABB, AMIT and ANA). The meaning of significant increases in ASS only in mobile home residents, of APC and AMIT in variable percents in different groups, of ABB in office workers, and of ANA in both mobile home and office occupants are difficult to interpret. Increases in ANA hadn't been found in occupational asthma (Newman-Taylor and Davies, 1981; Newman-Taylor and Tee, 1990; op cit.). Other possible mechanisms have been discussed previously. It is quite likely that eosinophilia may be part of the immunological picture in formaldehyde-induced sensitivity (Popa et al., 1969; Hoy and Cestero, 1979; Nagornyi et al., 1979; Orringer and Mattern, 1976; Rockel et al., 1989). It has been shown that aldehyde pyrolysates from thermal degradation of polyethylene wrap is characterized by eosinophilia as well as by nocturnal attacks of dyspnea, wheezing and cough (Skerfving et al., 1980). There are an increasing number of studies which suggests formaldehyde can induce an inflammatory bronchitis reactivity airway disease. Particulates will carry formaldehyde gas (LaBelle et al., 1955; Patterson et al., 1990; Takhirov, 1974). Thus formaldehyde might have its affects in tracheobronchial or alveolar regions. Thus, particulates or aerosols have synergistic effects (Krzyzanowski et al., 1990), or the respiratory response is potentiated with simultaneous exposure to particulates as well as to other irritative chemicals (NRC, 1981b), or the aerosolized HCHO produces both sensitization and hyperresponsiveness (Hendrick and Lane, 1975, 1977; Frigas et al., 1982; Burge et al., 1985). Ostapovich (1975) studied sensitization of formaldehyde (2, 7, 15 mg/m3) in rats and guinea pigs injected with Freund's adjuvant prior to priming in order to stimulate antibody formation. Intermittent exposure, to concentration of 7 mg/m3 were given. Sensitization was induced by unequivocal increases in leukocyte agglutination, agar gel precipitation and passive hemaglutination, decreases in all but cholinesterase activity, from concentrations of 2 and 7 mg/m3. The time required for leukocytosis was shorter in rats than in guinea pigs. Zaeva (1968) summarized similar findings in animals. Nagornyi et al. (1979) found similar results, and eosinophilia, in guinea pigs with chronic exposures to 0.5 mg/m3. Sensitization was enhanced by intermittent inhalation. One mechanism of sensitization measured by leukocyte activity was formulated by Jerne, whose modified technique was utilized in this above study. This mechanism states that somatic generation of large number of genes expressed in leukocytes initially are directed against the animal's own histocompatibility antigens, which in the process of reacting with the cells that can form antibodies against them, cause these cells to mutate early, giving rise to many dories of antibody-forming cells with new and different specificities (Rose et al., 1973). Other in vitro and animal studies (Adams et al., 1988; Damiani, 1989) have shown that formaldehyde (like SO2 and nickel salts) increase macrophage phagocytosis and increase hydrogen peroxide levels. The latter promotes lipid metabolism, increases inflammation, and decreases host resistance; these studies have shown little direct effect of HCHO on B cells/immunoglobulins. Activated macrophages are associated with increased IL-1 and subsequent enhanced inflammatory processes (also by secretion of lyric enzymatic chemo attractants). (Increased IL-1 also stimulates T cell migration.) Phagolysomes degrade foreign molecules and resulting peptides associated with class I or H major histocompability complex (MHC) glycoproteins (which secondarily are presented as antigens to T cells (Damiani, 1989). Allegra et al. (1989) indicate that HCHO-sensitized most cells in the epithelium are associated with increased progenitors in airway mucosa and thus symptom severity. Mast
cell promotion and differentiation in vitro occur by eytokines produced by such individual's epitherlium (with or without the presence of lymphokines) (op cit.)
Evidence of. Asthma in Humans
The first case of formalin asthma was in an occupational setting and was published by Vaughan in 1939. Large numbers of European case studies reported on formaldehyde-induced asthma, claiming that an allergic mechanism was responsible, but immunological data is not presented usually (Vaughan, 1939; Coulant, 1961; Gervais, 1966; Tara, 1966; Nova and Touraine, 1956; Paliard et al., 1949; Sakula, 1975). Baur and Fruhmann (1979) showed an IgG increase in one case. In formaldehyde resin makers and other formaldehyde workers, formaldehyde, including formalin (40% formaldhyde by volume, an aqueous solution of formaldehyde) will produce primary irritational or allergic hypersensitivity. Further, formaldehyde reaction products, for example, dimethylurea, can be sensitizers (Milby et al., 1964). These and other studies (Hendrick and Lane, 1975, 1977; Popa et al., 1969; Kuzmenski et al., 1975; Kratochovil, 1971; Spassovski, 1976; led an NAS Committee (1981b) to conclude that formaldehyde has been shown to cause bronchial asthma in humans. The Committee stated that asthmatic attacks related to exposures to formaldehyde at low concentrations are due specifically to formaldehyde sensitization in some cases; controlled inhalation studies with formaldehyde are positive in these instances (Hendrick and Lane, 1975, 1977; Frigas et al, 1982; Burge et al., 1985). Nordman et al. (1985) found that only 12 of 230 persons with explicit suspicion of formaldehyde asthma actually reacted to formaldhyde gas bronchoprovocation tests. They had significant dose-response reactions between 0.4-2.5 mg/m3 HCHO. Of 218 who didn't, 71 reacted to histamine bronchoprovocation. Cockroft et al. (1982) reported two cases of occupational asthma caused by urea formaldehyde particle board, with positive histamine bronchial challenge tests, but no increased PAST specific IgE antibodies directed against formaldehyde human serum albumin conjugate. A third asthmatic didn't have a response with exposure, indicating (to them) a specific sensitization to the agent. Formaldehyde also seems to act as a direct airway irritant in persons who have bronchial asthmatic attacks from other causes (op cit.) Hakim et al. (1984) also reported acute asthma-like symptoms of chest tightness and dyspnea in dialysis patients when formaldehyde-treated membranes were used. The symptoms were correlated with complement activation (C3a). Asthma-like symptoms and bronchospasm also were reported with similar exposure by Rockel et al.(1989), associated with increased eosinophilia, IgE, elevations, and complement activation (C3a, C5a). HCHO conjugated specific IgE (PAST) elevations were found in some; other agents were found to have similar effects (ethylene oxide, phthalates, isocyanate). Airway Obstructive Disease due to formaldehyde has been shown in various studies also (Yefremov, 1970; Kerfoot and Mooney, 1975; Schoenberg and Mitchell, 1975; Kilburn et al, 1985; Krzyzanowski et al., 1990). Most recently, a community population study (Krzyzanowski et al., 1990) showed higher rates of asthma and chronic bronchitis in children related to monitored formaldehyde exposure, with a synergistic contribution of ETS. Formaldehyde-induced pesistent bronchial responsiventeness by itself occurred primarily in the lower SES group (Quackenboss et al., 1989a). The monitoring used a modified LBL method with no temperature or humidity interferences (Quackenboss et al., 1989b). They also found acute changes in children, especially asthmatics, who were specifically exposed overnight in their bedrooms; morning PEF had decreased significantly with a demonstrable exposure-response relationship.
Skin Sensitization
Skin contact with formaldehyde has been reported to cause a variety of acutaneoud problems in humans, including irritation, allergic contact dermatitis (Type I allergy), and urticaria (Type IV allergy) (Odom and Maibach, 1977; Roth, 1969; Sneddon, 1968; NRC, 1981a; Consenses Workshop, 1984). Dermal exposure to formaldehyde may cause an acute and inflammatory reaction of the affected region concentrations of 1 to 3 ppm over minutes to hours produces irritant effects which may be tolerated by normal adults, but produces noticeable skin irritation from direct exposure (Pirila and Kilipo, 1949; Roth, 1969).
Current Levels of Knowledge
It is commonly accepted there is reasonable good knowledge of exposure assessment for formaldehyde, including: sources, monitoring instrumentation, and distribution of exposures (WHO/EURO, 1986, 1987, 1991) as represented by Tables 1, 3 and 4. The suggestions of the Consensus Workshop (1984) for future research are pertinent still. Knowledge levels are reasonable for certain effects: odor, sensory and mucosal irritation. More knowledge is needed about airway effects, including sensitization (Ibid.) and neurological effects (Kilburn et al., 1985). It is estimated that between I and 6% of individuals in the US are probably sensitive to low concentrations of formaldehyde (Moschandreas et al., 1980). Means of control have included technical and regulatory procedures (WHO/EURO, 1991); educational control methods have been recommended also.
TABLE 3
|
Formaldehyde Sources With Usual Range of Related Concentrations Indoors |
|
|
Primary: Particle/Press Board, |
0.1-2.0 rag/m3 |
|
Plywood (resin), Fabric |
|
|
Secondary: ETS |
5-50 Mg/ m3 |
|
Wood Burning |
|
|
Infiltration: Vehicles in garages |
? |
|
From outdoors |
?* |
|
* Expected to increase with oxygenated fuels, especially methanol. |
|
TABLE 4
|
Concentrations, Distribution of Exposures and Exposure-Response Relationships |
||||
|
Concentration |
People with exposure |
Levels of concern |
||
|
(mg/m3 |
|
|
|
|
|
Range (mg/m3 |
Low |
High |
Limited |
Definite |
|
|
most |
few |
< 0.03 |
0.10 |
|
0-20-0.1-2.0 |
|
|
|
|
|
Source: WHO/EURO 1991 (in press) The European Short-Term Air Quality Guideline (WHO/EURO, 1987) |
||||
References
Adams, D. O., J. G. Lewis, and J. H. Dean. 1988. Activation of mononuclear phagocytes by xenobiotics of environmental concern: analysis and host effects. Pp. 351-369 in Toxicology of the Lung, D. E. Gardner, J. D. Crapo, and E. J. Massaro, eds. New York: Raven Press.
Allegra, L., L. M. Fabbri, G. Picotti, and S. Mattoli. 1989. Bronchial epithelium and asthma. Eur Respir J 2(6 Suppl.):460S-468S.
Bard aria, Jr., E. J. 1980. Formaldehyde: hypersensitivity and irritant reactions at work and in the home. Immunol and Allergy Practice 2(3):11-23.
Barnes, P. J. 1991. Neuropeptides and asthma. Am Rev Respir Dis 143:828-S32.
Barnes, P. J., G. A. Fitzgerald, M. Brown, and C. T. Dollery. 1984. Nocturnal asthma and changes in circulating epinephrine, histamine and cortisol. N Engl J Med 303:263-267.
Barrett, K E., and D. D. Metcalfe. 1987. Heterogeneity of mast cells in the tissues of the respiratory tract and other organ systems. Am Rev Respir Dis 135:1190-1195.
Baseore, R., M. E. Baser, R. J. Thomas, J. F. Fisher, W. N. Yang, and J. H. Baker. 1990. Elevated serum IgE, eosinophilia, and lung function in rubber workers. 45(1):15.
Baur, X., and G. Fruhmann. 1979. Bronchial asthma of allergic or irritative origin as an occupational disease. Prax Klin Pneumol 1(Suppl. 33):317-22.
Bienenstock, J., G. Macqueen, P. Sestini, J. S. Marshall, R. H. Stead, and M. H. Perdue. 1991. Inflammatory cell mechanisms. Mast cell/nerve interactions in vitro and in vivo. Am Rev Respir Dis 143:S55-S58.
Blejer, H. P., and B. H. Miller. 1966. Occupational health report of formaldehyde concentrations and effects on workers at the Bayly Manufacturing Co, Visalia. Pp. 6 in Study Report No. S-1806. Los Angeles: State of California Health and Welfare Agency, Dept. of Public Health, Bureau of Occupational Health.
Bonini, Se, E. Adriani St. Bonini, G. De Petrillo, G. G. Loggi, A. Adabbo, M. Pani and F. Balsano. 1989. Allergy and asthma: distinguishing the causes from the triggers. An eye model for the study of inflammatory events following allergen challenge. Eur Respir J 2(6 Suppl):497S-501S.
Brooks, S.M. 1982. The evaluation of occupational airways disease in the laboratory and workplace. J Allergy Clin Immunol 70(1):56-66.
Bucca, C., and G. Rolla. 1989. Mucosal oedema and airway hyperreactivity. Eur Respir J 2(6 Suppl.):520S-522S.
Burdach, S. and K. Weschselberg. 1980. Damage to health at school. Complaints due to the use of materials which emit formaldehyde in school buildings [German]. Forstschritte
der Medizin 98(11):379-384.
Barge, P. S., M. G. Harries, W. K. Lain, I. M. O'Brien, and P. A. Patchet. 1985. Occupational asthma due to formaldehyde. Thorax 40:255-260.
Burge, P. S., I. M. O'Brien, and M. G. Haries. 1979. Peak flow records in the diagnosis of occupational asthma due to colophony. Thorax 34:308-312.
Burrows, B., F. D. Martinez, M. Halonen, R. A. Barbee, and M. G. Cline. 1989. Association of asthma with serum IgE levels and skin-test reactivity to allergens. New Eng J Med 320:271-277.
Bylin, G., G. Hedenstierna, T. Lindvall, and B. Sundin. 1988. Ambient nitrogen dioxide concentrations increase bronchial responsiveness in subjects with mild asthma. Eur Respir J 1:606-612.
Cain, W. S., T. Tosun, L. C. See, and B. Leaderer. 1987. Environmental tobacco smoke: sensory reactions of occupants. Atmospheric Environment 21(2):347-353.
Carson, S., R. Goldhamer, and R. Carpenter. 1966. Mucus transport in the respiratory tract. Am Rev Respir Dis 93 (Supl):86-92.
Cerasoli, Jr., F., J. Tocker, and W. M. Selig. 1991. Airway eosinophils from actively sensitized guinea pigs exhibit enhanced superoxide anion release in response to antigen challenge. Am J Respir Cell Mol Biol 4:355-363.
Chan-Yeung M. 1990. Occupational asthma. Chest 98(5 Suppl):148S-161S.
Coffin; D. L., and H. E. Stokinger. 1977. Biological effects of air pollutants. Pp. 231-360 in Air Pollution. Vol. II The Effects of Air Pollution, A. C. Stern, ed. New York: Academic Press, Inc.
Consensus Workshop. 1984. Report on the Consensus Workshop on Formaldehyde. Environ Health Perspect 58:323-381.
Coulant, M. P., and G. Lopes. 1961. L'allergie au formal et a ses derives. Archives des maladies professionelles, de medicine du travail et de securite sociale (Paris) 22:769.
Cralley, L. B. 1942. The effect of irritant gases upon the rate of ciliary activity. J Ind Hyg Tox 24:193-198.
Cockcroft, D. W., V. H. Hoeppner, and J. Dolovich. 1982. Occupational asthma caused by cedar urea formaldehyde particle board. Chest 1:49-53.
CPSC Ad Hoc Task Force. 1979. Formaldehyde. Washington, D.C.
Dalhamn T. 1956. Mucous flow and ciliary activity in the trachea of healthy rats and rats exposed to respiratory irritant gases. Acta Physiologica Scandinavica 36(Suppl. 123):5-161.
Damiani G. 1989. The role of macrophages and lymphocytes in bronchial asthma:
perspectives for new research models. Eur Respir J 6 (Suppl.):469S-472S.
Davies, R. J., and A. D. Blainey. 1983. Occupational Asthma. Pp. 205-213; 228-230 in Asthma, T. J. H. Clark and S. Godfred, eels. London: Chapman and Hall.
De Marzo, N., P. Di Blasi, P. Boschetto, C. E. Mapp, S. Sartore, G. Picotti, and L. M. Fabbri. 1989. Airway smooth muscle biochemistry and asthma. Eur Respir J 2(5 Suppl.):473S-476S.
Denburg, J. A., M. Ohnisi J. Ruhno, J. Bienenstock, and J. Dolovich. 1987. The effects of basophil/mast and eosinophil cell colony stimulating activities derived from human nasal polyp epithelial scrapings are T cell-dependent. J Allergy Clin Immunol 79(Suppl.), 167A.
Diaz-Sanchez, D., and D. M. Kemeny. 1990. The sensitivity of rat CD8+ and CD4+ T cells to ricin in vivo and in vitro and their relationship to IgE regulation. Immunology 69:71-77.
Dreher, D., and E. A. Koller. 1990. Circadian rhythms of specific airway conductance and bronchial reactivity to histamine: the effects of parasympathetic blockade. Eur Respir J 3:414-420.
Egle, J. L. 1972. Retention of inhaled formaldehyde, propionaldehyde and acreolein in the lung. Arch Environ Health 25:119-124.
EPA. August 1986. Evaluation of Integrated Health Effects Data for Ozone and Photochemical Oxidants. Chapter 13 in Air Quality Criteria Document for Ozone and Other Photochemical Oxidants, EPA-600/8-84-020eF, Washington, D.C.
Frigas, E., W. V. Filley, and C. E. Reed. 1982. UFFI dust. Non-specific irritant only? Cheat 82:511-512.
Gearhart, J. M. and R. B. Schlesinger. 1986. Sulfuric acid-reduced airway hyper-responsiveness. Fundamental and Applied Toxicology 7:681-689.
Gerblich, A. A., H. Salik, and M. R. Schuyler. 1991. Dynamic T-cell changes in peripheral blood and bronchoalveolar lavage after antigen bronchoprovocation in asthmatics. Am Rev Respir Dis 143:533-537.
Gervais, P. 1966. L'asthme professionel dans l'industrie des matieres plastiques. Poumon et le Coeur 22:2199.
Goh, K., and R. V. M. Cestero. 1979. Chromosomal abnormalities in maintenance hemodialysis patients. 1 Med 10:16/7.
Goldsmith, I. R., and L. T. Friberg. 1977. Effects of air pollution on human health. Pp. 457-610 in Air Pollution. Vol. II The Effects of Air Pollution, A. C. Stern, ed. New York: Academic 59 Press, Inc.
Gregg, I. 1983. Epidemiological Aspects. Pp. 242-294 in Asthma, T. J. Clarke and S. Godfrey, eds., 2nd ed., London: Chapman and Hall.
Hakin, R. M., J. Breillatt, J. M. Lazarus, and F. K. Port. 1984. Complement activation and hypersensitivity reactions to dialysis membranes. New Eng J Med 311(14):878-882.
Hetzel, M. R., and T. J. H. Clark 1983. Adult Asthma. Pp. 473-481 in Asthma, T. J. H. Clark and S. Godfred eds. London: Chapman and Hall .
Hendrick, D. J., and D. J. Lane. 1975. Formalin asthma in hospital staff. Brit Med J 1:607-608. 60.
Henririck, D. J., and D. J. Lane. 1977. Occupational formalin asthma. Brit J Ind Med 34:11-18.
Hoffmann, D. J., and E. L. Wynder. 1977. Organic particulate pollutants-chemical analysis and bioassays for carcinogenicity. Pp. 361-455 in Air Pollution. Vol. H The Effects of Air Pollution, A. C. Stern, ed. New York: Academic Press, Inc.
Hogg, J. C., A. L. James, and P. D. Pare. 1991. Evidence for inflammation in asthma. Am Rev Respir Dis 143:S39-S42.
Hoy, W. E., and R. V. M. Cestero. 1979. Eosinophilia in maintenance hemodialysis patients. J Dialysis 3(1):73-79.
Kenslet, C. J., and S. P. Battista. 1963. Components of cigarette smoke with ciliary-depressant activity. New Fag J Med 269:1161-61 1166.
Kerfoot, E. J., and T. F. Mooney. 1975. Formaldehyde and paraformaldehyde study in funeral homes. Am lad Hyg Assoc J 36:533-537.
Kilburn, K. H., R. Warshaw, C. T. Boylen, S. J. S. Johnson, B. Seidman, R. Sinclair, and T. Takaro. 1985. Pulmonary and neurobehavioral effects of formaldehyde exposure. Arch Environ Health 40:254-260.
Kotin, P. 1966. (need title) Can Cancer Conf 6:475. Kratochvil, I. 1971. Effects of formaldehyde on the health of workers in the crease-resistant clothing industry. Pracovni Lekarstvi 23(1):374-375.
Krzyzanowski, M., J. J. Ouackenboss, and M. D. Lebowitz. 1990. Chronic respiratory effects of indoor formaldehyde exposure. Environ Res 52:117-125.
Kuz'menko, N. M., et al. 1975. Study of the sensitizing action of formaldehyde under the conditions of plastic manufacture. Vrachebnoe Del 6:131-134.
LaBelle, C. W., J. E. Long, and E. E. Christofano. 1955. Synergistic effects of aerosols. AMA Arch Ind Health 11:297-304.
Lebowitz, M.D., R. Barbee, and B. Burrows. 1984. Family concordance of IgE, atopy and disease. J Allergy Clin Immunol 73:259-266.
Loomis, T. A. 1979. Formaldehyde toxicity. Arch Pathol Lab Med 103:321-324.
Malorney, R., N. Reitbrock, and M. Schneider. 1965. Die oxydation des formaldehyds zu ameisensaure im blur, ein beitrag zum stoffwechsel des formaldehyds. Naunyn-Schmiedebergs Arch Pharmacol 250:419-436.
Marone, G. 1989. The role of mast cell and basophil activation in human allergic reactions. Eur Respir J 2(6 Suppl.):446S-455S.
Marsh, D. G., L. M. Lichtenstein, and P.S. Norman. 1972. Induction of IgE-mediated immediate hypersensitivity to group 1 rye grass pollen allergen and allergoids in non-allergic man. Immunology 22:1013.
McGue, M., J. W. Gerrand, M. D. Lebowitz, and D. C. Rao. 1989. Commingling in the distributions of immunologulin levels. Human Heredity 39(4):196-201.
Milby, T. H., M. M. Key, R. L. Gibson, et al. 1964. Chemical Hazards. Pp. 63-242 in Occupational Diseases, WM. M. Gafafer, ed. U.S. Department of Health, Education, and Welfare, Publication No. 1097, Washington, D.C., U.S. Government Printing Office.
Morin, N. R., P. E. Zeldin, Z. O. Kubinski, P. K Bhattacharya, and H. Kubinski. 1977. Macromolecular complexes produced by chemical carcinogens and ultra-violet radiation. Cancer Res 37:3802-3814.
Moschandreas, D. J., R. H. Moyer, J. R. Ward, H. E. Rector, J. T. Schakenback, et al. 1980. An evaluation of formaldehyde problems in residential mobile homes. Pp. 1-146. Final Task Report I, GEOMET Report No.. ESF-797, April, for the Department of Housing and Urban Development. Washington, D.C.: EPA. 65
Nadel, J. A., and P. J. Barnes. 1984. Autonomic regulation of the airways . Am Rev Med 35:451-467. Nagai, S., H. Aung, M. Takeuchi, K. Kusume, and T. Izumi. 1991. IL-1 and IL-1 inhibitory activity in the culture supernatants of alveolar macrophages from patients with interstitial lung diseases. Chest 99:674-680.
Nagornyi, P. A, Z. A. Sudakova, and S. M. Schablenko. 1979. General toxic and allergenic action of formaldehyde. Gigiena Truda i professional'nye Zabolevaniya 1:27-30. National Research Council (NRC). 1981a. Formaldehyde and other aldehydes. National Academy Press, Washington, D.C.
National Research Council (NRC). 1981b. Indoor Pollutions. National Academy Press, Washington, D.C. Nemery, B. 1990. Metal toxicity and. the respiratory tract. Eur Respir J 3:202-219. Newman-Taylor, A. J., and R. J. Davies. 1981. Inhalation Challenge Testing. Pp. 143-168, in Occupational Lung Diseases, H. Weill and M. Turner-Warwick, eds. New York: Marcel Dekker. Newman-Taylor, A., and R. D. Tee. 1990. Environmental and occupational asthma. Exposure assessment. Chest 98(5 Suppl.):209S-215S.
Nordman, H., H. Keskinen, and M. Tuppurainen 1985. Formaldehyde asthma-rare or
overlooked? J Allergy Clin Immunol 75:81-99. Nova, M. M. H., and R. G. Touraine. 1956. Asthme au formol, Pp. 293-294 in Societe De Medecine Du Travail, Lyon.
Odom, R. G., and H. I. Maiback. 1977. 1977. Contact urticaria: a efferent contact dermatitis. Pp. 441-452 in Advances in Modern Toxicology, Vol. 4, Dermatotoxicology and Pharmacology, F. N. Marzulli and H. I. Maibach, eds. Washington, DC: Hemisphere Publishing.
Omini, C., G. Brunelli, A. Hernandez, and L. Daffonchio. 1989. Role of the mediators in pulmonary hyperreactivity: the cocktail interaction hypothesis. Eur Respir J 2(6 Suppl.):493S-496S. Orringer, E. P., and W. D. Mattern. 1976. Formaldehyde-induced hemolysis during chronic hemodialysis. New Eng J Med 294(26):1416-1420.
Ostapovich, I. K. 1975. Conditions of respiratory route exposure to sulfur dioxide and formaldehyde and subsequent sensitization. Gigyena I Sanitariya 2:9-13.
Otsuka, H., J. A. Denburg and J. Dolovich. 1985. Heterogeneity of metachromatic cells in human nose: significance of mucosal mast cells. J Allergy Clin Immunol 76:695-702.
Otsuka, K., J. Dolovich, A. D. Befus, et al. 1986. Basophilic cell progenitors, nasal metachromatic cells and peripheral blood basophils in ragweed allergic rhinitis. J Allergy Clin Immunol 78:365-371.
Otsuka, H., J. Dolovich, M. Richardson, J. Bienenstock, and J. A. Denburg. 1987. Metachromatic cell progenitors and specific growth and differentiation factors in human nasal mucosa and polyps. Am Rev Respir Dis 136:710-717.
Paggiaro, P. L., E. Bacci, N. Pulera, P. Bernard, F. L. Dente, D. Talini, and C. Giuntini. 1989. Vagal reflexes and asthma. Eur Respir J 2(6 Suppl.):502S-507S.
Patterson, R., L. C. Grammer, C. R. Zeiss, K. E. Harris, and M. A. Shaughnessy. 1990. Use of immunologic technology in the diagnosis of environmental and occupational immunologic lung disease. Chest 98(5 Suppl.):206S-208S.
Paliard, F., L. Roche, C. Exbrayat, and E. Sprunk. 1949. Chronic asthma due to formaldehyde. Arch Mal Prof 10:528.
Pirila, V., and O.Kilpio. 1949. On dermatitis caused by formaldehyde 70 and its compounds. Am Med Intern Fenn 38:38-51.
Popa, V., D. Teculescu, D. Stanescu, and N. Gavrilescu. 1969. Bronchial asthma and asthmatic bronchitis determined by simple chemicals. Dis Chest 56(5):395-404.
Postma, D. S., J. J. Keyzer, G. H. Koeter, H. J. Sluiter, and K. DeVries. 1985. Influence of the parasympathetic and sympathetic nervous system on nocturnal bronchial obstruction. Clin Sci 69:251-258.
Ouackenboss, J. J., M. D. Lebowitz, C. Hayes, and C. L. Young. 1989. Respiratory
responses to indoor/outdoor air pollutants: combustion products, formaldehyde, and particulate matter. Pp. 280-293 in Combustion Processes and the Quality of the Indoor Air Environment, J. Harper, ed. Pittsburg: Air Pollution Control Association.
Ouackenboss, J. J., M. D. Lebowitz, and C. Hayes. 1989a. Epidemiological study of respiratory response to indoor/outdoor air quality. Environ Int 15:493-502.
Ramsdale, E. H., M. M. Morris, R. S. Roberts, and F. E. Hargreave. 1985. Asymptomatic bronchial hyperresponsiveness in rhinitis. J. Allergy Clin. Immunol. 95(5):573-577.
Randera, B., M. H. Smolensky, B. Hsi, D. Albright, and S. Burge. 1987. Field survey of circadian rhythm in PEF of electronics workers suffering from colophony-induced asthma. Chronobiology International 4(2):263-271.
Reed, C. E. 1988. Basic mechanisms of asthma. Role of inflammation. Chest 94(1):175-177 Rockel, A., B. Klinke, J. Hertel, X. Baur, C. Thiel, S. Abdelhamid, P. Fiegel, and D. Walb. 1989. Allergy to dialysis materials. Nephrol Dial Transplant 4:646-652.
Rose, N. R., F. Milgram, and C. J. van Oss. 1973. Principles of Immunology. New York: MacMillan Publishing Co., Inc.
Rossi, G. A., O. Sacco, F. Vassallo, S. Lantero, A. Morelli, U. Benatti, and G. Damiani. 1989. Oxidative metabolism of human peripheral blood eosinophils and neutrophils: H2O production after stimulation with phorbol myristate acetate and immune complexes. Eur Respir J 2(6 Suppl.):435S-440S.
Roth, W. G. 1969. Tylotic palmer and planter eczema caused by steam ironing clothes containing formaldehyde. Berufs-Dermatosen 17:263-267.
Saetta, M., L. M. Fabbri, D. Danieli, G. Picotti, and L. Allegra. 1989. Pathology of bronchial asthma and animal models of asthma. Eur Respir J 2(6 Suppl.):477S-482S.
Sakula, A. 1975. Formalin asthma in hospital laboratory staff. Lancet 2:816.
Santiler, S. G., R. Sharon, M. Bush, M. Stroup, and B. Sabo. 1979. Formaldehyde-related antibodies in hemodialysis patients. Transfusion 19(6):682-687.
Semenzato, G. 1991. Immunology of interstitial lung diseases: cellular events taking place in the lung of sarcoidosis, hypersensitivity pneumonitis and HIV infection. Bur Respir J 4:94-102.
Semin, Y.A., E. N. Kolomyitseva, and A. M. Poverennyi. 1974. Effects of products of the reaction of formaldehyde and amino acids on nucleotides and DNA. Molekulyarnaya Biologiya 8:276.
Schlueter, D.P. 1982. Infiltrative lung disease hypersensitivity pneumonitis. J Allergy Clin Immunol 70(1):50-55.
Schoenberg, J. B., and C. A. Mitchell. 1975. Airway disease caused[ by phenolic (Phenol-formaldehyde) resin exposure. Arch Environ Health 30:574-577.
Shim, C., M. H. Williams, Jr. 1986. Effect of odors in asthma. Amer J Med 80:18-22.
Shipkovitz, H. D. 1968. Formaldehyde vapor emissions in the permanent-press fabrics industry. Report No. TR-52. U.S. Public Health Service, Cincinnati, Ohio.
Skerfving, S., B. Akesson, and B. G. Simonsson. 1980. ''Meatwrappers' asthma'' caused by thermal degradation products of polythylene. Lancet 1:211.
Sneddon, I. B. 1968. Dermatitis in an intermittent haemodialysis unit. Brit Meat J 1:183-184.
Spassovski, M. 1976. Health hazards in the production and processing of some fibers, resins, and plastics in Bulgaria. Environ Health Perspect 17:199-202.
Stokinger, H. E., and D. L. Coffin. 1968. A formaldehyde and its homologs, Pp. 483484 in Volume I Air Pollution, A. C. Stern, ed. New York: Academic Press.
Tara, M. S. 1966. Intolerance? Sensitization? Formaldehyde-induced pulmonary allergy? Arch Mal 17:66-68.
Tarlo, S. M., and I. Broder. 1989. Irritant-induced occupational asthma. Chest 96(2):297-300.
Tathirov, M. T. 1974. Experimental study of the combined effect of six atmospheric pollutants on the human body. Gig Sanit 5:100-102.
Thrasher, J. D., R. Madison, A. Broughthon, and Z. Gard. 1989. Building-related illness and antibodies to albumin conjugates of formaldehyde, toluene diisocyanate and trimellitic anhydride. Am J Ind Med 15:187-195.
Thrasher, J. D., A. Broughton, and R. Madison. 1990. Immune activation and auto-antibodies in humans with long-term inhalation exposure to formaldehyde. Archives Environ Hlth 45(4):217-223.
Thurmond, L. M., and J. H. Dean. 1988. Immunological responses following inhalation exposure to chemical hazards. Pp. 375-392 in Toxicology of the Lung, D. E. Gardner, J. D. Crapo, and E. J. Massaro, eds. New York: Raven Press.
Trinkler, H. 1968. Working with formaldehyde. Medizinische Laboratorium (Stuttgart) 21:283. Turner-Warwick, M., ed. 1978. Immunology of the Lung. London: E. Arnold (Publishers) Ltd.
Tuthill, R. W. 1984. Woodstoves, formaldehyde, and respiratory disease. Amer J Epidemiol 120(6):952-955.
Vaughn, W. T. 1939. The Practice of Allergy. P. 677. St. Louis: C. V. Mosby. Walker, J. F. 1964. Formaldehyde. Pp. 484. New York: Rinehold Publishing Corp.
Weber, A. 1984. Acute effects of environmental tobacco smoke. Eur J Resp Dis 68(Suppl. 133):98-108.
Wegman, D. H., and E. A. Eisen. 1990. Acute irritants. More than a nuisance. Chest 97(4):773-775.
Weill, H., and M. Turner-Warwick, eds. 1981. Occupational Lung Diseases: Research Approaches and Methods. New York: Marcel Dekker, Inc.
WHO/EURO. 1986. Indoor Air Quality: Radon and Formaldehyde. World Health Organization Regional Office for Europe, Environ Health
WHO/EURO. 1987. Air Quality Guidelines. World Health Organization Regional Office for Europe, Vol. 2, Chap. 5, Copenhagen/Geneva.
WHO/EURO. 1991. Indoor Air Quality:. Combustion Products, Copenhagen, in press. Yefremov, G. G. 1970. The state of the upper respiratory tract in formaldehyde production employees. Zh Ushn Nos Gorl Bolezn 30:11-15.
Zaeva, G. N., et al. 1968. Materials for the revision of the maximum 80 permissible concentration of formaldehyde in the interior atmosphere of industrial premises. Gigiena Truda Professional'nye Zab 12(7):16-20.






















