6
Exposure Assessment
Assessment of individual exposure to herbicides and 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) or other chemical compounds found in the herbicides used in Vietnam is a key element in determining whether specific health outcomes are associated with exposure to these compounds. For a variety of reasons, the committee placed particularly heavy emphasis on exposure assessment issues.
First, the committee's primary task is to review and evaluate the scientific literature to determine, if possible, whether there is a statistical association between various health effects and herbicide use, taking into consideration the strength of the scientific evidence and the appropriateness of the methods used to detect the association. Estimation of health risks associated with herbicide exposure consists of two primary activities: (1) exposure assessment and (2) assessment of the health effects in exposed individuals. The committee has found in its review that the weakest methodologic aspects complicating the interpretation of the available epidemiologic studies are the definition and quantification of exposure. This chapter describes the criteria used by the committee in assessing the quality and validity of exposure measures. It begins with a description of the role of exposure assessment in epidemiology, followed by a discussion and evaluation of the various approaches that have been used to measure exposure in studies of those occupationally and environmentally exposed to herbicides and TCDD. The next section describes exposure assessment in studies of Vietnam veterans and some of the problems of inaccurate exposure measurement in these studies.
Second, an additional task brought to this committee through its legislative charge was the determination of whether additional studies of Vietnam veterans are feasible. Drawing upon the committee's evaluation of the available literature and upon information on the military use of herbicides (see Chapter 3), this chapter summarizes what is known about exposure to herbicides in Vietnam in comparison to other populations with widely different types of exposure (e.g., in factories, of professional herbicide sprayers, from environmental accidents). Valid measures of exposure are critical to further epidemiologic studies, and this chapter proposes a method for developing such a measure for future studies of Vietnam veterans.
A third and related reason for the committee's concern about exposure assessment in epidemiologic studies is that these data are needed to draw inferences on the health effects of exposure in Vietnam veterans from studies of those occupationally and environmentally exposed. Studies of these other groups address the issue of whether herbicides are associated with particular health outcomes, but they have only an indirect bearing on the question of associations in veterans themselves. Exposure data in all groups are needed to translate the results of occupational and environmental studies to estimates of increased risk for Vietnam veterans.
AN OVERVIEW OF EXPOSURE ASSESSMENT FOR EPIDEMIOLOGY
When epidemiologists assess the potential health risks of exposure to a toxic chemical, they compare the disease experience of groups of people with different levels of exposure to the substance of interest. Accurate risk estimates depend on the ability to accurately identify those who are "exposed" and those who are not. When the concern is with low-level, possibly intermittent exposure to a chemical such as an herbicide, it becomes important not simply to assess exposure as its presence or absence, but to characterize the degree of exposure—its intensity and duration. At root there are three essential steps in epidemiology:
-
assembling a cohort of people with similar, well-defined exposures to some agent, and another cohort identical to the first but with members who lack exposure to the agent;
-
measuring and then comparing the disease experience of each of these cohorts; and,
-
drawing inferences from these comparisons about the risks that may derive from the exposure differences between cohorts.
Exposure assessment contributes to the epidemiologic study process in several ways. First, accurate measures of exposure are essential to a study's validity because if there is not a well-defined contrast in exposure among
the two (or more) groups being studied, then the results cannot be considered meaningful with respect to a judgment about the potential risk to an exposed individual or group. Second, very large groups must be studied in order to identify small risks, and conversely a relatively small study may be able to detect the effect of heavy or sustained exposure to a toxic substance. In this way, a study's precision is also linked to the extent of the exposure involved and the accuracy of its measurement.
The strength of an association between an exposure and a disease is just one of the criteria used in evaluating epidemiologic evidence, as described in Chapter 5. Another criterion often used in evaluating an association is whether or not there is evidence that as exposure increases, the risk of the disease also increases (Hill, 1971). This dose-response pattern can be detected only if there is some way to determine the degree of exposure among different cohorts or subcohorts of the study. Inaccurate assessment of exposure can obscure the existence of such a trend and thus make it less likely that a true risk will be identified as such. A recent review, for instance, has found evidence that studies of putative occupational carcinogens are more likely to identify these agents successfully when quantitative exposure data are used, instead of relying on qualitative categories or simply the duration of exposure (Blair and Stewart, 1992).
Once a given exposure-disease association has been established, it is often desirable to consider the implications of this risk for some exposed population other than the population with which the study was performed. In making this inference, it is critical to have accurate exposure assessments. If there is a risk of a certain disease in workers occupationally exposed to a herbicide, what would the risk be for a Vietnam veteran who was exposed only occasionally or for just a short period? The proper scale on which to compare these risks is the scale of quantitative exposure.
It is useful to view exposure to an environmental agent as a process involving a progression of events that links the chemical in the environment to the ultimate "target" tissue in the human body (Figure 6-1). It is particularly important to distinguish "exposure" from "dose"—words that are often used interchangeably but have quite different meanings. Exposure is the concentration of an agent in the environment in close proximity to a study subject. Depending on a variety of factors, some of this toxin may be taken up by the subject through inhalation, ingestion, or the skin. These routes of exposure then mediate uptake and, along with metabolic characteristics of the subject, determine the dose of the agent that reaches the target tissues in the body. With regard to Vietnam veterans, for instance, it has been suggested that troops who spent days in the field without the possibility of good personal hygiene may have received a larger effective dose from a given exposure. Although it is the toxic dose and not the exposure per se that causes a disease, this dose is rarely measurable, and often, exposure
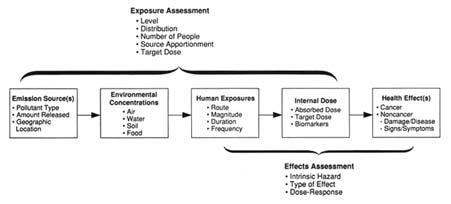
FIGURE 6-1 Progression of events from environmental exposure to health effects. Reprinted with permission from Heldref Publications.
SOURCE: Sexton et al., 1992.
must be used as a proxy for dose. The assumption that exposure approximates dose is not necessarily a bad one, but how strong this correlation actually is depends on the chemical and physical properties of the agent, the route of exposure, and the target tissue.
Hierarchy of Exposure Assessment Strategies
Exposure has been characterized in many different ways in epidemiologic studies, depending on the availability of data and the hypothesis being tested. One can usefully distinguish a few basic approaches to exposure assessment (Checkoway, 1986; Smith, 1987). The simplest approach compares the members of a class presumably exposed to an agent with the general population or with an "unexposed" group. Occupational studies are often of this type, comparing for example, herbicide production workers to the general population. Vietnam veterans have also been compared to veterans who served during the Vietnam era but did not serve in Vietnam. The advantages of this approach are its simplicity and the ease of interpretation of the results. If there is something fundamental to class membership that increases risk—for example, all "woodworkers" are exposed to wood dust—then studies of this type can effectively identify the increased morbidity or mortality in the group. If, however, only a small fraction of class members are actually substantially exposed to a toxic agent (in all likelihood, as discussed in this chapter, only a fraction of the estimated 2.6 million to 3.8 million veterans who served in Vietnam were substantially exposed to herbicides), then any increased risk from exposure in this subgroup may be lost entirely when the disease risk of the full class (all Vietnam veterans) is assessed.
A somewhat more refined method of exposure assessment assigns to
each cohort member a qualitative degree or level of exposure. This may be done in several different ways. For example, a cohort of herbicide production workers could be divided into subgroups in such a way that those who were likely to have been heavily exposed through their job assignments are placed in one group (e.g., "high exposure"); a second group might be identified who had sustained exposures, but not in those jobs or departments in direct contact with the toxin ("moderate exposure"); and finally, a residual group might contain those with little or no exposure who were nonetheless employed at the production facility ("low exposure"). The disease risk may then be calculated separately for each of these groups compared to a referent or ''unexposed" group. This method has several advantages over the simple exposed/unexposed comparison just described: it should (if the classification of exposure is done without serious errors, see below) yield less diluted risk estimates; provide support for a dose-response trend; and not necessarily require expensive and time-consuming measurements of the actual exposure of each cohort member.
Ideally, quantitative estimates should be available on the total exposure history of each subject in the study. When such data are available, it is possible to estimate quantitatively the relationship between a given level of exposure and the degree of risk that is expected to accrue. In occupational epidemiology studies, quantitative exposure data are sometimes developed through a process called historic exposure reconstruction, which is outlined in the following sections.
When quantitative estimates of the intensity of exposure are not available, it is sometimes possible to know the duration of exposure for each cohort member. Although less satisfactory, one may nevertheless assume that the intensity of exposure was relatively constant among cohort members so that the total exposure (sometimes called cumulative exposure) is proportional to its duration. Following these assumptions, one would hypothesize that a true risk would increase with the duration of exposure.
Epidemiologists generally think of these various exposure assessment strategies in a hierarchy of increasing accuracy: the exposed/unexposed approach being the least accurate, followed by the qualitative classification of level of exposure, and best of all, quantitative estimates of both the intensity and the duration of exposure. It is important to stress that all of these strategies may be valid, but they vary in their precision and in the degree to which they can contribute to the evidence for or against a particular exposure-disease association.
Exposure Assessment for Cohort Studies
In cohort studies it is sometimes possible to have other data sources available with which to estimate exposure for each study subject without
directly interviewing the subject. This is especially common in occupational cohort studies in which work records and industrial hygiene data may be available that cover the entire history of the factory being studied.
At minimum, it is often possible to know with considerable accuracy the length of time that each cohort member has spent in the industry. Somewhat more precise assessments may be possible if the cohort can be subdivided into those who were employed in one or more areas of the plant where the exposure of interest was heaviest.
A variety of approaches have been used to estimate the intensity of exposure in each job or department in an industry. Sometimes industrial hygiene measurements are available, in which statistical models can be used to "fill in the gaps" that inevitably occur in these data (Greife et al., 1988; Hornung, 1991). Often the potentially hazardous chemical is an essential component of an industry's production process, so the relative intensity of exposure can be estimated from data on production rates in each department of the factory being studied (Kalliokoski, 1990). Expert judgment has also been used to estimate relative intensity of exposure (Tankersley et al., 1991). Physical models of the workplace environment, the production process, and the location of workers may be used to estimate likely exposure levels (Schneider et al., 1991).
In recent years, considerable research has been done to develop systematic methods of historic reconstruction of exposure—the broad term covering all of these various strategies (Gamble and Spirtas, 1976; Smith, 1987; Bond et al., 1991; Rice, 1991; Smith et al., 1991; Stewart and Herrick, 1991; Tankersley et al., 1991). Three international symposia have been held on the subject (Rappaport and Smith, 1991; Stewart and Herrick, 1991; Axelson and Westberg, 1992). The essential features of historic exposure reconstruction applied to each member of a study cohort are (1) the use of surrogates of past exposure to toxic chemicals (if no measurements are available) to estimate the likely intensity of exposure an individual would have experienced in a particular location (often a job or industrial department) at a particular time; (2) the use of either work records, individual recollections, or a combination of the two to determine which locations or jobs the subject was in for which periods; this information can then be combined with the job/location exposure estimates to build up for each subject an estimated exposure history covering years to decades of past exposure; and (3) summation of individual exposure histories in parameters that can then be used in epidemiologic models to assess exposure-risk associations.
Historic exposure reconstruction is a lengthy and expensive process, and the field is still developing. There are however some recent examples of occupational epidemiologic studies in which exposure estimates derived from historic reconstruction have proven superior to those relying on simpler
measures such as the total duration of exposure (Dement et al., 1983; Stewart et al., 1986; Rinsky et al., 1987; Kriebel, 1988a,b; Seixas, 1990; Blair and Stewart, 1992).
Exposure Assessment for Case-Control Studies
Often case-control studies must rely on information provided by the study subjects—cases and controls—to estimate exposure histories. The problem of recall bias has already been mentioned, and it is a potentially serious one in case-control studies when subjects are asked directly to recall exposure to a potentially toxic chemical. Some case-control studies reduce the severity of this problem by gathering general data from study subjects, with which more specific exposures can be estimated, that are less likely to be recalled differentially by cases and controls. For example, in occupational case-control studies, it is common to collect from each subject a full occupational history including detailed job information, but not information on chemicals that may have been used in these jobs. Experts on industrial processes such as manufacturing engineers and industrial hygienists then independently develop a job exposure matrix (JEM), which estimates for each job the types of chemicals likely to have been used, and sometimes their likely intensity of use as well (Zahm et al., 1990; Gerin and Siemiatycki, 1991). There is doubtless a good deal of nondifferential misclassification that results from this approach because the JEM approximates the general use of chemicals in occupations and not the specifics of an individual subject's experience. However, recall bias is much less likely to occur because subjects are not asked directly about hazards.
An excellent example of this approach is provided by a large multicenter case-control study of hematolymphopoietic cancers being conducted in Italy and focusing on pesticides and solvents risk factors (Miligi and Masala, 1991). In the participating centers, which are located in different agricultural regions, detailed JEMs have been developed by agronomists with long experience in local pest control practices. Subjects who report employment in farming are asked detailed questions about the crops they grew and the pests they treated in the course of their working lives. Local agronomists who advise farmers on pesticide usage then can predict with some reliability which chemicals were used in that particular zone in a particular time period to combat the reported pest.
The committee recognized that case-control studies can only rarely provide quantitative estimates of exposure. Nevertheless, they can provide valid information on the existence of an association and the identification of a dose-response trend. An example of this comes from a recent study of non-Hodgkin's lymphoma (NHL) and the use of 2,4-dichlorophenoxyacetic acid (2,4-D) among Nebraska farmers (Zahm et al., 1990). Investigators
questioned cases and controls extensively about the use of all kinds of pesticides. One question asked how frequently the subjects changed clothes after spraying pesticides. The authors observed that for those reporting 2,4-D use, the risk of NHL was higher among those who tended to change clothes less often. There was a statistically significant trend in the odds ratio over three categories of clothes changing. Thus, although no conclusions can be drawn from this study about the amount of 2,4-D to which subjects were exposed, there are data that strengthen a finding of a statistical association.
It must be stressed that these methods of exposure assessment, like the historic reconstruction approach for cohort studies discussed previously, are rarely "confirmed" in a given study by comparison to some "true" measure, since such a measure generally does not exist. As explained below, however, it is unlikely that such methods, conducted in a systematic way, can produce false-positive associations (bias away from the null). On the other hand, it is entirely likely that estimates resulting from these methods may underestimate the true magnitude of risk.
Exposure Misclassification
Some degree of error is introduced into any exposure measurement. When a cohort member is incorrectly called "exposed," an individual is assigned to the "low-exposure" group when moderate exposure has actually occurred, or a quantitative estimate of exposure is highly inaccurate, then exposure misclassification occurs, which introduces bias into the study results. Epidemiologists distinguish two fundamental kinds of exposure misclassification.
Differential (or nonrandom) exposure misclassification occurs when errors in the assignment of exposure are unequally distributed between the diseased and nondiseased groups. A common example of this is recall bias, which may occur in case-control studies if cases and controls are asked directly about their exposure histories, and if cases recall their exposure differently than controls. It has been shown, for example, that mothers of newborns with birth defects (cases) are more apt to report exposure to many prescription and nonprescription drugs than are mothers of normal newborns (controls) (Werler et al., 1989). Differential misclassification is a particularly serious problem because the direction of the bias cannot be determined. That is, the effect of this kind of error in exposure assignment may be to increase artificially the magnitude of an association between exposure and disease (called a bias away from the null) or to reduce the apparent magnitude of association (a bias toward the null).
Nondifferential (or random) misclassification occurs when exposure is measured with error but the degree of error is the same for the diseased and
nondiseased groups. In well-designed epidemiologic studies, investigators are often able to reduce the likelihood that serious differential misclassification has occurred, but nondifferential misclassification is often inevitable because of the imprecision of any exposure measurement tool. It is often possible to determine with some confidence that the likely direction of the bias from nondifferential exposure misclassification will be toward the null (Fleiss and Shrout, 1977; Armstrong and Oakes, 1982; Kupper, 1984; Heedrik and Miller, 1988; Lagakos, 1988). That is, the magnitude of the association estimated with misclassified exposure data will be lower than that of the "true" exposure-disease association. One can think of this phenomenon as "diluting" the true effect, which occurs, for example, when all members of an occupational group are compared to nonmembers to estimate the risk of a toxin to which only some members are actually exposed.
For example, consider the relationship between Hodgkin's disease and exposure to Agent Orange, which was studied by the Centers for Disease Control (CDC) in the Selected Cancers Study (CDC, 1990b), but for which the primary exposure variable was Vietnam service, not something more closely related to herbicide exposure. The odds ratio relating Hodgkin's disease to Vietnam service was approximately 1.2, but it was not statistically significant. Suppose that (1) disease status is classified without error; (2) 100 percent of those truly exposed are classified as exposed; and (3) 95 percent of those truly not exposed are classified as not exposed, and this fraction is the same for Hodgkin's disease cases as for the controls in the Selected Cancers Study. Then, by using the method of Kleinbaum and colleagues (1982), the estimated relative risk adjusted for misclassification bias would be 1.6, which represents a tripling of the excess risk. If the percentages in assumptions (2) and (3) were further from 100, the adjusted relative risk would be even higher.
Several recent scientific papers have argued that the general principle of nondifferential misclassification producing bias toward the null may not always hold when ordered categorical exposure data (e.g., low, medium, high) are used; however, there is not yet agreement as to how frequent or how severe any bias away from the null may be (Dosemici et al., 1990; Myers and Ehrlich, 1990; Flegal et al., 1991; Wacholder et al., 1991; Birkett, 1992; Brenner, 1992; Delpizzo, 1992). There is general agreement that with continuous quantitative measures of exposure and with dichotomous measures (exposed/unexposed), nondifferential misclassification will bias toward the null.
A common method of exposure estimation in occupational cohort studies assigns exposure levels to each job in the factory; then each worker's lifetime exposure is estimated by multiplying the amount of time spent in each job by the exposure intensity for that job. The resulting lifetime cumulative exposure for each cohort member may not be seriously misclassified
even if there are substantial errors in the job exposure estimates on which the cumulative exposures are based. One reason that the practice of estimating exposure histories from data on the exposure levels in certain jobs and the amount of time spent in each job may be fairly robust against errors in exposure measurement is that if workers move from job to job over the course of their careers, the errors in each of the job exposure estimates tend to cancel one another. Often the process of reconstructing the exposure levels in jobs in the past is only approximate, but as long as the effect of the errors is to sometimes over- and sometimes underestimate the true exposure in the job, then a lifetime exposure estimate will not be strongly affected by these individual job errors.
One solution to the problem of misclassification of exposure is to conduct a small validation study in which elaborate and expensive measurements are performed alongside the quicker, more error-prone measurements that will be used in the full study. Analysis of the degree of correlation between the "true" and the approximate measures allows investigators to adjust the risk estimates from the full study for exposure misclassification (Fleiss and Shrout, 1977; Armstrong and Oakes, 1982; Marshall, 1990; Rosner et al., 1990).
Biomarkers
Biomarkers for TCDD
TCDD and other chlorinated dibenzo-p-dioxins and dibenzofurans are found in tissues of non-occupationally exposed humans at part-per-trillion (ppt; nanogram-per-kilogram) levels. Following absorption, TCDD is distributed to tissues with high lipid content. Adipose tissue appears to be the main site of accumulation, although TCDD has been found in all tissue samples that have been examined from autopsy (Ryan et al., 1986). On a whole-weight basis, adipose tissue contains the highest levels, followed by liver, muscle, and kidney; on a lipid basis, the concentrations vary less (Ryan et al., 1986). Normal exposure to TCDD leads to levels in the lipid stores of humans of about 5-6 ppt, as measured in the adipose tissue or the lipid portion of the blood (Needham et al., 1990).
Although exposure to TCDD from environmental sources, primarily food (Geyer et al., 1986; Byard, 1987), occurs on a continuing basis, both serum and fat biopsy samples taken from individuals with unusually high exposures indicate that TCDD may remain in the body for many years after exposure. For example, fat biopsy samples taken from three Vietnam veterans believed to have been exposed through herbicide use showed levels of TCDD up to 99 ppt approximately 10 years after exposure occurred. By comparison, veterans with no unusual herbicide exposure had TCDD levels
between 3 and 15 ppt (Gross et al., 1984). A number of other studies, including the Ranch Hand half-life study, the Missouri civilian study, and the National Institute for Occupational Safety and Health (NIOSH) study, suggest that current serum TCDD levels are useful for distinguishing between groups of individuals that were exposed to TCDD 15 to 20 years ago (CDC, 1989a). For example, Sweeney and colleagues (1990) measured TCDD levels in serum lipids of 143 workers at two chemical plants in New Jersey and Missouri. The median TCDD level in a group of 103 production workers at the New Jersey plant was 84 ppt compared to 11 ppt for a group of eight office workers.
The most reliable procedure for measuring serum lipid-associated TCDD was developed by the CDC in 1986 as a substitute for the considerably more difficult surgical procedure required to obtain fat biopsies (Patterson et al., 1987). This procedure involves an extremely sensitive mass spectroscopic technique that permits measurement of TCDD into the low part-perquadrillion levels. A study in which both serum and fat TCDD levels were compared in 50 individuals showed a very high correlation between the two, suggesting that TCDD levels in serum provide a valid measure of TCDD levels in the body (Patterson et al., 1988). The Pointman Project (Kahn et al., 1988), conducted by the New Jersey Agent Orange Commission, confirmed this result.
The pharmacokinetics of TCDD in humans—its distribution and passage through the body—are not fully understood, which makes individual serum TCDD levels difficult to interpret and also complicates the interpretation of epidemiologic studies relying on these measures of exposure. As described in Chapter 4, a complex, poorly understood process distributes dioxins among body tissues and slowly clears it from the body. There is evidence that this process is quite variable among humans, so it is difficult to model its behavior and thereby extrapolate backward to estimate the likely concentration of TCDD in fat or blood in the past. It is also assumed that TCDD is removed from the body according to first-order kinetics—that is, for a given period of time, a constant fraction of the TCDD body burden is eliminated—but some evidence suggests the process may be more complicated and may vary as conditions in the body change. Second, the metabolic processes governing this movement and disposition may not be relevant to the determination of the dose of TCDD to the brain or reproductive organs, for example. In the epidemiologist's view, there may be different "causal pathways" linking exposure to the biomarker and exposure to disease.
By measuring TCDD levels in 1982 and 1987 from serum samples of 36 Ranch Hand veterans, the median half-life of TCDD in humans was estimated to be 7.1 years, adjusted for background TCDD levels (with a 95 percent confidence interval of 5.8 to 9.6 years) (Pirkle et al., 1989). In this study, the background exposure level of TCDD in serum was taken to be 4
ppt on the basis of data from the Agent Orange Validation Study. Background levels appear to be log-normally distributed and to increase with age (Sielken, 1987). An expanded study of 337 Ranch Hand veterans, including the 36 from the previous study, estimated a median half-life between 11.5 and 12 years. The analytical measurement error (coefficient of variation) in these studies was about 22 percent (Pirkle et al., 1989). These data indicate that the half-life of TCDD is independent of its initial serum concentration, thereby supporting a first-order kinetics process for TCDD elimination. A follow-up study of this group collected a third set of serum samples in 1992, and a new estimate will be made when analyses are complete.
Variations in TCDD half-life estimates are likely to result from changes in weight and percentage of body fat. The normal aging process, as well as dietary changes, may play a role here. For example, an average 5 foot 10 inch male aged 20 to 24 years weighs approximately 70 kg, of which 15 to 18 percent is fat. If at age 40-49, this male weighs approximately 80 kg, fat content almost doubles. Thus, there may be an expected reduction in serum TCDD levels in Vietnam veterans just from the mechanisms of change in body composition with age (Albanese, 1991). If the percentage of body fat is found to significantly affect half-life estimates, with assumptions of a 6 to 12 year half-life and increased body fat characteristic of aging, it is possible that a small or moderate intake of dioxin among Vietnam veterans may no longer be detectable by using current serum TCDD level as a biomarker (Schlatter, 1991).
Still other confounding factors may complicate exposure assessment. In a study of 640 Vietnam veterans, researchers found that serum TCDD levels varied with several personal characteristics, including age, race, body mass, and region of residence (Devine et al., 1990). It has also been suggested that disease may affect serum TCDD levels (Michalek and Tripathi, 1992). Flanders and colleagues (1992) have shown, for instance, that reverse causality, in which health outcomes affect the measured serum TCDD level years after exposure, can better explain some relationships between serum TCDD and health outcomes in the Ranch Hand study than a direct model in which TCDD causes the outcome.
Based on these data, the committee concludes that serum TCDD measures are helpful in epidemiologic studies, but should not be taken as a "gold standard" of exposure. If there are group differences in serum TCDD levels, that probably does indicate a difference in exposure to TCDD between the two groups. However, even if there is a difference in TCDD exposure between two groups, it may disappear as subsequent serum levels fade to background levels with the passage of time between exposure and measurement. In particular, because of poorly understood variation among individuals in TCDD metabolism and relatively large measurement error, individual TCDD levels are usually not meaningful.
Although quantitative measures of exposure are highly desirable, it may still be quite valid to use approximate measures of exposures or even surrogates such as the amount of time spent in a job in which the toxin was likely to have been used. Thus a biomarker, especially one gathered years after exposure, is not necessarily better than qualitative exposure measures based on process records. Group differences in serum TCDD levels can be useful in confirming that occupational exposure measures reflect true differences in exposure, as has been done in the NIOSH study (Fingerhut et al., 1989, 1991; Sweeney et al., 1990) and other studies (see below).
Chloracne as a Biomarker
As discussed in Chapter 11, there is strong evidence indicating that chloracne develops in at least some individuals heavily exposed to TCDD. Some researchers have used chloracne as a clinical marker of TCDD exposure. As a biomarker, however, chloracne has several serious drawbacks.
Numerous studies of heavily exposed individuals have demonstrated that chloracne is neither a specific nor an exclusive indicator of TCDD exposure at the individual level (Suskind and Hertzberg, 1984; Bond et al., 1989; Zober et al., 1990; Mocarelli et al., 1991). Individual susceptibility to the activity of TCDD seems to vary, especially with respect to the seriousness of the manifestation (Del Corno et al., 1985). In Seveso, Italy, for instance, after the TCDD release in 1976, approximately half of the adults with the highest serum TCDD levels developed chloracne whereas the other half did not. Some of the individuals who did not develop chloracne had serum TCDD levels higher than those who did (Mocarelli et al., 1991). There are other causes of chloracne and chloracne-like conditions besides TCDD, so its presence does not indicate certain exposure (see Chapter 11). The etiology of chloracne can be attributed to a rather wide range of chemical compounds, and lesions mimicking chloracne, such as acne vulgaris, may also appear in subjects who have not been exposed to TCDD.
In summary, although chloracne has been used in epidemiologic studies as a biomarker for TCDD exposure, the data indicate that it is neither a sensitive nor exclusive indicator. It is usually not long lasting, is difficult to diagnose, and is not at all sensitive to exposure to herbicides that are not contaminated with TCDD.
EXPOSURE ASSESSMENT IN OCCUPATIONAL AND ENVIRONMENTAL STUDIES
The committee reviewed a large number of epidemiologic studies of occupationally or environmentally exposed groups for evidence of an association between health risks and exposure to TCDD and herbicides used in
Vietnam, especially the phenoxy herbicides 2,4-D and 2,4,5-T, chlorophenols, and other compounds. In reviewing these studies, two types of exposure were explicitly considered: exposure to TCDD per se and exposure to the various herbicides used, particularly 2,4-D and 2,4,5-T. This separate consideration is necessary because of the possibility that, for example, some health effects may be associated with exposure to 2,4-D in agriculture and forestry. The herbicide 2,4-D does not contain TCDD, although small quantities of other dioxins are present.
For TCDD-exposed populations, serum measures of TCDD concentrations can be made of a representative sample of those exposed. Serum biomarkers of TCDD exposure are sometimes used to estimate the degree of prior exposure of individuals; however, there are limitations to their use, as described in the previous section.
Occupational Studies
Problems exist in the estimation of exposures among nonveteran groups studied for health outcomes. In many of the studies reviewed, exposure to herbicides or TCDD was inferred simply by occupation as a surrogate measure. Types of occupations involving potential exposure include workers in herbicide or other chemical production plants, agricultural and forestry workers, herbicide and pesticide applicators, and paper and pulp mill workers. As noted in the beginning of this chapter, the problem with characterizing exposure based on occupation only is that misclassification can occur if those classified as exposed were actually unexposed, or vice versa. Any actual increased risk from exposure might not be detected in the entire group when exposure is classified simply by occupation. In studies of farmers, for example, outcome data may merely be related to the fact of being a farmer. A much more accurate measure would incorporate information on what specific herbicides the farmer had been exposed to, the number of days the farmer had been exposed to the herbicide in question, and the degree of direct contact the farmer experienced.
However, exposure measures in occupational studies depend on the nature of the available data. Data for occupational studies are obtained from a variety of sources with varying degrees of completeness and accuracy. Some studies have identified and followed cohorts of individuals based on company or union records, or government licensing registries. Others have relied upon detailed company records of duration of employment, or assignment to various jobs or to particular work locations within the plant, to construct exposure indices. In some cases, there has been additional qualitative information available regarding the type of herbicides used, the nature of the TCDD exposure, and whether other chemical exposures may have been involved. These studies, with supplemental information on herbicide
use, provide more refined measures of exposure than those based on membership in a particular occupational group.
In most studies, individual quantitative estimates of duration and intensity of exposure are unavailable and must be estimated from information on the number of years employed in a particular job, or through identification of areas or departments in the workplace that involved sustained or direct exposure to the herbicide or TCDD. Ideally, quantitative measures of both the intensity and the duration of exposure could be determined; although surrogate measures are less satisfactory, all of these strategies can contribute to the assessment of exposure-risk associations through the process of historic exposure reconstruction.
The process of historic exposure reconstruction has been applied to some occupational studies of industrial workers potentially exposed to herbicides or TCDD. The following examples illustrate some of the approaches used in the epidemiologic studies reviewed by the committee. Additional information on these studies is provided in Chapter 7.
Production Workers
One of the most extensive sets of data on workers engaged in the production of chemicals potentially contaminated with TCDD has been compiled by NIOSH. For 12 chemical companies, more than 5,000 workers were identified from personnel and payroll records indicating whether the worker had been involved in production or maintenance processes associated with TCDD contamination (Fingerhut et al., 1991). At each chemical plant, a review of operating conditions, job duties, and records of TCDD levels in industrial hygiene samples, intermediate reactants, products, and wastes was conducted. Exposure was estimated from job records according to duration of exposure in processes involving TCDD contamination and total length of employment at the plant; serum TCDD levels were measured in a sample of 253 workers. Data on current TCDD levels demonstrated a good correlation with duration of employment. Nonetheless, the authors note the possibility for misclassification of exposure. Workers were exposed concurrently to chlorophenols and phenoxy herbicides that were contaminated with TCDD, as well as numerous other chemicals during their employment.
A multisite study by the International Agency for Research on Cancer (IARC) involved 18,390 production workers and herbicide sprayers from 10 countries (Saracci et al., 1991). Exposure was estimated from a combination of factory records, work histories, spraying data, and questionnaires. The cohort included information according to whether individuals were exposed by spraying or during production, and by the type of chemical used or produced. Workers who sprayed chlorophenoxy herbicides or worked in
factory departments in contact with these chemicals were considered ''exposed" (N = 13,482); workers "probably exposed" had no job title but were judged to have been exposed (N = 416). Workers with no exposure status information were treated as having "unknown" exposure (N = 541), and those who never worked in factory departments with exposure to chlorophenoxy herbicides, or who never sprayed these chemicals, were considered "nonexposed" (N = 3,951). Exposure to TCDD was assumed to be possible for those who worked producing or spraying 2,4,5-trichlorophenol (TCP) and 2,4,5-T or related products. The degree of exposure to TCDD, however, is more uncertain than that of the NIOSH study since some of the cohorts of individuals either sprayed or produced compounds such as 2-methyl-4-chlorophenoxy-acetic acid (MCPA) or mecoprop (MCPP), which are unlikely to contain significant quantities of TCDD.
Several other occupational studies of workers involved in chemical production plants have relied upon job titles as recorded on individual work histories and company personnel records to classify exposure (Ott et al., 1980; Zack and Gaffey, 1983; Coggon et al., 1986, 1991; Cook et al., 1986; Zober et al., 1990). Similarly, exposure in chemical plant workers has been characterized by worker involvement in various production processes (i.e., synthesis, packaging, waste removal, shipping, plant supervision; Manz et al., 1991; Bueno de Mesquita et al., 1993).
Chloracne has been observed in some chemical plant workers and used as an indicator of prior TCDD exposure (Cook et al., 1980; Moses et al., 1984; Bond et al., 1987, 1989; Collins et al., 1993). Chloracne, however, is neither a sensitive nor an exclusive indicator of prior TCDD exposure.
Agricultural/Forestry Workers
Occupational studies of agricultural workers have estimated exposure to herbicides or TCDD using a variety of methods. In the simplest method, data on an individual's occupation are derived from death certificates, cancer registries, or hospital records (Burmeister, 1981). Although this information is relatively easy to obtain, it is not possible to estimate duration or intensity of exposure, or to determine the specific type of herbicide or chemical a worker was exposed to. Some studies of agricultural workers have attempted to investigate differences in occupational practices to identify subsets of workers who were likely to have had higher levels of herbicide exposure (Vineis et al., 1986; Wiklund and Holm, 1986; Musicco et al., 1988; Wiklund et al., 1988a; Hansen et al., 1992; Ronco et al., 1992). Other studies have used county of residence as a surrogate of exposure, relying upon agricultural censuses of farm production and chemical use to characterize exposure in individual counties (Gordon and Shy, 1981; Cantor, 1982; Blair and White, 1985). Still other studies attempted to refine
exposure estimates by categorizing the exposure based on the number of years employed in a specific occupation as a surrogate for exposure duration, obtaining supplier records on the amount of herbicides purchased to estimate the level of exposure, or estimating acres sprayed to quantify the amount used (Wigle et al., 1990; Morrison et al., 1992). In some cases, self-reported information on exposure was obtained, including direct handling of the herbicide, whether it was applied by tractor or hand-held spray, and what type of protective equipment was worn or what safety precautions were exercised, if any (Hoar et al., 1986; Zahm et al., 1990). Some studies attempted to validate self-reported information, based on verification using written records, signed statements, or telephone contacts with coworkers or former employers (Carmelli et al., 1981; Woods and Polissar, 1989).
Forestry workers and other outdoor workers, such as highway maintenance workers, are likely to have been exposed to herbicides and other chemicals to varying degrees. Exposure has been classified in a manner similar to other studies, for example, by number of years employed, job category, and occupational title.
Herbicide/Pesticide Applicators
Studies of herbicide sprayers are relevant because it can be presumed that applicators had more sustained exposure to herbicides; however, applicators were also likely to be exposed to a multiplicity of chemicals, complicating the assessment of any individual or group exposure to specifically phenoxy herbicides or TCDD. Some studies have attempted to quantify exposure of applicators based on information from work records on the number of acres sprayed or the number of days of herbicide spraying. Employment records can also be used to extract information on the type of chemicals sprayed.
One surrogate indicator of herbicide exposure is receipt of a license to perform spraying. Several studies have specifically identified licensed or registered pesticide and herbicide applicators (Smith et al., 1981, 1982; Blair et al., 1983; Wiklund et al., 1988b, 1989; Swaen et al., 1992). Individual estimates of the intensity and frequency of exposure were rarely quantified in the studies the committee examined, however, and often applicators were known to have applied many different kinds of herbicides, pesticides, and other chemicals. In addition, herbicide spraying is generally a seasonal occupation, and information may not be available on possible exposure-related activities during the rest of the year.
One study provided information on serum TCDD concentrations in herbicide sprayers, that of Smith et al. (1992). Blood from nine professional spray applicators in New Zealand, who first sprayed before 1960 and were also spraying in 1984, was analyzed. The duration of actual spray work
varied from 80 to 370 months. The serum levels ranged from 3 to 131 ppt TCDD on a lipid basis, with a mean of 53 ppt. The corresponding values for age-matched controls ranged from 2 to 11 ppt; the mean was 6 ppt. Serum TCDD levels were positively correlated with the number of months of professional spray application.
Several studies have evaluated various herbicide exposures during spraying in terms of type of exposure, routes of entry, and routes of excretion: Kolmodin-Hedman and Erne (1980), Kolmodin-Hedman et al. (1983), Lavy et al. (1980a,b), Ferry et al. (1982), Libich et al. (1984), Frank et al. (1985). Based on these studies, it would appear that the major route of exposure is through dermal absorption, with 2 to 4 percent of that on the skin being absorbed into the body during a normal work day. Air concentrations were usually less than 0.2 mg/m3. The absorbed phenoxy acid herbicides are virtually cleared within one day, primarily through urinary excretion. Typical measured excretion levels for ground crews ranged from 0.1 to 5 mg/day and less for air crews.
Paper and Pulp Mill Workers
Another occupational group likely to be exposed to TCDD and chlorinated phenols consists of paper and pulp mill workers. These workers are likely to have received varying degrees of exposure as part of the bleaching process in the production of paper and paper products. Pulp and paper production workers are also likely to be exposed to other chemicals in the workplace, which vary, for example, according to the type of paper mill or pulping operation, and the final product manufactured (Robinson et al., 1986; Henneberger et al., 1989; Solet et al., 1989; Jappinen and Pukkala, 1991).
Environmental Studies
Studies of environmental exposures related primarily to unintentional releases of TCDD into the environment at Seveso, Italy, and Times Beach, Missouri. In these cases, the simplest measure of exposure was classification according to place of residence. Intensity of exposure has been estimated by years of residence in a contaminated area; this measure does not take into account the concentration of TCDD or herbicide, or the frequency of individual contact with contaminated soil or water.
One of the largest industrial accidents involving environmental exposures to TCDD occurred in Seveso, in July 1976, as a result of an uncontrolled reaction during trichlorophenol production. A variety of indicators were used to estimate individual exposure; soil contamination by TCDD has been the most extensively used. On the basis of soil sampling, three
areas were defined about the release point. They were zone A, the most heavily contaminated, from which all residents were evacuated within 20 days; zone B, an area of lesser contamination that children and pregnant women in their first trimester were urged to avoid during daytime; and zone R, a region with some contamination, in which consumption of local crops was prohibited (Bertazzi et al., 1989). The samples so obtained are virtually unique in that they were numerous and were obtained prior to elimination and degradation of TCDD in the sample media.
Data on serum concentrations of zone A residents have been presented by Mocarelli et al. (1990, 1991) and earlier by the CDC (1988a). For those with severe chloracne (N = 10) the TCDD levels ranged from 828 to 56,000 ppt lipid weight. Those without chloracne (N = 10) had levels from 1,770 to 10,400 ppt. The levels in all controls but one were not detectable. The highest of these levels among TCDD-exposed workers exceeded any that had been estimated up to that time, based on backward extrapolation with a half-life of seven years. Data on nearby soil levels, number of days an individual stayed in zone A, and whether local food was consumed were considered in evaluating TCDD levels. None of these data correlated with serum TCDD levels, strongly suggesting that the exposure of importance was fallout on the day of the accident. The presence and degree of chloracne did correlate with TCDD levels; however, it appears that adults are much less likely to develop chloracne than children following an acute exposure, but surveillance bias may have played some role in this finding.
A number of reports have provided information on exposure to TCDD from environmental contamination in Missouri (Patterson et al., 1986; Andrews et al., 1989). In 1971, TCDD-contaminated sludge from a hexachlorophene production facility was mixed with waste oil and sprayed in various community areas for dust control. Soil contamination in some samples exceeded 100 parts per billion (ppb). One of the Missouri sites with the highest TCDD soil concentrations was the Quail Run mobile home park. Residents were considered exposed if they had lived in the park for at least six months during the time the contamination occurred (Hoffman et al., 1986). Other investigations of Times Beach have estimated exposure-risk based on residents' reported occupational and recreational activities in the sprayed area. Levels of exposure have been estimated from duration of residence and TCDD soil concentrations.
Andrews et al. (1989) provided the most extensive data on human adipose tissue levels for 51 persons who had ridden or cared for horses at arenas sprayed with TCDD-contaminated oil; persons exposed in residential areas where such oil had been sprayed; individuals involved in TCP production; persons exposed in TCP nonproduction activities, such as lab and maintenance workers; and 128 controls. Persons were considered exposed if they lived, worked, or had other contact for two years or more with
TCDD-contaminated soil at levels of 20-100 ppb or for six months or more with soil contaminated with TCDD at levels greater than 100 ppb. Of the exposed population samples, 87 percent of adipose tissue TCDD levels were less than 200 ppt; however, TCDD concentrations in 7 of the 51 exposed ranged from 250 to 750 ppt. For nonexposed persons, adipose tissue TCDD concentrations ranged from nondetectable to 20 ppt, with a median of 6 ppt. Based on a seven year half-life it is calculated that two of the study participants would have had adipose tissue TCDD levels near 3,000 ppt at the time of the last date of exposure.
Vietnamese Studies
Several studies have investigated exposure to herbicides among the residents of southern Vietnam (Constable and Hatch, 1985), comparing unexposed residents of the South to residents of the North. Other studies have attempted to identify veterans of northern Vietnam who served in the South during the Vietnam era. Records of herbicide sprays have been used to refine exposure measurements, comparing individuals who lived in sprayed villages in the South with those living in unsprayed villages. In some studies, residents of villages were considered exposed if a recorded herbicide mission passed within 10 km of the village center (Dai et al., 1990). Other criteria for classifying exposure included length of residence in a sprayed area and number of times the area had reportedly been sprayed.
A small number of studies provide information on TCDD concentrations in Vietnamese civilians exposed during the war. Schecter et al. (1986) detected TCDD in 12 of 15 samples of adipose tissue taken at surgery or autopsy in southern Vietnam during 1984. The concentrations in the positive samples were from 3 to 103 ppt. No detectable levels of TCDD were found in nine samples from residents of northern Vietnam who had never been in the South. The detection sensitivity was 2-3 ppt. Analysis of three breast milk samples collected in 1973 from Vietnamese women thought to have been exposed to Agent Orange varied from 77 to 230 ppt on a lipid basis.
Conclusions on Exposure Assessment in Occupational and Environmental Studies
In general, exposure assessment is the weakest aspect in most of the occupational and environmental studies that the committee has reviewed. The nature of the exposure is generally poorly characterized with regard to which of the herbicides or dioxins the subjects were exposed. Rarely is there any precise information on the intensity and duration of individual exposure; rather, surrogates such as the length of employment and job location
in the workplace are measured. Many studies use membership in a group to assign exposure, but some studies, such as the NIOSH and IARC studies of chemical production workers that evaluate subgroups with presumed higher, longer, or more relevant (to this study) exposure, are the exceptions to this pattern.
The range of occupational and environmental populations and the likely intensity and duration of their exposure to herbicides and TCDD are diverse. This provides an opportunity to compare results between studies to determine whether certain diseases are more common in populations likely to have higher exposures. Also, if one disease seems to be consistently associated with herbicides or TCDD among differently exposed groups, the evidence for an association is strengthened. Because of the complex pattern of exposures to the various herbicides and TCDD in the available epidemiologic studies, the committee was generally not able to differentiate among multiple chemical exposures to determine whether specific health effects were associated with any particular herbicide or TCDD.
EXPOSURE ASSESSMENT IN STUDIES OF VIETNAM VETERANS
Different approaches have been used in estimating exposure to herbicides in studies of Vietnam veterans. These studies generally rely on self-reported exposures, records-based exposure estimates, or biomarkers. Each of these approaches is limited in its ability to determine precisely the intensity and duration of individual exposure.
One of the simplest approaches to classifying Vietnam veterans' potential exposure to herbicides and TCDD assumes that service in Vietnam resulted in exposure to herbicides, so any adverse health effects associated with these substances would be discernible in veterans who served in Vietnam compared to Vietnam era veterans who did not serve there. In various studies, certain attributes of military service have been hypothesized to be associated with an increased likelihood of herbicide exposure. These include branch of service, combat experience, and military occupation. Another approach involves the reconstruction of herbicide exposure through a procedure of matching historical data on herbicide sprays with records of troop location and dates of service.
Self-Reports
Self-reported data are a common epidemiologic tool for assessing exposure, but they have a number of drawbacks when applied to studies of Vietnam veterans to determine herbicide and TCDD exposures. There include the general problem of recall and, in particular, the possible confusion of herbicide spraying with the spraying of insecticides, and underreporting
by individuals who unknowingly entered an area that had previously been sprayed and still contained residual herbicides or TCDD. On the other hand, some veterans who believe they were exposed to herbicides may not actually have been exposed. Veterans have been asked to recollect herbicide handling; or being in an area where there was ground or aerial spraying around base perimeters, or from boats, trucks, or helicopters; or being in an area that was previously sprayed. Self-reports of locations and dates of service have also been combined with a compilation of aerial spray data (Stellman and Stellman, 1986) to reconstruct an estimate of herbicide exposure. This approach, discussed in more detail below, is based on the assumption that Vietnam veterans recall the names and general time periods spent at various locations in Vietnam, but it is not clear how accurately this information can be reported many years later.
Records-Based Measures
Records-based classifications of the exposure of Vietnam veterans have been estimated by using information from military personnel records and historical documents on Vietnam service, branch of service, and military occupation. Levels of combat exposure have also been used as a surrogate measure of herbicide exposure. This military records-based approach is similar to reconstructing occupational exposure based on job classification. The inherent assumption is that specific jobs result in exposure or higher levels of exposure than other jobs.
Vietnam Service
As mentioned earlier, the advantage of using Vietnam service as an exposure measure is the simplicity of study design and analysis. The major limitation of Vietnam service as a measure of exposure is that not all men and women who served in Vietnam were actually exposed to herbicides or TCDD above global background levels, and therefore any epidemiologic study examining adverse health outcomes associated with herbicides or TCDD exposure would be diluted by the misclassification as exposed of individuals who served in Vietnam but were not exposed. Identifying such an outcome would therefore not be possible unless the exposure caused a very large excess in the incidence of disease.
Branch of Service
The rationale for using branch of service in classifying exposure is that the activities and locations of service of the Air Force, Army, Marines, and Navy varied, and therefore the potential for exposure to herbicides may also
vary. Among the factors that could differ among the four branches of service and potentially be related to the extent of exposure are troop mobility, the percentage of time spent outside of base camps, the availability of hygienic facilities, and the degree of reliance on local water and food. For example, it might be assumed that more mobile troops, spending more time in the field, having less access to hygienic facilities, and consuming more local food and water, would have greater potential exposures. However, those who operated primarily in the field cannot all be considered exposed because herbicide spraying was not distributed uniformly throughout the country. Also, support troops that operated from major bases might have been exposed because herbicides were used extensively around some base camps to remove tall grasses and heavy growth that obscured visibility (see Chapter 3). Since herbicides were used around base camps, military personnel could have received exposure in areas where no records of Operation Ranch Hand spraying exist. The violations of the simplifying assumptions would result in both false-positive and false-negative exposure assignments when "job classification" by branch of service is used. Classification of exposure by branch of service could be improved, for example, by stratifying Army troops by specialized duties or tactical operations and by including general geographical location information about individuals, units, or companies with estimates of the overall use of herbicides in those geographic areas.
Combat Experience
Classification using combat experience is based on the assumption that troops who had more frequent encounters with the enemy, and therefore engaged in combat more frequently, were more likely to enter areas that had been sprayed with herbicides. Since this was true only for some troops or regions in Vietnam, misclassification will occur. Military occupational specialties are sometimes used as a surrogate measure of combat. Individuals serving as riflemen or in artillery units are classified as having "combat-related" occupational specialties. Stratifying location of troops by geographic area, as well as by combat experience, as was suggested for the branch of service classification, would decrease exposure misclassification.
Military Occupation
Military occupation has been shown to be a valid exposure classification for two specific occupations that involved the direct handling and distribution of herbicides: first, the Air Force Ranch Hands, who were responsible for aerial spraying of herbicides and, second, the Army Chemical Corps, which performed ground and helicopter chemical operations. Biomarker
studies of the Ranch Hands are consistent with their exposure to TCDD as a group. When the Ranch Hand cohort was further classified by military occupation, a general increase in serum TCDD levels was detected with jobs that involved more frequent handling of herbicides. The median TCDD level for enlisted ground crew (24 ppt, range 0-618 ppt) was higher than the median level for enlisted flyers (18 ppt, range 0-196 ppt), and three times greater than the median level for officers (8 ppt, range 0-43 ppt) (AFHS, 1991).
The number of military personnel involved in these two units is small, however—approximately 1,000 for the Ranch Hands and, according to a recent abstract (Dalager and Kang, 1993), 2,954 for the Chemical Corps. Individuals serving in the Army Chemical Corps were also likely to be exposed to a number of chemicals other than herbicides, which can contribute to confounding in epidemiologic studies. Other classifications of general Vietnam veterans by military occupations are unlikely to improve the ability to predict exposure above what might be obtained from a category such as combat experience.
Reconstructing Estimated Exposure from Troop Location and Herbicide Spray Data
Another approach, the most detailed records-based classification scheme, matches troop location data with information on herbicide spray data from the HERBS and Services HERBS tapes to assign exposure categories based on the number of times a unit was within a defined time and distance from a documented herbicide spray. This strategy is similar to reconstructing occupational exposure based on the location of an employee within a plant during different time intervals and estimating the cumulative exposure associated with being in each location and performing specific job activities.
The available data on spraying missions have been compiled and stored on the HERBS and Services HERBS computer tapes. The HERBS tapes contain information on more than 6,500 herbicide missions and 17.6 million gallons of herbicides sprayed from August 1965 to February 1971. The tapes include information on the type of mission and the herbicide sprayed, the volume of herbicide used, the date of the herbicide mission, and the area sprayed. Data on helicopter defoliation missions are limited, and there is no information on ground herbicide sprays. Some of this information was compiled from various military documents and recorded on the Services HERBS tapes, prepared by the Department of Defense (DOD) Environmental Services Group (ESG). Because of the differences in reporting of Ranch Hand versus non-Ranch Hand sprays, it is likely that an unknown proportion of ground spray operations were not recorded. Further detail on the HERBS and Services HERBS tapes is provided in Chapter 3.
Data on herbicide sprays from the HERBS and Services HERBS tapes
can then be matched to information on troop location gathered either from self-reports of locations of military service or from military records. Military records, including morning reports, operational reports, and daily journals have been abstracted by the ESG for a subset of Army personnel to determine the location of their military units for a specified time period. Once individuals have been identified, information on their period of service can be verified from military personnel files archived at the National Personnel Records Center in St. Louis, Missouri.
Thus, at least in principle, one should be able to estimate the likelihood of herbicide exposure for a veteran or a military unit that served in Vietnam. This general approach has been tested, and different conclusions have been drawn as to the feasibility and utility of the estimates that result (see Appendix B). Like other exposure estimation strategies for retrospective studies, all of these strategies may be valid; however, they vary in their precision and in the degree to which they contribute to assessing a particular exposure-disease association.
The CDC Exposure Opportunity Index
The Centers for Disease Control (Erickson et al., 1984a,b) conducted a case-control study to determine if there was an increased risk of birth defects among the offspring of Vietnam veterans (see Chapter 9). The potential for an individual Vietnam veteran's exposure to Agent Orange ("exposure opportunity") was estimated by military records specialists of the Army Agent Orange Task Force without knowledge of case or control status. The exposure opportunity index (EOI) scores ranged from a value of 1 (minimum opportunities for exposure) to a value of 5 (most numerous opportunities for exposure). Higher values signify a greater likelihood of exposure but do not necessarily indicate a higher degree (duration or intensity) of exposure. An index score of 1, for example, was assigned to individuals who served in Vietnam prior to Agent Orange use or who were stationed offshore. Scores of 2 were assigned to men with noninfantry occupations stationed at selected bases with recorded perimeter spraying (e.g., a wireman serving in Chu Lai during 1968-1969). Individuals who served in selected noninfantry occupations at specified locations and times (e.g., an M.P. in Danang during 1968-1969) were allocated a score of 3. An index score of 4 was assigned to Army advisors serving during 1968-1969 and to infantry/combat arms serving in Tay Ninh in 1969 or 1970. The greatest opportunity for exposure, a score of 5, was assigned to Ranch Hand personnel and to individuals with service in specified areas at times of aborted Ranch Hand missions (during which herbicides may have been dumped on populated areas; for example, at Bien Hoa Air Force Base in July 1967 or November 1968) (see Table 6-1).
TABLE 6-1 Examples of Agent Orange Exposure Opportunity Index Scoresa
All individual veterans were given two index scores: one was derived from self-reported information on dates and location of service, and military duties, obtained during the interview; the second was developed based on a review of military records. The records-based EOI used unit location data determined from the Operational Report Lessons Learned (ORLL). The proximity of these general unit locations was compared to Agent Orange and other herbicide spray data by using the HERBS tapes and other data available on base perimeter sprays to construct the index scores.
Approximately 25 percent of interviewed Vietnam veterans reported that they had been exposed to Agent Orange. Fifty-two percent received the same score in both the index score and the self-reported Agent Orange exposure. A higher proportion of subjects who thought they had been exposed received scores of 4 or 5 than did subjects who thought they had not been exposed.
The CDC Agent Orange Study
In 1983, the Centers for Disease Control was assigned to conduct a study of the possible long-term health effects of Vietnam veterans' exposure to Agent Orange (see Chapter 2). The original design of the CDC Agent Orange Study (AOS) attempted to classify veterans' exposure to herbicides that occurred during military service. This was to be accomplished by determining the proximity of troops to Agent Orange spray using military records to track troop movement and the HERBS tapes to locate herbicide spraying patterns. Veterans' daily locations were to be abstracted from military records of company-sized units. The original study was to involve three cohorts, each containing approximately 8,500 men. Only Army units were to be studied. Two of the cohorts would consist of Army veterans serving at least 18 months in III Corps during 1967-1968. These cohorts would differ in their likelihood of herbicide exposure. The third cohort would be comprised of veterans from service support units stationed in other areas where there was evidence of little or no herbicide sprays. Participants were to have been draftees or single-term enlistees, had a rank of E1 through E5 at discharge, and served a single tour of duty in III Corps of Vietnam during 1967-1968. In addition, only individuals from infantry and artillery units were to be selected. These eligibility criteria and the exclusion of officers were intended to make the study groups as homogeneous as possible with respect to demographic characteristics and nonherbicide Vietnam-specific risk factors that could influence health.
The Environmental Support Group of DOD assisted CDC in the abstraction of military records on troop locations. According to the CDC protocol, 65 battalions were to be selected from III Corps. Herbicide exposure ''scores" would be calculated at the company (about 250 men) level,
based on a reported unit location occurring within a specified time and distance from a known herbicide application. Four different time and distance criteria were proposed for an encounter, or "hit," with Agent Orange: an application within 59 days and 2 km, within 1 day and 5 km, within 3 days and 7 km, or within 5 days and 7 km. Based on these analyses, the CDC initially concluded that "many veterans were in close enough proximity to applications of Agent Orange to be classified as highly likely to have been exposed to the herbicide" (CDC, 1985).
The CDC also proposed three exposure scores—E1 (short exposure), E2 (intermediate exposure), and E3 (chronic exposure)—to estimate an individual's likelihood of exposure. These scores attempted to account for variations in TCDD half-life, dispersion of herbicides, error in the calculated distances from spray lines, and uncertainties regarding the time between spraying and possible exposure, as well as whether the exposure could be viewed as acute, chronic, or intermediate. Preliminary results indicated substantial variability in exposure scores among units and among individual veterans (CDC, 1985).
The abstraction of military records is very labor-intensive, and a number of potential problems may arise. Some of the difficulties encountered by the ESG during its efforts for the AOS involved filling in gaps in the daily records of a unit's location; assigning a location to an individual who was not with the unit at the time of the recording; and tracking geographical dispersion among troops within any individual unit. There continues to be disagreement regarding how and whether these problems can be resolved (see Appendix B). Some participants in this research estimate, based on a 30 month study period for 35 different Army companies, that the location of company-sized units is available 85-90 percent of the time (Christian, 1992).
In testimony before the committee, varying views on these important points were presented, and the committee could neither verify nor refute these positions. The committee believes that the eligibility criteria developed for the study may, however, have decreased the range of exposures in the study and exacerbated the problem of missing troop location information.
In January 1986, a Science Panel of the White House Agent Orange Working Group (AOWG) was convened to evaluate the feasibility of the AOS. A subpanel was formed to review the records and information related to exposure assessment. The subpanel concluded that misclassification of an individual's exposure status was serious enough to warrant cancellation of the study. Two issues were specifically noted as influencing the degree of misclassification:
-
Unit dispersion—On a substantial number of days, personnel in combat units eligible for the AOS were not located together as a unit; rather they were dispersed geographically up to 20 km on any given day.
-
Incomplete records—An unknown, but apparently large, proportion of fire base perimeter spray operations were never recorded. The degree to which these "unrecorded" operations may have influenced exposure is unknown.
The panel recommended that "any study of ground troops, which is dependent on military records for the assessment of exposure to herbicides, not be conducted without an additional method to verify exposure" (emphasis added; Young et al., 1986).
The Stellmans' Study
Drs. Steven and Jeanne Stellman (1986) developed and published a similar but independent method for assessing veterans' exposure to herbicides in 1986. Because they did not have access to military troop location data, they relied on self-reports of locations and dates of service, but used the same HERBS tapes to identify spray missions. Like the CDC, they used these data to derive a series of exposure scores based on geographic and temporal proximity of a soldier to known spray missions. Several probabilistic exposure indices were developed to estimate exposure to herbicides for individual veterans. Three indices, C5, C10, and C15, provided counts of the number of times a veteran was located within a specified radius 5, 10, or 15 km from a recorded herbicide spray (there were no time restrictions). Three continuous exposure indices, E1, E2, and E3, accounted for the actual distance from each spray and for concurrent exposures plus potential exposures from residual herbicides from all previous spray missions that occurred nearby (Stellman and Stellman, 1993). Self-reported Agent Orange exposure was also determined based on a number of questions regarding whether subjects had sprayed, loaded, or handled herbicides, or had entered a sprayed area. According to the Stellmans, these measures "are to be regarded as approximations to population exposures in the same sense that past exposures to dusts and chemicals are often estimated for workers in industries for which industrial hygiene measurements of past conditions are not available" (Stellman and Stellman, 1986).
To test the method, the Stellmans selected a sample of 478 veterans participating in an outreach program sponsored by the Veterans Education Project of American University (Stellman and Stellman, 1986). They concluded that the approach was feasible and yielded estimates of exposure likelihood that would be useful in epidemiologic studies. Once again, no direct validation of the estimates was possible, but the indices appeared consistent with other measures or proxies of exposure.
The indices were also applied to an epidemiologic study of various health effects among a sample of 6,810 American Legionnaires (Stellman et
al., 1988). Those in the American Legion survey reporting a military occupation likely to involve heavy exposure (e.g., spraying herbicides from a helicopter, or loading or handling herbicides) had a higher mean exposure score than those reporting no direct exposure. Those who reported heavy combat experience were also more likely to have a higher estimated herbicide exposure. Because the exposure score was based on veterans' recall of dates and locations of service, rather than on recall of exposure per se, recall error was probably reduced.
Ranch Hand Exposure Index
The exposure index initially proposed in the Air Force Ranch Hand study relied upon military records of TCDD-containing herbicides (Agents Orange, Purple, Pink, and Green) sprayed as reported in the HERBS tapes for the period after July 1965, and military procurement records and dissemination information for the period prior to July 1965. A TCDD weighting factor (based on the concentration of TCDD in the herbicide and the time of spraying) was applied to the number of gallons of herbicides sprayed during each subject's tour of duty in Vietnam. The dates of each subject's tour(s) in Vietnam were determined by a manual review of military records. The HERBS tapes were used with quarterly operations reports to construct a table of gallons of TCDD-containing herbicides sprayed for each month during the Ranch Hand operation.
The exposure index for a Ranch Hand was defined as the product of the TCDD weighting factor and the number of gallons of TCDD herbicides sprayed during his tour of duty, divided by the number of Ranch Hands sharing such duties during his tour. The weighting factor served to separate from the total those who served in Vietnam prior to July 1965, a period in which TCDD levels in herbicides were known to be higher. Exposure levels were defined for five occupational categories: (1) officer-pilot, (2) officer-navigator, (3) officer-nonflying, (4) enlisted flyer, and (5) enlisted ground crew. Each Ranch Hand was placed in an exposure category (high, medium, or low) based on the value of the individual's exposure index.
This index was intended to distinguish between groups of individuals who served in Operation Ranch Hands units rather than as a measure of individual exposure. The measure did not account for individual variation in work habits or job duties—all enlisted flyers, for example, serving a similar tour of duty, received the same exposure classification. As noted in Chapter 3, concentrations of TCDD varied across herbicide lots—this index assumed that the relative concentration of TCDD was 2 ppm in Agent Orange, 33 ppm in Agent Purple, and 66 ppm in Agents Pink and Green. The index includes exposure from recorded Ranch Hand sprays only—the measure did not allow for other unrecorded herbicide exposures, such as chemical dumps
or perimeter sprays, or other non-Ranch Hand herbicide applications. In 1991, the exposure index was compared to the results of the Ranch Hand serum TCDD analysis. The exposure index and the TCDD body burden were weakly correlated.
Biomarkers of Exposure in Vietnam
Gross and colleagues (1984) reported on the results of a preliminary study designed to determine whether elevated levels of TCDD could be detected in adipose tissue of 20 Vietnam veterans who reported being exposed to Agent Orange. Investigators used gas chromatography and high-resolution mass spectrometry to measure dioxin levels in adipose tissue surgically removed from the abdominal area of volunteers. Three veterans who reported heaviest exposure had the highest levels of TCDD, ranging from 20 to 173 ppt. Veterans reporting less exposure did not differ appreciably in TCDD levels from a control group of veterans who had not served in Vietnam; mean TCDD levels in these groups were 5 to 6 ppt.
Several other studies to measure dioxin levels in adipose tissue of Vietnam veterans have been conducted. Some have found elevated TCDD levels among veterans thought to be exposed to herbicides (Schecter et al., 1987; Kahn et al., 1988), whereas others have not (Kang et al., 1991).
In the Pointman I Project (Kahn et al., 1988), conducted by the New Jersey Agent Orange Commission, 10 Vietnam veterans reported to be heavily exposed to Agent Orange were compared to matched controls in both blood and adipose tissue levels of TCDD. Nine of the ten veterans had handled herbicides regularly—five were members of Operation Ranch Hand, one was an Air Force freight handler, two were Army Chemical Corps specialists, and one was an Army helicopter crew chief who flew herbicide spray missions. Ten nonexposed Vietnam veterans and seven Vietnam-era veterans who did not serve in Vietnam were selected as controls. The mean TCDD level in the exposed men was reported to be about tenfold higher than the 4-5 ppt for controls. The levels of TCDD in the exposed men exceeded those in the pooled control subjects in both blood (p 8 .01) and adipose tissue (p 8 .001). Furthermore, blood TCDD levels were highly correlated with adipose tissue levels (r = +.89).
In an extension of the Pointman I study, Pointman II, researchers measured serum TCDD levels in 55 Vietnam veterans who did not handle herbicides directly but were assigned to areas known to be heavily sprayed and in 15 unexposed Vietnam veteran controls. The exposed veterans were representatives of an Army cavalry unit that served in III Corps, a Marine unit from I Corps, and a brown water Navy riverboat crew from IV Corps. The Marines had a mean serum TCDD level of 5 ppt, and the Navy riverine subjects had a mean level of 4 ppt, compared with a mean level of 3 ppt for
the controls. Army personnel had a mean level similar to the controls. Although some observers have argued that all of these measured TCDD levels are within a normal "background" level, project scientists maintain that the results suggest apparent group differences in military service characteristics that are important (Kahn et al., 1992; Lewis, 1993).
The Air Force Ranch Hand study examined serum TCDD levels in 866 Ranch Hands compared to 804 controls, who were Air Force veterans involved in C-130 aircraft missions in Southeast Asia (AFHS, 1991, vol. 1). Ranch Hands as a group had higher serum levels (median = 13 ppt, range 0-618 ppt) than comparison subjects (median = 4 ppt, range 0-55 ppt). Among Ranch Hands, there was a gradient for TCDD level: enlisted ground crew had the highest levels (median = 24 ppt, range 0-618 ppt), followed by flying enlisted personnel (median = 18 ppt, range 0-196 ppt) and then by officers (median = 8 ppt, range 0-43 ppt). All medians in the comparison group similarly stratified by rank and job were less than 5 ppt.
CDC Agent Orange Validation Study (AOVS)
To test the validity of several indirect methods for estimating exposure of ground troops to Agent Orange in Vietnam, in 1987 the CDC measured serum TCDD levels in a nonrandom sample of Vietnam veterans and Vietnam era veterans who did not serve in Vietnam (CDC, 1988b). Participants were chosen from a pool of 65 Army battalions that operated 18 months or longer in III Corps, a heavily sprayed region around Saigon, during 1967 and 1968. From this pool, veterans who served in combat companies, were discharged at pay grade E5 or lower, and had served a single term of enlistment, were included. Vietnam era veterans were selected from among those previously interviewed for the CDC's Vietnam Experience Study (but were not chosen for the physical examination). Men in the Vietnam cohort were chosen from among those who had served in a unit for which the ESG had locational data (as determined for the Agent Orange Study). Vietnam veterans were selected for further study based on their estimated number of Agent Orange hits, derived from the number of days for which at least one company location was within 2 km and 6 days of a recorded Agent Orange spray; the "low" exposure group included 298 veterans, the "medium" exposure group included 157 veterans, and 191 were included in the "high" exposure group. Blood samples were obtained from 66 percent of Vietnam veterans ( N = 646) and 49 percent of the eligible comparison group of veterans (N = 97). More than 94 percent of those whose serum was obtained had served in one of five battalions.
Five indirect exposure scores based on military records and two scores based on self-reports were used to rank veterans according to their likelihood of exposure to Agent Orange. The five indirect scores incorporated a
variety of assumptions concerning possible sources of TCDD exposure, the estimated half-life of TCDD in the environment, and the completeness of data on troop and spray location. Two Agent Orange exposure scores were calculated based on proximity to recorded Agent Orange sprays. Two similar scores were computed for recorded sprays of "unknown" agents. The fifth score, an area score, depended less on precise military unit location data than the other four scores. It was computed based on the number of days a company was in one of five heavily sprayed areas in III Corps during 1967 and 1968. Two self-assessed exposure scores were determined based on the number of days an individual reported direct and indirect exposure to herbicides during military service (CDC, 1988b).
The median TCDD level in Vietnam veterans was 4 ppt, with a range from less than 1 to 45 ppt and two having levels greater than 20 ppt; the distributions of these measurements were nearly identical to those for the control group of 97 Vietnam era veterans (Figure 6-2). None of the records-derived estimates of exposure, and neither type of self-reported exposure to herbicides, identified Vietnam veterans who were likely to have currently elevated serum TCDD levels (CDC, 1988b). The study concluded it is unlikely that military records can be used to identify a large number of U.S. Army veterans who might have been heavily exposed to TCDD in Vietnam.
When restricting the AOVS analysis to veterans with TCDD levels in the upper quartile and decile, the Stellmans (1993) reported a correlation between serum TCDD levels and military records-based scores. This finding results from one veteran whose serum TCDD concentration was 45 ppt. This veteran had an exposure score of 115 (1 Agent Orange hit and 114 unknown agent hits).
A draft of the AOVS was reviewed by an Institute of Medicine advisory committee in 1987 (IOM, 1987). The committee raised the concern that current serum levels (for this study, obtained in 1987) may not be related to an actual exposure that occurred some 20 years previously. Four possible scenarios for observing "background" levels in three hypothetical groups of Vietnam veterans (high, medium, and low exposure) who served in Vietnam in 1967 were suggested (see Figure 6-3): (1) All three veteran groups in the study could have been similarly exposed during the war, but their levels were not sufficiently high in the late 1960s to prevent them from decaying to background level in 1987. (2) All three exposed veteran groups could have had significantly higher blood levels than background before they were potentially exposed in Vietnam, but with the passage of time, all TCDD concentrations had decayed to similar levels. (3) The three groups could have been differentially exposed in Vietnam, and if their serum had been sampled then, variation in TCDD levels would have been detected. However, differences in decay rates and initial concentrations could result in all
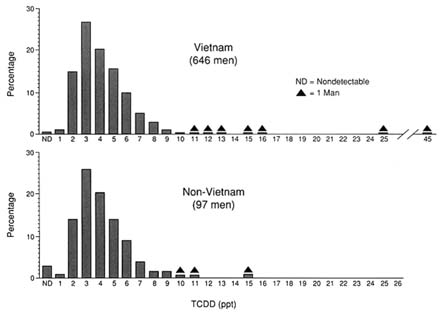
FIGURE 6-2 Distribution of serum dioxin levels in Vietnam veterans and non-Vietnam (Vietnam era) veterans, CDC Validation Study. Reprinted with permission from CDC, 1989a.
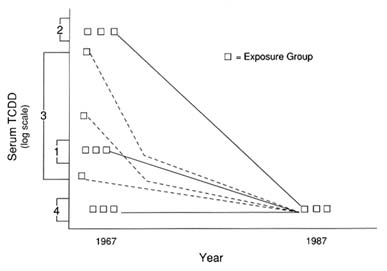
FIGURE 6-3 Possible scenarios for describing the relationship between current and prior serum TCDD levels, 1967-1987. SOURCE: Institute of Medicine, 1987.
demonstrating current levels at background. (4) None of the veterans ever had serum TCDD concentrations above background.
Without adequate knowledge of the decay rate and pharmacokinetics of TCDD in humans, it is not possible to distinguish among the alternative explanations. If initial concentrations of TCDD differed but then over the years fell to background, estimates based on reconstruction of troop locations and herbicide spraying activities might be more reliable indicators of exposure than current serum TCDD levels.
ESTIMATES OF EXPOSURE TO HERBICIDES AND TCDD DURING VIETNAM SERVICE
As the committee's review of the literature indicates, exposure assessment has been a weak aspect of epidemiologic studies of Vietnam veterans. Different approaches have been used in estimating the exposure of Vietnam veterans, and these studies generally rely on self-reported exposures, records-based exposure estimates, or biomarkers of TCDD exposure. Each of these approaches is limited in its ability to determine precisely the degree of individual exposure. Some studies rely on gross markers such as service in Vietnam, perhaps enhanced by branch of service, military region, military specialty, or exposure to combat as proxies for exposure to herbicides. The CDC Vietnam Experience Study and Selected Cancers Study, the Department of Veterans Affairs (DVA; formerly the Veterans Administration) mortality studies, and most state veterans studies are of this sort. This approach almost surely dilutes whatever health effects of herbicides exist. At the other extreme, some studies rely on fine details of military records on troop movements and herbicide spraying, perhaps combined with self-reported retrospective data, for individuals or small units on a daily basis. The Stellmans' study and the proposed but not completed CDC AOS are examples of this sort. Even though measures of this type may be accurate for many individuals, such fine detail may exceed the accuracy of a record system not designed for this purpose, and the accuracy of the resulting exposure measure cannot be guaranteed for all potential subjects.
Serum TCDD measurements are difficult to interpret with current knowledge, yet they may provide valuable information in some situations. They are most useful in defining differences in exposure levels among groups rather than among individuals, and particularly between more highly exposed groups and less exposed or unexposed groups. The groups must be of sufficient size to provide the statistical power to make meaningful assessments. In general, group differences in serum levels are likely to be reflective of differences in true past exposure to TCDD, but the failure to detect mean differences does not necessarily indicate that there were no differences in past exposure.
Current serum TCDD levels in Vietnam veterans, with the exception of measured levels in Ranch Hand veterans, suggest that exposure to TCDD in Vietnam was substantially less, on average, than that of occupationally exposed workers or of persons exposed as a result of the industrial explosion in Seveso, Italy (see Figure 6-4). The CDC's Validation Study found that study subjects could not be distinguished from controls based on serum levels. The median TCDD levels for Vietnam veterans and Vietnam era veterans were both 4 ppt. Serum TCDD levels obtained from Ranch Hand veterans in 1987, however, were able to distinguish among groups of Ranch Hand veterans (based on their military duties) with different prior exposures to TCDD, although the median levels for the Ranch Hand groups were lower than those of several occupational cohorts and Seveso residents. The NIOSH study of 253 workers measured serum TCDD 23 years (on average) after their occupational exposure. Therefore, although definitive data are lacking, the available evidence concerning herbicide exposure suggests that as a group, Vietnam veterans had lower exposure to TCDD than the subjects exposed occupationally or in Seveso.
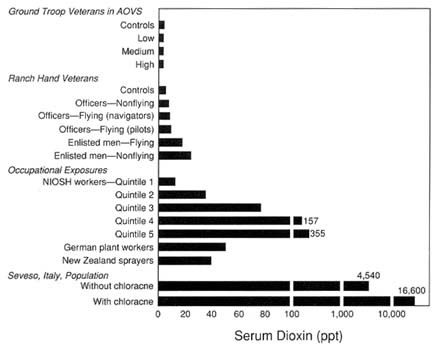
FIGURE 6-4 Median serum dioxin levels in selected populations. Reprinted with permission from Pirkle, 1993.
Historical data, personal testimony, and other information made available to the committee suggest that there was substantial variability in an individual's wartime experience, and that opportunities for exposure are also likely to have varied (see Chapter 3). Approximately 1.5 million U.S. military personnel served in South Vietnam from 1967 through 1969, the period of heaviest herbicide use; according to Air Force operations data, nearly 14 million gallons of herbicide were sprayed over Vietnam during this period. In addition to Ranch Hands, an unknown number of veterans were likely to have received varying degrees of exposure to herbicides. Localized spraying, while conducted on a smaller-scale than Ranch Hand spraying, may have resulted in higher degrees of exposure since it was done in close proximity to ground troops. Most military bases had vehicle-mounted and backpack spray units available for use in routine vegetation control programs. Approximately 750 personnel have been followed who were assigned to the Army Chemical Corps. They were responsible for the storage, preparation, and application of chemicals around the perimeters of base camps and, as a group, were likely to have been more highly exposed. U.S. Army Special Forces camps were often located in enemy territory throughout Vietnam and may have been in the target area of a Ranch Hand mission. Formation of Special Forces camps or fire support bases often required defoliation, so soldiers in these units may have been exposed to high levels of herbicides. Navy riverine units operating along the river and canals of the Mekong delta reportedly sprayed base perimeters with herbicides (Zumwalt, 1993).
Oral and written testimony presented before the committee also suggests that substantial numbers of veterans were exposed to Agent Orange and other herbicides. Results from a survey of U.S. military officers in Vietnam, across all branches of service (Army battalion commanders and advisors, Navy personnel and advisors involved in riverine operations, Air Force Ranch Hand personnel, chemical officers, Marine ground and air personnel, and advisors) indicated the utility of herbicides to their military operations (U.S. Army, 1972). In addition, surveys of veterans indicate that 25 to 55 percent believe they were exposed to herbicides while serving in Vietnam (Erickson et al., 1984a,b; Stellman and Stellman, 1986; CDC, 1989b). It appears that groups of veterans, other than those involved in Operation Ranch Hand, were likely to have been exposed to herbicides during their service in Vietnam.
It is clear that the military use of herbicides in Vietnam was not uniform either spatially or temporally, and that the movement and behavior of troops also varied, so one cannot assume that all troops were equally exposed to herbicides. In the committee's judgment, a sufficiently large range of exposures may exist among Vietnam veterans to conduct a valid epidemiological study for certain health outcomes. The difficulty (from the perspective
of epidemiological studies) is that the available data do not precisely quantify individual exposure. None of the measures that the committee has reviewed would be free of nondifferential misclassification bias. The effect of this bias on risk estimates would likely be to underestimate true effects if they existed, possibly to such an extent that these effects could be missed entirely by future studies. Thus, the committee recommends (see Chapter 12) the following set of activities.
EXPOSURE RECONSTRUCTION
The committee believes that it may be possible to develop a valid exposure reconstruction model for epidemiological studies based on existing records and structured interview data. Such a model would estimate the likelihood that each individual veteran was exposed to herbicides in Vietnam and could possibly quantify the likely degree of exposure. This model would incorporate information in existing military records about herbicide spraying (the HERBS and Services HERBS tapes) and troop movements. It would also include less formal sources of information on ground and perimeter spraying from records of herbicide shipments to various military bases, and would consider the type of terrain, typical foliage of the locations, and military mission of the bases and troops located there. Surveys and interviews of Vietnam veterans, stratified by location and period of service, might also provide useful information on situations in which herbicide spraying was prevalent and, if validated, may be incorporated into the exposure reconstruction model.
Development of the Exposure Reconstruction Model
This new effort would model the conditions under which herbicides were used in Vietnam and should consider the following information:
-
troop location—based on all available military records, including morning reports, daily journals, situation reports, intelligence summaries, ORLL, and combat operations after action reports;
-
aerial spray mission data—from the HERBS and Services HERBS tapes;
-
estimated ground spraying activity—not based solely on existing records (discussed in more detail below);
-
estimated exposure opportunity factors—including the identification of occupations involving the handling of herbicides, and considerations of how likely troops were to have heavier exposure through eating local food, bathing in or drinking local water, contact with contaminated soil, etc.;
-
military indications for herbicide use—systematic, historical reviews
-
of the conditions under which military use of herbicides was warranted, including information on typical use patterns in those situations such as the Army survey of military commanders conducted in 1971 (U.S. Army, 1972); and,
-
considerations of the composition and environmental fate of herbicides —including changes in the TCDD content of herbicides over time, the persistence of TCDD and herbicides in the environment, and the degree of likely penetration of the herbicides into the ground.
Once an exposure reconstruction model based on these data has been developed, it should be possible to estimate an exposure score for the large numbers of veterans needed for epidemiologic studies.
Considerable research has also been conducted on the environmental fate and persistence of TCDD and this information could be incorporated into the exposure model. TCDD deposited on foliage and the surface of soil could be photodegraded within a matter of days. Volatization could also be a significant removal mechanism. Once TCDD penetrates the surface, it could remain stationary for an indefinite period. It could persist essentially immobile in the soil for years. There is little translocation of TCDD within plants, although plant roots in contact with TCDD contaminated soil would, in turn, be contaminated.
Ground spraying, although probably representing a smaller quantity of herbicide than aerial spraying, may actually have resulted in heavier human exposures since it probably was done in closer proximity to ground troops, at higher application rates (i.e., number of gallons per acre), and potentially by less trained individuals than Ranch Hand spraying. This spraying was often performed around camp perimeters and along communication routes in response to enemy attack or during relocation to a new fire base. Thus, troops may often have remained in the area during spraying or passed through it soon afterward. Ground spraying could reportedly be approved by unit commanders at the Corps level, and the spraying does not appear to have been documented as carefully as the aerial missions of Air Force Ranch Hands. To incorporate estimates of exposure from ground spraying, the committee suggests the development of a subsidiary estimation model for this source of exposure. Because ground spraying data are incomplete, this model would be based on a series of factors likely to determine the extent of ground spraying in Vietnam. From testimony presented to the committee, it appears that the following factors might have been important determinants of this activity: region and date of military service, terrain, type of vegetation, intensity of enemy activity, any existing data on shipment of herbicides to different regions, and size of military base. Through structured interviews with military personnel who served in various regions and various military units, data would be gathered to permit the development of
a model predicting the likelihood and intensity of ground spraying around base perimeters, fire camps, and along roads for different areas and times. A partial check on the utility of this model could be accomplished by comparing its predictions to the limited ground spraying data that do exist in the Services HERBS tapes. This proposal incorporates ideas for exposure reconstruction previously described by others (Bricker, 1981; Erickson et al., 1984a; Stanton, 1989; Lewis, 1993).
Evaluation of the Exposure Reconstruction Model
The overall exposure reconstruction model can be evaluated in several ways. First, model developers should determine whether the data used in the exposure reconstruction model are internally consistent. This involves checking whether existing spraying records indicate more spraying in areas where it was likely to have been militarily useful from the point of view of terrain, foliage, and military mission. It would also be possible to cross-check the estimated spraying intensity data with a systematic survey of the recollections of veterans who served in particular areas.
In a second method of evaluation, exposure estimates based on the reconstruction model should be compared with serum TCDD measurements for a random sample of veterans, stratified according to records-based measures. Although the committee concludes that group differences can be useful in confirming that exposure measures reflect the differences in prior exposure, the absence of group differences cannot be interpreted to indicate that groups were not exposed earlier. Serum TCDD measurements should not, however, be regarded as a gold standard—a perfect measure of herbicide exposure. In addition to the problems with interpreting serum TCDD measures discussed above, some of the herbicides used in Vietnam such as Agent White did not contain TCDD, so it is possible for a veteran to have been exposed to a large amount of Agent White without having an elevated serum TCDD level at any time.
A third evaluation of the exposure estimation strategy would be to assess the association between the exposure reconstruction estimates and the incidence of health outcomes that are truly associated with herbicides. One would expect a positive association between the exposure reconstruction measure and those outcomes found in this report to have sufficient evidence for a statistical association. For instance, the non-Hodgkin's lymphoma data from the CDC Selected Cancers Study (CDC, 1990a) and the DVA case-control study (Dalager et al., 1991) can be combined and exposure measures calculated on the basis of the new exposure reconstruction model. Because there are sufficient data from occupational studies to suggest an association between herbicides and/or TCDD and NHL (see Chapter 8), one would expect to see a positive association between this cancer and the new
exposure reconstruction model data in the combined cases from the CDC and DVA studies. If such an association were found, it could be interpreted as positive evidence for the validity of the new exposure model. If no association were found, it would not be clear whether this was due to problems in the new exposure measure, to small sample sizes or low average herbicide exposure even in those exposed, or to the lack of a real association between herbicides and NHL.
Exposure reconstruction models for herbicides in Vietnam must be thoroughly evaluated before epidemiologic studies based upon them proceed. Chapter 12 contains recommendations about how this evaluation should be carried out by an independent, nongovernmental scientific panel with expertise in historic exposure reconstruction and epidemiology, and how the resulting exposure measures should be used in a program of epidemiologic studies of Vietnam veterans.
SUMMARY
The existing epidemiologic data base reviewed by the committee is severely lacking in quantitative measures of individual exposure to herbicides and TCDD. Assessment of the intensity and duration of individual exposures is a key component in determining whether specific health outcomes are associated with exposure to TCDD or other chemicals found in the herbicides used in Vietnam. Although different approaches have been used to estimate exposure in Vietnam veterans and in others exposed occupationally or environmentally, each of the approaches is limited in its ability to determine precisely the degree and level of individual exposure. New biochemical techniques that can detect small amounts of TCDD in the blood many years after exposure have some merit, especially for detecting group differences. However, because of common background exposures to TCDD, poorly understood variations among individuals in TCDD metabolism, and relatively large measurement errors, individual TCDD serum levels are usually not meaningful. Furthermore, because not all of the herbicides used in Vietnam contained TCDD, serum TCDD levels are not good indicators of overall exposure to herbicides.
Although definitive data are lacking, the available quantitative and qualitative evidence about herbicide exposure suggests that Vietnam veterans as a group had substantially lower exposure to herbicides and TCDD than the subjects in many occupational studies. The participants in Operation Ranch Hand are an exception to this pattern, and it is likely that others among the approximately 3 million men and woman who served in Vietnam were exposed to herbicides at levels associated with health effects. It is clear that the military use of herbicides in Vietnam was not uniform either geographically or temporally, and that the movement and behavior of troops also
varied, so one cannot assume that all troops were equally exposed to herbicides. Thus, in the committee's judgment, a sufficiently large range of exposures may exist among Vietnam veterans to conduct a valid epidemiologic study for certain health outcomes. The difficulty (from the perspective of epidemiologic studies) is that the available data do not precisely quantify individual exposure. None of the measures that the committee has reviewed would be free of nondifferential misclassification bias. The effect of this bias on risk estimates would likely be to underestimate true effects if they existed, possibly to such an extent that these effects could be missed entirely by future studies.
The committee believes that it may be possible to develop a valid exposure reconstruction model for epidemiologic studies based on existing records and structured interview data, using principles of historic exposure reconstruction developed by industrial hygienists. Such a model would estimate the likelihood that each individual veteran was exposed to herbicides in Vietnam, and could possibly quantify the likely degree of exposure. This model would incorporate information in existing military records about herbicide spraying (the HERBS and Services HERBS tapes) and troop movements. It would also include less formal sources of information on ground and perimeter spraying from records of herbicide shipments to various military bases, and would consider the type of terrain, typical foliage of the locations, and the military mission of the bases and troops located there. Surveys and interviews of Vietnam veterans, stratified by location and period of service, might also provide useful information on situations in which herbicide spraying was prevalent and, if validated, may be incorporated into the exposure reconstruction model.
REFERENCES
Air Force Health Study (AFHS). 1991. An Epidemiologic Investigation of Health Effects in Air Force Personnel Following Exposure to Herbicides. Serum Dioxin Analysis of 1987 Examination Results. Brooks AFB, TX: USAF School of Aerospace Medicine. 9 vols.
Albanese RA. 1991. The chemical 2,3,7,8-tetrachlorodibenzo-p-dioxin and U.S. Army Vietnam-era veterans. Chemosphere 22:597-603.
Andrews JS Jr, Garrett WA Jr, Patterson DG Jr, Needham LL, Roberts DW, Bagby JR, Anderson JE, Hoffman RE, Schramm W. 1989. 2,3,7,8-Tetrachlorodibenzo-p-dioxin levels in adipose tissue of persons with no known exposure and in exposure persons. Chemosphere 18:499-506.
Armstrong BG, Oakes D. 1982. Effects of approximation in exposure assessments on estimates of exposure-response relationships. Scandinavian Journal of Work, Environment, and Health Supplement 1:20-23.
Axelson O, Westberg H, eds. 1992. First seminar on occupational exposure assessment: on the concepts of exposure and dose. American Journal of Industrial Medicine 21:1-132.
Bertazzi PA, Zocchetti C, Pesatori AC, Guercilena S, Sanarico M, Radice L. 1989. Ten year mortality study of the population involved in the Seveso incident in 1976. American Journal of Epidemiology 129:1187-1200.
Birkett NJ. 1992. Effect of nondifferential misclassification on estimates of odds ratios with multiple levels of exposure. American Journal of Epidemiology 136:356-362.
Blair A, White DW. 1985. Leukemia cell types and agricultural practices in Nebraska. Archives of Environmental Health 40:211-214.
Blair A, Stewart P. 1992. Do quantitative exposure assessments improve risk estimates in occupational studies of cancer? American Journal of Industrial Medicine 21:53-63.
Blair A, Grauman DJ, Lubin JH, Fraumeni JF Jr. 1983. Lung cancer and other causes of death among licensed pesticide applicators. Journal of the National Cancer Institute 71:31-37.
Bond GG, Cook RR, Brenner FE, McLaren EA. 1987. Evaluation of mortality patterns among chemical workers with chloracne. Chemosphere 16:2117-2121.
Bond GG, McLaren EA, Brenner FE, Cook RR. 1989. Incidence of chloracne among chemical workers potentially exposed to chlorinated dioxins. Journal of Occupational Medicine 31:771-774.
Bond GG, Bodner KM, Olsen, GW, Burchfiel CM, Cook RR. 1991. Validation of work histories for the purpose of epidemiological studies. Applied Occupational and Environmental Hygiene 6:521-527.
Brenner H. 1992. Notes on the assessment of trend in the presence of nondifferential exposure misclassification. Epidemiology 3:420-427.
Bricker JG. 1981. Proposed Agent Orange Troop Exposure and Non-Exposure Cohort Selection Concept Paper. Memorandum to the Chairman, AOWG Science Panel. December 4, 1981. Washington, DC: Office of the Assistant Secretary of Defense, Health Affairs.
Bueno de Mesquita HB, Doornbos G, van der Kuip DA, Kogevinas M, Winkelmann R. 1993. Occupational exposure to phenoxy herbicides and chlorophenols and cancer mortality in the Netherlands. American Journal of Industrial Medicine 23:289-300.
Burmeister LF. 1981. Cancer mortality in Iowa farmers: 1971-1978. Journal of the National Cancer Institute 66:461-464.
Byard JL. 1987. The toxicological significance of 2,3,7,8-tetrachlorodibenzo-p-dioxin and related compounds in human adipose tissue. Journal of Toxicology and Environmental health 22: 381-403.
Cantor KP. 1982. Farming and mortality from non-Hodgkin's lymphoma: a case-control study. International Journal of Cancer 29:239-247.
Carmelli D, Hofherr L, Tomsic J, Morgan RW. 1981. A Case-Control Study of the Relationship Between Exposure to 2,4-D and Spontaneous Abortions in Humans. SRI International. Prepared for the National Forest Products Association and the U.S. Department of Agriculture, Forest Service.
Centers for Disease Control (CDC). 1985. Agent Orange Projects Interim Report Number 2: Exposure Assessment for the Agent Orange Study. Atlanta: CDC, Center for Environmental Health, Division of Chronic Disease Control, Agent Orange Projects.
Centers for Disease Control. 1988a. Preliminary report: 2,3,7,8-tetrachlorodibenzo-p-dioxin exposure in humans-Seveso, Italy. Morbidity and Mortality Weekly Report 37:733-736.
Centers for Disease Control. 1988b. Serum 2,3,7,8-tetrachlorodibenzo-p-dioxin levels in U.S. Army Vietnam-era veterans. Journal of the American Medical Association 260:1249-1254.
Centers for Disease Control. 1989a. Comparison of Serum Levels of 2,3,7,8-Tetrachlorodibenzo-p-Dioxin with Indirect Estimates of Agent Orange Exposure Among Vietnam Veterans: Final Report. Atlanta: U.S. Department of Health and Human Services.
Centers for Disease Control. Vietnam Experience Study. 1989b. Health Status of Vietnam Veterans. Atlanta: U.S. Department of Health and Human Services. Vols. I-V, Supplements A-C.
Centers for Disease Control. 1990a. The association of selected cancers with service in the U.S. military in Vietnam. I. Non-Hodgkin's lymphoma. Archives of Internal Medicine 150:2473-2483.
Centers for Disease Control. 1990b. The association of selected cancers with service in the U.S. military in Vietnam. III. Hodgkin's disease, nasal cancer, nasopharyngeal cancer, and primary liver cancer. Archives of Internal Medicine 150:2495-2505.
Checkoway H. 1986. Methods of treatment of exposure data in occupational epidemiology. medicina del Lavoro 77:48-73.
Christian R. 1992. Records-Based Measures of Exposure to Herbicides in Vietnam Veterans. Statement to the Institute of Medicine Committee to Review the Health Effects in Vietnam Veterans of Exposure to Herbicides. December 8, 1992.
Coggon D, Pannett B, Winter PD, Acheson ED, Bonsall J. 1986. Mortality of workers exposed to 2-methyl-4-chlorophenoxyacetic acid. Scandinavian Journal of Work, Environment, and Health 12:448-454.
Coggon D, Pannett B, Winter P. 1991. Mortality and incidence of cancer at four factories making phenoxy herbicides. British Journal of Industrial Medicine 48:173-178.
Collins JJ, Strauss ME, Levinskas GJ, Conner PR. 1993. The mortality experience of workers exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin in a trichlorophenol process accident. Epidemiology 4:7-13.
Constable JD, Hatch MC. 1985. Reproductive effects of herbicide exposure in Vietnam: recent studies by the Vietnamese and others. Teratogenesis, Carcinogenesis, and Mutagenesis 5:231-250.
Cook RR, Townsend JC, Ott MG, Silverstein LG. 1980. Mortality experience of employees exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). Journal of Occupational Medicine 22:530-532.
Cook RR, Bond GG, Olson RA. 1986. Evaluation of the mortality experience of workers exposed to the chlorinated dioxins. Chemosphere 15:1769-1776.
Dai LC, Phuong NTN, Thom LH, Thuy TT, Van NTT, Cam LH, Chi HTK, Thuy LB. 1990. A comparison of infant mortality rates between two Vietnamese villages sprayed by defoliants in wartime and one unsprayed village. Chemosphere 20:1005-1012.
Dalager NA, Kang HK. 1993. Mortality among Army Chemical Corps Vietnam veterans occupationally exposed to herbicides. Abstracts of the 26th Annual Meeting of the Society for Epidemiologic Research. Keystone, CO. June 16-18, 1993.
Dalager NA, Kang HK, Burt VL, Weatherbee L. 1991. Non-Hodgkin's lymphoma among Vietnam veterans. Journal of Occupational Medicine 33:774-779.
Del Corno G, Montesarchio E, Fara GM. 1985. Problems in the assessment of human exposure to tetrachlorodibenzodioxin (TCDD): the marker chloracne. European Journal of Epidemiology 1:139-144.
Delpizzo V. 1992. An apparently incongruous exposure-response relationship resulting from the use of job description to assess magnetic field exposure. Scandinavian Journal of Work, Environment, and Health 18:242-245.
Dement JM, Harris RL, Symons MJ, Shy CM. 1983. Exposures and mortality among chrysotile asbestos workers . Part II, Mortality. American Journal of Industrial Medicine 4:421-433.
Devine OJ, Karon JM, Flanders WD, Needham LL, Patterson DG Jr. 1990. Relationships between concentrations of 2,3,7,8-tetrachlorodibenzo-p-dioxin serum and personal characteristics in U.S. Army Vietnam veterans. Chemosphere 20:681-691.
Dosemici M, Wacholder S, Lubin JH. 1990. Does nondifferential misclassification of exposure always bias a true effect toward the null value? American Journal of Epidemiology 132:746-748.
Erickson JD, Mulinare J, McClain PW, Fitch TG, James LM, McClearn AB, Adams MJ. 1984a. Vietnam Veterans' Risks for Fathering Babies with Birth Defects. Atlanta: U.S. Department of Health and Human Services, Centers for Disease Control.
Erickson JD, Mulinare J, McClain PW, Fitch TG, James LM, McClearn AB, Adams MJ Jr. 1984b. Vietnam veterans' risks for fathering babies with birth defects. Journal of the American Medical Association. 252:903-912.
Ferry DG, Gazeley LR, Edwards IR. 1982. 2,4,5-T absorption in chemical applicators. Proceedings of the University Otago Medical School 60:31-34.
Fingerhut MA, Sweeney MH, Patterson DG Jr, Piacitelli LA, Morris JA, Marlow DA, Hornung RW, Cameron LW, Connally LB, Needham LL, Halperin WE. 1989. Levels of 2,3,7,8-TCDD in the serum of U.S. chemical workers exposed to dioxin-contaminated products: Interim results. Chemosphere 19:835-840.
Fingerhut MA, Halperin WE, Marlow DA, Piacitelli LA, Honchar PA, Sweeney MH, Greife AL, Dill PA, Steenland K, Suruda AJ. 1991. Cancer mortality in workers exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin. New England Journal of Medicine 324:212-218.
Flanders WD, Lin L, Pirkle JL, Caudill SP. 1992. Assessing the direction of causality in cross-sectional studies. American Journal of Epidemiology 135:926-935.
Flegal KM, Keyl PM, Nieto FJ. 1991. Differential misclassification arising from nondifferential errors in exposure measurement. American Journal of Epidemiology 134:1233-1240.
Fleiss JL, Shrout PE. 1977. The effects of measurement errors on some multivariate procedures. American Journal of Public Health 67:1188-1191.
Frank R, Campbell RA, Sirons GJ. 1985. Forestry workers involved in aerial application of 2,4-dichlorophenoxyacetic acid (2,4-D): exposure and urinary excretion. Archives of Environmental Contamination and Toxicology 14:427-435.
Gamble J, Spirtas R. 1976. Job classification and utilization of complete work histories in occupational epidemiology. Journal of Occupational Medicine 18:399-404.
Gerin M, Siemiatycki J. 1991. The occupational questionnaire in retrospective epidemiologic studies: recent approaches in community-based studies. Applied Occupational and Environmental Hygiene 6:495-501.
Geyer H, Scheunert I, Korte F. 1986. Bioconcentration potential of organic environmental chemicals in humans. Regulatory Toxicology and Pharmacology 6:313-347.
Gordon JE, Shy CM. 1981. Agricultural chemical use and congenital cleft lip and/or palate. Archives of Environmental Health 36:213-221.
Greife AL, Hornung RW, Stayner LG, Steenland KN. 1988. Development of a model for use in estimating exposure to ethylene oxide in a retrospective cohort mortality study. Scandinavian Journal of Work, Environment, and Health 14 (Supplement 1):29-30.
Gross ML, Lay JO, Lippstreu D, Lyon PA, Kangas N, Harless RL, Taylor SE. 1984. 2,3,7,8-Tetrachlorodibenzo-p-dioxin levels in adipose tissue of Vietnam veterans. Environmental Research 33:261.
Hansen ES, Hasle H, Lander F. 1992. A cohort study on cancer incidence among Danish gardeners. American Journal of Industrial Medicine 21:651-660.
Heedrik D, Miller BG. 1988. Weak associations in occupational epidemiology: adjustment for exposure estimation error. International Journal of Epidemiology 17(suppl):970-974.
Henneberger PK, Ferris BG Jr, Monson RR. 1989. Mortality among pulp and paper workers in Berlin, New Hampshire. British Journal of Industrial Medicine 46:658-664.
Hill AB. 1971. Principles of Medical Statistics, 9th ed. New York: Oxford University Press.
Hoar SK, Blair A, Holmes FF, Boysen CD, Robel RJ, Hoover R, Fraumeni JF. 1986. Agricultural herbicide use and risk of lymphoma and soft tissue sarcoma. Journal of the American Medical Association 256:1141-1147.
Hoffman RE, Stehr-Green PA, Webb KB, Evans RG, Knutsen AP, Schramm WF, Staake JL, Gibson BB, Steinberg KK. 1986. Health effects of long-term exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Journal of the American Medical Association 255:2031-2038.
Hornung R. 1991. Statistical evaluation of exposure assessment strategies. Applied Occupational and Environmental Hygiene 6:516-520.
Institute of Medicine. 1987. Review of Comparison of Serum Levels of 2,3,7,8-TCDD with Indirect Estimates of Agent Orange Exposure in Vietnam Veterans. Fifth Letter Report.
Washington, DC: IOM, Advisory Committee on the CDC Study of the Health of Vietnam Veterans.
Jappinen P, Pukkala E. 1991. Cancer incidence among pulp and paper workers exposed to organic chlorinated compounds formed during chlorine pulp bleaching. Scandinavian Journal of Work, Environment, and Health 17:356-359.
Kahn PC, Gochfeld M, Nygren M, Hansson M, Rappe C, Velez H, Ghent-Guenther T, Wilson WP. 1988. Dioxins and dibenzofurans in blood and adipose tissue of Agent Orange-exposed Vietnam veterans and matched controls. Journal of the American Medical Association 259:1661-1667.
Kahn PC, Gochfeld M, Lewis WW. 1992. Dibenzodioxin and Dibenzofuran Congener Levels in Four Groups of Vietnam Veterans Who Did Not Handle Agent Orange. The Pointman II Project. New Jersey Agent Orange Commission.
Kalliokoski P. 1990. Estimating long-term exposure levels in process-type industries using production rates. American Industrial Hygiene Journal 51:310-312.
Kang HK, Watanabe KK, Breen J, Remmers J, Conomos MG, Stanley J, Flicker M. 1991. Dioxins and dibenzofurans in adipose tissue of U.S. Vietnam veterans and controls. American Journal of Public Health 81:344-349.
Kleinbaum DG, Kupper LL, Morgenstern H. 1982. Epidemiologic Research: Principles and Quantitative Methods. London: Lifetime Learning Publications.
Kolmodin-Hedman B, Erne K. 1980. Estimation of occupational exposure to phenoxy acids (2,4-D and 2,4,5-T). Archives of Toxicology Supplement 4:318-321.
Kolmodin-Hedman B, Hoglund S, Akerblom M. 1983. Studies on phenoxy acid herbicides. I. Field study. Occupational exposure to phenoxy acid herbicides (MCPA, dichlorprop, mecoprop and 2,4-D) in agriculture. Archives of Toxicology 54:257-265.
Kriebel D, Sprince N, Eisen E, Greaves I. 1988a. Pulmonary function in beryllium workers: assessment of exposure. British Journal of Industrial Medicine 45:83-92.
Kriebel D, Sprince N, Eisen E, Greaves I, Feldman H, Greene R. 1988b. Beryllium exposure and pulmonary function: a cross-sectional study of beryllium workers. British Journal of Industrial Medicine 45:167-173.
Kupper LL. 1984. Effects of the use of unreliable surrogate variables on the validity of epidemiologic research studies. American Journal of Epidemiology 120:643-648.
Lagakos SW. 1988. Effects of mismodelling and mismeasuring explanatory variables on tests of their association with a response variable. Statistics in Medicine 7:257-274.
Lavy TL, Shepard JS, Mattice JD. 1980a. Exposure measurements of applicators spraying 2,4,5-trichlorophenoxy acetic acid in the forest. Journal of Agricultural and Food Chemistry 28:626-630.
Lavy TL, Shepard JS, Bouchard DC. 1980b. Field worker exposure and helicopter spray pattern of 2,4,5-T. Bulletin of Environmental Contamination and Toxicology 24:90-96.
Lewis W. 1993. The Vietnam Experience. Presentation to the Institute of Medicine Committee to Review the Health Effects in Vietnam Veterans of Exposure to Herbicides. February 8, 1993.
Libich S, To JC, Frank R, Sirons GJ. 1984. Occupational exposure of herbicide applicators to herbicides used along electric power transmission line right-of-way. American Industrial Hygiene Association Journal 45:56-62.
Manz A, Berger J, Dwyer JH, Flesch-Janys D, Nagel S, Waltsgott H. 1991. Cancer mortality among workers in chemical plant contaminated with dioxin. Lancet 338:959-964.
Marshall RJ. 1990. Validation study methods for estimating exposure proportions and odds ratios with misclassified data. Journal of Clinical Epidemiology 43:941-947.
Michalek JE, Tripathi RC. 1992. Predicting Checkmark Patterns in the Air Force Health Study. Brooks AFB, TX: Armstrong Laboratory.
Miligi L, Masala G. 1991. Methods of exposure assessment for community-based studies:
aspects inherent to the validation of questionnaires. Applied Occupational and Environmental Hygiene 6:502-507.
Mocarelli P, Patterson DG Jr, Marocchi A, Needham LL. 1990. Pilot study (phase II) for determining polychlorinated dibenzo-p-dioxin (PCDD) and polychlorinated dibenzofuran (PCDF) levels in serum of Seveso, Italy residents collected at the time of exposure: future plans. Chemosphere 20:967-974.
Mocarelli P, Needham LL, Marocchi A, Patterson DG Jr, Brambilla P, Gerthoux PM, Meazza L, Carreri V. 1991. Serum concentrations of 2,3,7,8-tetrachlorobdibenzo-p-dioxin and test results from selected residents of Seveso, Italy. Journal of Toxicology and Environmental Health 32:357-366.
Morrison HI, Semenci RM, Morison D, Magwood S, Mao Y. 1992. Brain cancer and farming in western Canada. Neuroepidemiology 11: 267-276.
Moses M, Lilis R, Crow KD, Thornton J, Fischbein A, Anderson HA, Selikoff IJ. 1984. Health status of workers with past exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin in the manufacture of 2,4,5-trichlorophenoxyacetic acid: comparison of findings with and without chloracne. American Journal of Industrial Medicine 5:161-182.
Musicco M, Sant M, Molinari S, Filippini G, Gatta G, Berrino F. 1988. A case-control study of brain gliomas and occupational exposure to chemical carcinogens: the risks to farmers. American Journal of Epidemiology 128:778-785.
Myers J, Ehrlich R. 1990. Does nondifferential misclassification of exposure always bias a true effect toward the null value? (letter). American Journal of Epidemiology 132:1429-1430.
Needham LL, Burse VW, Head SL, Korver MP, McClure PC, Andrews JS Jr, Rowley DL, Sung J, Kahn SE. 1990. Adipose tissue/serum partitioning of chlorinated hydrocarbon pesticides in humans. Chemosphere 20:975-980.
Ott MG, Holder BB, Olson RD. 1980. A mortality analysis of employees engaged in the manufacture of 2,4,5-trichlorophenoxyacetic acid. Journal of Occupational Medicine 22:47-50.
Patterson DG Jr, Hoffman RE, Needham LL, Roberts DW, Bagby JR, Pirkle JL, Falk H, Sampson EJ, Houk VN. 1986. 2,3,7,8-Tetrachlorodibenzo-p-dioxin levels in adipose tissue of exposed and control persons in Missouri. An interim report. Journal of the American Medical Association 256:2683-2686.
Patterson DG Jr, Hampton L, Lapeza CR Jr, Belser WT, Green V, Alexander L, Needham LL. 1987. High-resolution gas chromatographic/high-resolution mass spectrometric analysis of human serum on a whole-weight and lipid basis for 2,3,7,8-tetrachlorodibenzo-p-dioxin . Analytical Chemistry 59:2000-2005.
Patterson DG Jr, Needham LL, Pirkle JL, Roberts DW, Bagby J, Garrett WA, Andrews JS Jr, Falk H, Bernert JT, Sampson EJ, Houk VN. 1988. Correlation between serum and adipose tissue levels of 2,3,7,8-tetrachlorodibenzo-p-dioxin in 50 persons from Missouri. Archives of Environmental Contamination and Toxicology 17:139-143.
Pirkle JL, Wolfe WH, Patterson DG, Needham LL, Michalek JE, Miner JC, Peterson MR, Phillips DL. 1989. Estimates of the half-life of 2,3,7,8-tetrachlorodibenzo-p-dioxin in Vietnam Veterans of Operation Ranch Hand. Journal of Toxicology and Environmental Health 27:165-171.
Rappaport SM, Smith TJ, eds. 1991. Exposure Assessment for Epidemiology and Hazard Control. Chelsea, MI: Lewis Publishers.
Rice C. 1991. Retrospective exposure assessment: a review of approaches and directions for the future. In: Rappaport SM, Smith TJ, eds. Exposure Assessment for Epidemiology and Hazard Control. Chelsea, MI: Lewis Publishers. 185-198.
Rinsky RA, Smith AB, Hornung R, Filloon TG, Young RJ, Okun AH, Landrigan PJ. 1987. Benzene and leukemia: an epidemiologic risk assessment. New England Journal of Medicine 316:1044-1050.
Robinson CF, Waxweiler RJ, Fowler DP. 1986. Mortality among production workers in pulp and paper mills. Scandinavian Journal of Work, Environment, and Health 12:552-560.
Ronco G, Costa G, Lynge E. 1992. Cancer risk among Danish and Italian farmers. British Journal of Industrial Medicine 49:220-225.
Rosner B, Spiegelman D, Willett WC. 1990. Corrections of logistic regression relative risk estimates and confidence intervals for measurement error: the case of multiple covariates measured with error. American Journal of Epidemiology 132:734-745.
Ryan JJ, Schecter A, Sun W-F, Lizotte R. 1986. Distribution of chlorinated dibenzo-p-dioxins and chlorinated dibenzofurans in human tissues from the general population. In: Rappe C, Choudhary G, Keith L, eds. Chlorinated Dioxins and Dibenzofurans in Perspective. Chelsea, MI: Lewis Publishers. 3-16.
Saracci R, Kogevinas M, Bertazzi PA, Bueno De Mesquita BH, Coggon D, Green LM, Kauppinen T, L'Abbe KA, Littorin M, Lynge E, Mathews JD, Neuberger M, Osman J, Pearce N, Winkelmann R. 1991. Cancer mortality in workers exposed to chlorophenoxy herbicides and chlorophenols. Lancet 338:1027-1032.
Schecter A, Ryan JJ, Constable JD. 1986. Chlorinated dibenzo-p-dioxin and dibenzofuran levels in human adipose tissue and milk samples from the North and South of Vietnam. Chemosphere 15:1613-1620.
Schecter A, Constable JD, Arghestani S, Tong H, Gross ML. 1987. Elevated levels 2,3,7,8-tetrachlorodibenzodioxin in adipose tissue of certain U.S. veterans of the Vietnam war. Chemosphere 16:1997-2002.
Schlatter C. 1991. Data on kinetics of PCDDs and PCDFs as a prerequisite for human risk assessment . In: Gallo MA, Scheuplein RJ, van der Heijden KA, eds. Biological Basis for Risk Assessment of Dioxins and Related Compounds. Plainview, NY: Cold Spring Harbor Laboratory Press. Banbury Report 35. 215-227.
Schneider T, Olsen I, Lauersen B. 1991. Evaluation of other sources of exposure information. Applied Occupational and Environmental Hygiene 6:475-481.
Sielken RL Jr. 1987. Statistical evaluations reflecting the skewness in the distribution of TCDD levels in human adipose tissue. Chemosphere 16:2135-2140.
Seixas NS. 1990. Estimation of cumulative exposure for a cohort of coal miners with ''post-MSHA" exposures. Presented at the American Industrial Hygiene Conference, 17 May, 1990, Orlando, FL.
Sexton K, Selevan SG, Wagener DK, Lybarger JA. 1992. Estimating human exposures to environmental pollutants: availability and utility of existing databases. Archives of Environmental Health 47:398-407.
Smith AH, Matheson DP, Fisher DO, Chapman CJ. 1981. Preliminary report of reproductive outcomes among pesticide applicators using 2,4,5-T. New Zealand Medical Journal 93:177-179.
Smith AH, Fisher DO, Pearce N, Chapman CJ. 1982. Congenital defects and miscarriages among New Zealand 2,4,5-T sprayers. Archives of Environmental Health 37:197-200.
Smith AH, Patterson DG Jr, Warner ML, Mackenzie R, Needham LL. 1992. Serum 2,3,7,8-tetrachlorodibenzo-p-dioxin levels of New Zealand pesticide applicators and their implication for cancer hypotheses. Journal of the National Cancer Institute 84:104-108.
Smith TJ. 1987. Exposure assessment for occupational epidemiology. American Journal of Industrial Medicine 126:249-268.
Smith TJ, Hammond SK, Hallock M, Woskie SR. 1991. Exposure assessment for epidemiology: characteristics of exposure. Applied Occupational and Environmental Hygiene 6:441-447.
Solet D, Zoloth SR, Sullivan C, Jewett J, Michaels DM. 1989. Patterns of mortality in pulp and paper workers. Journal of Occupational Medicine 31:627-630.
Stanton SL. 1989. Area-scoring methodology for estimating Agent Orange exposure status of
U.S. Army personnel in the Republic of Vietnam. In: Centers for Disease Control, Veterans Health Study. Comparison of Serum Levels of 2,3,7,8-Tetrachlorodibenzo-p-dioxin with Indirect Estimates of Agent Orange Exposure Among Vietnam Veterans. Atlanta: CDC. Appendix A.
Stellman SD, Stellman JM. 1986. Estimation of exposure to Agent Orange and other defoliants among American troops in Vietnam: a methodological approach. American Journal of Industrial Medicine 9:305-321.
Stellman JM, Stellman SD. 1993. An Appraisal of Military Records Available for Research on the Health Effects of Herbicides Used During the Vietnam War. Prepared for the Institute of Medicine Committee to Survey the Health Effects in Vietnam Veterans of Exposure to Herbicides. February 4, 1993.
Stellman SD, Mager-Stellman J, Sommer JF Jr. 1988. Combat and herbicide exposures in Vietnam among a sample of American Legionnaires. Environmental Research 47:112-128.
Stewart PA, Herrick RF, eds. 1991. International workshop on retrospective exposure assessment for occupational epidemiologic studies. Applied Occupational and Environmental Hygiene 6:417-559.
Stewart PA, Blair A, Cubit D, et al. 1986. Estimating historical exposures to formaldehyde in a retrospective mortality study. Applied Industrial Hygiene 1:34-41.
Suskind RR, Hertzberg VS. 1984. Human health effects of 2,4,5-T and its toxic contaminants. Journal of the American Medical Association 251:2372-2380.
Swaen GMH, van Vliet C, Slangen JJM, Sturmans F. 1992. Cancer mortality among licensed herbicide applicators. Scandinavian Journal of Work, Environment, and Health 18:201-204.
Sweeney MH, Fingerhut MA, Patterson DG, Connally LB, Piacitelli L, Morris JA, Greife AL. 1990. Comparison of serum levels of 2,3,7,8-TCDD in TCP production workers and in an unexposed comparison group. Chemosphere 20:993-1000.
Tankersley WG, Ingle J, West C, Watson J, Crawford-Brown D. 1991. Guidelines for systematic assessment of occupational exposures in absence of monitoring data. Presented at Eighth International Symposium on Epidemiology in Occupational Health, Paris.
Thomas TL, Kang HK. 1990. Mortality and morbidity among Army Chemical Corps Vietnam veterans: a preliminary report. American Journal of Industrial Medicine 18:665-673.
U.S. Army. 1972. Herbicides and Military Operations. Vols I and II. Department of the Army, Engineer Strategic Studies Group, Office, Chief of Engineers.
Vineis P, Terracini B, Ciccone G, Cignetti A, Colombo E, Donna A, Maffi L, Pisa R, Ricci P, Zanini E, Comba P. 1986. Phenoxy herbicides and soft tissue sarcomas in female rice weeders. A population-based case-referent study. Scandinavian Journal of Work, Environment, and Health 13:9-17.
Wacholder S, Dosemici M, Lubin JH. 1991. Blind assignment of exposure does not always prevent differential misclassification. American Journal of Epidemiology 134:433-437.
Werler MM, Pober BR, Nelson K, Holmes LB. 1989. Reporting accuracy among mothers of malformed and nonmalformed infants. American Journal of Epidemiology 129:415-421.
Wigle DT, Semenciw RB, Wilkins K, Riedel D, Ritter L, Morrison HI, Mao Y. 1990. Mortality study of Canadian male farm operators: non-Hodgkin's lymphoma mortality and agricultural practices in Saskatchewan. Journal of the National Cancer Institute 82:575-582.
Wiklund K, Holm L-E. 1986. Soft tissue sarcoma risk in Swedish agricultural and forestry workers. Journal of the National Cancer Institute 76:229-234.
Wiklund K, Lindefors BM, Holm L-E. 1988a. Risk of malignant lymphoma in Swedish agricultural and forestry workers. British Journal of Industrial Medicine 45:19-24.
Wiklund K, Dich J, Holm L-E. 1988b. Soft tissue sarcoma risk in Swedish licensed pesticide applicators. Journal of Occupational Medicine 30:801-804.
Wiklund K, Dich J, Holm L-E. 1989. Risk of soft tissue sarcoma, Hodgkin's disease and non-Hodgkin lymphoma among Swedish licensed pesticide applicators. Chemosphere 18:395-400.
Woods JS, Polissar L. 1989. Non-Hodgkin's lymphoma among phenoxy herbicide-exposed farm workers in western Washington State. Chemosphere 18:401-406.
Young AL, Barnes DG, Blair A, Bricker JG, Christian RS, Fingerhut M, Kang H, Keller C, Muray JE, Shepard BM. 1986. Report of the Agent Orange Working Group Science Subpanel on Exposure Assessment. Submitted to the Chairman, Agent Orange Working Group. Washington, DC: Executive Office of the President, Office of Science and Technology Policy.
Zack JA, Gaffey WR. 1983. A mortality study of workers employed at the Monsanto company plant in Nitro, West Virginia. Environmental Science Research 26:575-591.
Zahm SH, Weisenburger DD, Babbitt PA, Saal RC, Vaught JB, Cantor KP, Blair A. 1990. A case-control study of non-Hodgkin's lymphoma and the herbicide 2,4-dichlorophenoxyacetic acid (2,4-D) in eastern Nebraska. Epidemiology 1:349-356.
Zober A, Messerer P, Huber P. 1990. Thirty four year mortality follow-up of BASF employees exposed to 2,3,7,8-TCDD after the 1953 accident. International Archives of Occupational and Environmental Health 62:139-157.
Zumwalt ER Jr. 1993. Letter to the Institute of Medicine Committee to Review the Health Effects in Vietnam Veterans of Exposure to Herbicides regarding draft version of the IOM chapter on the U.S. military and the herbicide program in Vietnam. May 20, 1993.


















































