Working Parents: The Impact of Day Care and Breast-Feeding onCytomegalovirus Infections in Offspring
SERGIO STAGNO AND GRETCHEN A. CLOUD
Human cytomegalovirus (CMV) is of medical importance because it is the leading cause of congenital infection in most parts of the world and because it can cause significant disease in immunocompromised patients. CMV is highly species specific, and humans are believed to be its only reservoir (1).
THE PROBLEM
Seroepidemiologic surveys have found CMV infection in every population that has been tested (2). Transmission occurs by direct or indirect person-to-person contact. Known sources of the virus include urine, oropharyngeal secretions, cervical and vaginal secretions, semen, milk, tears, and blood (3). CMV is not very contagious, and the spread of infection requires close or intimate contact with infected secretions. Infected children excrete virus for much longer periods of time (months to years) and in larger quantities than adults. Under special circumstances fomites may also play a role, since CMV has been shown to retain infectivity for hours on plastic surfaces and has been isolated from randomly selected toys and surfaces in day-care centers (4, 5). Restriction
Sergio Stagno is the Katharine Reynolds Ireland Professor and chairman of the Department of Pediatrics and professor of microbiology, and Gretchen A. Cloud is a biostatistician and research instructor in the Department of Microbiology and Biostatistics at the University of Alabama, Birmingham.
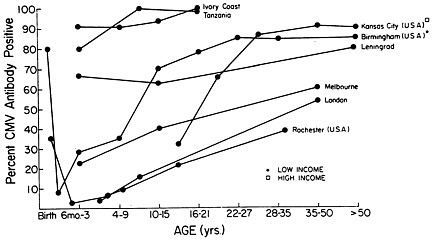
FIGURE 1 Age-related prevalence of antibody to CMV in various populations. [Reproduced with permission from ref. 6 (copyright 1981, C. A. Alford).]
enzyme analyses of CMV DNA have been used to demonstrate route of transmission of the virus in situations in which close contact occurs, such as breast-feeding, sexual activity, day care, and interaction between parents or caretakers and infected toddlers.
The prevalence of CMV infection increases with age, but, according to geographic, ethnic, and socioeconomic backgrounds, the patterns of acquisition of this infection vary widely among populations (6). As illustrated in Figure 1, CMV is acquired earlier in life in developing countries and among the lower socioeconomic strata of developed countries. Differences between populations can be particularly striking during childhood. Presumably, these significant differences are the reflection of factors that account for increased exposure to CMV such as crowding, breast-feeding, sexual practices, and certain rearing practices.
Studies carried out in the past 15 years indicate that breast-feeding and child-rearing practices singly or in combination are two of the most powerful factors influencing the rate of acquisition of CMV in the various populations (7, 8). Breast-feeding is the major factor during the first year of life with CMV excretion rates of >50% observed in countries where the majority of women are seropositive and breast-feed their infants. The rapid increase in the rate of infection that generally takes place after the first year of life is the result of close contact and exposure to children who acquired CMV infection from a maternal source (i.e., in utero, from exposure to genital secretions at birth, or from breast milk). In our view, the profound alterations that have occurred in breast-feeding
and child-rearing practices during the past several decades are largely responsible for the current regional differences in seroprevalence and will continue to exert a powerful effect in CMV-related morbidity in those areas where these practices undergo significant changes. Because of the public health importance of breast-feeding and child-rearing practices in the epidemiology of CMV this article will focus on their roles and the implications for the future. The data presented here cannot be extrapolated to all continents, or even across national boundaries. The situation we are reviewing applies principally to the United States and to a lesser extent to Canada and parts of Western Europe. However, it is likely that in many other countries a similar situation already exists or is likely to develop if these modifications take place. More studies are needed to better define the epidemiology of CMV on an international scale.
ROLE OF BREAST-FEEDING
Diosi et al. (9) were the first to report the isolation of CMV from human milk. The virus was found in 1 of 49 milk specimens collected from parturient women. Hayes et al. (10) subsequently reported the isolation of CMV from 14 milk specimens (27%) collected from 63 seropositive women. In addition, they found that virolactia was more common in samples collected >1 wk postpartum (50%) than in those collected during the 1st week (11%). Studies in our laboratory provided compelling evidence that CMV is indeed transmitted from mother to baby through breast milk and that the ensuing infection is almost always without clinical consequence (8, 11). Among seropositive women sampled serially, milk was the most common site for recovery of CMV, with 13 of 41 women (32%) having at least one positive sample. It is noteworthy that women excreting CMV from other sites and mothers of congenitally infected infants were more likely to have virolactia. The rate of CMV excretion changed during the course of lactation, with only 2 of 40 women (5%) shedding CMV in colostrum while 13 of 31 (42%) had positive milk specimens when sampled between 2 and 12 wk postpartum. As illustrated in Table 1, the acquisition of CMV by the infants of these seropositive women was significantly related to the duration of breast-feeding and the presence of detectable virus in the breast milk. In sharp contrast, no bottle-fed babies or infants born to women who shed CMV only in saliva or urine become infected. From this study, we can conclude that nearly 40% of all infants nursed for a month or longer by seropositive mothers acquire CMV postnatally (12) (Table 1). Since in most populations of the world the prevalence of CMV infection among women of childbearing age is very high (80–98%) and most women
TABLE 1 Breast-feeding and infant CMV infection from prospectively studied mother–infant pairs
|
Maternal status |
No. |
No. of infants infected (%) |
P value |
|
Seronegative |
17 |
0 |
0.006 |
|
Seropositive |
41 |
12 (30) |
|
|
Breast-fed <1 mo |
10 |
0 |
0.015 |
|
Breast-fed ≥1 mo |
31 |
12 (39) |
|
|
CMV excreted in milk |
13 |
9 (69) |
0.0007 |
|
CMV not excreted in milk |
28 |
3 (10) |
|
|
Data are from ref. 11. |
|||
breast-feed their infants for a period of 2–6 months, transmission through breast milk is certainly quite prevalent and accounts for the majority of infants infected in early life. A few years ago we reported that, of a cohort of 154 predominantly Black infants of low socioeconomic background who were prospectively followed for a 2-yr period to establish the rate of postnatal CMV infection, only 8% had acquired the infection by age 6 mo (8). The rate of seropositivity among mothers was 85%, yet only 8% of the infants had been breast-fed. As illustrated in Table 2, this is one of the lowest rates of perinatal transmission ever reported (13). What makes it more remarkable is that this rate is far
TABLE 2 Breast-feeding patterns and prevalence of CMV infection in young children from various nations
|
|
Breast-feeding rate |
|
% seropositive |
|
|
Nation |
Ever |
At 3 mo |
Mothers |
Children (age) |
|
Solomon Islands |
100 |
97 |
100 |
100 (5 mo to 4 yr) |
|
India (Vellore) |
96 |
64 |
98 |
80 (1 yr) |
|
India (Pondicherry) |
|
|
97 |
67 (1–5 yr) |
|
Barbados |
96 |
? |
77 |
62 (1–5 yr) |
|
Guatemala |
95 |
? |
98 |
47 (6 mo to 1 yr) |
|
Chile |
89 |
? |
92 |
42 (1–2 yr) |
|
Japan (Sapporo) |
? |
56 |
67 |
42 (6 mo to 2 yr) |
|
Japan (Sendai) |
|
|
85 |
38 (1 yr) |
|
Finland (Helsinki) |
95 |
50 |
55 |
28 (1 yr) |
|
USA (Houston) |
46 |
? |
48 |
15 (1 yr) |
|
USA (Birmingham) |
8 |
? |
85 |
8 (1 yr) |
|
France (Paris) |
85 |
? |
56 |
10 (10 mo) |
|
Canada (Nova Scotia) |
49 |
26 |
34 |
12 (6–12 mo) |
|
UK (Manchester) |
51 |
13 |
59 |
12 (3–11 mo) |
|
Data are from ref. 13. |
||||
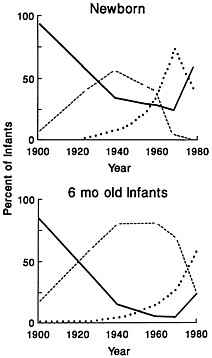
FIGURE 2 Feeding practices in the United States of newborns and infants 5–6 mo old from 1900 to 1980. Solid line, breast feeding; dotted line, combined feeding of formulas prepared from evaporated milk or whole cow's milk; dashed line, commercially prepared formulas. [Reproduced by permission of Pediatrics, Vol. 60, p. 654, copyright 1982 (12).]
below the rates seen in other populations in which the seroprevalence of CMV among mothers is much lower.
The popularity of breast-feeding in the industrialized world and particularly in the United States has changed dramatically during this century in terms of both frequency and duration (12). Trends in infant feeding practices within the United States since 1900 are shown in Figure 2. Breast-feeding within the hospital setting reached a nadir in 1970, when fewer than one in five mothers elected to breast-feed. Exclusive breast-feeding was reported by 19% of White and 9% of Black mothers. Abandonment of breast-feeding in the earlier part of this century started first within the upper socioeconomic levels and spread downward on the socioeconomic scale. After 1970, the resurgence of breast-feeding began at the upper socioeconomic levels, and it is spreading along the various strata in much the same manner as the abandonment of breast-feeding occurred.
In 1980 the National Center for Health Statistics reported a significantly higher percentage of both Black and White women exclusively breast-feeding their infants than in 1969 (14). For Whites, the percentage had increased to 51%, while for Black women it had increased only to
25%. The differences by race persist when education and parity are controlled. It is interesting to note that among White women a significantly higher percentage of breast-feeding was observed with increasing maternal education in 1969 as well as in 1980. In sharp contrast, among Black women with newborns in 1969 the rate of breast-feeding was significantly lower among those with a higher level of education. However, in 1980, a relationship similar to that observed in Whites appeared.
It is our belief that these changes in the popularity of breast-feeding have had a major effect on the epidemiology of CMV. The much lower prevalence of CMV infection that we and others have documented in the United States among women of the upper middle class when compared to women of low socioeconomic background living in the same geographic area is to some extent the result of this phenomenon. As fewer White, largely upper and middle class seropositve women elected to breast-feed their infants in the first half of this century, the perinatal transmission of CMV declined progressively. As the low socioeconomic sector followed in this trend a similar drop in perinatal transmission is now evident in this group.
IMPORTANCE OF DAY-CARE CENTERS
With the changing composition of the American family, many children live in single parent families in which the mother is the primary source of economic support (15). It is estimated that nearly 60% of the women with children 5 yr of age and under are employed. The National Center for Health Statistics reported that in 1988 two-thirds of children in the United States 5 yr of age and under (68% or 13.3 million) had been in child-care arrangements at some point in their lives (15). Two-thirds of them were cared for in a nursery, preschool, or day-care center. Twenty-four percent of children in formal child-care arrangements were cared for in groups of seven or more children. The number of family day-care or group home and day-care centers increased dramatically between 1977 and 1985 (15). The report from the Center for Health Statistics demonstrates that receipt of child care is strongly associated with socioeconomic status. Women of low socioeconomic strata are significantly less likely to use a child-care arrangement.
In 1971, Weller suggested that the high rate of seropositivity among Swedish children was probably due to the frequent use of day-care centers (1). Swedish children had a rate of infection that was 3–4 times higher than that observed in London or Rochester, New York. Similar high rates of CMV infection in young children were noted in the Solomon Islands and Israeli kibbutzim, where high hygienic standards
TABLE 3 Prevalence of CMV excretion among children in day-care centers
|
Investigator |
Year |
Location |
% infected |
|
Strangert |
1976 |
Stockholm |
35 (7/20) |
|
Strom |
1979 |
Stockholm |
72 (13/18) |
|
Pass |
1982 |
Birmingham, AL |
51 (36/70) |
|
Adler |
1985 |
Richmond, VA |
24 (16/66) |
|
Hutto |
1985 |
Birmingham, AL |
41 (77/188) |
|
MMWR |
1985 |
Birmingham, AL |
29 (66/231) |
|
Jones |
1985 |
San Francisco, CA |
22 (31/140) |
|
Murph |
1986 |
Iowa City, IA |
22 (9/41) |
|
Adler |
1987 |
Richmond, VA |
53 (55/104) |
|
MMWR, Morbidity and Mortality Weekly Report. Data are from ref. 18. |
|||
were maintained (16, 17). As illustrated in Table 3, high rates of CMV infection among children attending day-care centers were later confirmed in Sweden and have been reported in several studies in the United States (7, 18–26). The studies, which included a control group of children, confirmed that the rate of CMV infection was substantially higher among those in day care than in those who stayed at home (20, 21). The highest rates have been found consistently in children between 1 and 3 yr old. In the study of Pass et al. (7), in a group of 70 children of middle- to upper-income background whose ages ranged from 3 to 65 mo, the rate of CMV excretion in urine and saliva was 51%. The lowest rate of excretion (9%) occurred in infants <1 yr old, and the highest rate (88%) was among toddlers in their second year of life. Infants younger than 12 mo in group day care who excrete CMV are more likely to have acquired CMV congenitally or perinatally from maternal cervical secretions or breast milk. Twelve children whose mothers were seronegative excreted CMV, which indicated that their infection was not perinatally acquired. The findings of Pass et al. have been subsequently confirmed by Adler (18, 20, 23) and by Murph et al. (24). There is consensus that CMV infection acquired by young children is generally asymptomatic. No specific syndrome has been temporarily associated with CMV infection acquired in day-care centers. The high rates of infection combined with the large quantities of infectious virus excreted by these children create a potential for exposure that is difficult to encounter under any other circumstances.
There is now compelling evidence that the high rate of CMV infection among children in group day care is caused by horizontal transmission from child to child. The route of transmission that appears most likely is the transfer of virus that occurs through saliva on hands and toys. The
survival of CMV on fomites such as toys, diapers, and hands of day-care personnel also suggests a role for environmental contamination (4, 5, 27). No data have indicated CMV transmission via respiratory droplets.
The strongest evidence supporting child-to-child transmission was obtained by analysis of the restriction enzyme digestion patterns of CMV DNA of the isolates obtained from infected children attending day care. Adler (20) examined the restriction endonuclease patterns of isolates obtained from 16 children at a single day-care center. Four children older than 28 mo who shared a common room were shedding a common strain. A second strain was shed by seven children younger than 28 mo who had little contact with the older group but played with one another daily. Only five of the infected children were shedding unique strains. In a subsequent study, Adler (28) identified by endonuclease analysis 14 different strains of CMV among 104 children in a single day-care center who were monitored at 4-mo intervals for >26 mo. Three of these 14 strains infected 44 children, all of whom were <3 yr old. Of 75 initially seronegative children, 34 acquired day-care-associated strains, whereas 4 were infected with unique isolates. These findings have been confirmed in subsequent studies (24-26, 28, 29). These findings demonstrate that CMV is very efficiently transmitted from child to child in the day-care setting and that it is not unusual to find excretion rates as high as 20-80% in young toddlers. In many instances these rates of infection are substantially higher than the seroprevalence rates for the parents of the children and young adults in the cities where the studies were done.
COMBINED EFFECT OF BREAST-FEEDING AND DAY-CARE CENTERS
In the low socioeconomic population, the combined effects of a low rate of breast feeding and low utilization of child-care arrangements may result in a lowering of the prevalence of CMV. Over the past 20 years, as part of an ongoing study of the natural history of maternal CMV infection, we have surveyed the seroprevalence of CMV infection among nearly 6000 pregnant women attending a single prenatal public health clinic in the city of Birmingham, Alabama. All these women were enrolled at the time of the first prenatal visit. All deliveries took place at University Hospital. As illustrated in Figure 3A, in the past 2 decades the seroprevalence of CMV infection among these predominantly Black women of low socioeconomic background has significantly declined across all ages. The lower curve represents the last 6 yr of involvement (1986-1992), while the upper curve reflects the rate of seropositivity during the first 6 yr of involvement (1971-1977). It is particularly striking that in the 15- to 24-yr-old group, the rate of seropositivity has dropped
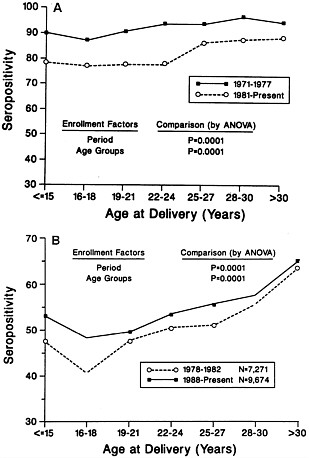
FIGURE 3 (A) Rate of seropositivity to CMV of two enrollment periods for women of low socioeconomic extraction. (B) Rate of seropositivity to CMV of two enrollment periods for middle and upper-middle income women.
nearly 10 percentage points. Since 1980 we have been screening this population for congenital CMV infection with a sensitive tissue culture method. As illustrated in Table 4 the rate of congenital infection has remained constant during the past 12 years. However, we have noticed an increase in the rate of symptomatic congenital infection from 4.3% to 10.2% (P = 0.09). This provocative observation may well be the result of more primary, as opposed to recurrent, infections experienced by young seronegative mothers in this socioeconomic group.
TABLE 4 Congenital CMV infection in low socioeconomic population
|
|
|
Congenital infection |
Symptomatic |
|
|
|
Interval |
Total newborn |
Total |
% |
Total |
% |
|
1980-1983 |
6,128 |
70 |
1.14 |
3 |
4.3 |
|
1984-1987 |
5,845 |
83 |
1.42 |
7 |
8.4 |
|
1988-1992 |
12,393 |
166 |
1.34 |
17 |
10.2 |
|
χ2 test for trend of percentage of symptomatic infection (P = 0.09). |
|||||
With the dramatic resurgence of breast-feeding in the middle and upper middle classes and a greater utilization of child-care arrangements, we anticipated the opposite phenomenon. As part of our ongoing studies of CMV in pregnant women since 1978, we have enrolled nearly 24,000 women of upper socioeconomic background from a single private obstetrical practice in Birmingham, Alabama. All women were enrolled and tested for CMV antibodies at the time of the first prenatal visit. All deliveries took place at a single private hospital. As predicted, the seroprevalence of CMV infection in women of middle and upper middle class has significantly increased, particularly in the 15- to 18-yr-old group (Figure 3B).
RISK TO CARE GIVERS
Several lines of evidence indicate that children excreting CMV often become the source of infection for serosusceptible parents and child-care personnel, particularly women of childbearing age. Using restriction endonuclease analysis, Spector and Spector (30) and Dworsky et al. (31) demonstrated transmission of CMV from infants with perinatal infections to a parent or other adult family members. Yeager (32) reported in 1983 that 7 (47%) of 15 seronegative mothers of infants who had acquired CMV in a nursery seroconverted within 1 yr. The same year, Dworsky et al. (33) reported that the rate of seroconversion for women with at least one child living at home was 5.5%, significantly higher than the 2.3% rate for women from the same clinic who were pregnant for the first time or the rates of susceptible nursery nurses and for physicians in training. Taber et al. (34) monitored the acquisition of CMV in 68 Houston families observed for a mean of 3.5 yr per family. The study showed a significant association between seroconversion among children and seroconversion among susceptible parents. Pass et al. (26, 35, 36) and Adler (23, 28, 37) have presented more compelling evidence linking the acquisition of CMV by children in day care with subsequent infection in their mothers and care givers. Pass et al. (35) did a
longitudinal serologic follow-up study of seronegative parents whose children attended a day-care center and of seronegative parents whose children did not attend day care. The groups were followed for a mean of 17 and 21 mo, respectively. The study revealed that 14 of 67 seronegative parents with children in day-care centers acquired CMV, compared with 0 of 31 serosusceptible parents whose children did not attend day care. More significant, all 14 parents of the day-care group who seroconverted had a child who was shedding CMV in saliva or urine. In fact, seroconversion occurred in 14–48 parents of children who shed CMV, compared with 0 of 21 whose children did not excrete CMV. The highest risk of seroconversion (45%) was for parents with a child shedding CMV who was 18 mo old or younger at enrollment. In 2 of the 14 cases, DNA analysis indicated the child as the source of CMV infection. In a subsequent study, this group of investigators also demonstrated by means of restriction enzyme analysis that infections acquired by a mother from a child can be transmitted to her fetus (26). In a very similar study, Adler (23) observed that of 18 seronegative mothers whose children shed CMV strains associated with day care, 6 seroconverted and excreted CMV strains identical to the strains shed by their children. On an average these mothers acquired the infection within 4.2 mo (range, 3–7 mo) after their children became infected.
In assessing the risk to day-care workers Adler (37) and Pass (38) have recently reported annual seroconversion rates of 11% and 20% among seronegative day-care workers. The risk could be different in smaller centers or in day-care homes where supervision and infection control measures are not well enforced. These rates are significantly higher than the 2–2.5% expected incidence from studies of large numbers of pregnant women or hospital workers of similar socioeconomic background (38). The risk of infection appears to be significantly higher among those caring for children who are <3 yr old. Results of restriction endonuclease analyses of CMV DNA from strains recovered from children attending day-care centers, from their parents, and from day-care workers provide compelling evidence that children transmit CMV to their care givers. Using this methodology, Adler (23) analyzed strains recovered from children, parents, and care givers at a single day-care center. He found that nine mothers, four fathers, and two child-care workers excreted strains of CMV that were identical to those excreted by children at the center. The same investigator in a more extensive study found that seven care givers from three different centers excreted CMV strains that by restriction enzyme analyses were identical to the strains isolated from one or more of the children they cared for.
From the data generated by these studies it is reasonable to expect that ≈50% of susceptible children between the ages of 1 and 3 yr who
TABLE 5 Rate of seroconversion during first and subsequent pregnancies in women of middle to upper socioeconomic background
|
Order of studied pregnancy |
Mean age, yr |
Seroconversions/no. seronegatives |
Annualized rate* |
|
Pregnancy 1† |
25.4 |
97/8194 (1.2%) |
2.0% |
|
Between 1 and 2 |
|
260/2236 (11.6%) |
4.5% |
|
Pregnancy 2 |
28.0 |
29/1597 (1.8%) |
3.1% |
|
Between 2 and 3 |
|
64/485 (13.2%) |
6.3% |
|
Pregnancy 3 |
29.3 |
5/352 (1.4%) |
2.4% |
|
Between 3 and 4 |
|
8/97 (8.3%) |
5.2% |
|
Pregnancy 4 |
30.0 |
2/73 (2.7%) |
4.6% |
|
* Based on an average gestational age of 2 mo at enrollment. † Does not equate with primigravida. |
|||
attend group day care will acquire CMV from their playmates and become an important potential source of infection for susceptible parents and care givers. Of particular concern is the risk to seronegative mothers who have children in group day care and who become pregnant. As part of our ongoing investigations of CMV infection in pregnancy, we have prospectively studied >2200 seronegative women from a high socioeconomic group during more than one pregnancy and have been able to establish the risk of seroconversion in successive pregnancies. In general, as illustrated in Table 5, the annualized rate of seroconversion increases with time from 2.0% for the first studied pregnancy to 4.6% during the fourth studied pregnancy. These observations combined with the results of the studies in day care underline the fact that susceptible pregnant women may acquire CMV infection introduced into the household by their young children. Given these figures and the data presented in the preceding paragraphs, it is obvious that the increased utilization of day-care centers is having a significant effect on the epidemiology of CMV.
CONCLUSIONS
In the United States between one-half and one-third of all women of childbearing age are susceptible to CMV infection. The rate of seronegativity is higher for middle to upper-middle class women than for women of low socioeconomic extraction. However, these differences appear to be narrowing and may reverse themselves if the current trends in breast-feeding practices and utilization of day-care centers continue. If 20–50% of all mothers elect to breast-feed their newborn infants, and considering that those who are seropositive transmit CMV
to their infants through breast milk at a rate of 40%, then somewhere between 10% and 15% of all infants can be expected to start shedding CMV before the age of 6 mo. In addition, the pool of infected infants must be expanded to include the estimated 0.5–2.0% of infants born with congenital infection and 2–4% of infants who acquire natal CMV infection from exposure to maternal genital tract secretion at delivery. Since a characteristic of CMV infections acquired in utero and early infancy is chronic viral shedding of large quantities of virus, grouping children for care will inevitably lead to horizontal spread to other children, parents, and other adults who have daily close contact with children. From the studies reviewed here, it is reasonable to anticipate that between one-half and two-thirds of all children who attend day care will become infected within 2 yr of enrollment and that these children will also shed CMV from urine and saliva for extended periods of time. The data also indicate that 30–50% of the parents of these children and those who care for them are susceptible to CMV and the evidence is persuasive that they are at increased risk for infection. In day-care centers of moderate size, the risk of transmission to parents is 15–30% and the risk to workers is 10–20% per yr. Because many mothers become pregnant while a previous child is still in day care, those that are susceptible to CMV are at a significant risk of acquiring CMV. When primary infection occurs during pregnancy, the risk of in utero transmission is ≈40% (6). Prospective studies have demonstrated that between 10% and 20% of congenitally infected infants whose mothers acquired a primary CMV infection during pregnancy develop sequelae (39). Fortunately, the risk of adverse effects for the fetus is extremely low for women who are seropositive before pregnancy.
For the immediate future, the implications of the data reviewed in this manuscript are quite serious for seronegative women of childbearing age who as parents or for work-related reasons are exposed to young children who receive group care outside their homes.
It is reasonable to anticipate that, over the next decade or so, many seronegative women of middle and upper-middle class background will become infected from exposure to children who acquired CMV infection in group day-care centers. This risk will be particularly serious for those women who become infected while pregnant as they will face the threat of transmission of virus to the fetus in utero, which carries a risk, albeit low, of serious fetal morbidity. In the more distant future, as more infants and young children acquire CMV in early life, the pool of seronegative individuals will decline progressively and the risk of primary infection for women of childbearing age will subside. The situation in the low socioeconomic sector is quite different. Few women in this group breast-feed their infants and the utilization of day-care
arrangements is also relatively low. Then it is to be expected that as there is less transmission of CMV the pool of seronegative individuals will actually increase and many more women will reach their reproductive age without the benefit of immunity to CMV.
If the risk of primary CMV infection during gestation increases, it is logical to anticipate more cases of severe congenital infection, as seems to be occurring in our community.
The sequence of these changes in the various socioeconomic groups will differ within any given geographic region principally in relation to the popularity of breast-feeding and the utilization of day-care centers. Since the data analyzed in this review were obtained from a limited number of studies carried out in only a few urban settings in the United States, we should not extrapolate to other settings without considering the many variables that obviously affect the epidemiology of CMV.
SUMMARY
The increase in the popularity of breast feeding and utilization of child care arrangements are having a major effect on the epidemiology of cytomegalovirus infections. The impact is greater for women of upper socioeconomic background who send their toddlers to day-care centers and for day-care workers. If primary cytomegalovirus infections occur during pregnancy it is logical to anticipate more cases of severe congenital infections.
These studies were supported by General Clinical Research Center Grant RR0032, Program Project Grant HD10699, Children's Hospital Research Center Grant HD28831, and University of Alabama, Birmingham, Comprehensive Cancer Center Grant CA13148.
REFERENCES
1. Weller, T. H. (1971) N. Engl. J. Med. 285, 203–214.
2. Krech, U., Jung, M. & Jung, F. (1971) Cytomegalovirus Infections of Man (Karger, Basel), p. 28.
3. Stagno, S. (1980) in Infectious Diseases of the Fetus and Newborn Infant, 3d Ed., eds. Remington, J. S. & Klein, J. O. (Saunders, Philadelphia), Vol. 6, pp. 241–281.
4. Hutto, S. C. & Pass, R. F. (1985) Pediatr. Res. 19, A202.
5. Faiz, R. G. (1973) J. Pediatr. (St. Louis) 106, 649–752.
6. Alford, C. A., Stagno, S., Pass, R. F. & Huang, E. S. (1980) in The Human Herpesviruses: Interdisciplinary Perspective, eds. Nahmias, A., Dowdle, W. & Schinazi, R. (Elsevier, New York), pp. 159–171.
7. Pass, R. F., August, A. M., Dworsky, M. E. & Reynolds, D. W. (1982) N. Engl. J. Med. 307, 477–479.
8. Stagno, S., Reynolds, D. W., Pass, R. F. & Alford, C. A. (1980) N. Engl. J. Med. 302, 1073–1076.
9. Diosi, P., Babusceac, L., Nevinglovschi, O. & Kum-Stoleu, G. (1967) Lancet 1, 1063.
10. Hayes, K., Danks, D. M., Gibas, H. & Jack, I. (1972) N. Engl. J. Med. 287, 177–178.
11. Dworsky, J., Yow, M., Stagno, S., Pass, R. F. & Alford, C. (1983) Pediatrics 72, 295–299.
12. American Academy of Pediatrics, Task Force Report (1982) Pediatrics 60, 654–661.
13. Pass, R. F. (1986) in Human Milk in Infant Nutrition and Health , eds. Howell, R. R., Morris, F. H., Jr., & Pickering, K. (Springfield, IL), pp. 205–224.
14. Setterly, K. & Graubard, B. (1984) Morbid. Mortal. Wkly. Rep. 33, 153–154.
15. Dawson, D. A. & Cain, V. S. (1990) Vital & Health Statistics of the National Center for Health Statistics 187, (Natl. Cent. Health Statistics, Rockville, MD), No. 187, pp. 1–12.
16. Lang, D. J., Garruto, R. M. & Gajdusek, D. C. (1977) Am. J. Epidemiol. 105, 480–487.
17. Sarov, B., Naggan, L., Rosenzveig, R., Katz, S., Haikin, H. & Sarov, I. (1988) J. Med. Virol. 10, 195–201.
18. Adler, S. P. (1988) Pediatr. Infect. Dis. J. 7, 279–285.
19. Strangert, L., Carlstrom, G., Jeansson, S. & Nord, C. E. (1976) Acta Paediatr. Scand. 65, 455–463.
20. Adler, S. P. (1985) J. Infect. Dis. 152, 760–768.
21. Hutto, C., Ricks, R., Garvie, M. & Pass, R. F. (1985) Pediatr. Infect. Dis. 4, 149–152.
22. Jones, L. A. & Duke-Duncan, P. M. (1985) J. Infect. Dis. 151, 953–955.
23. Adler, S. P. (1988) J. Pediatr. (St. Louis) 112, 366–372.
24. Murph, J. R., Bale, J. F., Murray, J. C., Stinsky, M. F. & Perlman, S. (1986) J. Pediatr. (St. Louis) 109, 35–39.
25. Grillner, L. & Strangert, L. (1986) Pediatr. Infect. Dis. 5, 184–197.
26. Pass, R. F., Little, E. A., Stagno, S., Britt, W. J. & Alford, C. (1987) N. Engl. J. Med. 316, 1366–1370.
27. Schupfer, P. C., Murph, J. R. & Bale, J. F., Jr. (1986) Pediatr. Infect. Dis. 5, 677–679.
28. Adler, S. P. (1986) Pediatr. Infect. Dis. J. 5, 315–318.
29. Hutto, C., Little, E. A., Ricks, R., Lee, J. D. & Pass, R. F. (1986) J. Infect. Dis. 154, 527–530.
30. Spector, S. A. & Spector, D. H. (1982) Pediatr. Infect. Dis. 1, 405–409.
31. Dworsky, M., Lakeman, A. & Stagno, S. (1984) Pediatr. Infect. Dis. 3, 236–238.
32. Yeager, A. S. (1983) Pediatr. Infect. dis. 2, 295–297.
33. Dworsky, M. E., Welch, K., Cassady, G. & Stagno, S. (1983) N. Engl. J. Med. 309, 950–953.
34. Taber, L. H., Frank, A. L., Yow, M. D. & Bagley, A. (1985) J. Infect. Dis. 151, 948–952.
35. Pass, R. F., Hutto, C., Ricks, R. & Cloud, G. (1986) N. Engl. J. Med. 314, 1414–1416.
36. Pass, R. F. & Hutto, C. (1986) Rev. Infect. Dis. 8, 599–605.
37. Adler, S. P. (1989) N. Engl. J. Med. 321, 1290–1300.
38. Pass, R. F. (1990) Semin. Pediatr. Infect. Dis. 1, 245–251.
39. Fowler, K. B., Stagno, S., Pass, R. F., Britt, W. J., Boll, T. J. & Alford, C. A. (1992) N. Engl. J. Med. 326, 663–667.
















