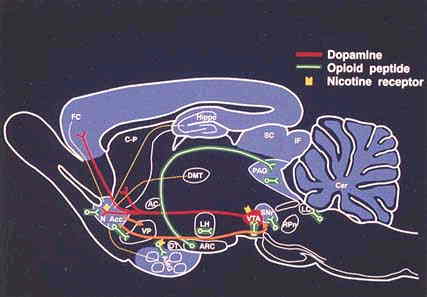
FIGURE 3.1 Sagittal rat brain section illustrating a drug (cocaine, amphetamine, opiate, and alcohol) neural reward circuit that includes a limbic-extrapyramidal motor interface. Yellow indicates limbic afferents to the nucleus accumbens (N Acc.), and orange represents efferents from the nucleus accumbens thought to be involved in psychomotor stimulant reward. Red indicates projection of the mesocorticolimbic dopamine system thought to be a critical substrate for psychomotor stimulant reward. This system originates in the A10 cell group of the ventral tegmental area (VTA) and projects to the N. Acc., olfactory tubercle, and ventral striatal domains of the caudate-putamen (C-P). Green indicates opioid peptidecontaining neurons, systems that may be involved in opiate and ethanol reward. These opioid peptide systems include the local enkephalin circuits (short segments) and the hypothalamic midbrain beta-endorphin circuit (long segment). Blue indicates the approximate distribution of GABA-A receptor complexes, some of which may mediate sedative/hypnotic (ethanol) reward, determined by both tritiated flumazenil binding and expression of the alpha, beta, and gamma subunits of the GABA-A receptor. Yellow refers to nicotinic receptors hypothesized to be localized on dopamine and opioid peptide systems. AC, anterior commissure; AMG, amygdala; Cer, cerebullum; DMT, dorsomedial thalamus; FC, frontal cortex; Hippo, hippocampus; LC, locus coeruleus; LH, lateral hypothalamus; OT, olfactory tract; PAG, periaqueductal gray; SNr, substantia nigra pars reticulata; VP, ventral pallidum. Modified with permission of Elsevier Science LTD., from Koob, 1992a.
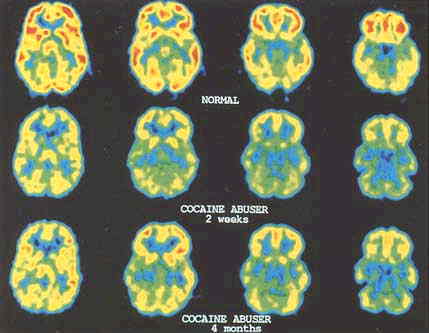
FIGURE 3.2 Metabolic images obtained with FDG in a normal control and in a cocaine abuser tested 3 months after cocaine discontinuation. Notice the reductions in metabolism in frontal brain regions when compared with the control. Reprinted with permission from N. Volkow, Brookhaven National Laboratory.
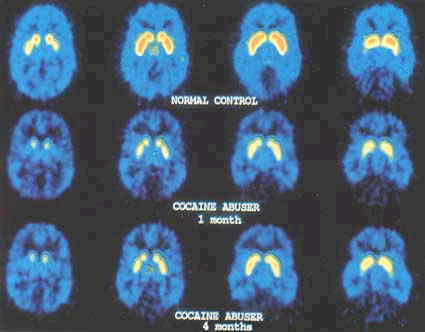
FIGURE 3.3 [18F]N-methylspiroperidol images in a normal control and in a cocaine abuser tested 1 month and 4 months after last cocaine use. The images correspond to the four sequential planes where the basal ganglia are located. Notice the lower uptake of the tracer in the cocaine abuser when compared with the normal control. Notice the persistence of the decreased uptake even after 4 months of cocaine discontinuation (Reprinted by permission of John Wiley & Sons, Inc. from Volkow et al., 1993).



