9 Postreproductive Survival
Steven N. Austad
Postreproductive Survival In Nature
Women in modern societies can expect to live nearly one-third of their adult lives in a postreproductive state. Whether this phenomenon is relatively new in human experience or whether it represents something that has been a part of human life for millennia may be relevant to understanding medical and social issues surrounding postmenopausal life. One approach to an understanding of the human significance of menopause is to examine it comparatively. How commonly is postreproductive life found in nature, and under what circumstances is it found? Is there a reason to assume that humans are somehow special in this regard?
Some of the special pleading for a novel evolutionary explanation for complete reproductive cessation and a long postreproductive life in human females derives from the misimpression that the more rapid senescent decline in reproductive capacity in women relative to men is a life-history feature unique to humans (Hill and Hurtado, 1991; Lancaster and King, 1992). On the contrary, such a pattern is common among well-maintained captive species such as laboratory rats (Austad, 1994) (see Figure 9-1).
This pattern is not surprising given the dynamics of oocyte formation and depletion in mammals. With possibly a few exceptions, female mammals do not exhibit continuous oocyte formation. Oocyte formation is terminated either before birth or shortly after, so there is a finite oocyte pool that is gradually depleted during life (see Finch, 1990, for details). Given this constraint on oocyte numbers, it would make evolutionary sense to assume that natural selection would ensure that sufficient oocytes were produced initially and that the rate of deple-
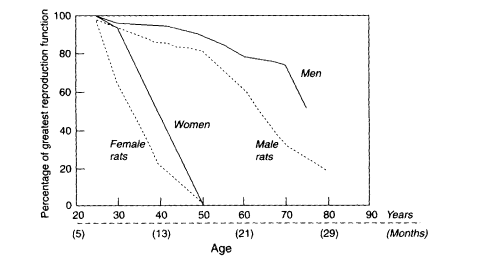
Figure 9-1
Comparison between humans and captive rats in sex-specific reproductive senescence.
tion is such that oocytes would be available at least as long as animals could expect to live under natural conditions (that is, under the conditions that this physiology evolved) unless there were some adaptive advantage to ceasing reproduction before death.
Consistent with this logic, physiologically postreproductive individuals are very rarely found in nature except in a few species (described below). This is not to say that long intensive field studies will not occasionally identify postreproductive individuals (e.g., Waser, 1978), only that they should always be rare. A typical example is the study of great tits (Parus major) in Wytham Woods near Oxford, U.K., where analysis of demographic data collected over 18 years suggests that 7-year-old females and 8-year-old males may be physiologically postreproductive. But for every thousand females or males fledged, only four females and two males will survive to postreproductive ages, respectively (McCleery and Perrins, 1988).
By contrast, male gamete production is continuous throughout life. Therefore under captive conditions in which the major sources of natural mortality are removed as life span is much extended, it is not surprising that males continue to be reproductively competent somewhat longer than females.
While it is true that menopause in nonhuman primates occurs only near the end of life under captivity (van Wagenan, 1972; Nozaki et al., 1995), this may reflect more about the current state of primate husbandry than about differences among primates in reproductive cessation.
A type of postreproductive condition that is much more common in nature
than physiological reproductive cessation, especially among strongly polygynous species, is age-related, behaviorally imposed reproductive cessation. For instance, when aging males can no longer successfully defend a harem against younger male competitors, reproduction ceases, not from senile sterility but from lack of opportunity. This is apparently true for red deer (Cervus elaphus) stags at age 11 or greater on the Scots island of Rhum (Clutton-Brock et al., 1988). In this type of case, postreproductive individuals are not necessarily expected to be rare. Thus, fully 20 percent of all red deer stags born on Rhum are still alive at an age when at least some individuals are behaviorally postreproductive (Clutton-Brock et al., 1988). The loss of blatantly physical competitive ability with age is explicable from standard analyses of the evolution of senescence. It requires no special interpretation and will not be discussed further.
In striking contrast to the rarity of physiologically postreproductive individuals in nature, such animals are often common in captivity (vom Saal and Finch, 1988). This fact is not surprising. Animals in captivity are protected from food and water shortages, environmental extremes, predators, and many pathogens. Consequently, they live much longer than in nature and, in fact, can frequently outlive the period over which natural selection has molded their reproductive physiology to remain intact. A well-documented example of this situation is the house mouse (Mus musculus), which in nature lives about 4 months, on average, and has 90 percent mortality by 9-10 months of age (Phelan and Austad, 1989). In the laboratory, of course, house mice average 2+ years of life (Myers, 1978), and females begin becoming postreproductive near the end of their first year of life, about the time they would almost certainly be dead in nature. Some information suggests that wild-type house mouse females can remain reproductively active until almost 2 years (Sacher and Hart, 1978).
Among our closest relatives, the other primates, a similar condition prevails—physiologically postreproductive individuals are rare to nonexistent in nature but occur in significant numbers in captivity. Japanese (Macaca fuscata) and rhesus macaques (Macaca mulatta), for instance, begin to experience female reproductive decline at about age 20 and undergo menopause at about 27 and 30 years, respectively (Nozaki et al., 1995; van Wagenan, 1972). But even in human-protected and food-provisioned semi-natural populations, median survival is less than 5 and 10 years, respectively, with maximum survival less than 25 years (Dunbar, 1986). Therefore, as in other mammals, the commonness of captive postreproductive survival is a consequence of increased captive longevity with no evolutionary significance. These animals were simply not physiologically designed for such a long survival.
Significantly though, there do seem to be at least a few species in nature in which postreproductive survival is relatively common. The best-documented example is female short-finned pilot whales, Globicephala macrorhynchus, in which 24 percent of all females born have been reported to live past the physiological end of reproduction (Marsh and Kasuya, 1984; see Table 9-1). The life
TABLE 9-1 Paleo-humans Compared with Cetaceans Known to Have a Natural Postreproductive Life
|
Trait |
Human |
Killer Whale |
Pilot Whale |
|
Proportion living to postreproductive ages (%) |
5-25a |
29 |
24 |
|
Life expectancy at end of reproduction (years) |
3-22 |
26 |
14 |
|
Age at maturity (years) |
15-19 |
15 |
9-18 |
|
Period of obligatory offspring care (years) |
10-12 |
? |
? |
|
aLower estimates for humans come from paleodemographers, who estimate age at death from osteological cues. whereas higher estimates come from the study of modern hunter-gatherers. Conclusions of these two groups of researchers dramatically differ. SOURCES: Marsh and Kasuya (1984). Kasuya and Marsh (1984), and Olesiuk et al. (1990). |
|||
expectancy of females at the end of their reproductive lives is 14 years (Kasuya and Marsh, 1984). Although not confirmed by ovarian histology, as in the pilot whales, detailed long-term observations and demographic analyses of resident killer whales along the northwestern coast of North America suggest that in a stable population about 30 percent of females born will live to become postreproductive and that their life expectancy at the end of reproduction will be more than 25 years (Olesiuk et al., 1990). A significant female postreproductive life is also suspected in several other species of toothed whales (Marsh and Kasuya, 1986). Life expectancy and maximum reported age is dramatically shorter in males relative to females in both pilot and killer whales.
In neither of these cetacean species is there any evidence that mortality rates have plunged recently, so this long period of postreproductive life does seem to be a product of natural selection rather than an artifact of a recent rapid increase in longevity.
Several hypotheses have been advanced to explain common postreproductive female survival, all assume that at some age it becomes more evolutionarily advantageous to redirect energy from continued personal reproduction to facilitating the survival and/or reproduction of existing offspring. Note that these hypotheses agree in predicting that postreproductive individuals will be actively involved in helping younger individuals.
The logic of hypotheses for adaptive postreproductive survival is that as the risk of reproductive failure and/or death increases with age in species with an
extended obligatory period of postnatal parental care, there will eventually come an age when the expected fitness gain of attempted personal reproduction will be lower than that available from redirecting energy toward rearing the last-born offspring to independence and/or assisting already-independent progeny with their reproduction. Specific quantitative models of adaptive menopause reveal that the conditions under which it is favored are stringent, however, requiring enormously effective assistance of young individuals by postreproductive females (Hill and Hurtado, 1991; Rogers, 1993).
Although relatively little is known about the social system of pilot whales or killer whales, existing knowledge is consistent with such adaptive hypotheses. Pilot whales live in breeding schools of a few to a few dozen individuals, with females thought to remain in their natal school throughout their lives. Therefore breeding schools are matrilineal kin groups. Females mature at 9 years of age (range: 7-12) and produce an average of four to five calves before ceasing reproduction at 30-40 years of age (Kasuya and Marsh, 1984). Males are probably functionally mature by age 16 but may not mate for several years after this. Calves of young females are nursed for 2+ years, but as females approach the end of reproduction, the duration of nursing increases substantially, so that postreproductive mothers wean their daughters at ages up to 8 years and wean their sons at up to 15 years. In either case, weaning occurs as the reproductive age of the calf is approached—long past the end of their mother's reproductive potential.
Killer whales similarly live in multigenerational groups. Pods averaged 1026 individuals in the northern and southern coastal study populations (Olesiuk et al., 1990), and neither males nor females are thought to emigrate from their natal pod (Bigg et al., 1990). Both sexes reach sexual maturity at about 15 years of age. Females produce a calf at about 6-year intervals and become postreproductive at about age 40. Information about lactational duration of older females specifically or other types of parental care in killer whales is lacking, although that lactation is known to last for only about 2 years in younger mothers, and calves travel and rest close to their mothers for at least 6 years. When a female has a second calf before the previous one is sexually mature, an ''auntie" (who may be postreproductive) will assume a pseudomaternal role and spend a great deal of time with the older calf (Haenel, 1986).
The potential growth and survival advantages of reliable maternal resources to their own offspring is self-evident, and postreproductive females are likely to have three or more generations of kin in the same social group. Simultaneous assistance to several generations of descendant and nondescendent kin is therefore possible. It is notable in this regard that one of the other whale species with suggestions of postreproductive female life (the sperm whale. Physeter macrocephalus ) also lives in stable cohesive multigenerational female kinship groups (Marsh and Kasuya, 1986).
Modern human females, of course, also have a widespread and extensive postreproductive life. However whether this postreproductive life is an adaptive
consequence of natural selection, as in short-finned pilot whales, or is a nonadaptive artifact of the rapid increase in longevity over the past few centuries analogous to the postreproductive life of captive animals has been controversial (for an alternative interpretation from that found here, see Nesse and Williams, 1994). The potential significance of this issue is not merely abstract or trivial. If menopause represents an artifact of modern life, then it also probably represents an uncontrolled degenerative loss of homeostasis associated with senescence. In this case, one cannot assume that the normal menopausal hyposteroidal state will facilitate optimum health. Hormonal intervention may be expected to improve overall postreproductive health. Indeed, the ability of postmenopausal hormone-replacement therapy to mitigate some life-threatening conditions, such as accelerated bone thinning and an accelerating risk of cardiovascular disease (Marshburn and Carr, 1994), and reduce overall mortality rate (Gura, 1995; Ettinger et al., 1996) might be considered evidence for this interpretation. In this case, there is also no reason to suppose that an uncontrolled degenerative loss of homeostasis will proceed similarly from species to species, and there is no reason to suppose that rodent or even primate models of the postreproductive state will provide revealing mechanistic details of the postreproductive state in humans.
On the other hand, if menopause is an adaptive physiological state molded by evolution to maximize fitness in the face of generalized senescence, then natural selection would presumably have tailored postreproductive physiology to the hyposteroidal state, suggesting that medical interventions to alleviate menopausal conditions should be approached with considerably more caution.
To what extent did humans in our evolutionary past have life histories similar to those of modern cetaceans with a known postreproductive existence (Table 9-1 )?
Any hypothesis of adaptive human menopause requires at least two types of supporting evidence. First, it must be established that humans before recent medical and public health advances lived long enough for menopausal years to be a common part of life. Otherwise, selection would have no opportunity to favor the putative adaptations of postmenopausal existence. Second, it must be established that postreproductive females can provide sufficient help to their younger kin to offset the reproduction they forfeited when menopause occurred. That there is considerable consistency in the age of menopause (about 45-50 years, on average) across cultures and environments does not seem relevant to this issue, given the rather deterministic dynamics of mammalian oocyte depletion.
The evidence relevant to preagricultural survival is dichotomous. Researchers studying living peoples without apparent access to the obvious benefits of modern medicine and hygiene such as the !Kung San of the Kalahari Desert or the Aché of the Paraguayan forests find that as many as 30-40 percent of women survive into their postreproductive years (Hill and Hurtado, 1991) (Figure 9-2) and that life expectancy at the age of menopause is about 20 years. On the other hand, paleodemographers, who estimate the demography of past human periods such as among Paleolithic hunter-gatherers (Weiss, 1973)or primitive agricultur-
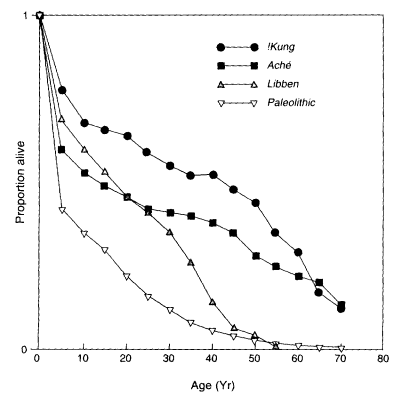
Figure 9-2
Survivorship estimates of some living (!Kung and Aché) and nonliving (Libben and Paleolithic) nontechnological populations. Estimates for living populations were made from personal interviews; estimates for nonliving populations were made from osteological examination of cemetary remains. SOURCES: Lovejoy et al. (1977), Howell (1979), Weiss (1973), and Hill and Hurtado (1991).
alists (Lovejoy et al., 1977) reach just as unanimous a conclusion that humans until recently did not live into their postreproductive years except very rarely. This disagreement may be unresolvable. Living nontechnological peoples may be more influenced by modernity than researchers imagine, or conversely there may be systematic errors in the age estimation of very old skeletal remains. With such a fundamental contradiction in evidence, this aspect of the adaptive menopause hypothesis must await further evidence.
The second aspect of the hypothesis—whether postreproductive females, if alive, could provide sufficient help to kin to offset their own potential reproduction—has been approached primarily from a theoretical perspective. For instance, a model by Hill and Hurtado (1991), combined with the demography they collected from the forest-dwelling Aché suggested that even given the higher esti-
mates of human survival that anthropologists studying modern hunter-gatherers find, postreproductive women did not provide enough assistance to offset the potential personal reproduction lost due to menopause. The model assumed that hypothetical women who did not undergo menopause would still experience a reproductive decline, such that fertility by age 65 would be 43 percent of maximum fertility and that increased maternal death risk at later ages had a trivial demographic impact. Nevertheless, the authors were hesitant to conclude that menopause was nonadaptive and theorized that if in the past older women could have improved the survival of their children and grandchildren more than they were doing at present, if they also improved the survival of relatives in addition to their own children and grandchildren, and if older women were more likely than observed to die in childbirth, then evolution might indeed have favored an adaptive menopause. A ringing endorsement of adaptive menopause this clearly isn't!
A somewhat different approach was used by Rogers (1993), who used the particularly detailed demographic information from Chinese farmers in turn-of-the-century Taiwan as a surrogate demographic model of Paleolithic human life to ask mathematically exactly how much help postreproductive mothers would have had to provide to their children for adaptive menopause to have evolved. His analytical model addressed effects on both survival and fertility for several generations of descendents. He assumed that the costs of continued fertility past menopause were due to either an increased probability of death during childbirth, a reduced ability of a mother to care for her older existing offspring, or a combination of both of these effects. His analysis revealed that the presence of a postmenopausal "mother" would have had to double the number of children all of her children had and completely eliminate infant deaths for evolution to have produced menopause by natural selection. As in Hill and Hurtado's model, the conditions favoring adaptive menopause seemed unlikely to hold. Unfortunately, whales have not been similarly analyzed to date.
Although this issue must remain unresolved at present, there is certainly no compelling reason to believe that human menopause is the adaptive result of selection for reproductive cessation. We seem to resemble laboratory mice more than killer whales.
The Role Of The Elderly In Nature
This thesis is not to say that elderly humans and other mammals do not have specialized roles in nature. But we need an instructive definition of what "elderly" means. If, as was formerly often assumed, animals did not exhibit senescence in nature (as measured by a mortality rate unrelated to age), then the concept of elderly is meaningless, because no matter what age an animal achieves, its expectation of further life remains undiminished. However, measurable senescence among mammals and birds now appears widespread in nature (Promislow, 1991; Holmes and Austad, 1995), so that even though a postre-
productive existence or the type of extreme senile debilitation found in captive animals and some elderly humans still appears rare, older animals often clearly have reduced expectation of life compared with younger animals. Thus in many natural populations, demographic studies allow the definition of ages beyond which animals will probably not survive, at which the likelihood of death is substantially increased, and those ages might sensibly be considered elderly. regardless of whether those animals are obviously debilitated at that time in life.
Given this definition of elderly, animals that acquire or accumulate important resources such as food caches, complex and costly burrow systems or nests, or foraging and breeding territories during their lifetime will inevitably influence their own reproductive success and the ultimate genetic structure of populations by the manner in which those resources are disposed of as they approach the end of life—specifically, whether those resources will be available to offspring.
The material value of some of these resources are considerable. For instance, all bannertail kangaroo rats (Dipodomys specabilis) require mounds in which to live. These mounds consist of an intricate burrow system and massive quantities of hoarded seeds. Mounds persist for much longer than the life of any individual, probably for decades. Construction of new mounds requires as long as 2 years (Best, 1972) and thus is rarely initiated (Holdenreid, 1957). Therefore sharing such a mound or somehow bequeathing it to one's offspring obviates their need to obtain a mound of their own.
Possible scenarios of the transfer of important material resources include (1) preventing adult offspring from making use of the resource at all, which I will call evicting them; (2) sharing the resource with adult offspring, which has the effect of bequeathing it upon their death; or (3) relinquishing resources to their offspring, while they are still alive and thereby sacrificing their own prospects to those of their offspring. Scenario 1 is most costly to offspring in terms of energy and risk associated with achieving independence. This risk can be substantial. particularly for small mammals. Female water voles (Arvicola terrestris), for instance, suffer 86 times the risk of predation when dispersing as when remaining in their natal home range (Leuze, 1980). Scenario 2 will be minimally costly to a parent and maximally beneficial to offspring if the expectation of continued parental life is low. Thus this pattern might be expected when overall mortality rates are high. In a high-mortality environment, the effect of resource transfer converges in scenario 2 and scenario 3. However, if a parent is likely to continue living, scenario 3 can be more or less costly than 2, depending upon the quality of the resources relinquished relative to those held. I will discuss representative examples of each of these scenarios in turn.
Eviction of Young from Resources Controlled by the Elderly
Any parentally forced dispersal from the parental home range would fall into this classification. Eviction not only has the effect of preventing one generation
from acquiring resources accumulated by the previous generation but also it places them at substantial risk while searching for new resources. In fact, when large amounts of resources are accumulated during one generation, parents will probably allow their offspring access to these resources well into adult life, unless their own life expectancy is still substantial. On the other hand, when life expectancy is still substantial, one might expect parental eviction of offspring to avoid direct competition for those resources. Indeed there is an encyclopedic literature (summarized in Baker, 1978) on the timing and extent of forced dispersal, but elderly individuals are by definition those with low life expectancy. Therefore, I expect that eviction of offspring will be rare among elderly animals. Systematic information on this sort of question has not been collected, mainly because field biologists have assumed that senescence is irrelevant to animals in the wild.
Bequeathing Resources to Young
Some mammals, at least occasionally, share access to valuable material resources with their offspring. For instance, bannertail kangaroo rat females frequently allow offspring, potentially of either sex, to share their home mounds (Jones, 1987). Thus a 3- or 4-year-old female who has a low life expectancy and shares a mound with her offspring is ultimately making a decision to bequeath them that mound. In a population in southeastern Arizona, 37 percent of males remained in their natal mound into adulthood, and 70 percent of these remained throughout their lives. Twenty-three percent of females remained in their natal mounds into adulthood, and 72 percent of these remained for their entire lives. Because mound improvement and renewing of cached seeds occur throughout life, resources acquired during the lifetime of females are passed along to their offspring. No information exists on whether males bequeath mounds to their offspring (Jones, 1987): however, it is improbable that males of this polygynous species can even recognize their own offspring.
In many species, this sort of resource bequeathing is sex-biased. As a gross generality, males are more apt to be bequeathed valuable resources such as nests, dens, or territories in birds, whereas females are more apt to inherit such resources in mammals (Greenwood, 1980). Thus in many communally breeding birds that defend valuable territories, territory ownership is passed down male lineages. For instance, among stripe-backed wrens (Campylorhynchus nuchalis), 60 percent of breeding males gained breeding status on their natal territory compared with 3 percent of breeding females (Rabenold, 1990). Conversely, among black-tailed prairie dogs (Cynomys ludovicianus) in Kansas, 98 percent of females bred in their natal coterie compared with 0 percent of males. The advantages of male-biased versus female-biased dispersal are still unresolved, but such dispersal is assumed to be related to aspects of mating system (Greenwood, 1980; Waser and Jones, 1983). It is generally assumed that dispersal of one sex is
necessary to avoid close inbreeding between parents and offspring or among siblings (Shields, 1982).
Relinquishing Resources to Offspring
Even if life expectancy is low, there is some finite chance of survival through one or more additional breeding episodes. In spite of this. adults of some species relinquish some or all of their existing resources to their offspring. For instance. adult female black bears (Ursus americanus) frequently shift their territories away from areas that overlap with their daughters, effectively reducing their own foraging area (Rogers, 1987). Daughters that are the recipients of territory in this manner grow at a faster rate than females that must acquire their own territory. Female wood rats sometimes completely abandon their middens to their young (Fitch and Rainey, 1956), apparently an even larger personal sacrifice than in black bears, but the consequences for either mother or daughter have not been investigated.
Because field biologists have generally not been interested in problems of aging, little evidence as to the adaptive nature of intergenerational resource transfer has accumulated. The possibilities are intriguing, however. For instance, if in a species such as the banner-tailed kangaroo rat, in which parents sometimes allow offspring to share their mound and sometimes do not, it is possible that such a decision is based on the life expectancy or senescent stage of that parent. Thus, parents that have begun to senesce and are consequently not likely to live much longer, would be more likely to allow mound sharing as a form of bequeathing the mound to their offspring. This sort of senescence-related question has yet to be specifically addressed by field biologists. To the extent that it is addressed in the coming years, a generalized theory of the ecology of resource transfer across generations may emerge.
References
Austad. S.N. 1994 Menopause: an evolutionary perspective. Experimental Gerontology 29:255-263.
Baker, R.R. 1978 The Evolutionary Ecology of Animal Migration. London: Hodder and Stroughton.
Best. T.L. 1972 Mound development by a pioneer population of the banner-tailed kangaroo rat Dipodomus spectabilis bailey Goldman, in eastern New Mexico. America Midland Naturalist 87:201206.
Bigg, M.A., P.F. Olesiuk, G.M. Ellis. J.K.B. Ford. and K.C. Balcomb III 1990 Social organization and geneology of resident killer whales (Orcinus-orca ) in the coastal waters of British Columbia Canada and Washington State USA. Report of the International Whaling Commission (Special Issue 12). Cambridge, England: International Whaling Commission.
Clutton-Brock, T.H., S.D. Albon, and F.E. Guinness 1988 Reproductive success in male and female red deer. In T.H. Clutton-Brock, ed., Reproductive Success. Chicago: University of Chicago Press.
Dunbar. R.I.M. 1986 Demography and reproduction. In B.B. Smuts. D.L. Cheney, R.M. Seyfarth, R.W. Wrangham, and T.T. Struhsaker, eds., Primate Societies. Chicago: University of Chicago Press.
Ettinger, B., G.D. Friedman, T. Bush, and C.P. Quesenberry, Jr. 1996 Reduced mortality associated with long-term postmenopausal estrogen therapy. Obstetrics and Gynecology 87:6-12.
Finch, C. E. 1990 Longevity, Senescence, and the Genome. Chicago: University of Chicago Press.
Fitch. H.S., and D.C. Rainey 1956 Ecological observations on the wood rat, Neotoma floridana. University of Kansas Publications. Museum of Natural History 8:499-533.
Greenwood P.J. 1980 Mating systems, philopatry and dispersal in birds and mammals. Animal Behavior 28:1140-1162.
Gura, T. 1995 Estrogen: key player in heart disease among women. Science 269:771-773.
Haenel, N.J. 1986 General notes on the behavioral ontogeny of Puget Sound killer whales and the occurrence of allomaternal behavior . In B.C. Kirkevold and J.S. Lockard, eds., Behavioral Biology of Killer Whales. New York: Alan R. Liss.
Hamilton, W.D. 1966 The moulding of senescence by natural selection. Journal of Theoretical Biology 12:12-45.
Hill, K., and A.M. Hurtado 1991 The evolution of premature reproductive senescence and menopause in human females: an evaluation of the "grandmother hypothesis." Human Nature 2:313-350.
Holdenreid, R. 1957 Natural history of the banner-tailed kangaroo rat in New Mexico. Journal of Mammalogy 38:330-350.
Holmes, D.J., and S.N. Austad 1995 Birds as animal models for the comparative biology of aging: A prospectus. Journal of Gerontlogy and Biological Science 50A:B59-B66.
Howell, N. 1979 Demography of the Dobe !Kung. New York: Academic Press.
Jones, W.T. 1987 Dispersal patterns in kangaroo rats. In B.D. Chepko-Sade and Z.T. Halpin, eds., Mammalian Dispersal Patterns. Chicago: University of Chicago Press.
Kasuya, T.. and H. Marsh 1984 Life history and reproductive biology of the short-finned pilot whale, Globicephala macrorhynchus, off the Pacific Coast of Japan. Report of the International Whaling Commission (Special Issue 6).
Lancaster, J.B., and B.J. King 1992 An evolutionary perspective on menopause. In V. Kerns and J.K. Brown, eds., In Her Prime. Urbana and Chicago: University of Illinois Press.
Leuze, C.C.K. 1980 The application of radio tracking and its effect on the behavioral ecology of the water vole, Arvicola terrestris. In C.J. Amlamer and D.W. MacDonald, eds., A Handbook on Biotelemetry and Radio Tracking. New York: Springer.
Lovejoy, C.O., R.S. Meindl, T.R. Pryzbeck, T.S. Barton, K.G. Heiple, and D. Kotting 1977 Paleodemography of the Libben Site, Ottawa County, Ohio. Science 198:291-293.
McCleery. R.H., and C.M. Perrins 1988 Lifetime reproductive success of the great tit, Parus major. In T.H. Clutton-Brock, ed., Reproductive Success. Chicago: University of Chicago Press.
Marsh. H., and T. Kasuya 1984 Changes in the ovaries of the short-finned pilot whale, Globicephala macrorhynchus, with age and reproductive activity. Report of the International Whaling Commission (Special Issue 6) .
1986 Evidence for reproductive senescence in female cetaceans. Report of the International Whaling Commission (Special Issue 8).
Marshburn, P.B., and B.R. Carr 1994 The menopause and hormone replacement therapy. In W.R. Hazzard, E.L. Bierman, J.P. Blass, W.H. Ettinger, and J.B. Halter, eds., Principles of Geriatric Medicine and Gerontology, 3rd ed. New York: McGraw-Hill.
Myers, D.D. 1978 Review of disease patterns and life span in aging mice: genetic and environmental interactions. Birth Defects: Original Article Series 14:41-53.
Nesse, R.M., and G.C. Williams 1994 Why We Get Sick: The New Science of Darwinian Medicine. New York: Times Books.
Nozaki, M., F. Mitsunaga. and K. Shimizu 1995 Reproductive senescence in female Japanese monkeys (Macaca fuscuata): Age- and season-related changes in hypothalamic-pituitary-ovarian functions and fecundity rates. Biology of Reproduction 52:1250-1257.
Olesiuk, P.F., M.A. Bigg, and G.M. Ellis 1990 Life history and population dynamics of resident killer whales (Orcinus orcu) in the coast waters of British Columbia and Washington State. Report of the International Whaling Commission (Special Issue 12). Cambridge, England: International Whaling Commission.
Phelan, J.P., and S.N. Austad 1989 Natural selection, dietary restriction, and extended longevity. Growth, Development and Aging 53:4-6.
Promislow, D.E.L. 1991 Senescence in natural populations of mammals: A comparative study. Evolution 45:1869-1887.
Rabenold, K.N. 1990 Campylorhynchus wrens: The ecology of delayed dispersal and cooperation in the Venezuelan savanna. In P.B. Stacey and W.D. Koenig, eds., Cooperative Breeding in Birds. Cambridge, U.K.: Cambridge University Press.
Rogers, A.R. 1993 Why menopause? Evolutionary Ecology 7:406-420.
Rogers, L.L. 1987 Factors influencing dispersal in the black bear. In B.D. Chepko-Sade and Z.T. Halpin, eds., Mammalian Dispersal Patterns. Chicago: University of Chicago Press.
Sacher, G.A., and R.W. Hart 1978 Longevity, aging and comparative cellular and molecular biology of the house mouse. Mus musculus. and the white-footed mouse, Peromyscus leucopus. Birth Defects: Original Article Series 14:71-96.
Shields, W. 1982 Philopatry. Inbreeding, and the Evolution of Sex. Albany. NY: State University of New York Press.
van Wagenan, G. 1972 Vital statistics from a breeding colony: Reproduction and pregnancy outcome in Macaca mulatta. Journal of Medical Primatology 1:3-28.
vom Saal, F.S., and C.E. Finch 1988 Reproductive senescence: Phenomena and mechanisms in mammals and selected vertebrates. In E. Knobil and J. Neill et al., eds., The Physiology of Reproduction. New York: Raven Press, Ltd.
Waser, P.M. 1978 Postreproductive survival and behavior in a free-ranging female mangabey. Folia Primatologica 29:142-160.
Waser. P.M., and W.T. Jones 1983 Natal philopatry among solitary mammals. Quarterly Review of Biology 58:355-390.
Weiss, K.M. 1973 Demographic models for anthropology. Society for American Archaeology Memoirs 27:11-86.














