7
Renal Disease
In the past, the development of severe renal disease signaled certain death within a short period of time. With the launch of hemodialysis for the management of end-stage renal disease (ESRD) in 1960 and subsequent developments such as peritoneal dialysis, home hemodialysis, and renal transplantation, the survival period for persons with severe renal disease has markedly increased. While the prognosis for persons with ESRD has improved, the number of patients continues to grow. As of December 31, 1995, the point prevalence of ESRD was 257,266 individuals (USRDS, 1997). As of December 31, 1997, the point prevalence grew to 304,083 (USRDS, 1999). Table 7.1 provides the demographic distribution of new cases of ESRD in 1997.
The above estimates indicate that the future economic burden will be great. In 1999, the total cost of care for patients with ESRD was approximately $15.64 billion (USRDS, 1999). This cost includes federal, state, and private sources of payment but does not include the cost of disability or Social Security monthly benefits. The estimated cost per person per year is $43,000 (USRDS, 1999).
Patients with ESRD experience significant morbidity and mortality and consume considerable health care resources. In 1995, dialysis patients over the age of 65 spent an average of 11.4 days per year in the hospital (USRDS, 1997). Medicare patients in general spent a mean of 7.1 days in the hospital per year (HCFA, 1999). Despite recent improvements in survival rates, the expected lifetime of a dialysis patient still ranges between 16 and 37 percent of the age-, gender-, and race-matched U.S. population
TABLE 7.1 Renal Disease Population as of December 31, 1997
|
|
End-Stage Renal Disease Patients |
Dialysis Patients |
||
|
|
Number |
Percent |
Number |
Percent |
|
Total |
304,083 |
100 |
221,596 |
100 |
|
Age Group |
||||
|
0–19 |
5,480 |
2 |
1,768 |
1 |
|
20–44 |
76,018 |
25 |
39,398 |
18 |
|
45–64 |
117,865 |
39 |
81,904 |
37 |
|
65–74 |
63,197 |
21 |
57,316 |
26 |
|
≥75 |
41,523 |
14 |
41,210 |
18 |
|
Race |
||||
|
Native American |
4,614 |
2 |
3,663 |
2 |
|
Asian/Pacific Islander |
10,795 |
4 |
7,885 |
4 |
|
African American |
97,503 |
32 |
82,624 |
37 |
|
Caucasian |
186,341 |
61 |
123,269 |
56 |
|
Other |
4,830 |
2 |
4,155 |
2 |
|
Gender |
||||
|
Male |
165,176 |
54 |
115,902 |
52 |
|
Female |
138,907 |
46 |
105,694 |
48 |
|
Cause of ESRD |
||||
|
Diabetes |
100,892 |
33 |
84,076 |
38 |
|
Hypertension |
72,961 |
24 |
61,673 |
28 |
|
Glomerulonephritis |
52,229 |
17 |
29,433 |
13 |
|
Other |
78,001 |
26 |
46,414 |
21 |
|
SOURCE: USRDS (1999). |
||||
(USRDS, 1997). The high morbidity and mortality among dialysis patients has stimulated research into potentially correctable factors that are associated with increased risk of death. The search for antecedent factors requires consideration of earlier stages of chronic renal disease.
Renal disease is marked by a decrease in the ability of the kidney to excrete metabolic waste and a subsequent build up of urea and other nitrogenous waste in the blood. For the purpose of this report, three stages of chronic renal disease are considered: chronic renal insufficiency, ESRD, and post-renal transplantation. Chronic renal insufficiency (CRI) is defined as the stage of renal disease associated with a reduction in renal function not severe enough to require dialysis or transplantation (glomerular filtration rate [GFR] 13–50 ml/min/1.73 m2). ESRD is defined as chronic renal disease that necessitates treatment by dialysis or renal trans-
plantation. Lastly, patients who have received a renal transplant are considered. Although many transplant recipients also have CRI, since the level of renal function usually remains subnormal after transplantation, for the purpose of this report, these patients are considered separately from those who have CRI, without a history of renal transplantation.
As described below, three key findings suggest that nutrition therapy may substantially lessen or delay the burden of chronic renal disease. First, dietary protein restriction in CRI may slow the onset of ESRD. Second, malnutrition during CRI is a major risk factor for death after the onset of ESRD. Third, patients at all stages of chronic renal disease are at high risk of cardiovascular disease, which may be prevented, in part, by treatment of traditional cardiovascular disease risk factors, such as hypertension, hyperlipidemia and diabetes.
CHRONIC RENAL INSUFFICIENCY
CRI can be detected and treated prior to the onset of ESRD. An elevation in serum creatinine concentration of approximately 1.5 mg/dL or greater indicates this stage of CRI. Diagnostic coding for individuals with CRI is not uniform and for this reason, the cost of providing care for these patients has not been analyzed.
Debate surrounds the question of the effectiveness of dietary protein restriction as a treatment for patients with moderate or early renal disease and as a method to delay the onset of ESRD. While convincing evidence in rats with reduced renal mass shows that deterioration is slowed with restriction of dietary protein (Ibels et al., 1978; Kleinknecht et al., 1979; Laouari et al., 1982), human studies have been less certain. During the past decade, a number of clinical trials on protein restriction in patients with CRI have been carried out.
Studies Including Patients with Diabetes
While more extensive experience is available in patients with nondiabetic renal disease, a few trials have been performed in patients with type 1 diabetes. Zeller and colleagues (1991) conducted a randomized trial in patients with type 1 diabetes (n = 35) who had greater than 1.0 g/ day urine protein excretion. Results demonstrated a slower decline in GFR in patients randomized to a low protein diet. Walker and colleagues (1989) also demonstrated a similar slowing in GFR decline in a sequential (non-randomized) trial of patients with type 1 diabetes (n = 19). Pedrini et al. (1996) performed a meta-analysis including these two studies, plus three others, which demonstrated a uniform finding of reduced progression of renal disease in patients assigned to dietary protein restriction.
Despite the results, the authors recommended caution because of the small number of patients included in the clinical trials and because of variation in study design and endpoints.
Modification of Diet in Renal Disease Study
Implemented in 1989, the Modification of Diet in Renal Disease (MDRD) Study was the largest clinical trial designed to determine the effects of a reduced protein and phosphorus diet on the progression of renal disease. While the multicenter, randomized trial included some persons over 65, the average age was 52 years (range 18–70 years). The MDRD Study was the first randomized clinical trial designed to determine the effects of a reduced protein and phosphorus diet on the progression of renal disease. It included two studies related to nutrition and the progression of renal disease:
-
Study A assessed low protein diets (0.58 g/kg body weight per day) combined with low phosphorus intakes (5 to 10 mg/kg body weight per day) in comparison to usual protein diets (1.3 g/kg body weight per day) combined with low phosphorus intakes (16 to 20 mg/kg body weight per day).
-
Study B assessed very low protein diets (0.28 g/kg body weight per day) combined with a low phosphorus intake (4 to 9 mg/kg body weight per day) in comparison to low protein diets (0.58 g/kg body weight per day) alone. The very low protein diet was supplemented with ketoacids, raising the total equivalent protein intake to 0.58 g/kg body weight per day.
In Study A, 585 patients with moderate renal disease, defined as a baseline GFR ranging from 25 to 55 mL/min per 1.73 m2 were randomly allocated to a low protein diet or a usual protein diet. Study B included 255 patients with advanced renal disease and a GFR of 13 to 24 ml/min per 1.73 m2 randomly assigned to a low protein diet or a very low protein diet. The primary outcome measure was the rate of change in GFR (Klahr et al., 1994).
This trial allowed careful monitoring of dietary potassium, sodium, and calcium intakes as well as corresponding biological indicators. For maximum patient adherence, participants were asked to record protein intake alone (MDRD Study Group, 1994). If phosphorus intakes were found by the dietitian to be out of range, he/she suggested changes in types of foods to reduce elevated levels. When other nutrient levels required adjustment, the nutritionist used laboratory values and food record
intakes to help patients change their types of foods in order to bring nutrient values back to normal ranges (Milas et al., 1995).
Initial results of the study showed that low protein dietary intervention was not conclusive in delaying the progression of renal disease (Klahr et al., 1994). For patients in Study A, the rate of decline in GFR over 36 months was not significantly different between the diet groups. It is proposed that this might be attributed to a nonlinear decline in GFR and a follow-up period that ended too quickly for dietary effects to be observed. For many patients, the MDRD diet was initially very difficult to follow. Many patients needed several months to acclimate to major changes in dietary habits. Data in Figure 7.1 show that patients in the low protein group demonstrated a faster rate of GFR decline in the first 4 months than those in the usual protein group. On the other hand, after 4 months the mean decline in GFR was slower in the low protein group compared to the usual protein group.
One of the limitations of the MDRD Study was the exclusion of subjects with diabetes. Presently, 38 percent of all ESRD patients have diabetes. A second limitation of the Study was its short duration, with an average intervention time of only 2.2 years.
Many have interpreted the failure of the MDRD Study to demonstrate a beneficial effect of protein restriction over a 2- to 3-year period as proving that low protein diets do not slow renal disease. Levey and coworkers (1999) state that this is an incorrect view and a result of misinterpretation of the inconclusive result as evidence in favor of the null hypothesis.
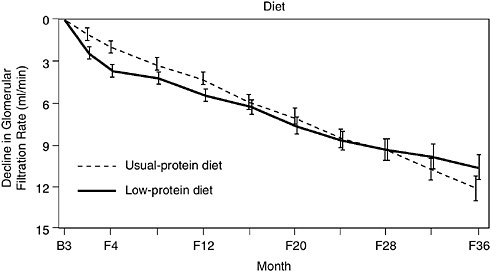
FIGURE 7.1 Estimated mean (±SE) decline in the glomerular filtration rate from base line (B) to selected follow-up time (F).
Following the initial analysis of MDRD Study data, secondary analyses have clarified the effect of protein restriction on the rate of decline in renal function, urine protein excretion, and onset of ESRD (Levey et al., 1999). Early analyses of patients in study B had showed a trend (p = 0.07) toward a slower progression of renal disease in the very low protein diet group versus the low protein group. Levey and colleagues (1996) conducted a secondary analysis of those Study B patients with advanced renal disease and demonstrated a correlation between achieving low protein intake and rate of decline in GFR. In this study, patients who complied very well with the diet were compared to those who had lower adherence. As described earlier, dietary intervention in MDRD included individualized counseling using feedback from dietary self-monitoring and laboratory data, modeling with low protein products, and dietitian support (Gillis et al., 1995).
More recently, a meta-analysis of five studies (Pedrini et al., 1996), which included the MDRD Study A, reported a beneficial effect of a low protein diet on the incidence of renal failure or death (relative risk [RR] = 0.67, 95 percent confidence interval [CI] = 0.50–0.89, p ≤ 0.01). All studies had a follow-up of more than 1 year, and information about the number of patients who died or developed renal failure was available. The prescribed protein intake in the low protein diet arms of these studies ranged from 0.4 to 0.6 g/kg per day. These results are consistent with those from an earlier meta-analysis (odds ratio [OR] = 0.54, 95 percent CI = 0.37–0.79, p < 0.002) (Fouque et al., 1992).
In a third meta-analysis, Kasiske and colleagues (1998) focused specifically on the rate of decline in renal function. In results pooled from 13 randomized, controlled trials along with 11 other nonrandomized, controlled trials, the effect of dietary protein restriction on rate of decline in renal function (GFR decreases in treatment subjects minus decrease in control subjects) was significantly less (p < 0.05) in randomized versus nonrandomized trials (regression coefficient 5.2 mL/min per year, 95 percent CI = –7.8 to –2.5 mL/min per year), relatively greater among patients with diabetes than those without (5.4 mL/min per year; 95 percent CI = 0.3–10.5 mL/min per year, p < 0.05); and although not statistically significant, demonstrated a trend toward greater effect with each additional year of follow-up (2.1 mL/min per year, 95 percent CI = –0.05 to 4.2 mL/ min per year, p > 0.05) (Kasiske et al., 1998).
Meta-analysis of randomized trials of protein restriction has both advantages and disadvantages. One advantage is the larger number of patients compared to results from a single clinical trial. Use of outcome measures such as ESRD or death can be used as a primary outcome measure, thus avoiding the problems of using surrogate outcomes based on the rate of decline in measures of renal function (Levey et al., 1999). Lastly,
using a time-to-event analysis may have more statistical meaning than comparisons of the rate of decline in renal function (Greene et al., 1995).
Potential disadvantages also exist in meta-analyses that use the onset of ESRD or death as the primary outcome. The disadvantage is that the analysis gives weight to patients with advanced renal disease or rapidly declining renal function. This occurs because these patients will develop ESRD more quickly during the relatively short follow-up times of these studies. A second disadvantage is that patients who are randomized to the low protein intervention group may be diagnosed with ESRD later than persons assigned to the usual protein intervention group. This results because the low protein intervention masks the symptoms of uremia or because of investigator bias in these unmasked studies. Finally, two well-recognized disadvantages of meta-analysis result from the possibility of non-uniformity of studies and publication bias (Levey et al., 1999).
Based on secondary analyses of the MDRD study results in conjunction with data from other randomized trials, it has been recommended that a protein intake of 0.6 g/kg/day for patients with chronic renal disease should be recommended (Levey et al., 1999). Because older individuals make up a large fraction of those with renal insufficiency and because there is motivation to follow this rather restricted diet because of the negative quality of life associated with dialysis treatment, older adults with renal insufficiency should be included in dietary treatment recommendations.
Nutritional Management of Chronic Renal Insufficiency Consensus Recommendations
In 1994, a conference in Bethesda, Maryland, supported by the National Institutes of Health, was held for the purpose of developing management recommendations for chronic renal disease. Panels of nephrology experts from around the world reviewed literature and developed practical recommendations for the prevention of progression of various stages of chronic renal disease. Recommendations for the nutritional management of CRI were as follows (Striker, 1995):
Nutritional Management, GFR 25–55 mL/min/1.73 m2
The evidence that the prescription of a low-protein diet slows the progression of renal failure in this group of patients is still inconclusive. Data from an additional year’s follow-up of the MDRD cohort shows even stronger trends in the direction of the benefit of a low-protein intake. However, until definitive information is available from longer-term studies, further group analysis of the MDRD or additional data from other studies, a standard protein intake should be prescribed (>0.8 g/kg
per day). If there is evidence of the progression of renal insufficiency or the development of uremic symptoms, an intake of 0.8 g/kg per day is appropriate.
Nutritional Management, GFR 13–25 ml/min/1.73 m2
Those who adhere to a diet containing 0.6 g/kg per day of protein have a reduced rate of loss of renal function, and the time to reach ESRD is prolonged. In addition, dietary protein restriction delays the onset of the uremic syndrome.
Nutritional Status during Chronic Renal Insufficiency
As will be discussed later, protein–energy undernutrition is a well-described important risk factor for morbidity and mortality in patients maintained on dialysis. Many studies have demonstrated a high prevalence of protein–energy undernutrition in patients beginning dialysis, suggesting that this disorder begins during the stage of CRI.
The causes of protein–energy undernutrition in patients with CRI are similar to those in ESRD (reviewed later). Of particular importance is decreased protein and energy intake. A recent cross-sectional analysis of the large number of patients enrolled in the MDRD Study baseline cohort clearly demonstrated reduced protein and energy intake in patients with GFRs as high as 50 ml/min per 1.73 m2, which became progressively impaired with further reduction in GFR (Kopple et al., in press). Progressive reduction in parameters of nutritional status including body weight, serum albumin and transferrin concentrations, skinfold thickness, arm muscle area, and urinary creatinine excretion accompanied these changes. These findings are particularly important because patients with type 1 diabetes and severe cardiovascular disease were excluded from this study. Thus, decreased nutrient intake and nutritional status did not appear to be the result of other coexisting conditions.
These data suggest that protein and energy intake, as well as nutritional status, begin to decline early during the stage of CRI, and worsen progressively as patients approach ESRD. It is reasonably hypothesized, but not yet proven, that preventing the development of malnutrition during this phase of CRI may reduce the consequences of malnutrition during ESRD. Thus, attention to diet in CRI is essential, not only to slow the progression of renal disease, but also to maintain nutritional status. Restriction of dietary protein, while maintaining adequate intakes of quality protein and total energy requires appropriate dietary prescription, counseling, and follow-up by a trained dietitian.
Renal Insufficiency as a Risk Factor for Cardiovascular Disease
Cardiovascular disease is the leading cause of death in patients with ESRD. In the general population, cardiovascular disease morbidity and mortality have declined substantially over the past three decades through risk-factor identification and reduction and more effective treatment of coronary artery disease. In 1997, the National Kidney Foundation convened a task force, consisting of experts in cardiovascular disease epidemiology, clinical trials, clinical cardiology, and nephrology, to consider whether strategies for the prevention and treatment of cardiovascular disease in the general population were applicable to patients with chronic renal disease. The task force considered four target populations of patients with chronic renal disease: patients with CRI, patients with ESRD treated by hemodialysis, patients with ESRD treated by peritoneal dialysis, and renal transplant recipients. Recommendations of the task force (Levey et al., 1998) especially relevant to consideration of nutrition therapy for Medicare beneficiaries with chronic renal disease follow:
Epidemiology of Cardiovascular Disease in Chronic Renal Disease
Patients with chronic renal disease should be considered in the highest risk group for subsequent cardiovascular disease events. They have a high prevalence of coronary artery disease and left ventricular hypertrophy, which are precursors of cardiovascular disease mortality and morbidity. They also have a high prevalence of congestive heart failure, which is an independent predictor of death in chronic renal disease. Treatment recommendations based on cardiovascular disease risk stratification should consider the “highest risk status” of patients with chronic renal disease.
Risk Factors
The excess risk for cardiovascular disease in chronic renal disease is caused, in part, by a higher prevalence of conditions that are recognized as risk factors for cardiovascular disease in the general population, such as older age, hypertension, hyperlipidemia, diabetes, and physical inactivity. The excess risk may also be caused, in part, by hemodynamic and metabolic factors characteristic of chronic renal disease, including proteinuria, increased extracellular fluid volume, electrolyte imbalance, anemia, and higher levels of thrombogenic factors and homocysteine than in the general population. Strategies for risk factor identification and reduction should target both the traditional coronary risk factors and specific factors related to chronic renal disease.
Recent studies have highlighted the excess risk of cardiovascular disease in patients with CRI, as well as in ESRD. A prospective study of
246 patients with CRI in France determined that the coronary artery disease event rates were three times higher than in the general population (Jungers et al., 1997). A cross-sectional study of 175 patients showed that the prevalence of left ventricular hypertrophy was inversely correlated with the level of renal function. The prevalence of left ventricular hypertrophy was 27 percent, 31 percent, and 45 percent, respectively, among patients with creatinine clearance of greater than 50, 25 to 50, and less than 25 ml/min (Levin et al., 1996). These studies suggest that CRI is a risk factor for both coronary artery disease and left ventricular hypertrophy.
CRI is also a risk factor for cardiovascular disease mortality. Secondary analysis of the Hypertension Detection and Follow-Up Program (a randomized clinical trial of two levels of blood pressure control in essential hypertension) found that an elevated serum creatinine concentration was an independent risk factor for total and cardiovascular disease mortality (Shulman et al., 1989). In that study, cardiovascular mortality was almost three times higher than mortality due to renal disease. Similar findings were reported in secondary analyses of the Program on the Surgical Control of the Hyperlipidemias (a randomized controlled trial of partial ileal bypass surgery versus medical therapy for hypercholesterolemic survivors of myocardial infarction) (Matts et al., 1993), and the Cardiovascular Health Study (a community-based observational study of factors associated with cardiovascular disease) (Fried et al., 1998). Preliminary data from the Framingham Study (a community-based observational study of factors associated with cardiovascular disease) also document an increased risk of fatal and nonfatal cardiovascular disease events in participants with elevated serum creatinine concentration, although the relationship was strongly related to age and coexisting cardiovascular disease risk factors (Culleton et al., 1999). Interpretation of these secondary analyses is limited by the diverse study populations, and the possibility of unique effects of the underlying disease on serum creatinine concentrations. Nonetheless, there appears to be consensus that an elevated serum creatinine concentration is a risk factor for cardiovascular disease.
Nutrition therapy is widely recommended as part of the medical regimen for the secondary prevention of cardiovascular disease in patients with hypertension, hyperlipidemia, and diabetes. The above data provide a strong basis for targeting patients with CRI for similar secondary preventive measures.
Chronic Renal Insufficiency: Summary and Recommendations
Overall the findings from the above randomized, controlled clinical
trial in adults suggest that there may be a beneficial effect over the long term from nutrition therapy in delaying the progression of renal disease. Two meta-analyses also support the benefits of nutrition therapy for these patients. Dietary interventions for patients with moderate renal disease, however, have not been widely adopted.
The committee recommends that dietary interventions be used in patients who are at risk for progression of renal failure in diabetic as well as in non-diabetic renal disease. Trained renal nutrition specialists are needed as part of the renal team and should be involved in the education and the monitoring of nutritional status for these patients. In addition, nutrition therapy is necessary to maintain adequate energy intake to prevent the onset of malnutrition, which is a risk factor for death after the onset of ESRD. Finally, nutrition therapy is necessary to address risk factors for cardiovascular disease which are highly prevalent in patients with CRI and ESRD.
Summary of Evidence—Nutrition Therapy for Chronic Renal Insufficiency (Pre-End Stage Disease)
|
Consensus statements: |
Recommended by 1994 consensus conference (Striker, 1995) |
|
Observational studies: |
Supportive evidence in animal studies (3 studies: Ibels et al., 1978; Kleinknecht et al., 1979; Laouari et al., 1982) |
|
Randomized trials: |
Supportive evidence (2 large studies: Klahr et al. 1994; Zeller et al., 1991; and numerous small studies) |
|
Meta-analysis of trials: |
Supportive evidence (3 studies: Fouque et al., 1992; Kasiske et al., 1998; Pedrini et al., 1996) |
|
Systematic reviews: |
Supportive recent evidence (Levey et al., 1999) |
END-STAGE RENAL DISEASE
ESRD requires complex nutritional management, which may include the modification of dietary intake of protein, sodium, potassium, phosphorus, magnesium, calcium, vitamin D, several other vitamins, trace minerals, and fluid (Renal Dietitians Dietetic Practice Group of the American Dietetic Association, 1993). Because many ESRD patients have underlying diabetes, hypertension, or cardiovascular disease, modification of calorie, carbohydrate, and fat intake is often required as well. Thus, the dietary interventions for ESRD patients are complex and are challenging for most to learn and follow. The consequences of dietary noncompliance
include increased frequency and duration of dialysis, increased morbidity, and possibly mortality.
The nutritional needs of patients may change as ESRD progresses and thus modifications of the diet prescribed are frequently required. For example, predialysis patients may need to restrict protein. On the other hand, patients who are stable on dialysis will require almost twice the amount of protein as predialysis patients. Depending on the adequacy of dialysis, underlying medical conditions, and patient compliance issues, protein intake may need to be adjusted periodically over the course of dialysis. Adjustments may also be necessary for other nutrients (e.g., sodium and potassium). Because of the complexity of the diet, each change in the dietary prescription requires reeducation of the patient and family. Patients dependent on dialysis may also need adjustments in nutrition therapy for other nutrition-related problems such as anemia, aluminum toxicity, and interactions between nutrients and medications. A number of complex interactions between medications and nutrients require close monitoring of medication administration and nutrition therapy (Sanders et al., 1994).
Undernutrition is common among patients with renal disease, especially those who are dependent on dialysis (Schoenfeld et al., 1983; Young et al., 1991). Several possible etiologies of undernutrition in renal failure have been explored, including the low protein diet restrictions (MDRD Study Group, 1994), altered taste in uremia resulting in anorexia (AtkinThor et al., 1978), and reduced energy intake (Monteon et al., 1986). The elevated risk of undernutrition may be related to losses of protein (Ikizler et al., 1994; Wolfson et al., 1982), carbohydrate (Gutierrez et al., 1994), and vitamins (Wolfson, 1988) during the dialysis procedure.
It has been suggested that dialysis is itself a catabolic process contributing to undernutrition (Borah et al., 1978). This hypothesis is supported by the work of Kopple and colleagues (1969, 1986) who found increased protein and possibly energy needs in dialysis patients compared to the normal population. Others have suggested that metabolic acidosis in renal failure may be associated with protein catabolism and that correction of the acidosis may reduce protein breakdown (Reaich et al., 1993).
Undernutrition has been identified as a predictor of mortality for patients dependent on dialysis. The prevalence of undernutrition in ESRD patients is appreciable. In a prospective study of 15,245 ESRD patients, 5,177 patients with a serum albumin concentration of less than 3.5 mg/dL on initiation of dialysis died within 91 days. A death rate of 29 percent has been reported for patients with a serum albumin concentration of less than 3.0 mg/dL (Soucie and McClellan, 1996). Although this is the largest study of this type, several smaller studies have reported similar findings (Canada-USA Peritoneal Dialysis Study Group, 1996; Iseki et al., 1993).
(Chapter 4 documents the limited utility of albumin as a nutritional status indicator. Since albumin is a strong proxy indicator of disease, inflammation, and injury, it is not surprising that dialysis patients with lower serum albumin concentrations face greater mortality risk.)
Dietitians have played a role in the nutritional management of ESRD patients since the inception of dialysis programs. As the knowledge regarding the effectiveness of nutrition intervention during dialysis has evolved, nutrition therapy has become increasingly complex. The advent of urea kinetic modeling has further contributed to the complexity of nutritional assessment and monitoring. Dietitians have also taken on roles as physician extenders in monitoring iron status, response to medication, and dosing of calcium, trace element supplements, and erythropoietin.
The expanding role of the dietitian has not always been accompanied by increased time to perform these tasks. A review of data submitted to the Health Care Financing Administration (HCFA) showed a decline in dietitian hours per renal patient from 0.315 to 0.245 (a decrease of 22 percent) from 1982 to 1987 (Held et al., 1990).
There are limited data concerning nutrition therapy providers for ESRD patients. Since the need for the involvement of dietitians in the provision of nutrition therapy to patients with ESRD was recognized before current reimbursement for services was implemented, there has been little incentive to study the provision of nutrition therapy. In one uncontrolled report, however, patients seemed to respond positively to individualized nutritional intervention by a dietitian (Hoover and Connelly, 1985). There are also small studies suggesting that counseling for appropriate protein intake may result in improved nutritional status (Borah et al., 1978; Kopple, 1994). Further studies are needed to identify the most appropriate diet for patients with ESRD, and to determine the frequency, type, and duration of nutrition therapy. Until further data are available, it is recommended that nutrition services be continued at their present level and that surveillance efforts continue to ensure that there are adequate numbers of trained renal nutrition specialists available to meet the needs of ESRD patients.
Supplemental essential amino acids are available for dialysis patients, however limited data are available to support their use. One study found improved plasma amino acid levels in a small series of patients who
received essential amino acid supplementation, but the study did not control for the protein intake of subjects (Phillips et al., 1978). A later study found no advantage to supplementing a 1 g/kg protein diet with essential amino acids (Hecking et al., 1978). More recently, liquid meal replacement formulas with lowered electrolyte levels have been marketed. Like essential amino acid supplements, these products are too expensive for many ESRD patients to purchase. No randomized trials of their use in augmenting the nutrition intake of undernourished patients with renal disease were available to review. Such trials should be conducted before use of these products is recommended.
Although undernourished ESRD patients should be candidates for enteral nutrition, this therapeutic modality has largely been ignored. Since no trials of enteral feeding in patients with ESRD were found, it is recommended that randomized, controlled trials of this therapy be conducted. The use of parenteral nutrition for acute renal failure patients in the acute hospital setting is discussed in chapter 10.
Intradialytic parenteral nutrition (IDPN) has been subject to preliminary study for use with ESRD patients receiving hemodialysis. A concentrated solution containing modest amounts of amino acids, dextrose, fat, electrolytes, vitamins, and minerals is typically administered several times weekly by infusion during hemodialysis treatments. The rationale for its development and use was to supplement the nutrient intake of patients at risk of undernutrition due to inadequate nutrient intake. Prospective case series suggest that there is opportunity for improved nutritional status in patients receiving IDPN, but none of these series include more than 21 patients, none were adequately controlled, and all were of short duration. One nonrandomized, retrospective review of 81 patients found improved survival (RR = 1.34, p < 0.01) in 50 patients receiving IDPN (Capelli et al., 1994). A large, retrospective analysis of 1,679 chronic hemodialysis patients (Chertow et al., 1994) found a decreased risk of death (OR for death = 3.14 in the IDPN group versus 4.93 in the untreated group, p < 0.01) in undernourished hemodialysis patients receiving IDPN. Prospective, appropriately randomized trials of IDPN in carefully selected patients should be conducted before Medicare reimbursement for IDPN is considered.
Economic and Regulatory Considerations
Presently, nutrition services for dialysis patients are provided by qualified dietitians who are employed by, or in a contractual relationship with, the ESRD facility. HCFA currently specifies that in consultation with the attending physician, the dietitian is responsible for assessing the nutritional needs of each ESRD patient, recommending therapeutic diets,
counseling patients and their families on prescribed diets, and monitoring adherence and response to diets. Dietitians are not currently reimbursed for services; rather, they are included through the monthly capitation payment. Medicare considers the provision of nutritional services to ESRD patients a “minimal services requirement,” which is a condition for coverage as part of comprehensive interdisciplinary care.
End-Stage Renal Disease: Summary and Recommendations
It is recommended that nutrition therapy for dialysis patients be continued at the present level and that surveillance continue to document that ESRD patients receive adequate nutrition therapy provided by a trained renal nutrition specialist.
NUTRITION THERAPY AFTER TRANSPLANTATION
There are many situations following kidney transplantation in which patients may benefit from nutrition therapy. The need for complex nutrient modifications is not always ameliorated with renal transplantation. Although individuals who have had a successful kidney transplant need encouragement to eat a normal diet, recommendations for nutrient intake will vary according to the posttransplant duration. The acute posttransplant phase lasts for 1 to 2 months. The chronic posttransplant phase follows. No difference in nutritional management is described; however, if graft failure occurs, further diet modifications are likely to be needed. In addition, several nutrition-related complications may occur in patients following transplant. These include obesity, hyperlipidemia, hypertension, hyperkalemia, and steroid-induced diabetes mellitus as well as bone disease and possibly hypervitaminosis (Rosenberg, 1986). Protein requirements also may change with adjustments in steroid dosing (Cogan et al., 1981; Seagraves et al., 1986). Corticosteroid medications (usually prednisone) cause decreases in lean body mass (due to increased gluconeogenesis) and increases in body fat deposition, glucose intolerance, hyperlipidemia, osteoporosis, hyperphagia, and steroid-induced diabetes.
Most posttransplant patients are at risk for excessive weight gain. In a study of 263 patients following transplant, Merion and colleagues (1991) found that each experienced weight gain. Results showed that in the 6 to 12 month period following transplant, the net 1-year weight gain was 14.2 ± 2.2 kg for obese patients (defined as > 120 percent of ideal body weight) and 8.9 ± 0.6 kg for nonobese patients.
Hyperlipidemia following kidney transplantation has been described
(Kritchevsky, 1987). In a survey of 500 patients treated with cyclosporine, 38 percent had hypercholesterolemia (> 250 mg/dL) and 15 percent had hypertriglyceridemia (> 300 mg/dL) (Rosenberg, 1986). The hyperlipidemia is thought to be steroid induced, but the withdrawal of steroids was not shown to change the total cholesterol-high-density lipoprotein (HDL) cholesterol ratio in a 2-month study (Hricik et al., 1992). A small trial by Shen and colleagues (1983) showed that diet modification may be helpful in decreasing cholesterol and triglyceride levels. Total cholesterol, but not HDL cholesterol or low-density lipoprotein cholesterol, was changed in another small study. Clearly, larger studies are needed to confirm the value of nutrition therapy, as are studies comparing dietary management with drug therapy.
As many as 19 percent of 173 posttransplant patients developed diabetes (Boudreaux et al., 1987). This observation was confirmed by Sumrani and colleagues (1991). It has been noted that the incidence of diabetes was greater in obese patients (Holley et al., 1990).
In patients who have been on dialysis for a long period of time before receiving a kidney transplant, dietary restrictions have become habitual and patients must re-establish normal eating patterns during the post-transplantation period. Given the short length of hospital stay following kidney transplantation (3 days), it is generally not feasible to adequately address nutrition counseling during the hospitalization. In addition, the need for nutrition interventions following transplantation often necessitates access to a nutrition professional for in-depth nutrition counseling.
Nutrition Therapy after Transplantation: Summary and Recommendations
The potential for nutrition-related complications following renal transplant, as well as the potential change in nutritional needs, makes this patient population a candidate that would likely benefit from nutritional therapy. Given the complexity of combined dietary modifications, nutrition therapy should be provided by a trained renal nutrition specialist. Insufficient data are available at this time to determine the appropriate type and frequency of nutritional intervention to maximize the nutritional status in these patients.
FUTURE AREAS OF RESEARCH
Randomized, controlled clinical trials designed to test interventions to maintain optimal nutrition status in predialysis, dialysis, and posttransplant patients are needed. Also necessary are prospective, randomized trials of IDPN in carefully selected undernourished patients before cover-
age for IDPN can be considered further. Specific research subjects to be addressed include the following:
-
although the calorie and protein needs of dialysis patients have been studied, there are gaps in knowledge about the optimum amounts of these and other nutrients in the diet of patients with renal disease;
-
the development of undernutrition as ESRD progresses in patients highlights the priority for further investigation of the underlying causes; the optimum type and frequency of nutrition intervention for these patients should be investigated;
-
the appropriate roles of oral nutritional supplements and enteral nutrition have not been defined and should be the focus of well-conceived clinical trials; and
-
large-scale, randomized trials of parenteral nutrition as a component of treatment for renal disease with clearly defined outcomes should be conducted.
REFERENCES
Atkin-Thor E, Goddard BW, O’Nion J, Stephen RL, Kolff WJ. 1978. Hypogeusia and zinc depletion in chronic dialysis patients. Am J Clin Nutr 31:1948–1951.
Borah MF, Schoenfeld PY, Gotch FA, Sargent JA, Wolfson M, Humphreys MH. 1978. Nitrogen balance during intermittent dialysis therapy of uremia. Kidney Int 14:491–500.
Boudreaux JP, McHugh L, Canafax DM, Asher N, Sutherland DE, Payne W, Simmons RL, Najarian JS, Fryd DS. 1987. Cyclosporine, combination immunosuppression, and posttransplant diabetes mellitus. Transplant Proc 19:1811–1813.
Canada-USA Peritoneal Dialysis Study Group. 1996. Adequacy of dialysis and nutrition in continuous peritoneal dialysis: Association with clinical outcomes. J Am Soc Nephrol 7:198–207.
Capelli JP, Kushner H, Camiscioli TC, Chen S-M, Torres MA. 1994. Effect of intradialytic parenteral nutrition on mortality rates in end-stage renal disease care. Am J Kidney Dis 23:808–816.
Chertow GM, Ling J, Lew NL, Lazarus JM, Lowrie EG. 1994. The association of intradialytic parenteral nutrition administration with survival in hemodialysis patients. Am J Kidney Dis 24:912–920.
Cogan MG, Sargent JA, Yarbrough SG, Vincenti F, Amend WJ Jr. 1981. Prevention of prednisone-induced negative nitrogen balance. Effect of dietary modification on urea generation rate in patients on hemodialysis receiving high-dose glucocorticoids. Ann Intern Med 95:158–161.
Culleton BF, Larson MG, Evans JC, Wilson PW, Barrett BJ, Parfrey PS, Levy D. 1999. Prevalence and correlates of elevated serum creatinine levels: The Framingham Heart Study. Arch Intern Med 159:1785–1790.
Fouque D, Laville M, Boissel JP, Chifflet R, Labeeuw M, Zech PY. 1992. Controlled low protein diets in chronic renal insufficiency: Meta-analysis. Brit Med J 304:216–220.
Fried LP, Kronmal RA, Newman AB, Bild DE, Mittelmark MB, Polak JF, Robbins JA, Gardin JM. 1998. Risk factors for 5-year mortality in older adults: The Cardiovascular Health Study. J Am Med Assoc 279:585–592.
Gillis BP, Caggiula AW, Chiavacci AT, Coyne T, Doroshenko L, Milas C, Nowalk MP, Scherch LK. 1995. Nutrition intervention program of the Modification of Diet in Renal Disease Study: A self-management approach. J Am Diet Assoc 95:1288–1294.
Greene T, Beck GJ, Kutner MH, Paranandi L, Wang S, MDRD Study Group, 1995. AASK Study Group: Comparison of time-to-event and slope-based analyses in nephrology clinical trials. Controlled Clin Trials 16:65S.
Gutierrez A, Bergström J, Alvestrand A. 1994. Hemodialysis-associated protein catabolism with and without glucose in the dialysis fluid. Kidney Int 46:814–822.
HCFA (Health Care Financing Administration). 1999. HCFA statitics: Utilization, Table 38. Available at: http://www.hcfa.gov/stats/hstats96/blustat3.htm. Accessed July 1999.
Hecking E, Köhler H, Zobel R, Lemmel EM, Mader H, Opferkuch W, Prellwitz W, Keim HJ, Muller D. 1978. Treatment with essential amino acids in patients on chronic hemodialysis: A double blind cross-over study. Am J Clin Nutr 31:1821–1826.
Held PJ, Brunner F, Odaka M, García JR, Port FK, Gaylin DS. 1990. Five-year survival for end-stage renal disease patients in the United States, Europe, and Japan, 1982 to 1987. Am J Kidney Dis 15:451–457.
Holley JL, Shapiro R, Lopatin WB, Tzakis AG, Hakala TR, Starzl TE. 1990. Obesity as a risk factor following cadaveric renal transplantation. Transplantation 49:387–389.
Hoover H, Connelly SV. 1985. The effect of diet intervention on 31 chronic hemodialysis patients. J Nephrol Nurs 2:244–249.
Hricik DE, Bartucci MR, Mayes JT, Schulak JA. 1992. The effects of steroid withdrawal on the lipoprotein profiles of cyclosporine-treated kidney and kidney-pancreas transplant recipients. Transplantation 54:868–871.
Ibels LS, Alfrey AC, Haut L, Huffer WE. 1978. Preservation of function in experimental renal disease by dietary restriction of phosphate. N Engl J Med 298:122–126.
Ikizler TA, Flakoll PJ, Parker RA, Hakim RM. 1994. Amino acid and albumin losses during hemodialysis. Kidney Int 46:830–837.
Iseki K, Kawazoe N, Fukiyama K. 1993. Serum albumin is a strong predictor of death in chronic dialysis patients. Kidney Int 44:115–119.
Jungers P, Massy ZA, Khoa TN, Fumeron C, Labrunie M, Lacour B, Descamps-Latscha B, Man NK. 1997. Incidence and risk factors of atherosclerotic cardiovascular accidents in predialysis chronic renal failure patients: A prospective study. Nephrol Dial Transplant 12:2597–2602.
Kasiske BL, Lakatua JD, Ma JZ, Louis TA. 1998. A meta-analysis of the effects of dietary protein restriction on the rate of decline in renal function. Am J Kidney Dis 31:954–961.
Klahr S, Levey AS, Beck GJ, Caggiula AW, Hunsicker L, Kusek JW, Striker G. 1994. The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. N Engl J Med 330:877–884.
Kleinknecht C, Salusky I, Broyer M, Gubler M-C. 1979. Effect of various protein diets on growth, renal function, and survival of uremic rats. Kidney Int 15:534–541.
Kopple JD. 1994. Effect of nutrition on morbidity and mortality in maintenance dialysis patients. Am J Kidney Dis 24:1002–1009.
Kopple JD, Shinaberger JH, Coburn JW, Sorensen MK, Rubini ME. 1969. Optimal dietary protein treatment during chronic hemodialysis. Trans Am Soc Artif Intern Organs 15:302–308.
Kopple JD, Monteon FJ, Shaib JK. 1986. Effect of energy intake on nitrogen metabolism in nondialyzed patients with chronic renal failure. Kidney Int 29:734–742.
Kopple et al. In press. J Am Soc Nephrol.
Kritchevsky D. 1987. Atherosclerosis: Diet and risk factors. Transplantation Proc 19:53–56.
Laouari D, Kleinknecht C, Gubler M-C, Ravet V, Broyer M. 1982. Importance of proteins in the deterioration of the remnant kidneys, independently of other nutrients. Int J Pediatr Nephrol 3:263–269.
Levey AS, Adler S, Caggiula AW, England BK, Greene T, Hunsicker LG, Kusek JW, Rogers NL, Teschan PE. 1996. Effects of dietary protein restriction on the progression of advanced renal disease in the Modification of Diet in Renal Disease Study. Am J Kidney Dis 27:652–663.
Levey AS, Beto JA, Coronado BE, Eknoyan G, Foley RN, Kasiske BL, Klag MJ, Mailloux LU, Manske CL, Meyer KB, Parfrey PS, Pfeffer MA, Wenger NK, Wilson PW, Wright JT Jr. 1998. Controlling the epidemic of cardiovascular disease in chronic renal disease: What do we know? What do we need to learn? Where do we go from here? Am J Kidney Dis 32:853–906.
Levey AS, Greene T, Beck GJ, Caggiula AW, Kusek JW, Hunsicker LG, Klahr S. 1999. Dietary protein restriction and the progression of chronic renal disease: What have all the results of the MDRD study shown? J Am Soc Nephrol 10:2426–2439.
Levin A, Singer J, Thompson CR, Ross H, Lewis M. 1996. Prevalent LVH in the predialysis population. Identifying opportunities for intervention. Am J Kidney Dis 27:347–354.
Matts JP, Karnegis JN, Campos CT, Fitch LL, Johnson JW, Buchwald H. 1993. Serum creatinine as an independent predictor of coronary heart disease mortality in normotensive survivors of myocardial infarction. POSCH Group. J Fam Pract 36:497–503.
MDRD (Modification of Diet in Renal Disease) Study Group. 1994. Reduction of dietary protein and phosphorus in the Modification of Diet in Renal Disease Feasibility Study. J Am Diet Assoc 94:986–990.
Merion RM, Twork AM, Rosenberg L, Ham JM, Burtch GD, Turcotte JG, Rocher LL, Campbell DA Jr. 1991. Obesity and renal transplantation. Surg Gynecol Obstet 172:367–376.
Milas NC, Nowalk MP, Akpele L, Castaldo L, Coyne T, Doroshenko L, Kigawa L, Korzec-Ramirez D, Scherch LK, Snetselaar L. 1995. Factors associated with adherence to the dietary protein intervention in the Modification of Diet in Renal Disease Study. J Am Diet Assoc 95:1295–1300.
Monteon FJ, Laidlaw SA, Shaib JK, Kopple JD. 1986. Energy expenditure in patients with chronic renal failure. Kidney Int 30:741–747.
Pedrini MT, Levey AS, Lau J, Chalmers TC, Wang PH. 1996. The effect of dietary protein restriction on the progression of diabetic and nondiabetic renal diseases: A meta-analysis. Ann Intern Med 124:627–632.
Phillips ME, Havard J, Howard JP. 1978. Oral essential amino acid supplementation in patients on maintenance hemodialysis. Clin Nephrol 9:241–248.
Reaich D, Channon SM, Scrimgeour CM, Daley SE, Wilkinson R, Goodship THJ. 1993. Correction of acidosis in humans with CRF decreases protein degradation and amino acid oxidation. Am J Physiol 265:E230–E235.
Renal Dietitians Dietetic Practice Group of the American Dietetic Association. 1993. National Renal Diet: Professional Guide. Chicago, Ill.: The American Dietetic Association.
Rosenberg ME. 1986. Nutrition and transplantation. Kidney 18:19–22.
Sanders HN, Rabb HA, Bittle P, Ramirez G. 1994. Nutritional implications of recombinant human erythropoietin therapy in renal disease. J Am Diet Assoc 94:1023–1029.
Schoenfeld PY, Henry RR, Laird NM, Roxe DM. 1983. Assessment of nutritional status of the National Cooperative Dialysis Study population. Kidney Int 23:S80–S88.
Seagraves A, Moore EE, Moore FA, Weil R 3rd. 1986. Net protein catabolic rate after kidney transplantation: Impact of corticosteroid immunosuppression. J Parenter Enteral Nutr 10:453–455.
Shen SY, Lukens CW, Alongi SV, Sfeir RE, Dagher FJ, Sadler JH. 1983. Patient profile and effect of dietary therapy on post-transplant hyperlipidemia. Kidney Int 24:S147–S152.
Shulman NB, Ford CE, Hall WD, Blaufox MD, Simon D, Langford HG, Schneider KA. 1989. Prognostic value of serum creatinine and effect of treatment of hypertension on renal function. Results from the hypertension detection and follow-up program. The Hypertension Detection and Follow-up Program Cooperative Group. Hypertension 13:I80– I93.
Soucie JM, McClellan WM. 1996. Early death in dialysis patients: Risk factors and impact on incidence and mortality rates. J Am Soc Nephrol 7:2169–2175.
Striker GE. 1995. Report on a workshop to develop management recommendations for the prevention of progression in chronic renal disease Bethesda (MD) April 1994. Nephrol Dial Transplant 10:290–292.
Sumrani NB, Delaney V, Ding ZK, Davis R, Daskalakis P, Friedman EA, Butt KM, Hong JH. 1991. Diabetes mellitus after renal transplantation in the cyclosporine era—an analysis of risk factors. Transplantation 51:343–347.
USRDS (U.S. Renal Data System). 1997. 1997 Annual Data Report. Bethesda, Md.: National Institute of Diabetes and Digestive and Kidney Diseases.
USRDS (U.S. Renal Data System). 1999. 1999 Annual Data Report. Bethesda, Md.: National Institute of Diabetes and Digestive and Kidney Diseases.
Walker JD, Bending JJ, Dodds RA, Mattock MB, Murrells TJ, Keen H, Viberti GC. 1989. Restriction of dietary protein and progression of renal failure in diabetic nephropathy. Lancet 2:1411–1415.
Wolfson M. 1988. Use of water soluble vitamins in patients with chronic renal failure. Semin Dial 1:28–32.
Wolfson M, Jones MR, Kopple JD. 1982. Amino acid losses during hemodialysis with infusion of amino acids and glucose. Kidney Int 21:500–506.
Young GA, Kopple JD, Lindholm B, Vonesh EF, De Vecchi A, Scalamogna A, Castelnova C, Oreopoulos DG, Anderson GH, Bergstrom J, DiChiro J, Gentile D, Nissenson A, Sakhrani L, Brownjohn AM, Nolph KD, Prowant BF, Algrim CE, Martis L, Serkes KD. 1991. Nutritional assessment of continuous ambulatory peritoneal dialysis patients: An international study. Am J Kidney Dis 17:462–471.
Zeller K, Whittaker E, Sullivan L, Raskin P, Jacobson HR. 1991. Effect of restricting dietary protein on the progression of renal failure in patients with insulin-dependent diabetes mellitus. N Engl J Med 324:78–84.




















