Antifouling
J. W. Costerton
Biofilms, which present problems in many different areas, result from the very tight adherence of bacteria to surfaces. Although bacteria are at risk and grow very poorly, when floating in water, they grow extremely well on surfaces. This characteristic has many consequences in different areas. I first studied the alpine environment, which probably has the cleanest water in any ecosystem, with sparse Pseudomonas aeruginosa growth of perhaps 8 bacteria/mL in the flowing water and abundant growth of at least 108 bacteria/cm² on the rocks. So it is in biofilms in any ecosystem that bacteria grow preferentially on surfaces.
A recent review in Science (Costerton and others 1999) illustrates the medical milieu to which Dr. Mittelman referred in his preceding introduction. The administration of antibiotics kills planktonic cells, which are also killed by specific antibodies and by white cells (Figure 1). We can deal with a certain number of planktonic bacterial cells in medical situations. If, however, biofilms have formed (which is the case much too regularly on foreign surfaces), then this biofilm is resistant to antibiotics and highly resistant to antibodies and white cells. The white cells are then unable to kill the bacteria because they cannot phagocytize them, and the “frustrated” white cells release enzymes that tend to digest the surrounding tissues. Thus a biofilm is essentially “bulletproof” against antibiotics and white cells. It withstands 1000 times as much antibiotic as floating (planktonic) cells of the same species. The Centers for Disease Control
Center for Biofilm Engineering, Montana State University, Bozeman, MT
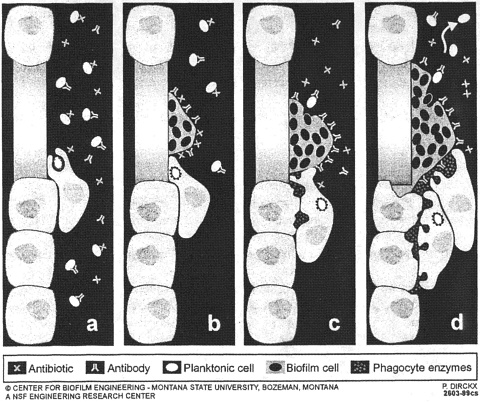
FIGURE 1. Diagrammatic representation of the very different effects of antibiotics and of host defense factors on bacteria growing in the planktonic mode of growth, as opposed to the biofilms mode of growth that characterizes very large numbers of modern bacterial infections. Reproduced with permission from Science (May 21, 1999).
have recently estimated that 65% of all infections seen by infectious disease specialists are biofilm infections (Costerton and others 1999).
Acute bacterial infections caused by planktonic bacteria have been largely controlled, primarily with antibiotics and vaccines; however, conditions such as children's middle ear infections persist until they are eventually outgrown. The persistent condition emerges as a very large concern. The NIH have recently issued three Requests for Applications (RFAs) in the biofilm area. In addition, and preceding the issuance of the RFAs, the Office of Naval Research and the engineering section of the National Science Foundation had funded research in this area.
Our ideas about biofilms have changed since 1993, when microbiologists viewed biofilms as a totally random accretion of cells on a surface.
In 1995, the idea of bacterial communication emerged and was called quorum sensing (Fuqua and others 1994). Bacteria floating in suspension were thought to produce signals. For example, in the case of Pseudomonasaeruginosa, two different signals are produced, and these simply diffuse through the cell envelope out into the surrounding milieu, and nothing happens. However, when planktonic cultures of bacteria reach a certain cell density (which Peter Greenberg calls a quorum), these signals begin to accumulate in relatively high concentration. They feed back through the membrane of the bacteria and activate their cognate receptor (R) protein to turn on the expression of certain genes. The first activation discovered was luminescence in Vibrio species, but the production of toxins, enzymes, and surfactants is also controlled by signals. Bacteria produce toxins, enzymes, and surfactant only when they achieve a certain concentration of cells. Some of the molecules that cause this controlled activity are called homoserine lactones (HSLs). Because we had a communication molecule and a phenomenon—biofilm formation—we then mutated the organisms so that they did not produce the communication molecule to learn what would happen to biofilm formation. The results were spectacular!
The wild type for a normal organism making both signals, the 3-oxydodecanoyl HSL signal and the butryl HSL, attaches to a surface, makes the exopolysaccharide, and eventually makes the biofilm. If we knock out the butyryl HSL, then it continues to make the polysaccharide and structured biofilms. If we knock out the 3-oxydodecanoyl HSL or both signals, the organisms that adhere to the surface cannot turn on polysaccharide production and cannot form biofilms. They adhere, but with a very small amount of surfactant or even with gentle stirring, they are removed from the surface. What was found, and published in Science in April 1998 (Davies and others 1998), was a signal that actually controls biofilm formation in P. aeruginosa.
People in the pharmaceutical industry are very interested in this area and in the possibility of developing resistance to biofilm blockage by HSLs. In the development of periodontitis, for example, the development of a deep trench between the tooth and the gum is caused not by one organism but by a whole community of microbes. People in the dental profession know that Fusobacterium nacleatum is the first microbe to get started; therefore, we are looking at the equivalent of the biofilm control HSL for this organism. The organism normally joins the plaque, which is perfectly harmless on the tooth but makes a deepening periodontal plaque by its presence. It is possible to simply reverse that process by adding the HSL-blocking analog to a mouthwash. Fusobacterium would thus be in the mouth, but it would not join the plaque deepening between the
gum and the tooth. We thus would have a preventative measure for periodontitis.
An intriguing marine manifestation of signaling was discovered in Sydney, Australia, by an American (Peter Steinberg) and a Swede (Staffan Kjelleberg), and it became very successful commercially. The idea they followed was one that Drs. Manyak and Mittelman discussed earlier: that certain marine organisms do not allow bacterial biofilms to foul their surfaces. However, marine algae have the most acute problem because they cannot carry out photosynthesis if they develop biofilms on their surface. They become covered in slime, which attracts clay and buries them. There is a red algal species that grows all across Botany Bay in Sydney Harbor (called Delcia pulcra, meaning “delicious” and “beautiful”) that occasionally fouls with macrophytes but virtually never with bacteria.
In the late 1980s, Staffan Kjelleberg and Pete Steinberg ground up some of these red algal fronds and found the active principle, a series of molecules called furanones. The furanones (which currently total 42) have been purified and added to boat coatings, to fishing nets (for tropical uses where they foul very badly), and to contact lenses. The latter use is the first medical application, which involves experimenting with resistance to bacterial fouling for long periods. This furanone panel (2) is located about 100 m from a sewage outfall in Sydney Harbor, where macrophyte fouling of control panels was evident 5 months after it was put in the base of the harbor, but the furanone-containing panel was virtually uncolonized. These natural furanone compounds have been shown (de Nys and others 1995) to block 3-oxydodecanoyl HSL, specifically at the level of the interaction of the signal with its cognate receptor (R) protein. This discovery does not require synthesizing new compounds, but naturally occurring compounds can be used to affect biofilm formation.
I believe that furanones have actually been sold to a very large number of companies, and I think we will start seeing the emergence of medical devices containing furanones in the next 3 or 4 years. In addition, we will see the use of signals that trigger the detachment of planktonic bacteria from mature biofilms, as butyryl HSL triggers detachment in biofilms of Pseudomonas aeruginosa. Consider what happens then if we put the butyryl HSL in high-level concentrations into a biofilm produced by P. aeruginosa: We should be able to induce the detachment of those cells from that surface. This would be the anticipated result—a natural compound or an analog that triggered the mechanism and then locked the position covalently so that it stayed in a permanent detachment mode. We could take a formed biofilm of Pseudomonas, insert the detachment analog, and pull the whole biofilm off the surface to have floating cells
4 hours later. A clinical use of this technology could be found in the mechanical heart valve. The valve is attached to the endocardium by a sewing cuff made of a plastic fabric material. During infection, Staphylococcus epidermidis enters and makes a biofilm on the sewing cuff. We would administer low-level concentrations of the detachment signal, which would be a peptide, in the case of Gram-positive cells like Staphylococcus epidermidis. It would be possible to dissolve the biofilm even after an infection had taken place and after the sewing cusp had been colonized, and this maneuver would be done in the presence of very high-level concentrations of specific antibiotics.
Thus, I have described the two areas that we now understand in microbiology and biofilm microbiology—from planktonic cells coming onto a surface and forming aggregates and changing into the true biofilm with the signal-dependent production of matrix. We can experiment with all of those signals.
I would like to point out that the furanones must inhibit the biofilm formation signals for thousands of species of bacteria. This panel (Figure 2) is in a sewage-contaminated marine environment, and thousands of bacterial species are present in this contaminated environment, but none

FIGURE 2. Photograph of a furanone-containing panel (center), with control panels, which had been exposed to the marine environment of Sydney Harbor for 5 months. Note the failure of marine biota in colonizing this material containing the natural blocking analog of the homoserine lactone signal that controls biofilm formation in many Gram-negative organisms. Reproduced with permission from Staffan Kjelleberg.
has become resistant and colonized the furanone panel. Also controlled by signals is the detachment event, in which these biofilm microcolonies simply dissolve and mobilize. We can now control these two activities, and a great deal of this work comes from the ONR program and from the NSF's concentration of resources in an engineering research center staffed by four-fifths engineers and one-fifth microbiologists in the Center for Biofilm Engineering (CBE). Together, these two groups are responsible for a very important discovery in medical, environmental, and industrial microbiology.
REFERENCES
Costerton JW, Stewart PS, Greenberg EP. 1999 Bacterial biofilms: A common cause of persistent infections. Science 284:13218-13220.
Davies DG, Parsek MR, Pearson JP, Iglewski BH, Costerton JW, Greenberg EP. 1998 The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science 280:295-298.
de Nys R, Steinberg PD, Willemsen P, Dworjanyn SA, Gabelish CL, King RJ. 1995 Broad spectrum effects of secondary metabolites from the red alga Delisea pulchra in antifouling assays. Biofouling 8:259-271.
Fuqua WC, Winans EP, Greenberg EP. 1994 Quorum sensing in bacteria: The Lux R-Lux I family of cell density-responsive transcriptional regulators. J Bacteriol 176:269-275.






