Research to Understand the Mechanisms Through Which Social and Behavioral Factors Influence Health
Bruce S. McEwen
The Rockefeller University
“What is a biologist doing in a meeting on social and behavioral science?” Dr. McEwen rhetorically asked. Two good reasons, he said: “Biology underlies individual and social behavior; and individual behavior and the social environment exert powerful effects on health.”
To illustrate the “gradients of health across the range of socioeconomic status,” he showed a graph that plotted a number of disorders—osteoporosis, chronic disease, hypertension, and cervical cancer—as a function of socioeconomic status, from its highest to lowest levels (see Figure A). Morbidity rates climbed sharply and steadily as socioeconomic status decreased.
The brain plays a central role here by interpreting and responding to environmental factors—work, neighborhood, relationships, family, major life events, trauma, and abuse—that influence people in every walk of life, Dr. McEwen said. As a neurobiologist, he is particularly interested in studying two brain areas—the amygdala, which has to do with fear and strong emotions; and the hippocampus, which affects spatial, declarative, episodic, and contextual memory. “We all know that we remember best the things that are connected to strong emotions, either positive or negative; the amygdala provides that emotional jolt,” he said. “The hippocampus provides the contextual memory—that is, where we were and what we were doing.”
These two brain areas regulate hypothalamic output to the autonomic nervous system and to the neuroendocrine system, which promote adaptation. “And we [use] the term ‘allostasis,’ which literally means maintaining
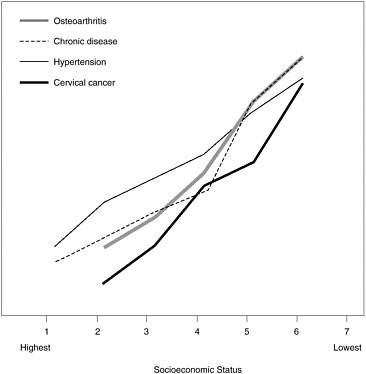
FIGURE A Morbidity rates by socioeconomic status level.
stability or homeostasis through active change, to refer to this process of adaptation,” Dr. McEwen said. If allostatic mechanisms remain on too long or are overused, they increase the “allostatic load,” a measure of the physiologic cost to different systems in the body from repeated burdens in one’s life.
Other contributions of biology to psychosocial studies include providing information on the biological basis of resilience, often referred to as “positive health”; on the early warning signs for the risk of disease, called “predisease pathways”; and of course on the human genetic code, and the expression of its genes, now in the process of being deciphered.
Biologists can provide information on the interactions between the body’s systems, Dr. McEwen noted. For example, diabetes, renal disease, and depression were the subjects of a workshop earlier this year at the National Institutes of Health. Other problems that need study across systems, he said, include cognitive disability in chronic pain and chronic fatigue; the consequences of sleep deprivation (which involve changes in immune func-
tion, metabolism, and cognition); and the wide-ranging results of anxiety disorder, which affects cardiovascular systems, metabolic systems, and immune systems.
Research using animal models has made many important contributions, Dr. McEwen said. Studies on monkeys, for example, “have taught us a lot about dominance and subordination and the rate of atherosclerosis and also suppression of immune function and the development of obesity.” Studies on rats show that quality of maternal care can result in lifelong patterns of increased or decreased anxiety states.
“Genes, early life events, experiences (some of which we refer to as stress), lifestyle, and individual behavior,” he said, are the important “players” here. They result in the cumulative wear and tear that is the cost of adaptation, referred to earlier as allostatic load, which “implies there are predisease pathways that we need to better understand in order to perform interventions before there is a real disease.”
The mechanisms, or “mediators,” involved in handling allostatic load include the autonomic nervous system, the neuroendocrine system, and the immune system, Dr. McEwen said. But while mediators in the short run have protective effects, in the long run they can exacerbate disease.
The best-known examples, he said, include cardiovascular disease, “where the acute activation of the sympathetic nervous system is involved in an animal fleeing from a predator and increasing its heart rate and also mobilizing energy stores. But chronic activation of the same system [can] accelerate atherosclerosis, particularly in a dominant animal vying for position in an unstable dominance hierarchy.” From metabolism, he added, “we know very well that the hormones cortisol and catecholamines are involved in mobilizing and replenishing energy stores. But these same hormones participate in the development of insulin resistance and obesity and increased risk for cardiovascular disease.”
To apply some of this information, the University of California at Los Angeles (UCLA)’s Theresa Seeman—like Dr. McEwen, a researcher in the MacArthur Network—and colleagues collected urinary cortisol and catecholamines samples, among others, in a group of subjects participating in the MacArthur Successful Aging Study. The researchers also devised a simple scoring system that correlated the measurements of these samples with the degree of allostatic load—the lower, the better. In that way, Dr. McEwen said, Seeman was able “to predict decline in both physical and cognitive function [and] predict new cardiovascular disease over two and a
half years. . . . She also found a higher score with lower income, a higher score with lower education, and a lower score with higher social ties.”
Other work, by Carol Ryff, Burton Singer, and colleagues in the Wisconsin Longitudinal Study, has found that the allostatic load in adulthood—among subjects in their 50s and 60s—is related to critical relationships. If subjects had at least one caring parent when growing up and a good-quality relationship with a spouse, this was defined as a positive pathway. Having uncaring or abusive parents when growing up, and/or a poor relationship with a spouse in adulthood, is a negative pathway. “The people with negative pathways, both men and women, had substantially higher allostatic load scores than people who had the more positive pathways,” Dr. McEwen said (see Figure B).
Future work, Dr. McEwen said, should draw on additional “primary mediators,” such as cytokines, anabolic hormones, and antioxidants, that have effects on many systems and are easy to measure. Similarly, we need “secondary outcomes,” like cholesterol, bone marrow density, and atrophy of brain structures like the hippocampus and prefrontal cortex, to serve as “functional markers” of immune, cardiovascular, and cognitive function.
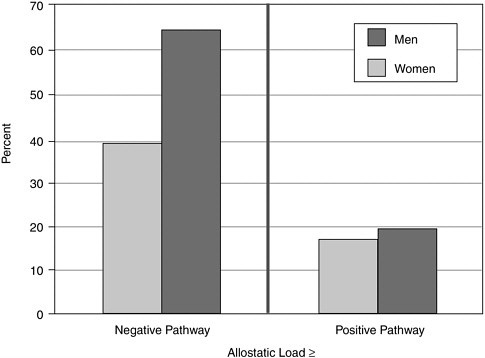
FIGURE B Relationship profiles and allostatic load.
These predisease markers can then “be used and measured longitudinally to find interventions that are effective in altering the course toward later disease,” he said. “The well-accepted model of preventing atherosclerosis in heart disease by exercise and lowering cholesterol needs to be expanded to other disorders.”
We also would benefit from more cross-system studies, he said. “We need to maintain a life-course perspective and think about early life events and their effects on later life outcomes—not just in aging, but in midlife. [We should] be particularly interested in understanding gene-environment interactions now that we understand more about the fact that it is not nature or nurture, but nature interacting with nurture. And of course, [we must] include behavioral and social environment as important factors in causing changes in biology.”
He noted that “it is somewhat ironic that the National Institutes of Health primarily studies disease, not the factors of positive health and resilience.” This highlights the biggest and most fundamental challenge, Dr. McEwen said, which is to promote “the establishment of working groups that will give scientists time to get acquainted with each other, learn about each other’s disciplines, and plan interdisciplinary studies.”





