4
The Patient Population
The congressional charge for the study of the Medicare ESRD program included the study of major epidemiologic and demographic changes in the ESRD patient population that may affect access to treatment, quality of care, or the resource requirements of the program. Chapters 4 and 5 address this charge.
This chapter presents data on trends in ESRD incidence, prevalence, and mortality in terms of age, gender, race, and primary diagnosis of the cause of renal failure (as reported by the patient's physician). These data include only treated ESRD patients, not all those reaching permanent renal failure; the difference between "treated" and "total" is not known. Mortality data are presented for this population and for various patient subgroups. The chapter also includes projections of incidence and prevalence of treated ESRD patients to the year 2000. Chapter 5 deals with the special needs and problems of pediatric, elderly, diabetic, hypertensive, and minority ESRD patients.
The data presented in these chapters are drawn primarily from the HCFA Program Management Medical Information System (PMMIS) files and the United States Renal Data System (USRDS) 1990 Annual Data Report. PMMIS and USRDS are described in detail in Chapter 13. In this report, patients are included in the data base from the time of diagnosis of ESRD rather than from the beginning of Medicare entitlement. This is consistent with the USRDS reports but differs from most HCFA reports. For incidence and prevalence, for example, HCFA usually counts patients only during their Medicare entitlement. For survival analyses, however, both HCFA and USRDS usually include patients from the time of diagnosis of ESRD.
Incidence is defined as new ESRD patients entering treatment during a given year. Incidence data are useful when considering issues of access,
patterns of referral to treatment, and disease prevention. Prevalence is defined as the total number of ESRD patients present in the population at a specific time; period prevalence refers to an interval, usually a year; point prevalence refers to the population on a given date, usually December 31. Prevalence data are useful for evaluating the health effects of the disease on society, estimating the costs of providing health care services, and determining what resources and manpower are necessary to provide these services.
INCIDENCE AND PREVALENCE
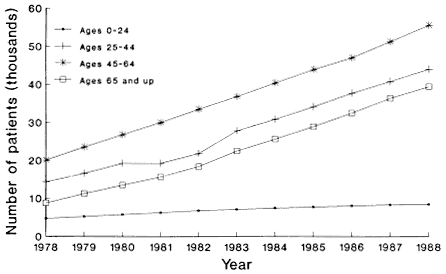
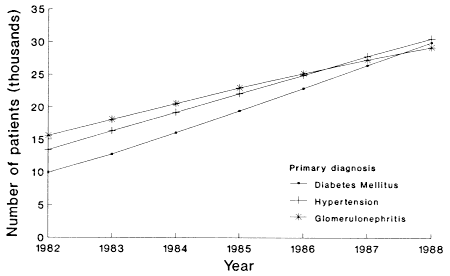
The incidence and prevalence of Medicare ESRD patients have increased dramatically from the start of the Medicare ESRD program (Table 1-1), and this pattern is projected to continue through the 1990s (Table 1-3). The highest rates of increase are among the aged (Figure 4-1) and diabetic (Figure 4-2) populations.
In 1967, the Report of the Committee on Chronic Kidney Disease estimated that a maximum of 700 to 1,000 patients started hemodialysis between March 1960 and March 1967. The age distributions of 247 patients in Public Health Service (PHS)-funded units who started dialysis during this period and of 231 Veterans Administration (VA) patients who began treatment during 1963–67 are shown in Table 4-1. Nearly three-quarters of the PHS patients and over 90 percent of the VA patients were 25–54 years old. Virtually no patients were under 15 or over 65 years of age.
By the early 1970s, the numbers of new patients had reached several thousand per year, the vast majority of whom were still between 25 and 55 years old. Although the rate of increase was dramatic—over 10-fold during this period—the absolute number of new patients was modest.
The Committee on Chronic Kidney Disease (1967) estimated the number of patients with renal failure who would be considered appropriate candidates for renal replacement therapy for the period from 1968 to 1977, based on the then-current criteria for acceptance into renal replacement treatment. Most eligible patients were in the 15-to 54-year age range. The projected population was expected to increase sharply during the early 1970s and then become relatively stable. The report estimated that the most probable level of new patients in 1977 would be about 8,400 and the total dialysis population about 40,000.
It was predicted, however, that as therapy became more readily available, acceptance criteria would be liberalized. In addition, the prevention of early death from diabetes and other diseases would probably lead to increased numbers of renal failure patients. After adjustment for age changes, a 5 percent increase in incident cases of chronic uremia was predicted for the end of the 1970s. The upper, lower, and most likely estimates are shown in Table 4-2.
TABLE 4-1 Age of New Dialysis Patients, 1960–67
What actually happened? After the introduction of the Medicare ESRD program in 1973, the number of newly treated patients increased dramatically. The age distribution of new patients began to shift upward; the proportion of new ESRD patients with chronic diseases such as diabetes mellitus and hypertensive vascular disease also began shifting upward. Before 1973, patients were disproportionately white, middle-class men with high educational status. After 1973, the proportion of women and racial minorities increased, and the distribution of patients tended to follow more closely the demographic features of the American population (Evans et al., 1981). The actual number of new Medicare ESRD patients in 1977 (USRDS, 1990) was 15,832 (estimated to be 90 to 93 percent of the total), or almost twice that projected by the Committee on Chronic Kidney Disease (1967).
COMPOSITION OF THE ESRD POPULATION
Not only has the size of the incident population increased over time, but the characteristics of the population also have changed. As mentioned above, the number and proportion of ESRD patients who are elderly have increased dramatically. Between 1978 and 1988, new Medicare ESRD patients over age 65 increased from 3,637 to 13,866 (USRDS, 1990), or from 24 percent to 38 percent of all new patients. In this same period, new patients over age 75 increased from 799 to 5,061, or from 5.4 percent to 14.0 percent of the total; those over age 85 increased from 54 to 531, or from 0.3 percent to 1.5 percent of the total. Table 4-3 shows these trends.
TABLE 4-2 1967 Projections of New End-Stage Renal Disease (ESRD) Patients, 1968–77
|
Year |
Lower Limit |
Most Probable |
Upper Limit |
|
1968 |
5,978 |
6,958 |
8,152 |
|
1969 |
6,104 |
7,105 |
8,324 |
|
1970 |
6,233 |
7,255 |
8,499 |
|
1971 |
6,364 |
7,408 |
8,678 |
|
1972 |
6,498 |
7,564 |
8,861 |
|
1973 |
6,635 |
7,723 |
9,048 |
|
1974 |
6,775 |
7,886 |
9,239 |
|
1975 |
6,918 |
8,052 |
9,434 |
|
1976 |
7,065 |
8,222 |
9,633 |
|
1977 |
7,214 |
8,395 |
9,836 |
|
SOURCE: Committee on Chronic Kidney Disease, 1967, pp. 125–127. |
|||
More liberal acceptance criteria and possibly decreased death rates from nonrenal complications have led to a notable increase in the diabetic and hypertensive ESRD population. Between 19821 and 1988, the reported number of new Medicare ESRD patients with diabetes as the primary cause of renal failure increased from 5,019 to 11,034 per year (USRDS, 1990), an increase from 23 percent to 31 percent of all new patients.
The incidence of black patients increased at a greater rate than that of whites; between 1978 and 1988, incidence increased an average of 10 percent per year for blacks compared to 8 percent per year for whites. The incidence of Native Americans and Asians/Pacific Islanders increased more rapidly, with average yearly increases of 19 percent and 25 percent, respectively, between 1981 and 1988. Before 1981, reporting for the racial groups Native Americans and Asians was highly incomplete.
More men than women are patients, and the proportions have remained fairly stable between 1978 and 1988.
Incidence rates, defined as the number of newly treated ESRD patients per million population, after adjustment for age, show differences between blacks and whites and between men and women that cannot be seen in the unadjusted data (Table 4-4). In all age groups, except 0–5 years, blacks have higher rates of treated ESRD than do whites. Starting in the teenage years, rates among blacks increase rapidly with age. By age 40, the incidence of ESRD among blacks of both sexes is up to four times greater than that of whites.
For both races, men have higher rates than women, with the exception that among blacks aged 55–84 years, rates of men and women are similar. Among whites of all ages, incidence rates for males are higher than those
TABLE 4-3 New End-Stage Renal Disease (ESRD) Patients by Age, Gender, Race, and Primary Diagnosis, 1978–89
|
|
Year |
|
||||||||||
|
|
1978 |
1979 |
1980 |
1981 |
1982 |
1983 |
1984 |
1985 |
1986 |
1987 |
1988a |
1989a |
|
AGE GROUP (years) |
|
|||||||||||
|
0–4 |
37 |
39 |
60 |
77 |
101 |
99 |
110 |
120 |
116 |
122 |
115 |
112 |
|
5–14 |
288 |
279 |
307 |
262 |
314 |
281 |
317 |
296 |
309 |
305 |
285 |
273 |
|
15–24 |
1,101 |
1,112 |
1,080 |
1,108 |
1,164 |
1,103 |
1,159 |
1,186 |
1,181 |
1,231 |
1,254 |
1,267 |
|
25–34 |
1,763 |
1,851 |
2,086 |
2,178 |
2,460 |
2,466 |
2,624 |
2,707 |
2,975 |
2,837 |
3,052 |
3,286 |
|
35–44 |
2,038 |
2,080 |
2,240 |
2,368 |
2,601 |
2,838 |
3,017 |
3,382 |
3,647 |
3,959 |
4,287 |
4,615 |
|
45–54 |
2,842 |
3,116 |
3,172 |
3,241 |
3,546 |
3,755 |
3,877 |
4,222 |
4,422 |
4,834 |
5,318 |
5,758 |
|
55–64 |
3,464 |
3,951 |
4,378 |
4,737 |
5,292 |
5,773 |
6,283 |
6,902 |
7,099 |
7,733 |
8,227 |
8,671 |
|
65–74 |
2,850 |
2,267 |
3,785 |
4,006 |
4,631 |
6,067 |
6,190 |
7,078 |
7,703 |
8,606 |
9,028 |
10,272 |
|
75–84 |
748 |
1,074 |
1,239 |
1,310 |
1,644 |
2,519 |
2,696 |
3,166 |
3,695 |
4,167 |
4,624 |
5,520 |
|
85 & up |
56 |
83 |
100 |
101 |
170 |
250 |
275 |
347 |
407 |
474 |
543 |
702 |
|
GENDER |
|
|||||||||||
|
Male |
8,522 |
9,462 |
10,332 |
10,779 |
12,139 |
13,863 |
14,719 |
16,047 |
17,356 |
18,634 |
19,998 |
22,063 |
|
Female |
6,669 |
7,494 |
8,120 |
8,612 |
9,788 |
11,292 |
11,833 |
13,372 |
14,205 |
15,639 |
16,745 |
18,434 |
|
RACE |
|
|||||||||||
|
Native American |
37 |
40 |
70 |
131 |
196 |
260 |
265 |
271 |
335 |
345 |
450 |
486 |
|
Asian |
31 |
41 |
59 |
133 |
312 |
306 |
384 |
507 |
508 |
556 |
644 |
683 |
|
Black |
3,920 |
4,591 |
4,862 |
5,064 |
5,976 |
7,074 |
7,491 |
8,284 |
8,728 |
9,666 |
10,412 |
11,425 |
|
White |
10,431 |
11,417 |
12,528 |
13,295 |
15,260 |
17,132 |
18,200 |
20,098 |
21,577 |
23,194 |
24,588 |
26,981 |
|
Other/unknown |
772 |
867 |
933 |
768 |
183 |
273 |
212 |
259 |
413 |
512 |
649 |
922 |
|
PRIMARY DIAGNOSIS |
|
|||||||||||
|
Diabetes |
1,430 |
1,635 |
2,258 |
3,649 |
5,050 |
5,913 |
7,112 |
8,192 |
9,290 |
10,234 |
11,247 |
12,610 |
|
Glomeruloneph. |
1,918 |
2,116 |
2,244 |
3,500 |
5,118 |
5,480 |
5,775 |
6,181 |
6,263 |
6,500 |
6,560 |
6,577 |
|
Hypertension |
1,845 |
2,042 |
2,528 |
3,965 |
5,404 |
5,791 |
6,459 |
7,362 |
7,772 |
8,829 |
9,725 |
10,801 |
|
Missing |
6,931 |
7,802 |
7,802 |
3,729 |
1,691 |
2,897 |
1,852 |
1,894 |
2,239 |
2,315 |
2,806 |
4,271 |
|
Other |
1,737 |
1,918 |
2,125 |
2,881 |
3,229 |
3,642 |
3,853 |
4,615 |
4,393 |
4,564 |
4,652 |
4,764 |
|
Unknown |
1,330 |
1,443 |
1,495 |
1,667 |
1,460 |
1,432 |
1,501 |
577 |
1,604 |
1,831 |
1,753 |
1,474 |
|
TOTAL |
15,191 |
16,956 |
18,452 |
19,391 |
21,927 |
25,155 |
26,552 |
29,419 |
31,561 |
34,273 |
36,743 |
40,497 |
|
a Data incomplete for this year. SOURCE: HCFA, 1990b. |
||||||||||||
TABLE 4-4 New End-Stage Renal Disease (ESRD) Patients per Million Population by Age, Gender, and Race, 1987
for females: after age 75, the incidence among white men is more than twice that of white women between ages 75 and 85, and four times greater after age 85.
The incidence of the underlying cause of ESRD also differs substantially among the principal gender-race subgroups (Table 4-5).2 Among white men, ESRD rates attributed to diabetes mellitus and hypertension are quite similar, with these two diagnoses accounting for the majority of patients. Diabetes mellitus is the most frequently reported underlying cause of ESRD among white women, with incidence rates nearly as high as among white men. Hypertension and glomerulonephritis contribute about 60 percent and 40 percent as many patients, respectively, as diabetes.
For each diagnosis, incidence rates are higher among blacks than among whites. Hypertension accounts for nearly half of all ESRD cases among black men, and the rate of hypertensive ESRD is more than five times higher among black than white men. Compared to white men, black men have over twice the incidence of ESRD due to diabetes and glomerulonephritis, respectively. Compared to white women, black women have incidence rates seven, four, and three times higher for ESRD attributed to hypertension, diabetes, and glomerulonephritis. Among the races, Native Americans have the highest rates of ESRD attributed to diabetes and glomerulonephritis; nearly two-thirds of ESRD among Native Americans is attributed to diabetes. Clearly, the rates and causes of ESRD differ substantially between the races, and the burden of ESRD falls more heavily on blacks and Native Americans than on whites.
TABLE 4-5 New End-Stage Renal Disease (ESRD) Patients per Million Population by Primary Diagnosis, Gender, and Race, 1988
The distribution of prevalent treated ESRD patients generally resembles that of incident patients (Table 4-6). However, because of considerably higher mortality rates among elderly and diabetic ESRD patients compared to other age and diagnostic groups, both represent a considerably smaller proportion of the prevalent compared to the incident population. For example, in 1988 the incidence rate of diabetic ESRD was over twice that of glomerulonephritic ESRD, but the prevalence was very similar. Similarly, in 1988, those over age 65 accounted for 38 percent of the incident population but only 26 percent of the December 31 prevalent population.
MORTALITY ISSUES
The mortality experience of the ESRD program, as a matter of clinical, epidemiologic, and policy concern, raises three basic issues: First, has ESRD mortality changed over time? Second, what factors are causally related to observed patterns of mortality? Third, what are the clinical and policy implications of the first two issues. This section addresses mainly the first and second issues; Chapter 10 deals with the third issue.
HCFA is the primary source of mortality data in the ESRD program. The annual HCFA research report on ESRD presents patient survival data that are generally grouped by age, gender, race, and primary disease. The methods of analysis of these data have changed over time, reflecting a continuing search for improved ways to analyze and present the data (HCFA, 1986, 19879, 1988, 1989). The USRDS, in its first two annual data reports, published extensive summaries of HCFA ESRD data (USRDS, 1989, 1990). The second report made several methodological changes to improve its reporting of the data. Other analyses of ESRD mortality have been published, including analysis of gross mortality (Hull and Parker, 1990) and regional and local analyses (Acchiardo et al., 1983; Blagg et al., 1983; Collins et al.,
TABLE 4-6 Prevalent End-Stage Renal Disease (ESRD) Patients by Age, Gender, Race, and Primary Diagnosis, 1978–89
|
|
Year |
|
||||||||||
|
|
1978 |
1979 |
1980 |
1981 |
1982 |
1983 |
1984 |
1985 |
1986 |
1987 |
1988a |
1989a |
|
AGE GROUP (Years) |
|
|||||||||||
|
0–4 |
57 |
68 |
87 |
130 |
175 |
214 |
241 |
274 |
284 |
301 |
309 |
301 |
|
5–14 |
736 |
817 |
913 |
951 |
1,079 |
1,167 |
1,254 |
1,303 |
1,391 |
1,484 |
1,551 |
1,624 |
|
15–24 |
3,937 |
4,349 |
4,722 |
5,095 |
5,157 |
5,686 |
5,922 |
6,067 |
6,308 |
6,460 |
6,591 |
6,704 |
|
25–34 |
6,990 |
8,231 |
9,627 |
10,890 |
12,193 |
13,382 |
14,569 |
15,730 |
16,959 |
17,832 |
18,757 |
19,752 |
|
35–44 |
7,906 |
9,029 |
10,196 |
11,501 |
13,136 |
15,002 |
16,886 |
18,986 |
21,248 |
23,417 |
25,731 |
28,394 |
|
45–54 |
10,105 |
11,510 |
12,715 |
13,953 |
15,311 |
16,605 |
18,157 |
19,730 |
21,275 |
23,331 |
25,802 |
28,404 |
|
55–64 |
10,935 |
12,940 |
14,937 |
16,883 |
19,018 |
21,063 |
26,061 |
24,964 |
26,547 |
28,739 |
30,715 |
32,903 |
|
65–74 |
7,147 |
8,937 |
10,565 |
12,075 |
13,986 |
16,617 |
18,594 |
20,616 |
22,888 |
25,230 |
27,226 |
30,009 |
|
75–84 |
1,567 |
2,232 |
2,836 |
3,441 |
4,221 |
5,613 |
6,682 |
7,827 |
9,065 |
10,427 |
11,622 |
13,334 |
|
85 & up |
86 |
140 |
204 |
261 |
367 |
499 |
602 |
759 |
884 |
1,128 |
1,327 |
1,592 |
|
GENDER |
|
|||||||||||
|
Male |
27,732 |
32,477 |
37,199 |
41,672 |
46,983 |
52,891 |
58,533 |
64,038 |
69,658 |
75,688 |
81,700 |
88,941 |
|
Female |
21,734 |
25,776 |
29,603 |
33,508 |
37,960 |
42,957 |
47,435 |
52,319 |
57,191 |
62,661 |
67,931 |
74,076 |
|
RACE |
|
|||||||||||
|
Native American |
102 |
137 |
201 |
320 |
482 |
686 |
856 |
985 |
1,144 |
1,291 |
1,507 |
1,729 |
|
Asian |
102 |
143 |
202 |
333 |
598 |
848 |
1,102 |
1,424 |
1,708 |
2,013 |
2,353 |
2,717 |
|
Black |
12,979 |
15,616 |
18,021 |
20,419 |
23,480 |
27,061 |
30,350 |
33,683 |
36,784 |
40,356 |
43,972 |
47,918 |
|
White |
33,956 |
39,562 |
45,176 |
50,718 |
57,319 |
64,303 |
70,879 |
77,572 |
84,456 |
91,765 |
98,574 |
106,858 |
|
Other/unknown |
2,327 |
2,795 |
3,202 |
3,390 |
3,064 |
2,950 |
2,781 |
2,693 |
2,757 |
2,924 |
3,225 |
3,795 |
|
PRIMARY DIAGNOSIS |
|
|||||||||||
|
Diabetes |
3,113 |
3,861 |
2,258 |
7,120 |
9,938 |
12,743 |
16,073 |
19,457 |
22,969 |
26,567 |
30,265 |
34,611 |
|
Glomeruloneph. |
8,670 |
9,991 |
2,244 |
13,844 |
17,454 |
20,946 |
24,297 |
27,609 |
30,479 |
33,151 |
35,538 |
37,746 |
|
Hypertension |
5,229 |
6,424 |
2,528 |
10,242 |
13,431 |
16,373 |
19,189 |
22,099 |
24,883 |
27,827 |
30,679 |
33,854 |
|
Other/unknown |
32,454 |
37,977 |
7,802 |
43,974 |
44,120 |
45,786 |
46,409 |
47,192 |
48,518 |
50,804 |
53,149 |
56,806 |
|
TOTAL |
49,466 |
58,253 |
66,802 |
75,180 |
84,943 |
95,848 |
105,968 |
116,357 |
126,849 |
138,349 |
149,631 |
163,017 |
|
a Data incomplete for this year. SOURCE: HCFA, 1990b. |
||||||||||||
1990; Degoulet et al., 1982; Lowrie and Lew, 1990; Parker et al., 1983; Shapiro and Umen, 1983; Wolfe et al., 1990).
Various factors complicate the discussion of mortality trends. First, data accuracy and completeness have varied over time. However, HCFA has steadily improved its data collection procedures, and current data are more reliable than those available in the 1970s and early 1980s. These problems limit the ability to compare the mortality experience between time periods.
Second, differences in methodology among analysts and over time for a given analyst make the interpretation of mortality complex. In particular, no standard convention or protocol is used for calculating ESRD mortality rates. For example, such calculations may or may not adjust for differences in the composition of the patient population, such as age or race, even though adjustment provides a more appropriate basis for comparing rates between different populations. In addition, populations may be variously described as all patients present at a point in time (point-prevalent population), in a time period (period-prevalent population), or new patients during a time interval (incident cohort). Mortality rates also differ as a function of the method of calculating mortality. Consequently, the study commissioned a paper by Robert A. Wolfe (see Appendix D of this report) to clarify methodological issues and recommends the formation of a technical working group (Chapter 14) related to ESRD patient mortality.
Third, it is necessary to differentiate between mortality rates for an entire patient population (unadjusted as well as adjusted mortality) and the risk of death attributable to various patient or treatment characteristics such as age, gender, race, diagnosis leading to ESRD, treatment modality, or treatment year. The former may be increasing at the same time that the latter is stable or decreasing.
Fourth, three current hypotheses of major factors causing changes in mortality rates in the ESRD patient population are patient characteristics (e.g., increasing age and complexity), treatment characteristics (e.g., inadequate dialysis), and eroding quality of care resulting from reimbursement reductions. Although it is difficult to distinguish among these factors in most analyses, the practical importance of the conclusions about the causes of mortality varies greatly between providers and the government. This tends to infuse the technical discussions with subjective considerations.
The Medicare ESRD program mortality experience can be summarized as follows:
-
The unadjusted mortality (or gross mortality) rate, defined as the rate of death in all patients treated for ESRD during a year, has been increasing over time.
-
Unadjusted mortality rates in incident cohorts, however, were stable during 1978–82, jumped upward from the 1982 to the 1983 incident cohort, and have remained fairly stable since then.
-
Mortality for the 1978–88 incident and prevalent cohorts, when adjusted for age, gender, race, and primary diagnosis, has been quite stable.3
-
Analyses of subgroups of the ESRD patient population show the mortality of some groups decreasing (i.e., survival improving), some stable, and some increasing.
-
International data show that the United States has higher gross mortality and adjusted mortality rates than some European countries, although these data must be interpreted cautiously because cross-national comparisons have substantial limitations.
Unadjusted Mortality
This section presents data for annual incident and prevalent cohorts of ESRD patients and discusses the effect of increasing incidence on temporal trends of unadjusted mortality. The following section deals with mortality adjusted for various patient characteristics. The purpose of the discussion is to clarify various published analyses of temporal changes in mortality within the program.
In the literature, ESRD patients have been categorized in several ways for mortality analysis. These include the mortality of all patients treated during a given year, often referred to as gross mortality (Hull and Parker, 1990),4 the mortality of incident patient cohorts (HCFA, 1986, 1988, 1989; USRDS, 1989, 1990), and the mortality of those patients who have survived at least one year of ESRD treatment (HCFA, 1987). Each method provides a different result for the Medicare ESRD program mortality experience. The importance of these differences will be explored.
Unadjusted annual mortality of the prevalent ESRD population, also referred to as gross mortality, may be defined as the rate of death among all ESRD patients treated during a year. It has been increasing over time as shown in Table 4-7 for the period 1978–88.5 This trend is in the same direction, although not as large, as that shown by Hull and Parker (1990) for all dialysis patients (Medicare plus non-Medicare).
Various patient characteristics contribute to higher unadjusted or gross mortality, including increasing age of the patient population and more severe illness (e.g., diabetes and other comorbid conditions). The age effect is clear: As with mortality in general, the mortality of ESRD patients increases with age. Increased severity of illness also contributes directly to mortality risk. The increase in many comorbid conditions, although not well documented, has been reported in several studies (Collins et al., 1990; Kjellstrand et al., 1990).
Trends in unadjusted mortality of annual incident patient cohorts are shown in Table 4-7. Mortality of incident patient cohorts is commonly reported in the literature (Disney, 1990; Eggers, 1990; HCFA, 1986, 1988,
TABLE 4-7 Gross and Unadjusted One-Year Mortality for All End-Stage Renal Disease (ESRD) Patients, 1978–88
|
Year |
Gross Mortalitya (%) |
Prevalent Cohort Unadjusted Mortalityb (%) |
Incident Cohort Unadjusted Mortalityc (%) |
|
1978 |
14.0 |
11.8 |
18.8 |
|
1979 |
13.7 |
11.8 |
19.1 |
|
1980 |
14.2 |
12.2 |
18.8 |
|
1981 |
14.0 |
12.5 |
18.6 |
|
1982 |
13.8 |
12.1 |
18.8 |
|
1983 |
14.2 |
12.1 |
21.3 |
|
1984 |
14.7 |
12.1 |
21.2 |
|
1985 |
15.3 |
13.0 |
21.6 |
|
1986 |
15.5 |
13.3 |
21.6 |
|
1987 |
15.4 |
13.2 |
22.5 |
|
1988 |
15.8 |
13.4 |
21.7 |
|
a Calculated for all patients treated during reference year, life-table method with censoring at end of year (HCFA, 1990a). b One-year mortality during reference year for prevalent patients who had survived at least one year before start of the index year, life-table method (HCFA, 1990a.) c One-year mortality after first 90 days of ESRD for all patients by year of incidence, Kaplan-Meier method (USRDS, 1990). SOURCES: HCFA, 1990a; USRDS, 1990. |
|||
1989a; USRDS, 1989, 1990). Unadjusted mortality analyses indicate three things: First, in the period from 1978 through 1982, mortality was stable; second, an abrupt increase in recorded mortality occurred during the 1982–83 period; and third, mortality has remained fairly stable since 1983 (Table 4-7).
The abrupt upward shift in unadjusted incident cohort mortality reported from 1982 to 1983 (Eggers, 1990; USRDS, 1989, 1990) represents a puzzle to analysts of ESRD mortality data that is unlikely to be fully resolved. The increase is partly due to a sharp increase during 1983 in reported new patients in two high-risk groups—the elderly and the Medicare disabled (those who had qualified for disability before kidney failure). It also appears to reflect an artifact of the data reporting system. Evidence supporting this view includes recent analyses by Eggers (P.W. Eggers, HCFA, unpublished data, 1990) showing that a large number of cases, most of whom were elderly, were reported to HCFA on July 1, 1983, and that more incident ESRD cases were recorded in the HCFA PMMIS data set for 1983 than reported by the facilities, a virtual impossibility.
In addition to mortality for the total population, most of these unadjusted incident patient cohort analyses usually present data grouped by patient
characteristics such as age, gender, race, and/or primary diagnosis. Mortality for the subgroups usually differs from the total unadjusted mortality.
It is also clear that mortality of new ESRD patients is considerably higher than mortality of those continuing treatment (e.g., those who have already survived for at least one year on treatment) or of the total patient population (gross mortality) (Table 4-7). For example, unadjusted incident cohort mortality for 1988 was 21.7 percent (USRDS, 1990), whereas it was 13.4 percent for one-year survivors. Mortality rates are generally highest during the period immediately after initiation of ESRD treatment, largely because of earlier death of more vulnerable patients.
Adjusted Mortality
Although mortality rates of a patient population may change over time, the risk of death for particular groups of patients does not necessarily follow the same trend. Gross mortality for ESRD patients has increased over time, largely because of an increased proportion of higher risk patients, but there is no a priori reason to believe that the risk of death, for example, of a white male patient, age 45, with no major comorbid conditions, increased between 1978 and 1988.
In order to assess whether the risk of death has changed over time, mortality data must be adjusted for changes in the patient population. For ESRD patients, mortality risk differs by age, race, gender, primary diagnosis, time since diagnosis of renal failure, and comorbid conditions: Various statistical methods, including a Cox proportional hazards model, Poisson regression, and subgroup analysis, can be used to adjust or control for these variables so that populations with different distribution of these variables can be more appropriately compared. HCFA data permit adjustment for age, gender, race, primary diagnosis, and time-related covariates such as year of treatment and time since renal failure. They do not permit adjustment for comorbid conditions and severity of illness, although these factors have important effects on mortality.
ESRD mortality rates for annual incident patient cohorts, when adjusted for age, race, gender, and primary diagnosis, were stable during the 1980s (Eggers, 1990; USRDS, 1990) as shown in Table 4-8. Mortality for prevalent (all) ESRD patients, adjusted for age, was stable or improving.
Subgroup Mortality
Mortality data grouped by patient characteristics generally indicate the following patterns (Table 4-9). Mortality increases with age, males have slightly higher mortality than females, and whites have higher mortality than blacks. By the major primary disease leading to ESRD, diabetes has
TABLE 4-8 Mortality for Medicare End-Stage Renal Disease (ESRD) Patients (ever entitled) Adjusted for Age, Race, Gender, and Primary Diagnosis, 1978–88 Patient Cohorts
|
|
Adjusted Mortality (%) |
|
|
Year |
Incident Cohortsa |
Prevalent Cohortsb |
|
1978 |
21.0 |
13.8 |
|
1979 |
20.3 |
13.5 |
|
1980 |
20.1 |
13.9 |
|
1981 |
20.9 |
14.2 |
|
1982 |
20.7 |
13.2 |
|
1983 |
21.7 |
13.2 |
|
1984 |
21.6 |
12.0 |
|
1985 |
21.6 |
13.8 |
|
1986 |
21.1 |
13.7 |
|
1987 |
21.5 |
13.6 |
|
1988 |
20.6 |
13.7 |
|
a One-year adjusted mortality calculated from day 91 to 1 year + 90 days for patients, by year of incidence of ESRD. Adjustment is by years of age (1–19, 20–44, 45–64, 65–74, 75 plus), race, gender, and primary diagnosis, Kaplan-Meier method (USRDS, 1990). b One-year mortality calculated for patients who had survived at least I year before start of the index year. Adjustment is by 5-year age groups, life-table method (HCFA, 1990a). |
||
the highest mortality, hypertension is next, and glomerulonephritis and polycystic disease have the lowest mortality. Mortality rates for transplant patients are lower than those for dialysis patients. The extent to which these differences reflect patient selection is not known.
In spite of the general stability in adjusted mortality, changes have occurred for different subgroups of the ESRD population between 1978 and 1988. There has been general improvement in survival of younger patients and diabetic patients on all modalities of treatment (USRDS, 1990, Tables E.69–E.89). In older patients, race-and diagnosis-adjusted survival is generally steady except for a step increase between 1982 and 1983. Mortality rates, especially in older patients, vary considerably depending on the method of analysis. For example, using age groups with 10-year spans shows a larger trend toward increased mortality than using groups with a span of 5 years or less. This is because age is less adequately controlled as the span increases.
By treatment, there has been improvement in survival for transplant patients of all ages. For example, one-year survival percentages for cadaveric transplant patients in the age groups 0–19, 20–44, and 45–64 increased from 72 to 78, 87 to 94, and 68 to 89, respectively, between 1978 and 1988
TABLE 4-9 One-Year Mortality (percent) for All End-Stage Renal Disease (ESRD) Patients, at Year of Incidence, by Age, Gender, Race, and Primary Diagnosis, 1978–88
|
|
Year |
|
|||||||||
|
|
1978 |
1979 |
1980 |
1981 |
1982 |
1983 |
1984 |
1985 |
1986 |
1987 |
1988 |
|
AGE GROUP (years) |
|
||||||||||
|
0–19a |
6.8 |
5.8 |
5.4 |
4.0 |
4.9 |
3.9 |
4.8 |
5.2 |
4.0 |
4.1 |
4.6 |
|
20–44 |
12.9 |
10.7 |
11.0 |
10.0 |
8.9 |
9.6 |
9.5 |
9.3 |
8.7 |
9.5 |
8.4 |
|
45–64 |
18.7 |
18.5 |
17.3 |
18.4 |
18.2 |
19.1 |
19.6 |
19.0 |
18.3 |
18.0 |
17.5 |
|
65–74 |
25.5 |
24.8 |
27.6 |
27.8 |
27.5 |
29.8 |
29.0 |
30.6 |
29.8 |
29.6 |
28.5 |
|
75 and older |
37.2 |
35.6 |
34.2 |
38.6 |
38.1 |
41.1 |
37.5 |
38.2 |
38.3 |
41.7 |
39.2 |
|
GENDER |
|
||||||||||
|
Female |
18.4 |
17.3 |
15.6 |
17.9 |
17.6 |
18.4 |
17.9 |
18.9 |
18.4 |
18.2 |
17.3 |
|
Male |
22.4 |
21.7 |
21.9 |
22.4 |
22.0 |
23.3 |
23.3 |
22.9 |
22.3 |
22.0 |
22.0 |
|
RACE |
|
||||||||||
|
Black |
20.2 |
20.1 |
19.8 |
20.5 |
19.8 |
20.9 |
20.4 |
20.8 |
19.5 |
19.3 |
19.3 |
|
Nonblack |
21.7 |
20.7 |
20.4 |
21.2 |
21.5 |
22.4 |
22.5 |
22.3 |
22.3 |
21.8 |
21.8 |
|
PRIMARY DIAGNOSIS |
|
||||||||||
|
Diabetes |
28.5 |
27.2 |
27.5 |
28.9 |
29.6 |
28.7 |
27.1 |
29.5 |
27.6 |
24.8 |
24.8 |
|
Hypertension |
17.9 |
16.9 |
18.2 |
18.5 |
18.9 |
20.1 |
21.0 |
19.3 |
19.9 |
20.5 |
20.5 |
|
Kidney diseases |
14.4 |
12.3 |
13.0 |
14.0 |
14.1 |
14.3 |
13.9 |
14.8 |
4.6 |
14.9 |
14.9 |
|
Other |
22.9 |
23.4 |
22.3 |
21.5 |
20.3 |
24.4 |
24.4 |
25.2 |
22.8 |
21.7 |
21.7 |
|
TOTAL |
21.0 |
20.3 |
20.1 |
20.9 |
20.7 |
21.7 |
21.6 |
21.6 |
21.1 |
20.6 |
20.6 |
|
NOTE: Mortality calculated from day 91 to 1 year + 90 days. a Because of possible data errors for pediatric patients (ages 0–19 years), unadjusted mortality has been used for this group. Because measured differences in incidence patterns for the pediatric population have been very small, the use of unadjusted in place of adjusted data should not have a meaningful effect. SOURCE: USRDS, 1990, Tables E-52 and E-53. |
|||||||||||
(USRDS, 1990). Dialysis patient survival, however, does not show consistent time trends (Table 4-10), partly because of greater variability among patient age groups. In addition, it is difficult to interpret dialysis mortality because it is influenced by the outward flow of healthier patients to transplantation.
Nevertheless, by age, adjusted by gender, race, and primary diagnosis, there appears to be improved survival among dialysis patients under 45 years old and steady survival for those between ages 45 and 64. Over age 65, there is greater variability in the data; however, a trend toward higher mortality is evident in Table 4-10 for both the 65–74 and the over 75 age groups.
State and Regional Mortality Data
State and regional data, although they often differ from national data, offer insight into the experience of various patient groups. Wolfe and coworkers (1990) reported mortality outcomes for 2,754 dialysis patients using data from the Michigan Kidney Registry for patients between the ages of 20 and 60. Using a Cox regression model, they found a 6 percent per year increase in mortality between annual incident patient cohorts starting center hemodialysis (CH) treatment between 1980 and 1987. (They defined treatment as the modality of dialysis at six months after the onset of ESRD, without regard to whether patients changed to another modality after six months.) This represents a 50 percent increase in the mortality of new patient cohorts during the study years. (Time-dependent covariates, such as treatment year and time since renal failure, were not included in the study; see Wolfe, Appendix D.)
Wolfe and colleagues suggest several possible factors contributing to the observed trend: acceptance of sicker patients for ESRD therapy; selection of healthier patients to treatment modes other than CH, leaving sicker patients on CH (increased proportions of patients were treated with transplantation and peritoneal dialysis during the period of this study); and a degradation of quality of CH therapy. The investigators conclude: ''If these trends continue, or if the most recent results are maintained, then future studies must confront the increase in mortality rates among center hemodialysis patients and determine its cause'' (Wolfe et al., 1990, p. 439). This report differs sharply from the national pattern for the same period and it should be followed up with studies designed to disentangle patient characteristics and treatment modality effects on mortality.
Collins and associates (1990) analyzed the mortality experience of 2,985 hemodialysis patients treated within the Regional Kidney Disease Program in Minnesota. A comparison of the 1976–82 and 1983–87 periods showed that the proportion of patients starting hemodialysis with no major risk
TABLE 4-10 One-Year Mortality (percent) for Dialysis Patients at Year of Incidence by Age, Gender, Race, and Primary Diagnosis, 1978–88
|
|
Year |
|
|||||||||
|
|
1978 |
1979 |
1980 |
1981 |
1982 |
1983 |
1984 |
1985 |
1986 |
1987 |
1988 |
|
AGE GROUP (years) |
|
||||||||||
|
0–19a |
15.8 |
14.4 |
11.7 |
10.0 |
11.3 |
9.4 |
9.7 |
9.6 |
6.3 |
6.4 |
6.4 |
|
20–44 |
21.1 |
19.8 |
19.5 |
17.3 |
16.9 |
16.6 |
14.9 |
13.9 |
12.2 |
12.2 |
9.9 |
|
45–64 |
21.0 |
20.7 |
19.2 |
20.8 |
20.2 |
21.1 |
21.6 |
20.9 |
20.1 |
19.1 |
18.4 |
|
65–74 |
24.9 |
25.5 |
24.8 |
27.7 |
27.8 |
27.5 |
29.8 |
29.1 |
30.6 |
29.8 |
29.7 |
|
75 and older |
37.2 |
35.6 |
34.2 |
38.6 |
38.1 |
40.1 |
37.5 |
38.2 |
38.3 |
41.7 |
39.2 |
|
GENDER |
|
||||||||||
|
Female |
22.7 |
22.6 |
22.3 |
22.7 |
21.8 |
23.0 |
22.1 |
22.2 |
20.8 |
21.7 |
19.8 |
|
Male |
25.3 |
24.5 |
23.7 |
24.5 |
24.6 |
25.4 |
25.2 |
24.8 |
24.1 |
22.7 |
22.7 |
|
RACE |
|
||||||||||
|
Black |
20.1 |
19.8 |
17.4 |
19.5 |
19.6 |
20.1 |
19.5 |
20.1 |
19.2 |
17.6 |
17.6 |
|
Nonblack |
26.0 |
25.3 |
25.3 |
25.6 |
24.9 |
26.3 |
25.8 |
27.2 |
24.2 |
22.9 |
22.9 |
|
PRIMARY DIAGNOSIS |
|
||||||||||
|
Diabetes |
31.9 |
32.7 |
31.6 |
33.1 |
32.9 |
32.1 |
30.1 |
32.4 |
30.3 |
25.8 |
25.8 |
|
Hypertension |
20.5 |
20.0 |
20.8 |
20.8 |
21.1 |
22.3 |
22.6 |
20.6 |
21.2 |
21.0 |
21.0 |
|
Kidney diseases |
17.2 |
15.2 |
15.4 |
16.7 |
16.4 |
16.7 |
15.7 |
16.4 |
15.9 |
15.5 |
15.5 |
|
Other |
25.6 |
26.1 |
24.9 |
23.7 |
22.4 |
26.2 |
26.2 |
27.1 |
24.2 |
22.5 |
22.5 |
|
TOTAL |
24.1 |
23.5 |
23.0 |
23.7 |
23.4 |
24.3 |
23.8 |
23.7 |
22.6 |
21.4 |
21.4 |
|
NOTE: Mortality calculated from day 91 to 1 year + 90 days. a Because of possible data errors for pediatric patients (ages 0–19 years), unadjusted mortality has been used for this group. Because measured differences in incidence patterns for the pediatric population have been very small, the use of unadjusted in place of adjusted data should not have a meaningful effect. SOURCE: USRDS, 1990, Tables E-68 and E-69. |
|||||||||||
factors (atherosclerotic heart disease, cerebrovascular disease, peripheral vascular disease, chronic obstructive lung disease, and nonskin malignancies) decreased from 41 percent to 28 percent. Those with two or more such risk factors increased from 59 percent to 72 percent: diabetics increased from 29 percent to 43 percent; and the percentage of patients over age 75 nearly doubled. Between 1982–84 and 1985–88, the average annual unadjusted mortality rate for all dialysis patients (hemodialysis and peritoneal dialysis) increased from 16.7 percent to 25.6 percent, a 55 percent increase.
In spite of increased comorbidities, mortality among diabetics remained stable up to age 75 but increased among older patients. Mortality decreased among nondiabetic patients with no major comorbidities in these years; nondiabetic patients with increased numbers of major risk factors had increased mortality. Thus, higher comorbidity correlated with increased mortality. The investigators conclude that "the increase in the annual gross mortality rate is highly predicted based on the change in the diabetic population and the increase in single and multiple comorbid conditions in the nondiabetic population" (Collins et al., 1990, p. 422).
These regional studies show increased mortality rates among some groups of dialysis patients and point out the importance of controlling for comorbidities in the analysis of mortality. At present, however, HCFA data do not include these comorbidities.
Cross-National Mortality Data
There are usually two major objectives of international comparisons of mortality rates. (See Wolfe, Appendix D.) The first is to document the existence of differences in mortality rates or to evaluate the relative risk of mortality between countries. The second is to identify the reasons for such differences, if they exist. Using the data currently available from different national registries, it is difficult to arrive at a definitive answer to the first objective, and it is impossible to arrive at an answer to the second.
It has been reported that the mortality of U.S. dialysis patients is higher than in many other countries. The measure most commonly used is gross mortality, which has major limitations as described above. Held and co-workers (1990), however, reported that the mortality of U.S. dialysis patients is much higher even after age adjustment. They compared 5-year survival (the inverse of mortality) of new ESRD patients accepted for treatment in the U.S. Medicare program (150,862 patients), the European Dialysis and Transplant Association (EDTA) countries (124,796 patients for 1982–87), and Japan (66,244 patients for 1983–87). The patients over age 65 represented 37, 24, and 30 percent of the patients for the United States, EDTA, and Japan, respectively; the percentages diabetic were 27, 10, and 19, respectively).
Compared to the U.S. experience, survival rates, adjusted for age and
diabetes as a primary diagnosis, were higher in the EDTA for all ages above 25 and in Japan for all ages above 15. Five-year survival rates for the ESRD populations, adjusted to the U.S. population age structure, are shown in Table 4-11. The authors suggest that the differences in mortality may be due to one or more of the following: patient selection rates (the U.S. accepts more patients per million for renal replacement treatment); the completeness and representativeness of the different registries; patient compliance; and the adequacy of treatment.
Kjellstrand and associates (1990) reported important factors that influenced mortality rates in U.S. and Swedish dialysis centers. Data included 2,004 cases from the Regional Kidney Disease Program at Hennepin County Medical Center in Minneapolis and 274 patients at the Karolinska Hospital in Stockholm, Sweden. Focusing on 10 comorbid conditions (arteriosclerotic heart disease, chronic heart insufficiency, peripheral vascular disease, cerebral stroke, pulmonary disease, gastrointestinal disease, cirrhosis, malignancy, hypertensive cardiomegaly, and presence of infection), the investigators found that the number of patients without complications remained stable in both centers between the early 1970s and the mid-1980s. Almost all the increase in the number of patients was among patients over age 50 with complications. Survival analyses stratified by patient comorbid conditions showed little difference between the U.S. and the Swedish experiences.
In an analysis of survival in five European areas (Benelux; British Isles—UK and Ireland; Nordic—Denmark, Norway, Sweden, and Finland; Latin—Italy, Spain and France; and Germany and Austria) and the United States, Kjellstrand and associates (1990) reported that over 90 percent of differences in cumulative survival rates among younger dialysis patients correlated with acceptance and transplantation rates. The analysis reported 4-year cumulative survival in dialysis patients aged 15–44 years. This analysis points to effects of comorbid conditions, acceptance rates, and transplantation rates on survival rates in national populations.
Wolfe (Appendix D) has identified several limitations that apply to international comparisons of mortality rates: (1) data are not collected and reported in a consistent way; (2) the level of mortality observed in a national registry is strongly related to the criteria for patient acceptance for treatment; (3) patient-specific data are not available in a unified data base on which analyses can be performed, resulting in the comparison of dissimilar data; (4) etiologies and comorbidities are not adequately reported; (5) age adjustment tends not to fully adjust for age differences—if patients are older in one nation, then they will tend to be older within each age category as well; (6) cross-national differences in withdrawal from treatment and in the reporting of withdrawal may also be relevant since withdrawal is a major cause of death among elderly U.S. patients.
In spite of these limitations, international comparisons provide important information and hypotheses for further analysis which may lead to improve-
TABLE 4-11 Five-Year Survival of Dialysis Patients by Country and Diagnosis of Diabetes
|
Country |
% Non-diabetic |
% Diabetic |
|
United Statesa |
45 |
26 |
|
Europe, 1982–87b |
55 |
31 |
|
United Statesa |
44 |
26 |
|
Japan, 1983–87c |
60 |
40 |
|
a Years and age groupings differ for the age-adjusted comparisons with EDTA and Japan. Therefore, U.S. data are calculated twice to match European and Japanese data. b Europe consists of the countries of the European Dialysis and Transplant Association. Age ranges for Europe are 0–14, 15–24, 25–34, 35–44, 45–54, 55–64, 65–74, and 75–84 years. c Age ranges for Japan are 0–14, 15–29, 30–44, 45–59, 60–74, and 75–89 years. SOURCE: Held et al., 1990. |
||
ments in provision of treatment to ESRD patients. It is important to evaluate more precisely which subgroups (both by patient and by treatment characteristics) of ESRD patients have higher, similar, or lower mortality rates than in other countries. Is quite possible that a lower overall mortality rate is achievable in the United States and that international comparisons may contribute to attaining this goal.
PROJECTIONS TO THE YEAR 20006
In 1984, Eggers and coworkers (1984) suggested that the Medicare ESRD population was unlikely to level off in the near future. They projected that program enrollment would rise to 94,400 by 1990, 117,200 by 2000, and 162,100 by 2030. By 1988, however, the total Medicare enrollment had already increased to more than 133,900 people. The primary reason for Eggers and coworkers' underestimation was an unanticipated increase in the incidence of ESRD patients.
Eggers, at the request of the IOM ESRD study, prepared an estimate of the Medicare ESRD patient population for the year 2000 for this study, updating previous projections (Eggers, 1989) by incorporating new information on incidence and mortality for dialysis as well as transplant patients. His model, based on current eligibility criteria, generates low, middle, and high projections of the ESRD patient population for December 31, 2000, as presented in Table 4-12.
The middle projection anticipates that over 240,000 ESRD patients will be receiving treatment on December 31, 2000. This scenario estimates that
TABLE 4-12 Medicare End-Stage Renal Disease (ESRD) Population Projections, Year 2000
|
|
Estimates |
|
|
|
Category |
Low |
Middle |
High |
|
Incidence of ESRD patients |
47,949 |
56,877 |
68,472 |
|
24,915 |
14,445 |
8,237 |
|
|
Cadaver donors |
20,186 |
11,653 |
6,594 |
|
Living donors |
4,729 |
2,792 |
1,643 |
|
Dialysis patientsc |
122,953 |
182,037 |
231,401 |
|
Functioning graft patientsc |
86,993 |
59,414 |
42,927 |
|
Patients leaving Medicare |
5,256 |
3,566 |
2,562 |
|
Deathsa |
34,164 |
43,357 |
49,351 |
|
Total patientsc |
209,946 |
241,452 |
274,328 |
|
a During calendar year 2000. b Underexisting statutory authority, transplant recipients are limited to 3 years of Medicare eligibility. Increased rates of transplantation, therefore, reduce the projected Medicare patient population, creating the counterintuitive results shown in the table. c End of calendar year 2000. SOURCE: Paul W. Eggers, HCFA, unpublished data, 1990. |
|||
almost 57,000 new patients will enter the program during the year 2000, an increase of 57 percent from 1988. The low scenario projects that almost 210,000 patients will be undergoing treatment at the end of the year 2000, the high scenario nearly 275,000 patients.
The model is sensitive to underlying assumptions, particularly incidence rates. The projection based on the highest incidence rate results in almost 65,000 more ESRD patients than that using the lowest rate. Changes in the number of transplants performed do not influence the projections of total patient population very much but do affect the distribution by mode of therapy. Failure to increase transplants above the current levels will lead to a decline in the percentage of all ESRD prevalent patients with a functioning graft.
Although these projections are unlikely to predict the future of the Medicare ESRD enrollment with precision, they provide a reasonable estimate of future enrollment based on current treatment and outcome trends. Even the low scenario indicates that historical growth of the ESRD population will continue well into the next century, as shown in Figure 4-3.
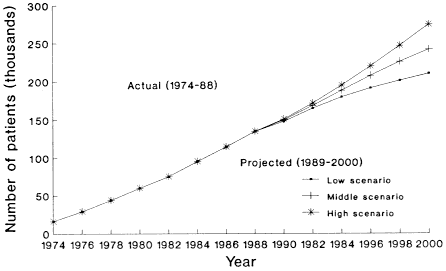
FIGURE 4-3
Number of ESRD Patients, Actual and Projected, 1974–2000
NOTE: As of December 31. SOURCE: Paul E. Eggers, HCFA, unpublished data, 1990.
NOTES
REFERENCES
Acchiardo SR, Moore LW, Latour PA. 1983. Malnutrition as the main factor in morbidity and mortality of hemodialysis patients. Kidney Int 24(Suppl 16):S99–S203.
Blagg CR, Wahl PW, Lamers JY. 1983. Treatment of chronic renal failure at the Northwest Kidney Center, Seattle, from 1960 to 1982. ASAIO J 6:170–175.
Collins AJ, Hanson G, Umen A, Kjellstrand C, Keshaviah P. 1990. Changing risk factor demographics in end-stage renal disease patients entering hemodialysis and the impact on long-term mortality. Am J Kidney Dis 15:422–432.
Committee on Chronic Kidney Disease. 1967. Report of the Committee on Chronic Kidney Disease to U.S. Bureau of the Budget.
Degoulet P, Legrain M, Reach I. 1982. Mortality risk factors in patients treated by chronic hemodialysis: Report of the Daiphane Collaborative Study. Nephron 31:103–110.
Disney APS. 1990. Dialysis treatment in Australia, 1982 to 1988. Am J Kidney Dis 15:402–409.
Eggers, PW. 1989. Projections for the end-stage renal disease population to the year 2000. Proceedings of the 1989 Public Health Conference on Records and Statistics. U.S. Department of Health and Human Services, DHHS Publ. No. PHS 90-1214. November, pp. 121–126.
Eggers PW. 1990. Mortality rates among dialysis patients in Medicare's End-Stage Renal Disease Program. Am J Kidney Dis 15:414–421.
Eggers PW, Connerton R, McMullan M. 1984. The Medicare experience with end-stage renal disease: Trends in incidence, prevalence, and survival. Health Care Financing Rev 5:69–88.
Evans RW, Blagg CR, Bryan FA, Jr. 1981. Implications for health care policy: A social and demographic profile of hemodialysis patients in the United States. JAMA 245:487–491.
HCFA (Health Care Financing Administration). 1986. Health Care Financing Research Report: End-Stage Renal Disease, 1984. Baltimore, Md.
HCFA. 1987. Health Care Financing Research Report: End-Stage Renal Disease, 1985. Baltimore, Md.
HCFA. 1988. Health Care Financing Research Report: End-Stage Renal Disease, 1986. Baltimore, Md.
HCFA. 1989. Health Care Financing Research Report: End-Stage Renal Disease, 1987. Baltimore, Md.
HCFA. 1990a. ESRD Program Management and Medical Information System. March update. Baltimore, Md.
HCFA. 1990b. ESRD Program Management and Medical Information System. August update. Baltimore, Md.
Held PJ, Brunner MD, Odaka MD, Garcia BS, Port FK, Gaylin DS. 1990. Five-year survival for end-stage renal disease patients in the United States, Europe, and Japan, 1982 to 1987. Am J Kidney Dis 15:451–457.
Hull AR, Parker TF. 1990. Introduction and summary: Proceedings from the Morbidity, Mortality, and Prescription of Dialysis Symposium (Dallas, Tex., Sept. 15–17, 1989). Am J Kidney Dis 15:375–383.
Kjellstrand CM, Hylander B, Collins AC. 1990. Mortality on dialysis—On the influence of early start, patient characteristics, and transplantation and acceptance rates. Am J Kid Dis 15:483–490.
Lowie EG, Lew NL. 1990. Death risk in hemodialysis patients: the predictive value of commonly measured variables and an evaluation of death rate differences between facilities. Am J Kidney Dis 15:458–482.
Parker T, Laird NM, Lowrie EG. 1983. Comparison of the study groups. The National Cooperative Dialysis Study. A description of morbidity, mortality and patient withdrawal. Kidney Int 23(Suppl 13):S42–S49.
Shapiro FL, Umen A. 1993. Risk factors in hemodialysis patient survival. ASAIO J 6:176–184.
USRDS (U.S. Renal Data System). 1989. Annual Data Report. National Institute of Diabetes and Digestive and Kidney Diseases, Baltimore, Md.
USRDS. 1990. Annual Data Report. National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, Md.
Wolfe RA, Port FK, Hawthorne MV, Guire KE. 1990. Comparison of survival among dialytic therapies of choice: In-center hemodialysis versus continuous ambulatory peritoneal dialysis at home. Am J Kidney Dis 15:433–440.