The Greening of Industrial Ecosystems. 1994.
Pp. 23-37. Washington. DC:
National Academy Press.
Industrial Metabolism: Theory and Policy
ROBERT U. AYRES
The word metabolism, as used in its original biological context, connotes the internal processes of a living organism. The organism ingests energy-rich, low-entropy materials (food), to provide for its own maintenance and functions, as well as a surplus to permit growth or reproduction. The process also necessarily involves excretion or exhalation of waste outputs, consisting of degraded, high-entropy materials. There is a compelling analogy between biological organisms and industrial activities—indeed, the whole economic system—not only because both are materials-processing systems driven by a flow of free energy (Georgescu-Roegen, 1971), but because both are examples of self-organizing ''dissipative systems" in a stable State, far from thermodynamic equilibrium (Ayres, 1988).
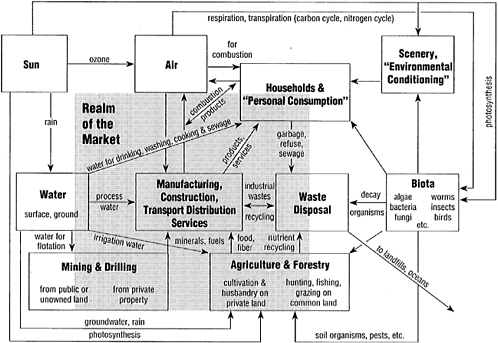
At the most abstract level of description, then, the metabolism of industry is the whole integrated collection of physical processes that convert raw materials and energy, plus labor, into finished products and wastes in a (more or less) steady-state condition (Figure 1). The production (supply) side, by itself, is not self-regulating. The stabilizing controls of the system are provided by its human component. This human role has two aspects: (1) direct, as labor input, and (2) indirect, as consumer of output (i.e., determinant of final demand). The system is stabilized, at least in its decentralized competitive market form, by balancing sup
NOTE: This paper is reprinted with minor editorial changes from Industrial Metabolism, R. Ayres and U. Simonis, eds, United Nations University Press, Tokyo, Japan, 1993, all rights reserved.
ply of and demand for both products and labor through the price mechanism. Thus, the economic system is, in essence, the metabolic regulatory mechanism.
Industrial metabolism can be identified and described at a number of levels below the broadest and most encompassing global one. Thus, the concept is obviously applicable to nations or regions, especially "natural" regions such as watersheds or islands. The key to regional analysis is the existence of a well-defined geographical border or boundary across which physical flows of materials and energy can be monitored.
The concept of industrial metabolism is equally applicable to another kind of self-organizing entity, a manufacturing enterprise or firm. A firm is the economic analogue of a living organism in biology. 1 Some of the differences are interesting, however. In the first place, biological organisms reproduce themselves; firms produce products or services, not other firms (except by accident). In the second place, firms need not be specialized and can change from one product or business to another. By contrast, organisms are highly specialized and cannot change their behavior except over a long (evolutionary) time period. In fact, the firm (rather than the individual) is generally regarded as the standard unit of analysis in economics. The economic system as a whole is essentially a collection of firms, together with regulatory institutions and worker-consumers, using a common currency and governed by a common political structure. A manufacturing firm converts material inputs, including fuels or electric energy, into marketable products and waste materials. It keeps financial accounts for all its external transactions; it is also relatively easy to track physical stocks and flows across the "boundary" of the firm and even between its divisions.
THE MATERIALS CYCLE
A third way in which the analogy between biological metabolism and industrial metabolism is useful is to focus attention on the "life cycle" of individual "nutrients." The hydrological cycle, the carbon cycle, and the nitrogen cycle are familiar concepts to earth scientists. The major way in which the industrial metabolic system differs from the natural metabolism of the earth is that the natural cycles (of water, carbon/oxygen, nitrogen, sulfur, etc.) are closed, whereas the industrial cycles are open. In other words, the industrial system does not generally recycle its nutrients. Rather, the industrial system starts with high-quality materials (fossil fuels, ores) extracted from the earth, and returns them to nature in degraded form.
This point particularly deserves clarification. The materials cycle, in general, can be visualized in terms of a system of compartments containing stocks of one or more nutrients, linked by certain flows . For instance, in the case of the hydrological cycle, the glaciers, the oceans, the freshwater lakes, and the groundwater are stocks, while rainfall and rivers are flows. A system is closed if there are no
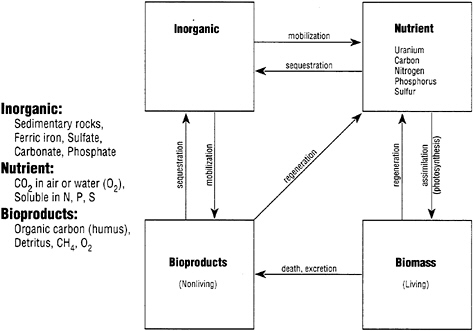
FIGURE 2
Box scheme for bio-geo-chemical cycles.
external sources or sinks. In this sense, the Earth as a whole is essentially a closed system, except for the occasional meteorite.
A closed system becomes a closed cycle if the system is also in steady-state, that is if the stocks in each compartment are constant and unchanging, at least on the average. The materials balance condition implies that the material inputs to each compartment must be exactly balanced (on the average) by the outputs. If this condition is not met, for a given compartment, then the stock in one or more compartments must be increasing, while the stocks in one or more other compartments must be decreasing.2
It is easy to see that a closed cycle of flows, in the above sense, can be sustained indefinitely only by a continuous flow of free energy. This follows immediately from the second law of thermodynamics, which states that global entropy increases in every irreversible process. Thus, a closed cycle of flows can be sustained as long as its external energy supply lasts. An open system, on the contrary, is inherently unstable and unsustainable. It must either stabilize or collapse to a thermal equilibrium state in which all flows, that is, all physical and biological processes, cease.
It is sometimes convenient to define a generalized four-box model to describe materials flows. The biological version is shown in Figure 2, while the analogous industrial version is shown in Figure 3. To revert to the point made at the beginning of this section, the natural system is characterized by closed cycles, at least
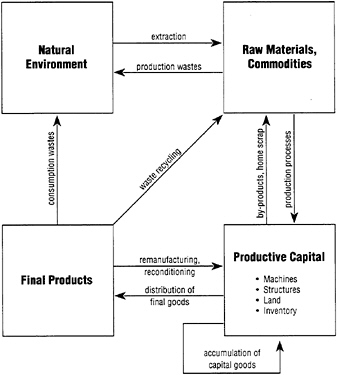
FIGURE 3
Box scheme for industrial material cycles.
for the major nutrients (carbon, oxygen, nitrogen, sulfur)—in which biological processes play a major role in closing the Cycle. By contrast, the industrial system is an open one in which "nutrients" are transformed into "wastes" but are not · significantly recycled. The industrial system of today is therefore unsustainable.
At this stage, it should be noted that nothing can be said about open cycles (on the basis of such simple thermodynamic arguments, at least) with respect to any of the really critical questions. These are as follows: (1) Will the industrial system stabilize itself without external interference? (2) If so, how soon, and in what configuration? (3) If not, does there exist any stable state (i.e., a system of closed materials cycles) short of ultimate thermodynamic equilibrium that could be reached with the help of a feasible technological "fix"? (4) If so, what is the nature of the fix, and how costly will it be? (5) If not, how long do we have until the irreversible collapse of the bio-geosphere system makes Earth uninhabitable? (If the time scale is a billion years, we need not be too concerned. If it is a hundred years, civilization, and even the human race, could already be in deep trouble.) It is fairly important to try to find answers to these questions. Needless to say, we do not aspire to answer all these questions in the present paper.
It should also be pointed out that the bio-geosphere was not always a stable
system of closed cycles. Far from it. The earliest living cells on Earth obtained their nutrients, by fermentation, from nonliving organic molecules whose origin is still not completely understood. At that time the atmosphere contained no free oxygen or nitrogen; it probably consisted mostly of water vapor plus some hydrogen and hydrogen-rich gases such as methane, hydrogen sulfide, and ammonia. The fermentation process yields ethanol and carbon dioxide. The system could have continued only until the fermentation organisms used up the original stock of "food" molecules or choked on the carbon dioxide buildup. The system stabilized temporarily with the appearance of a new organism (blue-green algae, or cyanobacteria) capable of recycling carbon dioxide into sugars and cellulose, thus again closing the carbon cycle. This new process was anaerobic photosynthesis.
However, the photosynthesis process also had a waste product: namely, oxygen. For a long time (over a billion years) the oxygen generated by anaerobic photosynthesis was captured by dissolved ferrous iron molecules, and sequestered as insoluble ferric oxide or magnetite, with the help of another primitive organism, the stromatolites. The resulting insoluble iron oxide was precipitated on the ocean bottoms. 3 (The result is the large deposits of high-grade iron ore we exploit today.) The system was still unstable at this point. It was only the evolutionary invention of two more biological processes, aerobic respiration and aerobic photosynthesis, that closed the oxygen cycle as well. Still other biological processes—nitrification and denitrification, for instance—had to appear to close the nitrogen cycle and others.
Evidently biological evolution responded to inherently unstable situations (open cycles) by "inventing" new processes (organisms) to stabilize the system by closing the cycles. This self-organizing capability is the essence of what has been called Gaia. However, the instabilities in question were slow to develop, and the evolutionary responses were also slow to evolve. It took several billion years before the biosphere reached its present degree of stability.
In the case of the industrial system, the time scales have been drastically shortened. Human activity already dominates natural processes in many respects. While cumulative anthropogenic changes to most natural nutrient stocks still remain fairly small, in most cases,4 the rate of nutrient mobilization by human industrial activity is already comparable to the natural rate in many cases. Table 1 shows the natural and anthropogenic mobilization (flow) rates for the four major biological nutrients, carbon, nitrogen, phosphorus, and sulfur. In all cases, with the possible exception of nitrogen, the anthropogenic contributions exceed the natural flows by a considerable margin. The same is true for most of the toxic heavy metals, shown in Table 2.
Based on relatively crude materials cycle analyses, at least, it would appear that industrialization has already drastically disturbed, and therefore destabilized, the natural system.
TABLE 1 Anthropogenic Nutrient Fluxes, Teragrams/year (Tg/yr)
|
|
Carbon |
Nitrogen |
Sulfur |
Phosphorus |
||||
|
|
Tg/yr |
% |
Tg/yr |
% |
Tg/yr |
% |
Tg/yr |
% |
|
To atmosphere, total |
7,900 |
4 |
55.0 |
12.5 |
93 |
65.5 |
1.5 |
12.5 |
|
Fossil fuel combustion and smelting |
6,400 |
|
45.0 |
|
92 |
|
|
|
|
Land clearing, deforestation |
1,500 |
|
2.6 |
|
1 |
|
1.5 |
|
|
Fertilizer volatilizationa |
|
|
7.5 |
|
|
|
|
|
|
To soil, total |
|
|
112.5 |
21 |
73.3 |
23.4 |
15 |
7.4 |
|
Fertilization |
|
|
67.5 |
|
4.0 |
|
15 |
|
|
Waste disposalb |
|
|
5.0 |
|
21.0 |
|
|
|
|
Anthropogenic acid deposition |
|
|
30.0 |
|
48.3 |
|
|
|
|
Anthropogenic (NH3, NH4) deposition |
|
|
10.0 |
|
|
|
|
|
|
To rivers and oceans, total |
|
|
72.5 |
25 |
52.5 |
21 |
5 |
10.3 |
|
Anthropogenic acid deposition |
|
|
55.0 |
|
22.5 |
|
|
|
|
Waste disposal |
|
|
17.5 |
|
30.0 |
|
5 |
|
|
a Assuming 10% loss of synthetic ammonia-based fertilizers applied to land surface (75 Tg/yr). b Total production (= use) less fertilizer use, allocated to landfill. The remainder is assumed to be disposed of via waterways. |
||||||||
TABLE 2 Worldwide Atmospheric Emissions of Trace Metals (1,000 tonnes per year)
MEASURES OF INDUSTRIAL METABOLISM
There are only two possible long-run fates for waste materials: recycling and reuse or dissipative loss.5 (This is a straightforward implication of the law of conservation of mass.) The more materials are recycled, the less will be dissipated into the environment, and vice versa. Dissipative losses must be made up by replacement from virgin sources. A strong implication of the analysis sketched above is that a long-term (sustainable) steady-state industrial economy would necessarily be characterized by near-total recycling of intrinsically toxic or hazardous materials, as well as a significant degree of recycling of plastics, paper, and other materials whose disposal constitutes an environmental problem. Heavy metals are among the materials that would have to be almost totally recycled to satisfy the sustainability criterion. The fraction of current metal supply needed to replace dissipative losses (i.e., production from virgin ores needed to maintain a stable level of consumption) is thus a useful surrogate measure of "distance" from a steady-state condition, that is, a condition of long-run sustainability.
Most economic analysis in regard to materials, in the past, has focused on availability. Data on several categories of reserves (economically recoverable, potential, etc.) are routinely gathered and published by the U.S. Bureau of Mines, for example. However, as is well known, such figures are a poor proxy for actual reserves. In most cases the actual reserves are much greater than the amounts actually documented. The reason, simply, is that most such data are extrapolated from test borings by mining or drilling firms. There is a well-documented tendency for firms to stop searching for new ore bodies when their existing reserves exceed 20 to 25 years' supply. Even in the case of petroleum (which has been the subject of worldwide searches for many decades) it is not possible to place much reliance on published data of this kind.6
However, a sustainable steady state, as discussed above, is less a question of resource availability than of recycling or reuse efficiency. As commented earlier, a good measure of unsustainability is dissipative use. This raises the distinction between inherently dissipative uses and uses for which the material could be recycled or reused, in principle, but is not. The latter could be termed potentially recyclable. Thus, there are really three important classes of materials use: (1) uses that are economically and technologically compatible with recycling under present prices and regulations; (2) uses that are not economically compatible with recycling but where recycling is technically feasible, for example, if the collection problem were solved; and (3) uses for which recycling is inherently not feasible. Admittedly there is some fuzziness in these classifications, but it should be possible for a group of international experts to arrive at some reconciliation.
Generally speaking, it is arguable that most structural metals and industrial catalysts are in the first category; other structural and packaging materials, as well as most refrigerants and solvents, fall into the second category. This leaves coatings, pigments, pesticides, herbicides, germicides, preservatives, flocculants, anti-
freezes, explosives, propellants, fire retardants, reagents, detergents, fertilizers, fuels, lubricants, and the like in the third category. In fact, it is easy to verify that most chemical products belong in the third category, except those physically embodied in plastics, synthetic rubber, or synthetic fibers.
From the standpoint of elements, if one traces the uses of materials from source to final sink, it can be seen that virtually all sulfur mined (or recovered from oil, gas, or metallurgical refineries) is ultimately dissipated in use (e.g., as fertilizers or pigments) or discarded, as waste acid or as ferric or calcium sulfites or sulfates. (Some of these sulfate wastes are classed as hazardous). Sulfur is mostly (75-80 percent) used, in the first place, to produce sulfuric acid, which in turn is used for many purposes. But in every chemical reaction the sulfur must be accounted for—it must go somewhere. The laws of chemistry guarantee that reactions will tend to continue either until the most stable possible compound is formed or until an insoluble solid is formed. If the sulfur is not embodied in a "useful" product, it must end up in a waste stream.
There is only one long-lived structural material embodying sulfur: plaster-of-Paris (hydrated calcium sulfate) which is normally made directly from the natural mineral gypsum. In recent years, sulfur recovered from coal-burning power plants in Germany has been converted into synthetic gypsum and used for construction. However, this potential recycling loop is currently inhibited by the very low price
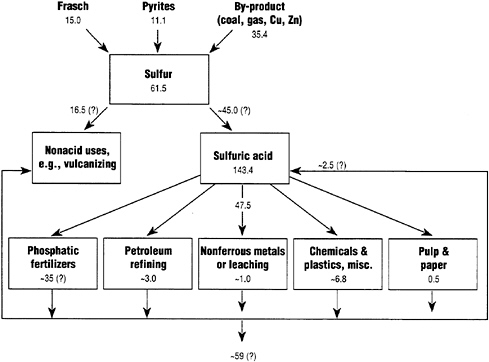
FIGURE 4
Dissipative uses of sulfur, 1988 (millions of metric tons).
TABLE 3 Examples of Dissipative Use
|
Substance |
106 T |
Dissipative Uses |
|
Other chemicals |
|
|
|
Chlorine |
25.9 |
Acid, bleach, water treatment, PVC solvents, pesticides, refrigerants |
|
Sulfur |
61.5 |
Acid (H2SO4), bleach, chemicals, fertilizers, rubber |
|
Ammonia |
24.0 |
Fertilizers, detergents, chemicals |
|
Phosphoric acid |
93.6 |
Fertilizers, nitric acid, chemicals (nylon, acrylics) |
|
NaOH |
35.8 |
Bleach, soap, chemicals |
|
Na2CO3 |
29.9 |
Chemicals (glass) |
|
Heavy metals |
|
|
|
Copper sulfate (CuSO4*5H2O) |
0. 10 |
Fungicide, algicide, wood preservative, catalyst |
|
Sodium bichromate |
0.26 |
Chromic acid (for plating), tanning, algicide |
|
Lead Oxides |
0.24 |
Pigment (glass) |
|
Lithopone (ZuS) |
0.46 |
Pigment |
|
Zinc Oxides |
0.42 |
Pigment (tires) |
|
Titanium Oxide (TiO2) |
1.90 |
Pigment |
|
Tetraethyl lead |
? |
Gasoline additive |
|
Arsenic |
? |
Wood preservative, herbicide |
|
Mercury |
? |
Fungicide, catalyst |
of natural gypsum. Apart from synthetic gypsum, there are no other durable materials in which sulfur is physically embodied. It follows from materials balance considerations that sulfur is entirely dissipated into the environment. Globally, about 61.5 million metric tons of sulfur as sulfur—not including gypsum—were produced in 1988. Of this, less than 2 million tons were recycled (mainly as waste sulfuric acid), as indicated schematically in Figure 4.
Very little is currently used for structural materials. Thus, most sulfur chemicals belong in class 3. Following similar logic, it is easy to see that the same is true of most chemicals derived from ammonia (fertilizers, explosives, acrylic fibers), and phosphorus (fertilizers, pesticides, detergents, fire retardants). In the case of chlorine, there is a division between class 2 (solvents, plastics, etc.) and class 3 (hydrochloric acid, chlorine used in water treatment, etc.).
Chlorofluorocarbon (CFC) refrigerants and solvents are long-lived and non-reactive. In fact, this is the reason they pose an environmental hazard. Given an appropriate system for recovering and reconditioning old refrigerators and air conditioners, the bulk of the refrigerants now in use could be recovered, for either reuse or destruction. Hence, they belong in class 2. However CFCs used for foam-blowing are not recoverable. Table 3 shows world output of a number of materials—mostly chemicals—whose uses are, for the most part, inherently dissipative (class 3). It would be possible, with some research, to devise measures of the inherently dissipative uses of each element, along the lines sketched above.
TABLE 4 Scrap Use in the United States
|
|
Total Consumption(million short tons) |
% of Total Consumption in Recycled Scrap |
||||
|
Material |
1977 |
1982 |
1987 |
1977 |
1982 |
1987 |
|
Aluminum |
6.49 |
5.94 |
6.90 |
24.1 |
33.3 |
29.6 |
|
Copper |
2.95 |
2.64 |
3.15 |
39.2 |
48.0 |
39.9 |
|
Lead |
1.58 |
1.22 |
1.27 |
44.4 |
47.0 |
54.6 |
|
Nickel |
0.75 |
0.89 |
1.42 |
55.9 |
45.4 |
45.4 |
|
Iron/steel |
142.40 |
84.00 |
99.50 |
29.4 |
33.4 |
46.5 |
|
Zinc |
1.10 |
0.78 |
1.05 |
20.9 |
24.1 |
17.7 |
|
Paper |
60.00 |
61.00 |
76.20 |
24.3 |
24.5 |
25.8 |
|
SOURCE: Institute of Scrap Recycling Industries (1988). |
||||||
Sustainability, in the long run, would imply that such measures decline. Currently, they are almost certainly increasing.
With regard to materials that are potentially recyclable (classes 1 and 2), the fraction actually recycled is a useful measure of the approach toward (or away from) sustainability. A reasonable proxy for this, in the case of metals, is the ratio of secondary supply to total supply of final materials: see, for example, Table 4. This table shows, incidentally, that the recycling ratio in the United States has been rising consistently in recent years only for lead and iron or steel. For lead, the ban on using tetraethyl lead as a gasoline additive (an inherently dissipative use) is entirely responsible.
Another useful measure of industrial metabolic efficiency is the economic output per unit of material input. This measure can be called materials productivity. It can be measured, in principle, not only for the economy as a whole, but for each sector. It can also be measured for each major ''nutrient" element, such as carbon, oxygen, hydrogen, sulfur, chlorine, iron, and phosphorus. Measures of this kind for the economy as a whole are not reliable indicators of increasing technological efficiency, or Progress toward long-term sustainability. The reason is that increasing efficiency—especially in rapidly developing countries—can be masked by structural changes, such as investment in heavy industry, which tend to increase the materials (and energy) intensiveness of economic activity. On the other hand, within a given sector, one would expect the efficiency of materials use—or materials productivity—to increase, in general.7
Useful aggregate measures of the state of the environment in relation to sustainability can be constructed from physical data that are already collected and compiled in many countries. To derive these aggregates and publish them regularly would provide policymakers with a valuable set of indicators at little cost.
It is clear that other interesting and useful measures based on physical data are also possible. Moreover, if similar data were collected and published at the
sectoral level, it would be possible to undertake more ambitious engineering-economic systems analyses and forecasts—of the kind currently possible only for energy—in the entire domain of industrial metabolism.
POLICY IMPLICATIONS OF THE INDUSTRIAL METABOLISM PERSPECTIVE
It may seem odd to suggest that a mere viewpoint—in contradistinction to empirical analysis—may have policy implications. But it is perfectly possible. In fact, there are two implications that come to mind. First, the industrial metabolism perspective is essentially "holistic" in that the whole range of interactions between energy, materials, and the environment is considered together—at least, in principle. The second major implication, which virtually follows from the first, is that from this holistic perspective it is much easier to see that narrowly conceived or short-run (myopic) "quick fix" policies may be far from globally optimum. In fact, from the larger perspective, such policies can be harmful.
The best way to explain the virtues of a holistic view is by contrasting it with narrower perspectives. Consider the problem of waste disposal. It is a consequence of the law of conservation of mass that the total quantity of materials extracted from the environment will ultimately return thence as some sort of waste residuals or "garbo-junk" (Ayres and Kneese, 1969, 1989). Yet environmental protection policy has systematically ignored this fundamental reality by imposing regulations on emissions by medium. Typically, one legislative act mandates a bureaucracy that formulates and enforces a set of regulations dealing with emissions by "point sources" only to the air. Another act creates a bureaucracy that deals only with waterborne emissions, again by "point sources." And so forth.
Not surprisingly, one of the things that happened as a result was that some air pollution (e.g., fly ash and SOx from fossil fuel combustion) was eliminated by converting it to another form of waste, such as a sludge to be disposed of on land. Similarly, some forms of waterborne wastes are captured and converted to sludges for land disposal (or, even, for incineration). Air and water pollution were reduced, but largely by resorting to land disposal. But landfills also cause water pollution (leachate), and air pollution, due to anaerobic decay processes. In short, narrowly conceived environmental policies over the past 20 years and more have largely shifted waste emissions from one form (and medium) to another, without significantly reducing the totals. In some cases, policy has encouraged changes that merely dilute the waste stream without touching its volume at all. The use of high stacks for coal-burning power plants, and the building of longer sewage pipes to carry wastes farther offshore exemplify this approach.
To be sure, these shifts may have been beneficial in the aggregate. But the costs have been quite large, and it is only too obvious that the state of the environment "in the large" is still deteriorating rapidly. One is tempted to think that a
more holistic approach, from the beginning, might have achieved considerably more at considerably less cost.
In fact, there is a tendency for suboptimal choices to get "locked in" by widespread adoption. Large investments in so-called clean coal technology would surely extend the use of coal as a fuel—an eventuality highly desired by the energy establishment—but would also guarantee that larger cumulative quantities of sulfur, fly ash (with associated toxic heavy metals), and carbon dioxide would be produced. The adoption of catalytic converters for automotive engine exhaust is another case in point. This technology is surely not the final answer, since it is not effective in older vehicles. Yet it has deferred the day when internal combustion engines will eventually be replaced by some inherently cleaner automotive propulsion technology. By the time that day comes, the world's automotive fleet will be two or three times bigger than it might have been otherwise, and the cost of substitution will be enormously greater.
The implication of all these points for policymakers, of course, is that the traditional governmental division of responsibility into a large number of independent bureaucratic fiefdoms is dangerously faulty.8 Yet the way out of this organizational impasse is far from clear. Top-down central planning has failed miserably and is unlikely to be tried again soon. On the other hand, pure "market" solutions to environmental problems are limited in cases where there is no convenient mechanism for valuation of environmental resource assets (such as beautiful scenery) or functions (such as the ultraviolet radiation protection afforded by the stratospheric ozone layer). This is primarily a problem of indivisibility. Indivisibility means that there is no possibility of subdividing the attribute into "parcels" suitable for physical exchange. In some cases this problem can be finessed by creating exchangeable "rights" or "permits," but the creation of a market for such instruments depends on other factors, including the existence of an effective mechanism for allocating such rights, limiting their number, and preventing poaching or illicit use of the resource.
SUMMARY
Needless to say, the policy problems have economic and sociopolitical ramifications well beyond the scope of this paper. However, as the Chinese proverb has it, the longest journey begins with a single step. Developing industrial metabolism as an analytic tool certainly represents one critical step in understanding industrial ecology systems and effecting change toward sustainability.
NOTES
REFERENCE
Ayres, Robert U. 1988. Self organization in biology and economics. International Journal on the Unity of the Sciences 1(3)(Fall) [also IIASA Research Report #RR-88-1, 1988].
Ayres, Robert U., and Allen V. Kneese. 1969. Production, Consumption and Externalities. American Economic Review, June [Reprinted in Benchmark Papers in Electrical Engineering and Computer Science, Daltz and Pentell, eds., Dowden, Hutchison and Ross, Stroudsberg 1974 and Bobbs-Merrill Reprint Series, N.Y.].
Ayres, Robert U., and Allen V. Kneese. 1989. Externalities: Economics and thermodynamics. In Economy and Ecology: Towards Sustainable Development, Archibugi and Nijkamp, eds. Netherlands: Kluwer Academic Publishers.
Georgescu-Roegen, Nicholas. 1971. The Entropy Law and the Economic Process. Cambridge, Mass.: Harvard University Press.
Institute of Scrap Recycling Industries. 1988. Facts—1987 Yearbook. Washington, D.C.: Institute of Scrap Recycling Industries.
Nriagu, J. O. 1990. Global metal pollution. Environment 32(7):7-32.
Rogner, Hans-Holger. 1987. Energy in the world: The present situation and future options. In Proceedings of the 17th International Congress of Refrigeration, August 24-28, 1987.