4
EFFECT SEVERITY
THIS chapter presents the subcommittee's evaluation of the definition of severity categories for LATRA. In the first section of this chapter, the subcommittee reviews several precedents for severity descriptors, including the concept of severity as used by toxicologists, and the severity descriptors used by the U.S. Environmental Protection Agency (EPA) and the American Thoracic Society (ATS). The relationship between severity and sensitivity also is discussed. In the second section of this chapter, the subcommittee evaluates the LATRA model severity categories in light of those precedents. The last section provides the subcommittee's conclusions and recommendations on how severity categories can be used and how to define mild, moderate, and severe effects.
PRECEDENT FOR SEVERITY DESCRIPTORS
The concept of severity of effect is commonly used in toxicology and other health sciences. Sometimes, ratings, such as mild, moderate, and severe, are used to describe the severity of a particular outcome of exposure. In other instances, the term "adverse" is used to distinguish between outcomes that are detrimental to an organism and outcomes that are temporary physiological responses with no detrimental impact. The reversibility of an outcome is often related to severity; generally, an outcome is considered more severe if it is irreversible. The remainder of this section describes how toxicologists, EPA, and the American Thoracic Society, have used severity descriptors.
SEVERITY OF EFFECT IN TOXICOLOGY
In toxicology, two types of dose-response curves are used (Eaton and Klaassen 1996). One type relates dose to the incidence of an effect in a group of subjects. For each subject in the group, the effect either occurs or does not occur (binary response). The second type of dose-response curve relates dose to the severity of response of an individual subject. The severity of response of a subject is "graded" according to objective or subjective criteria. Objective severity descriptors can be obtained by direct measurement (e.g., degree of cholinesterase inhibition by organophosphates). Subjective grading can be performed by an investigator, or in human experimentation, the subject might be asked to judge the severity of response. Examples of responses that often are subjectively graded are erythema and eye irritation. Toxicologists generally are referring to the second type of dose-response curve when discussing severity of effect.
Figure 4-1 illustrates the two types of dose-response curves and shows how more-and less-sensitive individuals are depicted in each type. To incorporate severity into an incidence dose-response curve, health effects would need to be categorized into discrete categories so that an individual could be classified as either showing that severity of effect or not. Figure 4-1A depicts that approach using three severity levels for coughing: mild, severe, and incapacitating. The proportion of individuals experiencing a given severity of coughing is plotted against dose. More-sensitive individuals are at the lower-left end of the curve for a specified severity of coughing, and less-sensitive individuals at the upper-right end of the curve. The curves are likely to be overlapping; some of the more-sensitive individuals might experience severe coughing at doses that only produce mild coughing in others.
For a severity dose-response curve (Figure 4-1B), severity of response is represented as a continuum. In this case, more-sensitive individuals are represented by curves starting at lower exposure concentrations, and the population is represented by a series of individual severity dose-response curves. The individual curves might be parallel (i.e., equal slopes), or the slope of the dose-response curves might be more steep for more-sensitive individuals. In either case, the severity of response in a sensitive individual would be greater than the severity of response in a normal individual at a given dose, as was shown for SO2 in Chapter 3. The proportion of the population experiencing a given
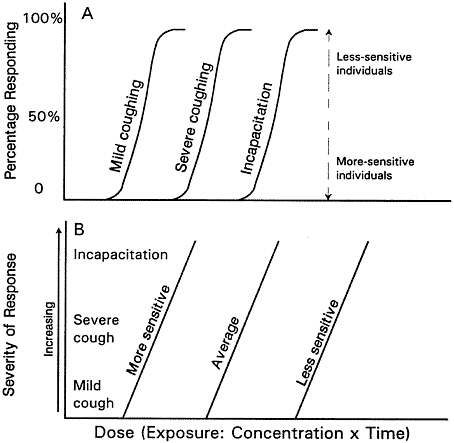
FIGURE 4-1 (A) Incidence dose-response curve. (B) Severity dose-response curve.
severity of effect at a given dose could be determined by identifying the severity level on the y-axis, the dose on the x-axis, and determining the proportion of the total number of curves representing individuals in the population that fall to the left of that point, assuming such data were available. It is more likely that a relationship between severity of response and dose would be established from a sample of individuals by regression analysis (severity of response against dose). The proportion of the population responding would be reflected in the variance of the response around the mean response predicted by the regression line. Estimating the proportion responding from that model is awkward, however, and in general, toxicologists do not use severity dose-response curves to estimate the incidence of effects.
EPA DEFINITION OF ADVERSE EFFECTS
Describing exposure effects as adverse or not adverse is a discrete severity scale for rating outcomes according to their degree of interference with an organism's normal functioning. Although carefully defining the criteria for classifying the effects of exposure to toxic substances is desirable, such severity descriptors often are loosely applied. For instance, the Clean Air Act of 1970, Section 108A, uses the term "adverse health effect" when referring to the consequences of air-pollution exposure without defining the term "adverse." To clarify this issue, EPA proposed that eye, nose, and throat irritation associated with urban smog or photochemical oxidant exposure are not medically important and, thus, should not be considered an adverse health effect (see ATS 1985). EPA developed a spectrum of biological responses ranging from the trivial to the fatal as follows: pollutant burdens, physiological changes of uncertain significance, pathophysiological changes, morbidity, and, finally, mortality. EPA considered the last three of those five categories to represent adverse responses (see ATS 1985).
AMERICAN THORACIC SOCIETY DEFINITION OF ADVERSE EFFECTS
To provide guidance in interpreting the epidemiological literature, the American Thoracic Society (ATS) defined adverse respiratory health effects as "medically significant physiologic and pathologic changes generally evidenced by one or more of the following: (1) interference with normal activity of the affected person or persons, (2) episodic respiratory illness, (3) incapacitating respiratory injury, and/or (4) progressive respiratory dysfunction" (ATS 1985). ATS also listed respiratory health effects from most severe to least severe, as shown in Appendix C.
Several aspects of the ATS discussion are particularly relevant to brief exposures, such as those possibly encountered from release of rocket propellants. First, the ATS considers certain reversible effects to be adverse. For instance, ATS considers asthmatic attacks to be adverse because they interfere with a person's normal activities, but ATS does not consider a small, brief reduction in pulmonary function to be adverse. Some measurable changes in pulmonary function due to exposure might be so slight that they are not noticeable, even for those with asthma, and ATS considers those changes to be medically insignificant.
Coughing or excess phlegm production are considered generally annoying and would be considered adverse effects only in extreme cases.
LATRA'S USE OF THE TERM ''SEVERITY"
In keeping with risk estimates for other types of launch hazards of concern to the Air Force (e.g., explosions with flying debris), LATRA is designed to predict the incidence of effects, i.e., how many people would suffer an effect. To provide information to the wing commander on the seriousness of effects, the LATRA model estimates the incidence of at least mild effects and at least serious effects separately. (The definitions of mild and serious effects for LATRA are presented in Chapter 2.) Thus, in LATRA, severity of effect is incorporated in discrete categories (similar to the paradigm depicted in Figure 4-1A) rather than in a continuum from mild to severe as might be expressed by a single individual (as depicted in Figure 4-1B). In that way, the number of people likely to suffer at least a mild effect and the number of people likely to suffer at least a severe effect can be estimated.
This use of severity descriptors is fundamentally different from those used by toxicologists. In toxicology, the severity of a subject's response is graded for a single health effect (e.g., mild-to-severe coughing; see Figure 4-lB). In the LATRA paradigm, diverse potential health end points (e.g., tearing, coughing, or bronchospasm) are identified with specific severity categories. To link specific health effects with specific severity categories, the Air Force made value judgments to determine the relative significance of various health effects, but the rationale for those judgments has not been documented.
In evaluating the Air Force's use of severity descriptors, the sub-committee considered the relation between the definition of the signs and symptoms of mild and serious effects (see Chapter 2) and ATS's definition and examples of adverse effects (Appendix C). The subcommittee noted a few inconsistencies. For example, some of the responses categorized by the Air Force as severe (e.g., coughing and excess sputum) are categorized as adverse by ATS only if they are extreme. Also, many of the responses categorized as mild for LATRA are considered adverse by ATS if they either interfere with normal activities or require medical attention. Those inconsistencies point out that a specific sign, symptom, or effect by itself is not a good indicator of severity unless
qualified by phrases such as "interferes with normal activities" or "requires medical attention."
CONCLUSIONS AND RECOMMENDATIONS
The subcommittee agrees that categorizing health effects into severity categories is possible and offers several advantages. The subcommittee recommends, however, a somewhat different approach to categorize effect severity than the one currently used in LATRA.
In the LATRA-ERF model, all relevant health end points are categorized as either mild or severe and grouped accordingly. That approach provides several advantages for risk communication. Those who must ultimately use the results of LATRA are not health professionals. Thus, the use of severity categories translates the risks of specific outcomes (e.g., bronchoconstriction) to more easily understood response levels (e.g., mild response). In addition, the aggregation of data for many different health end points into a few severity categories increases the amount of data available to derive each ERF relative to the amount of data available to derive an ERF for a single end point (e.g., coughing). (How the ERFs are developed for each severity category is evaluated in Chapter 5.)
The subcommittee believes, however, that effect-severity descriptors are best defined by (1) the impact on a person's ability to perform normal activities, (2) the impact on organ function, (3) the need for medical attention, and (4) the reversibility of the effect. Although some health end points are restricted to one severity category, they more often can span all severity categories. For instance, bronchoconstriction can range from mild to severe depending on its intensity. The term "adverse" has been used to describe the severity of air-pollution effects by EPA and ATS. The subcommittee has chosen not to use that descriptor because its meaning is based on the more-fundamental effect characteristics listed above. The subcommittee recommends the following broad definitions for severity of effect for use by the Air Force. To respond to the U.S. Air Force's request for a "moderate" category in between mild and serious, the subcommittee defines three severity categories as follows. Mild effects are reversible within 48 hr and do not interfere with normal ac-
tivity or require medical attention. Moderate effects are irreversible effects that do not alter organ function or interfere with normal activity, or they are reversible effects that alter organ function or interfere with normal activity. Persons experiencing moderate effects might seek medical attention. Severe effects are irreversible effects that alter organ function or interfere with normal activities. Severe effects usually require medical attention. Those definitions were developed specifically for exposures to rocket emissions and might not be applicable to other exposure scenarios or toxicants.
Table 4-1 attempts to classify the signs, symptoms, and effects of exposure of humans to rocket-emission toxicants as found in the literature into the three severity categories. Severity categories were checked if the subcommittee determined that a sign, symptom, or effect could conform to the definition of that category presented above. It is clear that many signs, symptoms, and effects cover more than one severity category and, thus, do not uniquely define the severity of an exposure outcome. Judging severity must be recognized as fundamentally a subjective process in which the values of the evaluator play a key role. Other stakeholder groups (e.g., persons living near launch areas, Air Force personnel, or individuals with asthma) might categorize these end points differently. Further difficulties with developing ERFs for severity categories are described in the next chapter. The subcommittee suggests that the Air Force be especially aware to avoid making certain value judgments based on an incomplete or limited data base. Such limitations make it difficult to evaluate or predict accurately the degree to which a specific human subpopulation might be more sensitive to air contaminants than others.
Finally, the subcommittee notes that variation in the sensitivity of individuals to toxic substances generally is accounted for in dose-response curves of the type illustrated in Figure 4-1A rather than by separate dose-response curves for more-and less-sensitive groups. That issue also is described in more detail in the next chapter.
TABLE 4-1 Classification of Potential Signs, Symptoms, and Effects of Human Exposure to Rocket-Emission Toxicants
|
Outcome |
Mild |
Moderate |
Severe |
|
Perception of odor or taste |
√ |
|
|
|
Headache |
Nonrecurring |
Increased frequency |
Incapacitating |
|
Eye irritation |
Discomfort |
Burning sensation |
|
|
Tearing (by itself) |
√ |
|
|
|
Corneal Opacity |
|
|
√ |
|
Nasal irritation |
Discomfort |
Burning sensation |
|
|
Sneezing |
√ |
|
|
|
Nasal catarrh |
√ |
|
|
|
Rhinorrhea |
√ |
|
|
|
Miosis |
√ |
|
|
|
Cough |
Infrequent |
Frequent |
Incapacitating |
|
Burning throat |
|
|
√ |
|
Spasm of larynx |
|
|
√ |
|
Tightness in chest |
√ |
|
|
|
Increased airway resistance |
√ |
√ |
√ |
|
Increased airway responsiveness |
√ |
√ |
√ |
|
Increased sputum production |
√ |
√ |
|
|
Wheezing, in absence of cold |
|
√ |
√ |
|
Bronchospasm |
|
|
√ |
|
Sloughing of airway lining |
|
√ |
√ |
|
Rhonchi |
|
|
√ |
|
Bronchiolitis |
√ |
√ |
√ |
|
Focal pneumonia |
√ |
√ |
|
|
Diffuse pneumonia |
|
√ |
√ |
|
Chemical pneumonitis |
|
√ |
√ |
|
Chest pain |
|
|
√ |
|
Shortness of breath |
|
√ |
|
|
Difficulty breathing |
|
√ |
√ |
|
Increase in methemoglobin |
√ |
√ |
√ |
|
Increase in blood pressure |
√ |
√ |
√ |
|
Increase in blood glutathione levels |
√ |
|
|
|
Decrease in CO diffusion capacity |
√ |
√ |
√ |
|
Decrease in lung compliance |
√ |
√ |
√ |
|
Decrease in earlobe blood PO2 |
√ |
√ |
√ |
|
Decrease in alveolar O2 partial pressure |
√ |
√ |
√ |
|
Pulmonary edema |
|
√ |
√ |
|
Asthma attack |
√ |
√ |
√ |
|
Disorientation |
|
√ |
√ |
|
Exacerbation of chronic cardiopulmonary disease |
|
|
√ |








