3
Electrolytic Chromium-Metal Production
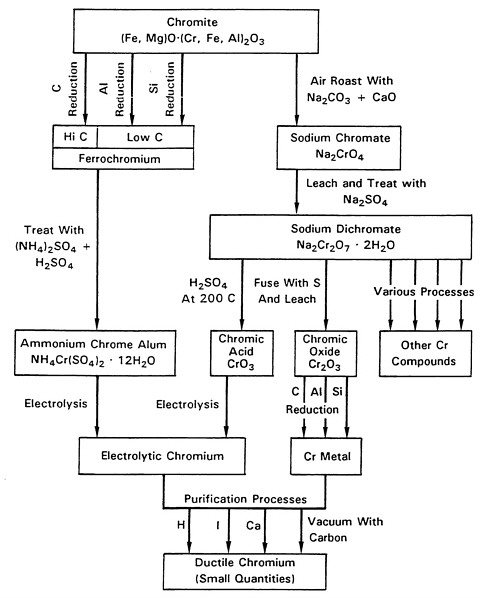
The previous chapter focused on the use of high-purity chromium metal for high-performance alloys in jet-engine applications. Technical emphasis in this chapter focuses on the two main variations of the electrolytic process that have traditionally produced high-purity chromium metal: the trivalent chrome-alum electrolyte method and the chromic-acid electrolyte method. Also discussed are the final vacuum-degassing process that is used to refine electrolytic chromium metal further for aerospace applications and the environmental and occupational health considerations of these methods. Other potential methods for producing high-purity chromium metal are detailed in Chapter 4. An overview of possible treatment schemes available to produce metallic chromium metal is presented in Figure 3-1.
TRIVALENT CHROME-ALUM BATH ELECTROLYTE METHOD
The main electrolytic method used for the production of large tonnages of chromium metal is the chromium (III) or trivalent chrome-alum bath electrolyte method. This process was developed by the U.S. Bureau of Mines in 1950, following a long-term study on chromium-metal electrowinning.
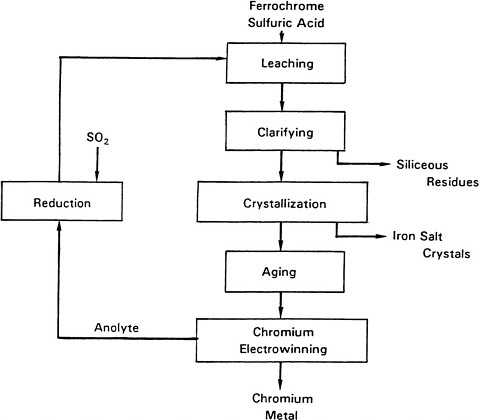
Solutions suitable for producing chromium metal can be derived from ore by oxidative roasting in alkali or dissolution of chromite in sulfuric acid. However, the preferred starting material is milled high-carbon ferrochrome that is leached in recycled anolyte and make-up H2SO4. A simplified flow sheet showing the essential steps in the process is given in Figure 3-2. The 20-mesh ferrochrome (67 percent chromium) is leached at about 90 °C in roughly 3-metric-ton batches without agitation. Approximately 95 percent of the solids dissolve.
The hot-acid leach solution is clarified, and the undissolved solids, such as silica, are separated by filtration. The filtrate is cooled using mother liquor from the ferrous ammonium sulfate circuit and the mixture is conditioned at 80 °C, causing the transformation to the green, non-alum form of chromium. Upon further cooling, the crude iron sulfate crystals form, are separated for further conditioning, and are recrystallized as ferrous ammonium sulfate.
The mother liquor from the crude ferrous sulfate crystallization contains the bulk of the chromium metal that is then sent to the aging circuit. Aging and crystallization are conducted at relatively low temperatures. The kinetics are rather sluggish, but the desired purple ammonium chromium alum eventually forms. The crystal slurry is then filtered, washed, and pumped to the leach circuit. The washed chromium-alum crystals are dissolved in hot water and filtered to produce cell feed.
The key to successful, efficient electrodeposition of metallic chromium is ensuring that the proper chemistry is maintained at the cathode. This requires using a diaphragm cell, which separates the anolyte and catholyte, to minimize the migration of chromium (VI) to the anode and assist in controlling the pH. If chromium (VI) is present, its reduction to Cr+2 is detrimental to current efficiency. If the Ph is too low, hydrogen evolution causes low current efficiency. If the Ph is too high, however, the undesirable precipitation of Cr2O3·xH2O occurs. Thus, control of electrolyte composition and Ph at about 2.1–2.4 is essential. The current efficiencies obtained are usually about 45 percent.
Figure 3-3 shows the desired reactions during electrolysis; these reactions are listed below.
At the cathode:
Cr+3 + 1e− → Cr+2
Cr+2 + 2e− → Cr0
2H+ + 2e− → H2
At the anode:
2H2O → 4H+ + O2 + 4e−
2Cr+3 + 7H2O → Cr2O7−2 + 14H+ + 6e−
The approximate electrolyte composition in both compartments of the diaphragm cell and typical electrode reactions are shown in Figure 3-3. Other operating data are listed in Table 3-1.
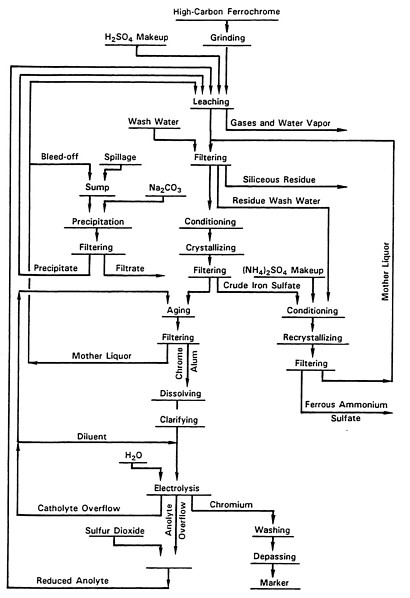
After 72 hours of plating, the brittle chromium metal is removed in pieces from the cathode blanks by hammering. The metal at this point contains a number of impurities and must be refined by vacuum reduction before final use. A more complete flow sheet for producing electrolytic chromium metal by the chrome-alum process is given in Figure 3-4.
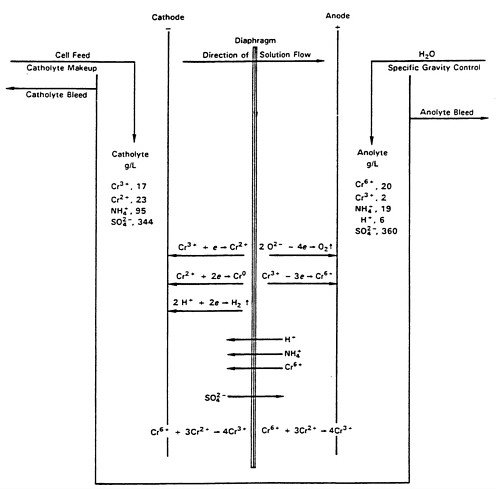
FIGURE 3-3 Principal electrolytic cell reactions in chromium production by the chrome-alum process. Reprinted courtesy of Grayson (1985) and Elkem Metals Company.
CHROMIC-ACID ELECTROLYTE METHOD
Referred to as the chromium (VI) or hexavalent process, the chromic-acid electrolyte method is used primarily for surface finishing and electroplating applications. Chromium trioxide, CrO3, is obtained from sodium dichromate. Small, catalyzing additions of halogens or sulfate are essential for electrolysis. The current efficiency of the process is very low (e.g., in the range of 10 percent) because of the low hydrogen overpotential on chromium. The excessive hydrogen evolution may result in the precipitation of chromium hydroxides
TABLE 3-1 Operating Data for Electrowinning of Chromium Metal from Chrome Alum
|
Cathode Current Density (A/M2) |
753 |
|
Cell Potential (V) |
4.2 |
|
Current Efficiency (%) |
45 |
|
Electrical Consumption (MJ/Kg) |
67 |
|
pH of Catholyte |
2.1 - 2.4 |
|
Catholyte Temperature (°C) |
53 ± 1 |
|
Deposition Time (h) |
72 |
|
Cathode Material |
Type 316 stainless steel |
|
Anode Material (wt %) |
1.99 Ag-Pb |
because of the higher localized Ph at the cathode surface. Passivation of the reactions may then occur, making efficient processing even more difficult. Some of the cell conditions for chromium electrowinning from chromic acid are given in Table 3-2.
The low overall efficiency obtainable with hexavalent chromium makes it nonviable as an alternative method for the production of bulk, high-purity chromium metal for alloys. Thus, this method has not been used when large tonnages are required.
TABLE 3-2 Cell Conditions for the Chromic Acid Process for Electrowinning Chromium Metal
|
Bath Composition (g/L) |
300 CrO3, 4 sulfate ion |
|
Temperature (°C) |
84-87 |
|
Current Density (A/m2) |
9,500 |
|
Current Efficiency (%) |
6-7 |
|
Plating Time (h) |
80-90 |
VACUUM-DEGASSING OF CHROMIUM METAL
The following process is used to produce vacuum-grade chromium metal by the Elkem Metals Company (trade name Elchrome). It is the final refinement step for the production of chromium metal for aerospace applications.
The irregular plates of chromium metal removed from the electrodes are first milled to 100 µm and then blended into a briquetting mixture with finely divided carbon, tin, and a polymeric binder. The quantities of these additives will depend on the composition of the feed chromium metal. This mixture is wetted and formed into small briquets, which are allowed to dry and placed in separate lots on a long flatbed railcar. The railcar is placed in a long, cylindrical, resistance-heated vacuum oven, which is closed and evacuated with a steam extractor. The oven is heated to approximately 1400 °C (2600 °F) at a rate sufficiently slow to accommodate offgassing without excessive increases in pressure. When the maximum temperature is reached, it is held until a constant vacuum of less than 100 µm of mercury can be maintained. It is then allowed to cool while a stream of argon gas is admitted. Final purging is with helium. The briquets are then removed, analyzed, and packaged for shipment in 1-ton boxes.
An entire cycle requires approximately one week. During the process, nitrogen and lead are volatilized, and sulfur is removed as tin sulfide. Oxygen is also removed as carbon dioxide to levels lower than 0.05 percent. The use of carbon as an oxygen getter places lower limits on carbon content. Some chromium metal is volatilized and recondensed on the cooler parts of the oven at the highest temperatures and lowest pressures. Thus there is a tradeoff between the additional purity obtained by continued treatment and the loss of chromium metal. All of the chromium metal produced electrolytically can be refined to aerospace quality by degassing.
ENVIRONMENTAL AND OCCUPATIONAL HEALTH CONSIDERATIONS
Chromium-metal manufacture by any method is energy intensive. Electric power is consumed in both the electrowinning and vacuum-degassing processes. To the extent that this power is generated by the burning of fossil fuels, it contributes a burden to the local environment. In the aluminothermic process, electric power is consumed externally to the process proper (in the smelting of aluminum reductant used in the process) but is nevertheless comparable to the electrolytic process in terms of energy consumption.
The major direct by-products of electrolytic chromium-metal manufacture are ferrous ammonium sulfate and ammonium sulfate, which are formed during ammonia neutralization of the sulfuric acid leachate of ferrochrome. Ferrous ammonium sulfate is subjected to two recrystallization steps to remove as much entrained chromium as possible and then pumped to settling ponds on site for indefinite storage. Excess ammonium sulfate is discharged to the Ohio River under an EPA permit. Currently, research and development efforts are being made at Elkem to develop an alternative method for removal of iron that does not introduce ammonia into the process stream. While the iron removal step is not needed in the aluminothermic process since it employs chromium (III) oxide rather than ferrochrome as the main feedstock, iron removal must be performed at some point in the path between chromite ore and high-purity chromium metal.
In general, chromium-containing solutions that exit the process stream are mixed with the main process stream at the ferrochrome leaching stage, a practice driven largely by the cost of chromium units. These solutions include mother liquors from ferrous ammonium sulfate recrystallization and anolyte containing chromium (VI) from the electrolytic process, reduced to the chromium (III) state with sulfur dioxide before re-addition to the leach. Thus, there are no significant costs associated with disposal of chromium-bearing hazardous wastes from the electrolytic process.
One concern in the chromium process is in the milling, blending, and briquetting operations that occur prior to the vacuum-degassing step. This generates and releases a considerable quantity of very finely divided particulates. This dust is known to constitute an explosion hazard under certain conditions, and safety precautions are observed. These particulates may cause health and environmental problems as well, but no special efforts are apparently being expended to contain this material. Similar considerations apply to the aluminothermic process. Metallic chromium recovered as floor sweepings can be reintroduced into the process stream at the leaching step.
The vacuum-degassing process itself results in production of gaseous stack emissions, largely consisting of carbon monoxide, although these are insignificant in relation to the initial smelting of chromite ore. This process will be performed in a similar manner in both electrolytic and aluminothermic processes.
In conclusion, the production of electrolytic chromium metal as carried out at the Elkem plant in Marietta, Ohio, appears to have few opportunities for major environmental improvements other than the effort to remove ammonia from the process stream. In addition, neither the electrolytic nor the aluminothermic process offers a significant advantage over the other in terms of its environmental impact.