2
Labor and Delivery*
Normal pregnancy is a carefully programmed sequence of events, which can be divided into stages that culminate with labor and delivery (see Figure 2.1). Following fertilization, a series of events is set in motion, with the sequence unfolding in a manner and speed determined by both the genome and the environment—that is, by both nature and nurture. As with most biological processes, the duration of pregnancy is gene influenced, rather than gene determined. With the hope of understanding the mechanisms involved in labor and delivery, and ultimately preterm birth, researchers have spent many years looking for the trigger that starts parturition without success. In the normal pregnancy, there is no trigger for parturition, according to Peter Nathanielsz, Cornell University. “In one very real sense, the only certain signal to parturition is fertilization,” meaning that during the stages of pregnancy, there are backup mechanisms and fail-safe devices to ensure a successful pregnancy.
In the normal pregnancy, there is no trigger for parturition. The only certain signal to parturition is fertilization.
Peter Nathanielsz
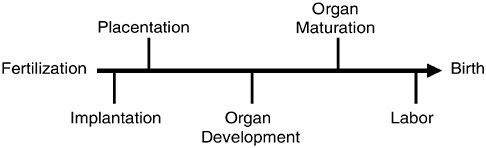
FIGURE 2.1 Normal pregnancy is often divided into various stages, which has led to research into finding a “trigger” or switch resulting in the initiation of labor and delivery. Currently there are no known triggers to move to the next stage, the process is a continuum resulting in delivery of the infant.
SOURCE: Nathanielsz, unpublished, October 3, 2002. Used with permission.
UNDERSTANDING THE SWITCH FROM PREGNANCY TO LABOR AND DELIVERY
Labor and delivery is a multifactorial process that involves fetal, placental, and maternal mechanisms. It involves the recruitment of interactive positive feedback loops and the removal of pregnancy maintenance mechanisms. Changes in these stimulatory and inhibitory mechanisms exhibit critical tissue-specific time relationships to each other. Both fetal and maternal roles are involved in these processes, and it is the interaction of these two roles that determines when birth will occur. Because parturition is a multifactorial system, it is difficult to determine precisely what initiates the cascade of events that leads to delivery. The three indispensable processes involved in normal parturition are (1) a switch in myometrial contractility pattern from contractures to contractions (2) the rupture of the fetal membranes, and (3) the dilation of the cervix.
Myometrium
The onset of labor requires both the activation and the stimulation of the myometrium to generate the intense and coordinated contractions needed to bring about the delivery of the newborn. Animal studies have also provided clues to the mechanisms of myometrial activity or contraction, which has both a fetal and a maternal component. In all mammalian species studied, myometrial activity throughout pregnancy is of the contracture type, exemplified by long-lasting, low-frequency epochs of activity that have a very different temporal and amplitude pattern from contractions (see Figure 2.2).
At labor and delivery, contractures switch to contractions to produce efficient delivery of the fetus. In sheep, this switch occurs once, generally at night, and the ewe proceeds to delivery. In monkey, this switch occurs and augments
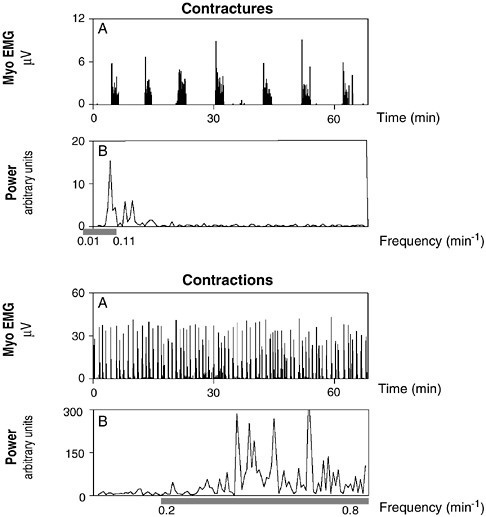
FIGURE 2.2 The amplitude and frequency of uterine activity vary during the course of pregnancy. During pregnancy, myometrial activity is of the contracture type—long-lasting, low-frequency epochs of activity. The activity switches to a high-frequency burst at the onset of labor, which is necessary to deliver the fetus.
SOURCE: Nathanielsz, unpublished, October 3, 2002. Used with permission.
for a few hours each night until delivery occurs after several transitions. By understanding the switch in myometrial activity and the interactions with environmental factors, researchers may better understand mechanism involved in preterm birth. Despite these observations, available evidence suggests that home uterine monitoring is of no value in predicting risk for preterm birth, especially since there are no effective interventions currently.
Genetic Factors
In closely inbred groups of animals maintained under carefully regulated nutritional and environmental conditions, the duration of pregnancy is very homogeneous, said Nathanielsz, suggesting that in such precisely defined environments the onset of labor is precisely regulated by the genome. According to Stephen Lye, University of Toronto, it is the fetal genotype that determines the onset of labor. In cattle bred for longer and shorter gestations, researchers observed that if an embryo from one breed was implanted into the other breed, the length of pregnancy was determined by the fetal genotype. If the cattle were bred so that the fetus had a mixed genotype, then the length of the pregnancy was halfway between the two. Lye presented evidence to suggest that the process of myometrial activation results from the synchronous increased expression of a cassette of genes encoding “contraction-associated proteins,” or CAPs, including ion channels which regulate the resting membrane potential of myocytes and hence their excitability; agonist receptors, which enhance myometrial responsiveness to stimulatory hormones; and gap junctions, which provide for enhanced cell–cell communication and therefore synchronization of uterine contractions (see next section).
Maternal Endocrine Factors
Myometrial activation and the onset of labor are ultimately controlled by endocrine and mechanical signals. Progesterone is clearly important for the maintenance of pregnancy, and in most species progesterone levels fall prior to the onset of labor. Animal studies also provide evidence for increased fetal adrenal function in late gestation. In addition, an increase in estrogen production in late gestation has been reported in sheep and various primates, including baboons, rhesus monkeys, and humans (Figure 2.3). These endocrine changes are central to the initiation of labor.
In a series of studies in the pregnant rhesus monkey, Nathanielsz observed that situations resulting in elevated maternal estrogen concentrations in late pregnancy were generally accompanied by a switch in myometrial activity from the contracture to the contraction mode. Such situations included laparotomy and fetal catheterization surgery, hypoglycemia induced by food withdrawal, and the administration of androstenedione to the pregnant monkey. Other investigators have been unable to induce preterm delivery in the pregnant rhesus monkey by the administration of estrogen. Thus, researchers hypothesize that under normal circumstances, estrogen exerts both paracrine effects at its site of generation from androgens and classical endocrine effects.
When Nathanielsz carried out androgen infusion into the pregnant rhesus monkey to ensure that estrogen was produced at its normal locus, labor occurred in the following sequence: a switch of myometrial contractures to contractions,
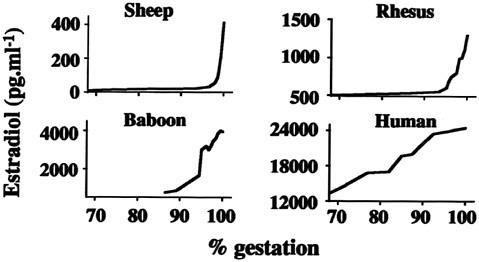
FIGURE 2.3 In most mammalian species studied, the onset of final stages of parturition coincides with a rise in the production of estrogen.
SOURCE: Nathanielsz, unpublished, October 3, 2002. Used with permission.
rupture of the fetal membranes, dilation of the cervix, and delivery of live young. In this experimental paradigm, the switch is prevented if aromatase inhibitors are administered with the androgen to inhibit estrogen production. Although the infusion of androgen was continuous and maternal plasma estrogen concentration was elevated throughout the day, the switch from contractures to contractions occurred only around the hour of darkness, suggesting a role for oxytocin (Mecenas et al., 1996). Maternal plasma oxytocin concentrations in late gestation show a pronounced 24-hour rhythm, with a peak in the early hours of the evening, coincident with the switch from contractures to contractions. A similar rhythm has been shown in pregnant women in late gestation. Further, oxytocin antagonists such as Atosiban will inhibit both the normal-term switch from contractures to contractions and the transition that is produced (prematurely) by androgen infusion.
The rise in estrogen is the central key for normal labor and delivery, a context within which it becomes interesting to look at environmental factors and environmental disruptions, including stress hormones.
The action of estrogen is complex, but one site of regulation is the expression of CAP genes. As discussed in the previous section, CAP genes are involved in the onset of labor and their expression increases as myometrial activation occurs. Lye suggested that estrogen increases the expression of CAP genes,
while progesterone suppresses their expression. The mechanisms by which these steroids act remain to be determined, although evidence suggests that specific transcription factors contribute to the activation of CAP genes. Interestingly, in humans, progesterone levels do not decrease at term, suggesting that another mechanism exists in humans to block progesterone signaling. This would in essence induce a functional withdrawal of progesterone, permitting activation of CAP genes, myometrial activation, and the onset of labor. The rise in estrogen is the central key for normal labor and delivery, a context within which it becomes interesting to look at environmental factors and environmental disruptors, including stress hormones.
Fetal Endocrine Factors
The fetal endocrine system plays a crucial role in the initiation of labor and delivery. In classic experiments in animal physiology, Liggins found that interruption of the fetal hypothalamic–pituitary–adrenal (HPA) axis at any level prolongs pregnancy in sheep. This occurred if the fetal adrenal glands or the pituitary were removed, or if a wax plate was placed between the hypothalamus and the pituitary (see Figure 2.4).
Conversely, it has been found that infusion of adrenocorticotropic (ACTH) hormone or cortisol to the ovine fetus will shorten the length of pregnancy. These findings have led some to believe that the fetus determines the length of ovine pregnancy. The endocrine pathway involves activation of the fetal HPA axis leading to increased synthesis and release of cortisol from the fetal adrenal gland. Cortisol in turn induces the expression of PGHS-2, the type 2 isomer of prostaglandin H synthase, in the placenta, which leads to the production of fetal prostaglandin E2 (PGE2). As PGE2 levels rise, an induction of cytochrome P450C17 activity occurs, which ultimately leads to the metabolism of maternal progesterone to estrogen.
The increased production of prostaglandins, which also can stimulate the myometrium, coincides with the end of gestation. William Gibb of the University of Ottawa described work in experimental animals in which a decrease in the concentration of progesterone in the maternal plasma has been observed prior to the onset of labor. In parturition, PGHS-2 is particularly important in this pathway, and its expression has been found to increase in term and preterm labor. In fetal sheep, increased cortisol regulates PGHS-2 expression in the placenta in an estrogen-independent manner, resulting in increased levels of PGE2 in fetal circulation. Later increases in maternal uterine expression of PGHS-2 require estrogen and lead to increased concentrations of PGF2α in the maternal circulation. In women, the fetal membranes surrounding the amniotic cavity are thought to be an important source of prostaglandins involved in parturition. Formation of pros-
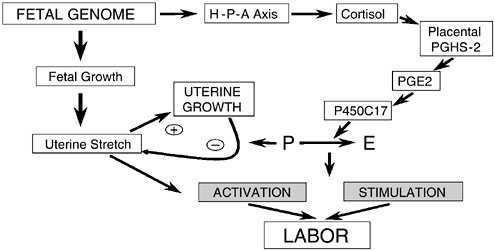
FIGURE 2.4 The fetal genome stimulates labor through two pathways: the endocrine system and uterine stretch. The fetal adrenal gland releases cortisol, which through a molecular pathway results in a rise of cytochrome P450C17. The stimulation of cytochrome P450C17 shifts the hormonal balance from progesterone to estrogen, a key step for the initiation of labor. The second pathway leading to the activation of labor is fetal growth. As the fetus grows, the uterus stretches. In the presence of progesterone, the uterus is allowed to increase the size of the myometrial cells to accommodate the growing fetus. At the end of pregnancy with the shift toward estrogen, the inhibitory effects of progesterone, and resulting in stimulation of labor.
SOURCE: S. Lye, unpublished. Reprinted with permission.
taglandins in these tissues is regulated by paracrine and autocrine mechanisms. Thus, cortisol can contribute to increased prostaglandin production in fetal tissues through upregulation of PGHS-2 and downregulation of 15 hydroxyprostaglandin-dehydrogenase (PGDH), the principal enzyme involved in the metabolism of prostaglandins. The effect of cortisol on chronic expression of PGDH reverses a tonic stimulatory effect of progesterone and likely occurs through the glucocorticoid receptor. The 11β-hydroxysteroid dehydrogenase enzyme appears to be important in regulating the effect of cortisol on prostaglandin metabolism in the placenta and chorion. In turn, this enzyme is upregulated by the fetal membranes by prostaglandins, forming a feedback loop leading to increased prostaglandin production. Cortisol also increases expression of placental corticotropin-releasing hormone, which can in turn increase prostaglandin production. Other agents such as pro-inflammatory cytokines similarly upregulate PGHS-2 and decrease expression of PGDH, indicating the presence of several mechanisms by which labor at term or preterm may be initiated.
Mechanical Factors
Although endocrine pathways are necessary for the initiation of labor, they are not sufficient, according to Lye. Mechanical signals are also required. Previous work has shown that the stretch of the uterus could induce an increase in gap junction production (see below) that may be related to the onset of labor. Fetal growth puts tension on the wall of the uterus, and it has been hypothesized that this tension could activate the same genes within the myometrium as the endocrine pathway. Rats have two uterine horns and can be manipulated such that they are pregnant only in one horn (the other horn is empty). At the time of labor, the endocrine changes occur as normal; however, the labor genes are turned on only in the horn with fetuses. The genes in the empty horn were not turned on even though the empty horn was exposed to the endocrine changes, reported Lye, but if a tube was inserted in the empty horn to stretch the muscle, then all the labor genes were turned on.
Although endocrine pathways are necessary for the initiation of labor, they are not sufficient. Mechanical signals are also required.
Stephen Lye
Interestingly, if stretch activates genes that cause labor, the question remains why animals don’t trigger labor as the fetus grows. Lye suggested that it is the presence of progesterone that is a preventive mechanism. As the fetus grows, the stretch in the presence of progesterone causes an increase in the size of the myometrial cells. This increases the growth of the uterus, which results in less tension being applied as a result of fetal growth. It is a protective mechanism that allows the fetus to grow without inducing preterm labor, and it occurs as long as progesterone is present.
Molecular Mechanisms and Cellular Signaling Pathways Associated with Parturition
During parturition, maternal and fetal compartments express the signaling pathways and produce the molecules that stimulate the myometrium to contract. The regulation of myometrial contraction is of overriding importance for the maintenance of pregnancy and for parturition, and understanding this regulation involves delineating the pathways that control contraction and relaxation and defining their interaction. The cellular mechanisms regulating uterine quiescence, the switch to contractivity, and the contractivity itself are complex and constitute an area of ongoing research. The speakers summarized some of the key pathways, receptors, and molecules that may be important.
Cellular Depolarization
The fundamental mechanism that drives uterine contraction is the smooth muscle cells of the myometrium. The myometrium has a number of pacemaker cells that propagate electrical signals to other myometrial cells to produce a forceful contraction. The rhythmic electrical firings are grouped together in bursts, which dictate the contraction patterns of the uterus. The frequency of these bursts dictates the frequency of contractions, while the duration of the bursts dictates this duration. The magnitude of contraction is dependent on the extent of propagation of these electrical signals throughout the uterus to recruit additional smooth muscle cells.
In order to have these smooth, coordinate muscle contractions, the myometrial cells must have rhythmic coupling to provide the contractility necessary for delivery. One way in which this occurs is through gap junctions, which allow the cytoplasm from one cell to be contiguous with the cytoplasm of a second cell. Thus, the channel provides a low-resistance contact between the cells and allows current to spread with minimal delay between cells. More than 20 years ago, Garfield measured the number of gap junctions at various stages of pregnancy and parturition. During pregnancy when the uterus is in a stage of relaxation, there are few gap junctions. They begin to increase in numbers prior to delivery, peak during delivery, and decline thereafter. They play a necessary role for normal labor and delivery in all species, including humans, which have been studied. If the gap junctions form early, preterm delivery results, and if their formation is blocked, delivery will not occur.
Cellular Pathways
Prior to parturition, a transition period occurs during which a series of events prepare the myometrium to respond to contractant signals and to be less responsive to relaxant signals (Table 2.1), said Barbara Sanborn of the University of Texas Medical School, Houston. This requires conformational changes in the actin and myosin molecules, which result in a shortening of the myocytes. Calcium is a key mediator in this process, acting through the calcium—calmodulin and myosin light chain kinase pathway (Figure 2.5).
The pathway to myometrial contraction begins with an increase in intracellular calcium via calcium channels in the cellular membrane or from intracellular calcium stores in the endoplasmic reticulum, which activates calmodulin. The calcium–calmodulin complex then binds to the myosin light chain kinase (MLCK), where it induces a conformational change in MLCK. This conformation change is necessary for the activation of actin and myosin through phosphorylation by MLCK. The calcium concentration is then lowered from the intracellular space by calcium pumps on the extracellular membrane or the endoplasmic reticulum, which returns the cells to homeostasis.
TABLE 2.1 Myometrial Signaling Components in Late Pregnancy
|
Contraction |
Relaxation |
|
Oxytocin receptors ↑ |
β-adrenergic receptors ↓ |
|
α1-adrenergic receptors ↑ |
CGRP receptors ↓ |
|
Gαq ↑ |
Gαi ↑, Gαs ↓ |
|
PLCb, no change or ↑ |
Adenylate cyclase, no change or ↓ |
|
Inhibition of PLC by PKA ↓ |
↑ PDE4-2B |
|
CGRP = calcitonin gene-related peptide; Gαq, Gαs, Gαi = variants of the G-protein alpha subunit; PDE4-2B = a type of phosphodiesterase; PKA = protein kinase A; PLC = phospholipase C. |
|
|
SOURCE: B. Sanborn, unpublished. Reprinted with permission. |
|
Although calcium–calmodulin mediates the major pathway associated with uterine contractions, there are opportunities for other pathways to influence the activities of various enzymes. The uterine relaxant pathways involve activation of cGMP cyclic guanosine 5′-monophosphate) and cAMP(cyclic adenosine 5′monophosphate), which in turn activate protein kinase G (PKG) and protein kinase A (PKA), respectively. PKG and PKA phosphorylate several proteins associated with promoting relaxation. They phosphorylate C (PLC), which decreases activation by GTP phospholipase (guanosine 5′-triphosphate)-binding proteins. In addition, circulating hormones such as oxytocin and prostaglandins increase intracellular calcium in part through the PLC/–IP3 (inositol 1, 4, 5-triphosphate) pathway.
The A-kinase anchoring proteins (AKAPs) are critical to PKA regulation. AKAP 9/150 brings PKA and PP2B (protein phosphatase 2B; the phosphatase that dephosphorylates PKA and other molecules) into close proximity with the PLC/–IP3 pathway. In fact, there is a switch in the ratio of PKA/–PP2B bound to AKAP 150 in myometrial plasma membrane between days 19 and 21 of gestation in the rat (see Figure 2.6). This switch, in essence, upregulates the effectiveness of the PLC/–IP3 pathway, while downregulating one of the feedback pathways. Thus, the PLC/–IP3 pathway is more poised to respond to a signal with an elevation of calcium. It is not yet clear if similar changes in the association of PKA with a plasma membrane AKAP are seen in humans, sheep, and baboons during gestation.
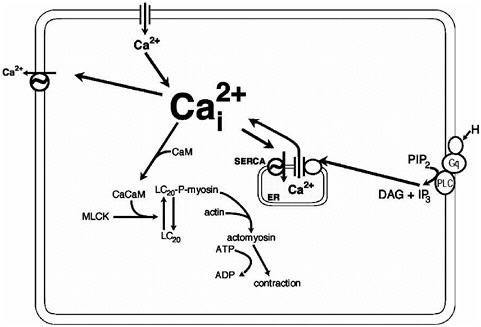
FIGURE 2.5 The rise in intracellular calcium via release from internal stores or through Ca2+ channels plays a central role in producing contraction and the initiation of labor. CaCaM = calcium–calmodulin complex; CaM = calmodulin; DAG = diacylglycerol; IP3 = inositol 1,4,5-trisphosphate; LC20 = myosin light chain; MLCK = mysoin light chain kinas; PIP2 = phosphatidylinositol bisphosphate; SERCA = sarcoendoplasmic reticulum Ca2+–adenosine triphosphatases (ATPases).
SOURCE: B. Sanborn, unpublished. Reprinted with permission.
Nitric Oxide
Nitric oxide (NO) exists as a highly reactive free radical in the gas or solution form and is highly diffusible—that is, there is no need for a receptor or transporter to diffuse it. It was first discovered in 1972, but rose to prominence in the 1990s as a mediator of short-term physiological function. Researchers suggest that it may be involved in blood pressure regulation, male impotence, learning and memory, stroke, and so forth. In 1993, Yallampalli and colleagues, at the University of Texas Medical Branch in Galveston reported the presence of the NO system in the uteri of a variety of animals (including humans), suggesting that the endogenous NO synthesized in the uterus could play a role in uterine quiescence during pregnancy.
Several studies have reported increases in NO synthesis in the uterus and in NO-induced uterine relaxation during pregnancy. Both NO synthesis and uterine sensitivity to NO are substantially reduced at term, indicating a role for NO in uterine quiescence during pregnancy and labor (Figure 2.7).
The extent to which uterine NO is necessary for maintaining uterine quies-
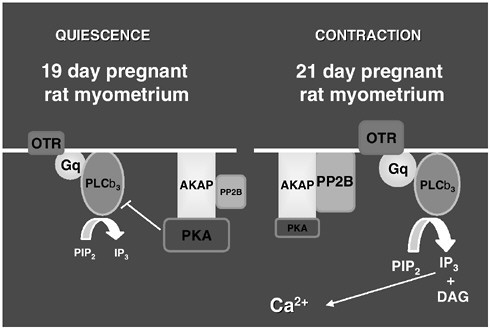
FIGURE 2.6 The A-kinase anchoring protein (AKAP) plays a pivotal role in the shift of the uterus from quiescence to contraction. In the rat between days 19 and 21, there is a decrease in the ratio of bound protein kinase A (PKA) to protein phosphatase 2B (PP2B). The change in ratio results in upregulation of the IP3 pathway to generate an increase in intracellular calcium.
SOURCE: B. Sanborn, unpublished. Reprinted with permission.
cence during normal pregnancy is under investigation. Studies in the rat and sheep indicate that inhibition of NO synthesis prior to term does not result in parturition; however, in mice, parturition occurs. Moreover, uterine NO synthesis has been shown to be regulated by estradiol and progesterone, key hormones in pregnancy. Progesterone not only enhances NO synthase expression and NO synthesis in the rat uterus but also enhances NO-induced uterine relaxation. On the other hand, antiprogesterones decrease NO synthase enzymes, inhibit NO synthesis, and reduce uterine relaxation responsiveness to NO. Yallampalli suggested that during pregnancy there is a significant increase in NO production driven by the elevated progesterone/estrogen ratio, which switches during normal-term labor. Thus, NO might interact with other uterotonins in the maintenance of uterine quiescence during pregnancy.
Calcitonin Gene-Related Peptide
The calcitonin gene-related peptide (CGRP) also acts through the relaxant pathway. This peptide is synthesized and transferred to many organs, such as the
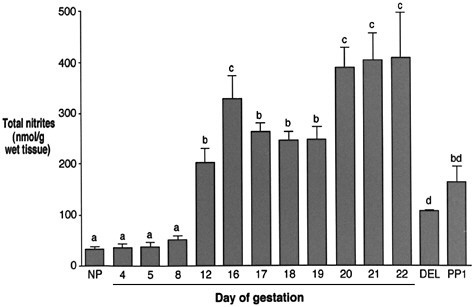
FIGURE 2.7 The role of nitric oxide in maintenance of the rat pregnancy is still under investigation. The concentration of NO rises during pregnancy, but decreases at the onset of labor and delivery. This suggests that NO concentration may be important for maintaining quiescence.
SOURCE: Yallampalli et al., 1998. © 1998 Society of Gynecologic Investigation. Reprinted with permission.
uterus, and to the vasculature. Yallampalli’s research has shown that the level of CGRP is increased during pregnancy and falls at the end of gestation, suggesting that the CGRP levels follow relaxation (Figure 2.8). He has concluded that (1) CGRP inhibits both spontaneous and induced contractility of uterus in both rat and human, (2) sensitivity to CGRP is increased with pregnancy and decreased at term, and (3) CGRP binding to myometrium is increased with pregnancy and decreased with labor. Moreover, CGRP receptor levels in myometrium parallel CGRP binding and relaxation responses.
Rupture of the Fetal Membrane and Cervical Ripening
The ripening of the cervix and the rupture of the fetal membranes are both necessary for labor and delivery to occur. In the cervix and fetal membranes, the preparatory step during term delivery is temporally different from that of the uterus, indicating separate control systems.
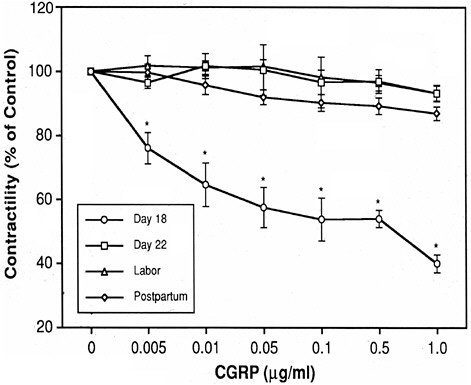
FIGURE 2.8 The calcitonin gene-related peptide (CGRP) may be important for maintaining uterine quiescence. In the rat prior to delivery, the uterus is sensitive to CGRP which has a dose-dependent effect on uterine contractility. As labor and delivery near, CGRP has little effect on contractility as the uterus changes its sensitivity to the peptide.
SOURCE: Dong et al., 1998. Reprinted with permission.
Cervical Ripening
To prepare for labor and delivery, the cervix, which is closed and rigid throughout most of pregnancy, must soften and dilate, according to Robert Garfield, University of Texas Medical Branch. This occurs independently of uterine contractivity and is necessary for an uncomplicated delivery. There are three phases to cervical ripening: softening, effacement, and dilation. The softening phase is chronic, while the effacement and dilation phases are acute—occurring immediately before delivery.
Although there are a number of smooth muscle cells, fibroblasts, and mast cells in the cervix, it is primarily comprised of collagen-type connective tissue. At the time of parturition, the breakdown in the cervical matrix coincides with a decrease in the amount of collagen and a subsequent increase in the water content. Further it is the change in type I collagen from an insoluble to a soluble
form that is crucial to ripening. The two forms of collagen have a different fluorescent pattern that can be monitored by a collascope. Garfield reported that in humans and rats there is a decline in the fluorescent pattern of the cervix as parturition nears, which corresponds to the change in type I collagen. Further, they were able to show in patients that when the fluorescent patterns decrease due to changes of collagen content, the cervix softens and the patient will deliver preterm.
The biochemical pathway resulting in the breakdown in the cervical matrix has been extensively studied (see Uldbjerg 1989; Leppert,1995, for review). These pathways can be activated by a number of external compounds and provide possible sites of interactions for environmental factors to play a role. The activation of prostaglandins and the subsequent activation of compounds such as NO constitute an area of ongoing research. The role of NO appears to have different effects in activating the myometrium or softening the cervix, according to Garfield. NO levels are elevated in the uterus (as discussed previously) during pregnancy and are associated with uterine quiescence. At the onset of labor, production of NO decreases, resulting in an increase in uterine contractility. In contrast, NO production increases in the cervix as labor begins and appears to be involved in softening of the cervix. Similar results (cervical softening) have been obtained by locally applying NO agonists to the cervix.
Fetal Membranes
The fetal membranes, which surround the amniotic cavity, are composed of two layers: the amnion and the chorion. The primary function of the membranes is to retain the amniotic fluid, to secrete substances into the fluid and the uterus, and to protect the developing fetus from infection. Rupture of the membranes is the third event necessary for delivery, and it occurs shortly before parturition begins. Preterm rupture of the membranes occurs in approximately 1 percent of all pregnancies and is associated with 30–40 percent of all preterm deliveries.
Preterm rupture of the membranes occurs in approximately 1 percent of 23 pregnancies and is associated with 30–40 percent of all preterm deliveries.
Fetal fibronectin is a protein, which is part of the extracellular matrix between the membranes and the decidua. It is used as a marker of impending labor, especially preterm birth. When an inflammation or breakdown of the extracellular matrix occurs, fibronectin leaks down into the cervix and the vagina. Goldenberg stated that women in early preterm labor with a negative fetal fibronectin test have a less than 1 percent chance of delivering in the subsequent two weeks.















