4
Chemical Characteristics of Bombs
COMPOSITION OF BOMBS
While few chemicals find use as military explosives (Table 4.1), these can be combined with platiscizers and other materials to create a plethora of formulations.1,2 The problem of terrorism and suicide bombers, however, narrows the focus to high explosives. Several such explosives, as well as some plasticizers and taggants found in plastique explosives, are listed in Table 4.1 along with their abbreviations. The devastating shock wave that accompanies detonation of a high explosive (HE), results in widespread damage and loss of life. High explosives consist of an intimate mixture of oxidant and reductant, either within a single molecule, such as nitroglycerin, pentaerythritol tetranitrate (PETN), trinitrotoline (TNT), or triacetone triperoxide (TATP), or within an ionic solid, such as ammonium nitrate, when mixed with fuel oil. Mixtures of high explosives are frequently used. For example, Semtex is a blend of cyclomethylenetrinitramine (RDX) and PETN. Reductants (e.g., aluminum powder, fuel oil)
may be added to solids such as ammonium nitrate, which have excess oxidizing power, in order to increase the explosive yield.
The diversity of molecular features found in explosives suggests that a consideration of the elemental compositions might lead to new or improved detection approaches. Table 4.1 provides a summary of high explosives that would be relatively simple to prepare or that could reasonably be obtained by a determined individual. It also contains some other explosive types for comparison. If elemental formulations are considered, few common chemicals would be mistaken for explosives. The empirical formulas of all of the high explosives in Table 4.1 were entered in the online Aldrich catalog of common laboratory and industrial chemicals and polymers. Only two explosive elemental compositions had other isomers among the 90,000 chemicals in the catalog. One was TNT, which has the same composition as dinitroanthranilic acid. The latter compound is carcinogenic and was a former dye intermediate that is being phased out. The diacetone and triacetone peroxides (e.g., TATP) pose the greatest problem for a detection scheme based solely on elemental constituents. These explosives have the same elemental composition as several organic compounds, including the specialty polymer poly(propylene adiponate). However, the high volatilities of these compounds might make it feasible to detect the vapor plume by molecular spectroscopic techniques, such as microwave or infrared (IR) spectroscopy. For example, the carbonyl stretching absorption in the infrared spectrum at 1740 cm-1 is intense and diagnostic of acetone.
All explosives must contain both oxidizing and reducing agents. Strong oxidizing agents require the use of the most electronegative elements nitrogen, oxygen, fluorine, and chlorine. Therefore, one common aspect of HE compositions is a large percentage of the more electronegative elements nitrogen and oxygen. Chlorine and fluorine are used less often in explosives because of its difficult chemistry and greater expense. Also, fluorine’s extreme oxidizing power may lead to unstable explosive formulations. The preponderance of highly electronegative elements in explosives is one reason why their detection by IMS (ion mobility spectrometry), which employs electron attachment to neutral explosive molecules, succeeds.
The light elements carbon and hydrogen usually serve as the reducing components of HE formulations. Occasionally, metal powders of the lighter elements (aluminum or magnesium) are added as supplemental reducing agents in explosive mixtures. Black powder, which is a less energetic material, uses both charcoal and elemental sulfur as reductants.3
TABLE 4.1 Some Representative Common High Explosives and Their Compositionsa
|
Explosives Based on Nitrogen |
Formula |
wt % C |
wt % H |
wt % N |
wt % O |
Sum N + O |
|
Ammonium nitrate (AN) |
H4N2O3 |
0 |
5.04 |
35.01 |
59.97 |
94.98 |
|
Ammonium picrate (Expl D) |
C6H6N4O7 |
29.28 |
2.46 |
22.76 |
45.5 |
68.26 |
|
Cyclonite (RDX) |
C3H6N6O6 |
16.22 |
2.72 |
37.84 |
43.22 |
81.06 |
|
Ethylenediamine dinitrate |
C2H10N4O6 |
12.91 |
5.42 |
30.1 |
51.58 |
81.68 |
|
Guanidine nitrate |
CH6N4O3 |
9.84 |
4.95 |
45.89 |
39.32 |
85.21 |
|
Hexamethylenetriperoxide diamine (HMTD) |
C6H12N2O6 |
34.62 |
5.81 |
13.46 |
46.11 |
59.57 |
|
Hexanitrohexaazaisowurtzitane (HNIW or CL20) |
C6H6N12O12 |
16.45 |
1.38 |
38.36 |
43.82 |
82.18 |
|
Hydrazine nitrate |
H5N3O3 |
|
5.3 |
44.2 |
50.09 |
94.29 |
|
Mannitol hexanitrate |
C6H8N6O18 |
15.94 |
1.78 |
18.59 |
63.69 |
82.28 |
|
Monomethylamine nitrate |
CH4N2O3 |
13.05 |
4.38 |
30.43 |
52.14 |
82.57 |
|
Nitrocellulose |
C6H7N3O11 |
24.24 |
2.37 |
14.14 |
59.23 |
73.37 |
|
Nitroglycerin (NG) |
C3H5N3O9 |
15.87 |
2.22 |
18.5 |
63.41 |
81.91 |
|
Nitrotriazolone (NTO) |
C2H2N4O3 |
18.47 |
1.55 |
43.08 |
36.9 |
79.98 |
|
Octogen (HMX) |
C4H8N8O8 |
16.22 |
2.72 |
37.84 |
43.22 |
81.06 |
|
Pentaerythritol tetranitrate (PETN) |
C5H8N4O12 |
19 |
2.55 |
17.72 |
60.73 |
78.45 |
|
Picric acid |
C6H3N3O7 |
31.46 |
1.32 |
18.34 |
48.88 |
67.22 |
|
Tetrazene |
C2H8N10O |
12.77 |
4.29 |
74.44 |
8.5 |
82.94 |
|
Tetryl |
C7H5N5O8 |
29.28 |
1.76 |
24.39 |
44.58 |
68.97 |
|
Trinitrobenzene (TNB) |
C6H3N3O6 |
33.82 |
1.42 |
19.72 |
45.05 |
64.77 |
|
Trinitrotoluene (TNT) |
C7H5N3O6 |
37.02 |
2.22 |
18.5 |
42.26 |
60.76 |
|
Triaminoguanidine nitrate (TAGN) |
CH9N7O3 |
7.19 |
5.43 |
58.67 |
28.72 |
87.39 |
|
Triaminotrinitrobenzene (TATB) |
C6H6N6O6 |
27.92 |
2.34 |
32.55 |
37.19 |
69.74 |
|
1,3,3-Trinitroazetidine (TNAZ) |
C3H4N4O6 |
18.76 |
2.1 |
29.17 |
49.98 |
79.15 |
|
Trinitrochlorobenzene |
C6H2ClN3O6 |
29.11 |
0.81 |
16.97 |
38.78 |
55.75 |
|
Trinitropyridine |
C5H2N4O6 |
28.05 |
0.94 |
26.17 |
44.84 |
71.01 |
|
Urea nitrate |
CH5N3O4 |
9.76 |
4.09 |
34.14 |
52 |
86.14 |
|
Average (%) |
|
20.29 |
2.98 |
30.81 |
46.14 |
76.95 |
|
Standard deviation |
|
±8.15 |
±1.38 |
±11.01 |
±8.01 |
±8.47 |
|
Maximum/minimum (%) |
|
37/0 |
5/0.8 |
58/13 |
63/9 |
95/56 |
|
Other Explosive Types |
||||||
|
Ammonium perchlorate |
H4NO4Cl |
0 |
3.43 |
11.92 |
54.47 |
66.39 |
|
Lead styphnate |
C6H3N3O9Pb |
15.39 |
0.65 |
8.97 |
30.75 |
39.72 |
|
Triacetone triperoxide (TATP) |
C9H18O6 |
48.64 |
8.16 |
0 |
43.2 |
43.2 |
|
Black powder |
|
10 |
0 |
10.4 |
35.6 |
46 |
|
Tetraamminecopper perchlorate |
H24Cl2CuN4O8 |
|
7.06 |
16.35 |
37.35 |
53.7 |
|
Average |
|
|
|
11.91 |
40.27 |
49.80 |
|
Standard deviation |
|
|
|
±2.23 |
±6.85 |
±8.19 |
|
aFor additional listings of explosives considered important in forensic investigations, see TWGFEX: The Technical Working Group for Fire and Explosions, http://www.twgfex.org. |
||||||
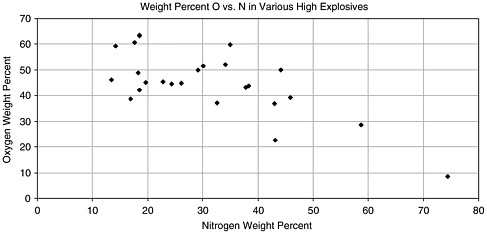
FIGURE 4.1 Plot of the weight percent of oxygen versus nitrogen for the high explosives based on nitrogen listed in Table 4.1.
These general observations suggest that a focus on the percentage composition of the most electronegative elements might be a useful identifier of explosive formulations.
The majority of high-explosive formulations use inorganic or organic nitrate or nitro functional groups as the oxidant. The correlation between nitrogen and oxygen content is roughly linear, as shown in the scatter plot in Figure 4.1. The nitrogen content of a wide variety of nitrogen-containing explosives is 31 ± 12%. Oxygen composition is even more constant at 45 ± 8%. This suggests that dual analysis of nitrogen and oxygen content might provide a more reliable indication of high explosives than techniques based on nitrogen content alone. Indeed, it has been stated, “A measurement of the oxygen and nitrogen densities, to an uncertainty of ±20%, gives a unique separation of explosives from other compounds.”4 Note, however, that this analysis will fail for certain explosives that do not contain nitrogen, for example TATP.
While analysis such as this is not useful for energetic materials such as TATP, it may be quite useful for more “common” explosive materials such as ammonium nitrate/fuel oil, or black and smokeless powders. Ammonium nitrate (AN) in particular is readily available and when mixed with fuel oil is capable of producing widespread explosive dam-
age. Therefore, the ability to exploit the nitrogen and oxygen content in AN may provide a useful means of detection of this material in explosive devices.
From a chemical point of view, the other possible elements of high electronegativity that might be employed in explosives are chlorine and fluorine. For example, perchlorate and chlorate salts are used in certain energetic materials formulations. Ammonium perchlorate, which is mixed with a powdered aluminum-polymer binder, finds use as a solid rocket fuel. Metal powder-potassium chlorate mixtures are used in fireworks. Chlorine-based explosives could be detected by an elemental analysis approach, since a high chlorine-oxygen-nitrogen content is indicative of such species. The explosives employed by terrorists and suicide bombers will continue to evolve as military establishments worldwide strive to build more efficient and/or more energetic materials. As terrorists and other potential bombers become more sophisticated, both in their choice of explosive materials and in the way these materials are procured, transported, and concealed, detection methods must be changed concomitantly.
For example, in the near term, a new class of energetic materials, ionic liquids (e.g., 4-diamino-1,2,4-triazolium dinitroamide [(NH2TazN(NO2)2], is just appearing in the open literature.5 The syntheses of these liquids or low-melting salts from readily available compounds are very straightforward. Most of them could be identified with a technique that relies on N + O content for identification.
Other new explosive materials are fluorine-containing derivatives of the familiar RDX and octogen (HMX).6 These tend to be more dense and thus have greater impact per unit mass. Although many of these compounds could probably be detected with the same techniques used for RDX and HMX (e.g., negative polarity IMS) or those that use bulk properties, as the percentage of fluorine vis-à-vis oxygen increases, an analysis based on nitrogen and oxygen content becomes problematic. This could be corrected with the addition of fluorine to the list of electronegative elements scanned.
Recommendation: Improved detection systems will lead to development of new explosives. Research is needed on the identifica-
tion and characterization of new chemical explosives that do not utilize nitrogen and have very low vapor pressures, for example, ionic liquids.
PLUMES AND VAPOR PRESSURES
Existing explosive detection approaches for luggage rely on estimating physical characteristics, such as the density and approximate elemental nitrogen content, by using X-ray scattering (e.g., X-ray computer tomographic [CT] analysis).7 The reason for the existing focus on analysis of solid materials is that the low volatility of many explosives precludes detection of emanating molecular vapors (see Figure 4.2). Vapors that are emitted from a bomb may be present at concentrations two to four orders of magnitude less than the equilibrium vapor pressures shown in the figure, both because of enclosure in a bomb package and because explosive compositions containing other compounds may have lower vapor pressures than those shown for the pure explosive compounds.
In some cases, more volatile impurities (e.g., dinitrotoluene [DNT] in TNT) can be employed in specific explosive detection applications, such as landmine detection. Some energetic plasticizers8 or taggants9 used in plastique explosives, such as mononitrotoluene (MNT), diglycol dinitrate (DEGN), dimethyldinitrobutane (DMNB), ethylene glycol dinitrate (EGDN), or butanetriol trinitrate (BTTN), are volatile enough for vapor detection of these species to be used as an indicator of the presence of explosive compounds. Nitroglycerin, a high explosive and constituent of dynamite, is another volatile species that might be detected directly in the vapor phase. The vapor pressures of less volatile explosives are very temperature dependent. For example, the vapor pressure of TNT increases by approximately a factor of four between 20 and 30°C.10 This means that the probability of detecting explosives will depend strongly on the ambient
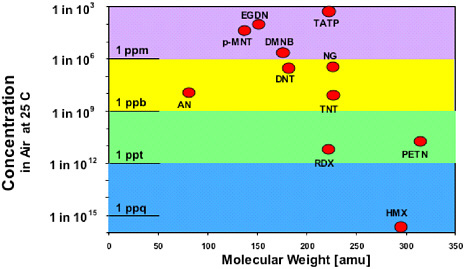
FIGURE 4.2 Vapor pressures of high explosives and additives. (Courtesy of J. Parmeter et al., Sandia National Laboratory)
temperature. Another variable to consider is the tendency of high explosives to adsorb strongly on surfaces. It is well known in land mine detection studies that both dogs and chemical sensors show improved performance when soil moisture content is high.11 Water competes with TNT and DNT for binding on soil particles, thereby releasing more TNT and DNT into the soil airspace where these molecules can diffuse out for improved detection.
ATOMIC AND MOLECULAR PROPERTIES
Several different atomic and molecular properties might be exploited in explosives detection. Two excellent detailed descriptions exist,12 so the following discussion focuses on general categories.
Atomic Properties
Although atomic absorption spectroscopy and atomic fluorescence can be used to determine elemental composition (after atomization of a sample in a flame or plasma), these methods require direct sampling and work best for heavier elements. They have not been considered for standoff detection of explosives. Several neutron techniques have been explored13,14,15 and some are being considered commercially for land mine detection and cargo screening. Gamma rays, which are emitted from radioactive nuclei that form after neutron bombardment, can provide a unique signature for each element. Techniques that measure transmitted, attenuated, or scattered neutrons can provide imaging, as well as information about elemental composition. Being electrically neutral, neutrons do not interact strongly with matter. Their penetrating nature is an asset for screening cargo and luggage; however, the inconvenience, expense, and hazards of radioactive, accelerator, and reactor neutron sources limit their application to settings in which human exposure can be prevented with certainty. The photonuclear reaction employs resonant gamma-ray absorption to produce an unstable nucleus that is subject to radioactive decay. This is another method that has been applied to determining elemental nitrogen content.16
Core electron ionization and subsequent characteristic X-ray emission gives rise to another broad class of methods, which might potentially be used for atom identification. X-ray absorption edge behavior can also provide element-specific detection. The above techniques mainly diagnose the type of elements present, rather than being strongly influenced by molecular properties. Techniques that involve X-ray emission are advantageous, since computer tomography can be used to create a three-dimensional image of the target.
Besides the characteristic emissions, X-ray scattering has been employed to broadly distinguish light elements from heavier elements in imaging applications; however, the light elements carbon, nitrogen, and
|
13 |
Yinon, J. Forensic and Environmental Detection of Explosives; John Wiley & Sons: Chichester, UK, 1999. |
|
14 |
Bruschini, C. Commercial Systems for the Direct Detection of Explosives for Explosive Ordnance Disposal Tasks; EPFL-DI-LAP Internal Note, February 2001, 68 pp.; http://diwww.epfl.ch/lami/detec/detec.html#Detec_doc. |
|
15 |
Singh, S.; Singh, M. Explosives detection systems (EDS) for aviation security; Signal Processing 2003, 83, 31-55. |
|
16 |
Habiger, K.W.; Clifford, J.R.; Miller, R.B.; McCullough, W.F. EXDEP/CTX: An explosive detection system for screening luggage with high energy X-rays; Proc. IEEE Particle Accelerator Conf. 1994, 4, 2622-2624. |
oxygen are not easily differentiated from each other. Cargo screening equipment using both transmitted and backscattered X-rays for imaging has been developed by several vendors.
Molecular Properties
Spectroscopic methods, which reflect structure and type, provide means that are capable of identifying specific explosives being used. These span the range of methods from those that are relatively unspecific for large molecules, such as ultraviolet-visible (UV-Vis) spectroscopy, to highly specific identifiers, such as mass spectrometry. It is useful to examine the various techniques in the context of whether they are applied to detect small amounts of vapors emanating from bulk solid explosives or trace particulates.
Molecular Vapors Emitted by Explosives
As noted above, the vapor plume from an explosive may contain from as much as 1000 parts per million (ppm) to fractions of a part per trillion (ppt) of the molecular constituents, impurities, or decomposition products. This provides an opportunity for identification by gas-phase molecular spectroscopies. Molecular spectroscopic techniques can be used to uniquely identify explosive molecules in the vapor phase, but the low vapor pressure of many explosives means that this will often not be feasible. Most packaged explosives (nitroglycerin, EGDN-based dynamites, and TATP being notable exceptions) emit so little material into the gas phase that detection is feasible only near the surface of these materials. Chemically specific spectroscopic probes therefore require direct sampling, near-proximity instrumentation measurements, and perhaps preconcentration. There is an inherent problem with increasingly more sensitive means of molecular detection. Ultrasensitive detection methods could give rise to high nuisance alarms due to the trace residues from the use of recreational firearms or medical use of nitroglycerin as a heart medication. Ultrasensitive detection approaches would be problematic in explosive-rich military environments and could also lead to false alarms due to the presence of chemical interferents.
Some techniques applied for the identification of explosive molecules in the vapor phase include UV-Vis, infrared, and microwave absorption. Fluorescence in the UV-Vis has also been employed. For large molecules, the UV-Vis and fluorescence characteristics of electronic spectra are broad, so specificity is low. The presence of other compounds that absorb or emit in similar spectral regions limits the usefulness of these methods. Infrared spectra probe characteristic vibrations of a molecule, and the presence of
the −NO2 group in many high explosives offers a characteristic spectral feature that can indicate the presence of an explosive. Alternative spectroscopic (e.g., photoacoustic) probes, as well as laser techniques, such as cavity ring-down spectroscopy and LIDAR(light detection and ranging) for UV absorption or fluorescence have been explored.17 The LIDAR techniques offer the possibility of standoff detection; however, they suffer from the breadth of electronic spectral features for large molecules. For that reason, selective luminescent granular sensors (e.g., “smart dust”) have been developed, whose luminescence is affected by adsorption of explosive molecules and can be probed remotely by LIDAR.18 The application of nanosensors to explosives detection is an area yet to be explored.
Direct sampling ionization methods19 include gas chromatography (GC) interfaced with electron capture detectors (ECD). Mass spectrometry can provide unique identification, and when interfaced with GC even complex mixtures can be analyzed. Gas chromatography-mass spectrometry (GC-MS) is the gold standard for chemical analysis; however, it requires bulky, expensive, and delicate equipment as well as direct sampling. The development of MEMS (microelectromechancial systems) approaches to GC separations and interest in developing fieldable mass spectrometers may lead to advances that make these approaches better adapted for field deployment. The chief ion method currently used is IMS, which employs an electron source at ambient pressures to create negative ions of explosive vapors and characterize them by their drift times in a fixed electric field. It is not as selective as GC-MS, but it has been widely deployed for several reasons, including relatively low cost and simplicity of instrumentation.
Recommendation: Research into the vapor space surrounding the bomber may lead to improved means of explosives detection. An increased quantitative understanding of vapor plume dynamics is required for application to explosives with high-volatility components such as TATP.
Vapor plume research is particularly required for high-volatility components such as TATP both because this high volatility could be exploited with detection techniques that are not applicable to low-volatility components, and, especially in the case of TATP, its lack of nitrogen may make it undetectable by some traditional detection techniques.
Bulk Explosives
Typical high explosives used by terrorists, such as PETN and RDX, have total densities between 1.2 and 1.8 g/cm3.20 X-ray imaging approaches combined with computer tomography allow estimation of densities of objects and form the basis for the airport CTX luggage scanners. The rate of false positives from a simple density determination could be improved by additional elemental or molecular information. Nuclear quadrupole resonance (NQR) is being explored as an adjunct method for nitrogen detection. Since nuclear quadrupole energy levels are perturbed by the chemical environment, the technique can offer explosive-specific information. However, the sensitivity toward different nitrogen-based explosives is variable.21,22 Nuclear magnetic resonance (NMR) is a technique that has not yet been widely utilized in the detection of explosives. Pulsed NMR to examine H-N coupling via T1/T2 measurements has been tried. This approach is complicated by the requirement of a strong homogeneous auxiliary magnetic field.23 Even though the resonance frequencies in NQR are low and hence the technique is a less sensitive technique, nonetheless NQR has been demonstrated to be useful in practice for explosives detection.
Diffracted X-rays may provide information that can be used to detect crystalline explosive compounds such as RDX and some other high explosives. Measurement of low-angle X-ray scattering has the potential to detect such materials with high specificity, since the diffraction peaks arise from the regular order of atoms within molecules arranged in a periodic lattice.24
Microwave and terahertz imaging are being explored as standoff methods for detecting concealed solid explosives,25 but these techniques lack chemical specificity. Some resonant absorption occurs in the low-frequency microwave region, which may be exploited to give useful specificity.
Trace Particulate or Adsorbed Explosives
The propensity of explosive vapors and explosive particles of low volatility to adsorb strongly to bulk surfaces26 or dust particles is the basis for several explosive detection schemes. Fine particulates of explosives may also be dislodged during the bomb making process and adhere to packing material, as well as the clothing and skin of a bomb maker. Raman microspectroscopy using fiber-optic techniques has been employed to determine characteristic vibrational spectral features, which can be used to identify explosives particles or surfaces contaminated with explosives.27 In one common application of IMS, a probe with an affinity for explosive particulates is rubbed on potentially contaminated luggage or shoe surfaces. Heating the probe to about 200°C in the IMS inlet vaporizes low-volatility solid explosives, such as RDX, for detection.
Catalytic or oxidative decomposition of many explosives yields NO2 that can be detected by highly sensitive chemiluminescence schemes. Immunoassay has been shown to be a viable detection method for several nitrogen-based explosives.28 The ability of explosive molecules to quench the luminescence of conjugated polymers has been explored as a sensitive method for trace detection.29 Specificity can be improved by adopting an
“electronic nose” approach with an array of sensors with differing responses to analyte.30
FUTURE POSSIBILITIES
For standoff detection one has either to examine a characteristic physical emission, to probe with a beam of particles or radiation and observe a characteristic emission, or else to collect a sample remotely. Various bulk material properties can be used to indicate the probable presence of explosives. Properties, such as density and effective atomic number, are presently used to indicate the presence of an explosive, but are not unique identifiers. False-alarm rates occur because such diagnostics lack true chemical specificity. A detailed analysis of the weight percent of all elements present in a solid would offer greater specificity if it could be engineered in a practical system. In the absence of an analysis of all elements present, a determination of nitrogen and oxygen content would identify a wide variety of nitrogen-based explosive materials with a high degree of certainty. An analysis of just the nitrogen content is useful but less specific. At present, neutron techniques seem to be the techniques most likely to provide such information for concealed explosives.
Trace detection methods must surmount the issue of sample collection to be useful in a standoff mode. Vapor and particle collection booths, which preconcentrate samples for analysis, have been prototyped for walk-in passenger screening. LIDAR techniques show promise for standoff applications; however, the issue of selectivity becomes problematic when detecting complex explosive molecules with broad spectroscopic features. Sensor array detectors (e.g., resistive, fluorescent) or “electronic noses” present the possibility of specificity with relatively inexpensive instrumentation. Key issues that have to be faced include sample collection and concentration. The application of MEMS technology to explosive sensing shows promise, as does the application of nanotechnology (e.g., smart dust). Explosives are also molecules with high exotherms for anaerobic thermal decomposition, which might be exploited in microcalorimetry using MEMS technology.31 The development of inexpensive se-
lective miniature sensors offers the possibility of deploying sensor networks that could monitor large areas and perhaps react to a mobile source of trace explosives.
Biology is also expected to contribute to new sensing approaches. Chemically specific higher biological systems, such as dogs, require close proximity to the explosive material being sensed, but the sensor is mobile. Similarly, bees have been trained to locate specific explosives.32 For example, spiking sugar water with TNT trains them to associate food with the odor of TNT. Robotics or unmanned air vehicles provide an engineered analogue to these approaches. Many nitro-based explosives are also potent vasodilators (e.g., nitroglycerin for angina), which suggests biological detection schemes. Other novel approaches to explosive detection are considered in Chapter 7.















