8
Propylenimine1
Acute Exposure Guideline Levels
PREFACE
Under the authority of the Federal Advisory Committee Act (FACA) P. L. 92-463 of 1972, the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances (NAC/AEGL Committee) has been established to identify, review and interpret relevant toxicologic and other scientific data and develop AEGLs for high priority, acutely toxic chemicals.
AEGLs represent threshold exposure limits for the general public and are applicable to emergency exposure periods ranging from 10 minutes (min) to 8 hours (h). Three levels—AEGL-1 and AEGL-2, and AEGL-3—will be developed for each of five exposure periods (10 and 30 min, 1 h, 4 h, and 8 h) and will be distinguished by varying degrees of severity of toxic effects. It is believed that the recommended exposure levels are applicable to the general population including infants and children, and other individuals who may be susceptible. The three AEGLs have been defined as follows:
AEGL-1 is the airborne concentration (expressed as parts per million or milligrams per cubic meter [ppm or mg/m3]) of a substance above which it is predicted that the general population, including susceptible individuals, could
experience notable discomfort, irritation, or certain asymptomatic, non-sensory effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure.
AEGL-2 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience irreversible or other serious, long-lasting adverse health effects or an impaired ability to escape.
AEGL-3 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience life-threatening health effects or death.
Airborne concentrations below the AEGL-1 represent exposure levels that could produce mild and progressively increasing but transient and nondisabling odor, taste, and sensory irritation, or certain asymptomatic, non-sensory effects. With increasing airborne concentrations above each AEGL, there is a progressive increase in the likelihood of occurrence and the severity of effects described for each corresponding AEGL. Although the AEGL values represent threshold levels for the general public, including susceptible subpopulations, such as infants, children, the elderly, persons with asthma, and those with other illnesses, it is recognized that individuals, subject to idiosyncratic responses, could experience the effects described at concentrations below the corresponding AEGL.
SUMMARY
Propylenimine is an aziridine compound used to modify latex surface coating resins to improve adhesion and to modify bonding properties of textiles, paper, and dyes. It is also used in photography, in the pharmaceutical industry, in gelatins, and organic syntheses. Propylenimine is a colorless oily liquid that has an odor similar to that of ammonia. It is flammable and is an explosion hazard. Propylenimine is similar in structure and toxicity to ethylenimine.
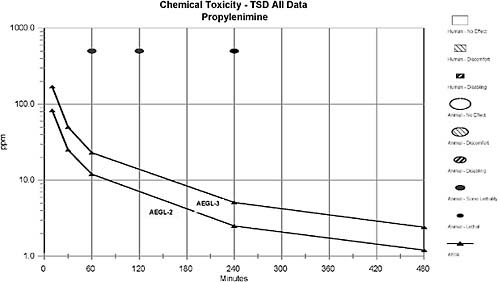
No data were found concerning toxicity or the odor detection threshold for propylenimine in humans. A time-response study conducted in rats and guinea pigs showed that one of six guinea pigs died after exposure to 500 ppm for 60 min and none of the six died after exposure to the same concentration for 30 min. Five of six rats died after exposure to 500 ppm for 240 min and none of the six died after exposure to the same concentration for 120 min. No concentration-response data were available for deriving AEGL values from animal studies. Therefore, a relative potency approach was used to derive AEGL-2 values based on lethality data for propylenimine and ethylenimine. Propylenimine was 4 to 8 times less toxic than ethylenimine depending on the species: 4 or 5 times less toxic to the guinea pig and 8 times less toxic to the rat. Tumors developed at multiple sites in rats treated orally with propylenimine for 28 or 60 weeks; therefore, IARC classified propylenimine as Group 2B (possibly carcinogenic to humans). Propylenimine is mutagenic in Salmonella and Drosophila.
Data are not available for deriving AEGL-1 values for propylenimine; therefore, no AEGL-1 values were recommended. The absence of AEGL-1 values does not imply that exposures below AEGL-2 are without adverse health effects. In addition, data are not available for estimating the level of distinct odor awareness (LOA) for propylenimine.
Data consistent with AEGL-2 end points were not available for propylenimine; therefore, the AEGL-2 values were derived based on a relative toxicity approach in which the inhalation toxicity of propylenimine was compared with that of ethylenimine. A relative potency factor of 5, which is the geometric mean of the three relative toxicity values calculated from the inhalation studies in rats and guinea pigs exposed to the same concentrations and/or durations, was applied to the AEGL-2 values for ethylenimine. Use of the geometric mean is common practice in toxicology when calculating means of values associated with risk assessments or other expressions of comparative toxicity. It is typically used because it does not give excessive weight to extreme values (“outliers”). It draws outliers toward the center of the distribution, decreasing the sensitivity of the parameter to undue influence of the outlier (Gad 2005). The geometric mean was used for the propylenimine AEGL-2 assessment because of the variability in relative potency values between ethyleneimine and propylenimine (relative potencies were 8-fold in a rat study and 4- and 5-fold in two guinea pig studies). The AEGL-2 values for ethylenimine based on a no-observed effect level (NOEL) for extreme respiratory difficulty in guinea pigs were 33, 9.8, 4.6, 1.0, and 0.47 ppm for 10 min, 30 min, 1 h, 4 h, and 8 h, respectively. In addition, a modifying factor of 2 was applied to account for the deficient database for propylenimine. The resulting AEGL values for propylenimine are 83, 25, 12, 2.5, and 1.2 ppm for exposure durations of 10 min, 30 min, 1 h, 4 h, and 8 h, respectively.
A single exposure of guinea pigs to 500 ppm of propylenimine for 30 min was a no-observed effect level (NOEL) for lethality and this concentration was used as the point of departure for deriving AEGL-3 values. An uncertainty factor of 10 (3 for interspecies sensitivity and 3 for intraspecies variability) was applied to the NOEL for lethality. An interspecies uncertainty factor of 3 was selected because propylenimine is a reactive direct-acting alkylating agent, and the effects of acute toxicity are expected to be confined to the respiratory tract. Propylenimine-induced respiratory tract damage appears to be due to a direct effect of the alkylating agent on the respiratory epithelium; this mechanism is expected to be similar among species. An uncertainty factor of 3 was applied for intraspecies variability because the effects appear to involve direct contact of the eyes or respiratory epithelium with a very reactive alkylating agent and these effects are not expected to differ considerably among members of the population. Studies have shown that DNA damage is probably the initiating step in a cascade of events leading to cell damage after exposure to alkylating agents, and DNA damage persists in respiratory and systemic organs following inhalation exposure to these agents. This mechanism is not expected to be different among individuals in the population or among various species. Time scaling was based
on the equation, Cn × t = k, where n = 0.91 was derived by probit analysis of LC50 data for guinea pigs exposed to ethylenimine. The AEGL values are summarized in Table 8-1.
1.
INTRODUCTION
Propylenimine is an aziridine compound similar to that of ethylenimine (Ham 1981). Propylenimine is the second most important aziridine, the first being ethylenimine (Ham 1981). Propylenimine is a colorless oily liquid. One source stated that the odor of propylenimine is similar to that of aliphatic amines (fishy) (Trochimowicz et al. 1994), whereas other sources stated that propylenimine has an odor similar to that of ammonia (Ham 1981; RTECS 2008). Propylenimine is flammable, and it is an explosion hazard (Trochimowicz et al. 1994; HSDB 2006). Propylenimine is used to modify latex surface coating resins to improve adhesion, and propylenimine and its derivatives are used to modify bonding properties of textiles, paper, and dyes. It is also used in photography, in pharmaceutical industries, as an oil additive, in gelatins, and in organic syntheses (IARC 1975; Lewis 1993, Trochimowicz et al. 1994).
The data concerning the toxicity of propylenimine are very limited, necessitating a relative toxicity approach for deriving AEGL-values. Therefore, the AEGL values derived for ethylenimine served as the basis for deriving AEGLs for propylenimine. The physical and chemical properties of propylenimine are presented in Table 8-2.
2.
HUMAN TOXICITY DATA
2.1.
Acute Lethality
No reports were found on the acute lethality due to exposure to propylenimine.
2.2.
Nonlethal Toxicity
No epidemiologic or experimental studies, case reports, or anecdotal data concerning potential nonlethal toxicity, developmental/reproductive toxicity, cancer, or genotoxicity were located in the literature searched. However, the acute toxicity of propylenimine is likely to be similar to that of ethylenimine because of their similar chemical and physical properties. Humans exposed to ethylenimine in air experience skin, eye, and respiratory tract irritation, nausea, vomiting, headache, dizziness, and shortness of breath (Trochimowicz et al. 1994). However, concentrations, exposure durations, and specific routes of exposure were not presented in this report.
TABLE 8-1 AEGL Values for Propyleniminea,b [ppm (mg/m3)]
|
Classification |
10 min |
30 min |
1 h |
4 h |
8 h |
End Point (Reference) |
|
AEGL-1c (Nondisabling) |
Not recommendedd |
|
|
|
|
|
|
AEGL-2c (Disabling) |
83 (200) |
25 (58) |
12 (28) |
2.5 (5.8) |
1.2 (2.8) |
NOEL for extreme respiratory difficulty (Carpenter et al. 1948) |
|
AEGL-3c (Lethal) |
170 (398) |
50 (120 ) |
23 (54 ) |
5.1 (12) |
2.4 (5.6 ) |
Lethality threshold, Carpenter et al. 1948 |
|
aAEGL-2 and -3 values do not account for the potential cancer risk due to inhalation exposure to propylenimine. bEffects including death, irritation to eyes, and irritation to the respiratory tract may be delayed until after cessation of exposure. cAEGL values for propylenimine = AEGL for ethylenimine × 5 (relative potency factor) ÷ 2 (modifying factor). dThe absence of AEGL-1 values does not imply that exposures below the AEGL-2 levels are without adverse health effects. |
||||||
TABLE 8-2 Physical and Chemical Data for Propylenimine
2.3.
Summary
No human toxicity data were available for propylenimine. However, because of its structural and physical and chemical similarity to ethylenimine, propylenimine is expected to cause damage to the eye, respiratory tract, and skin, with onset of effects being delayed depending on the exposure concentration and duration.
3.
ANIMAL TOXICITY DATA
3.1.
Acute Lethality
3.1.1.
Rats
Carpenter et al. (1948) exposed groups of six rats (weight range 90-120 g) to 500 ppm of propylenimine for 5, 10, 15, 30, 60, 120, or 240 min; only five rats were exposed for 30 min. Although the report did not state it specifically, the animals were probably observed for 14 days, as was reported for studies with ethylenimine. No mortality occurred in groups exposed ≤120 min. Five of six rats exposed to 500 ppm for 240 min died. Other manifestations of toxicity were not specifically described; nevertheless, the authors stated that toxicity of propylenimine was similar to but only one-eighth as severe as that of ethylenimine. Toxicity of ethylenimine was manifested by extreme respiratory difficulty at all concentrations ≥25 ppm, but only after exposure for at least 3 h at 25 ppm. Prostration was observed at 250 ppm (3 h) or 500 ppm (2 h). Death was delayed in all cases with some animals dying more than 10 days after exposure. Gross examination revealed lung congestion and hemorrhage and congestion of all internal organs in animals that died. Microscopic examination of tissues and organs revealed necrosis of the renal tubular epithelium, congested lungs with leakage of fluid and blood into the bronchioles of animals that died. Cloudy swelling was observed in the kidneys of survivors.
3.1.2.
Mouse
The lethal concentration for propylenimine reported for mice exposed by inhalation was >2 g/m3 (856 ppm) for a 10-min exposure (RTECS 2008). No other details are known.
3.1.3.
Guinea pig
Groups of six guinea pigs (weight range 250-300 g) were exposed to 500 ppm of propylenimine for 5, 10, 30, 60, 120, or 240 min; only five guinea pigs were exposed for 120 min (Carpenter et al. (1948). Although the investigators did not state specifically, the animals were probably observed for 14 days, the
same as reported for studies with ethylenimine. No mortality occurred in groups exposed for 30 min. One of six, three of five, and six of six guinea pigs died after exposure for 60, 120, or 240 min, respectively. Other manifestations of toxicity were not specifically described; nevertheless, the report stated that toxicity of propylenimine was similar to but only one-fourth as potent as that of ethylenimine. Exposure to ethylenimine caused by extreme respiratory difficulty at all concentrations ≥25 ppm, but only after exposure for at least 3 h at 25 ppm. Prostration was observed after exposure to 250 ppm for 3 h or 500 ppm for 2 h. Deaths of guinea pigs exposed to ethylenimine were delayed in all cases with some animals dying more than 10 days after exposure. Gross examination revealed pulmonary congestion and hemorrhage and congestion of all internal organs in animals that died. Microscopic examination of tissues and organs revealed necrosis of the renal tubular epithelium, pulmonary congestion with leakage of fluid and blood into the bronchioles of guinea pigs that died, and cloudy swelling in the kidneys of survivors.
3.2.
Nonlethal Toxicity
No additional data were available on nonlethal effects of exposure to propylenimine other than discussed above.
3.3.
Developmental and Reproductive Toxicity
No data were available on potential developmental and reproductive toxicity after inhalation exposure to propylenimine in experimental animals.
3.4.
Carcinogenicity
The overall incidence of malignant tumors was markedly increased in groups of 26 male and 26 female Charles River CD rats (6 weeks of age) administered propylenimine by gavage (Ulland et al. 1971). The animals were administered doses of 20 or 10 mg/kg body weight twice weekly for 28 or 60 weeks, respectively; all animals were killed at week 60. At 20 mg/kg, 28 tumors were found in 22/52 (males and females combined) rats killed after 60 weeks: gliomas, ear-duct squamous cell carcinomas, intestinal adenocarcinomas, and leukemia in males and mammary tumors (primarily adenocarcinomas), gliomas, and miscellaneous tumors in females. At 10 mg/kg, 45 tumors were found in 37/52 rats killed at the end of the treatment period: gliomas, ear-duct squamous cell carcinomas, intestinal adenocarcinomas, leukemia, and miscellaneous tumors in males and mammary tumors, gliomas, ear-duct squamous cell carcinomas, and miscellaneous tumors in females. Six male and six females serving as controls were killed at 61 weeks; one pituitary adenoma was found. IARC (1975, 1999) evaluated the carcinogenicity data for propylenimine and concluded that the evidence for carcinogenicity was sufficient in experimental ani-
mals and classified the chemical propylenimine as possibly carcinogenic to humans (Group 2B).
3.5.
Genotoxicity
Speck and Rosenkranz (1976) demonstrated that propylenimine (1.5 μg/plate) was mutagenic in Salmonella typhimurium strain TA100 incubated under aerobic or anaerobic conditions. Vogel and Nivard (1997) determined that propylenimine was mutagenic in the sex-linked recessive lethal assay using repair-deficient Drosophila melanogaster.
3.6.
Summary
Propylenimine was lethal in rats after exposure to 500 ppm for 240 min, whereas it was lethal in guinea pigs after exposure to 500 ppm for 60, 120, or 240 min. The effects of propylenimine were similar to but less severe than those of ethylenimine. The data were not adequate for calculating LC50 values for either rats or guinea pigs. Propylenimine administered orally induces malignant tumors at multiple sites in rats exposed for 28 or 60 weeks. IARC (1975) classified propylenimine as Group 2B (possibly carcinogenic to humans) based on sufficient evidence in animals. Propylenimine is genotoxic in a variety of in vitro assay systems, including Salmonella typhimurium and Drosophila. No studies were available on nonlethal, developmental toxicity, or reproductive toxicity in animals.
4.
SPECIAL CONSIDERATIONS
4.1.
Metabolism, Disposition, and Kinetics
No data were available on absorption, tissue distribution, metabolism, or elimination of propylenimine in humans or experimental animals. However, ethylenimine is excreted primarily in urine after intraperitoneal injection of radiolabeled compound, with a small percentage eliminated in expired air (Wright and Rowe 1967). Ethylenimine showed a two-compartment elimination pattern; one compartment had a half-time of 16 h and the other had a half-time of 56 days. Tissue distribution showed uptake by all tissues with the greatest uptake (specific activity) in liver followed by cecum, spleen, kidneys, intestines, and bone marrow. Ethylenimine was metabolized to unknown substances primarily by a route that did not involve oxidation. In addition, either the parent compound or a metabolite that retained the aziridine ring reacted with tissue components (Wright and Rowe 1967). Absorption, distribution, and excretion of propylenimine are expected to be qualitatively similar to that of ethylenimine because the two compounds have similar structures, physical/chemical properties, and biological effects.
4.2.
Mechanism of Toxicity
No data were available on the mechanism of toxicity of propylenimine; however, because of its structural similarity to ethylenimine, propylenimine is likely a reactive alkylating agent, with signs of toxicity being delayed until after exposure is terminated (insidious) depending on the exposure concentration. The mechanism of toxicity of propylenimine is expected to be similar to that of ethylenimine and mustard compounds.
4.3.
Structure–Activity Relationship
Propylenimine is structurally similar to ethylenimine, and data for ethylenimine have been summarized (see Chapter 4). Briefly these data showed that exposure of an individual to a high, but unknown concentration of ethylenimine for no more than 5 min caused eye irritation, respiratory tract irritation, salivation, vomiting, breathlessness, pulmonary edema, and death (Gresham and West 1975). Exposure of humans to a nonlethal concentration for 1.5 to 2 h caused clinical signs that were delayed in onset: photophobia, very severe vomiting, and coughing. Clinical observations associated with acute exposure to ethylenimine included fever, conjunctival irritation, evidence of liver inflammation, transitory hemoconcentration, eosinophilia, mild albuminuria, extensive respiratory irritation manifested by decreased respiratory function, and ulceration of the posterior nasal cavity (Weightman and Hoyle 1964).
The effects in animals exposed to propylenimine by inhalation are similar to those described for ethylenimine. Morality results and LC50 values for rats and guinea pigs exposed to ethylenimine are presented in Table 8-3. Mice died after exposure to 1170 ppm ethylenimine for 10 min; the LC50 was 2236 ppm for a 10-min exposure (Silver and McGrath 1948).
4.4.
Other Relevant Information
4.4.1.
Species variability
Data were not available to assess the sensitivity of different species exposed to propylenimine. However, data for ethylenimine showed very little difference in response between rats and guinea pigs exposed to ethylenimine at similar concentrations and durations of exposure. Mice, rats, and guinea pigs showed the characteristic delayed mortality response after inhalation exposure to ethylenimine, and the clinical signs of toxicity in the three species were similar. Because of the chemical similar structure and physical properties of propylenimine and ethylenimine, the toxicity of propylenimine is also expected to be similar across species.
TABLE 8-3 Effects of Acute Exposure of Wistar Rats and Guinea Pigs to Ethylenimine
|
Exposure Duration (min) |
Species |
Exposure Concentration (ppm) |
Mortality Response |
LC50a(ppm) |
|
5 |
Rat |
100 |
0/6 |
2558 |
|
|
|
250 |
0/6 |
|
|
|
|
500 |
1/6 |
|
|
|
|
1000 |
1/5 |
|
|
|
|
4000 |
4/6 |
|
|
|
Guinea pig |
250 |
0/6 |
2906 |
|
|
|
500 |
0/6 |
|
|
|
|
1000 |
0/6 |
|
|
|
|
4000 |
4/6 |
|
|
10 |
Rat |
500 |
2/6 |
1407 |
|
|
|
1000 |
4/6 |
|
|
|
|
2000 |
1/6 |
|
|
|
|
4000 |
5/6 |
|
|
|
Guinea pig |
2000 |
1/12 |
2824 |
|
|
|
4000 |
6/6 |
|
|
15 |
Rat |
100 |
0/6 |
545 |
|
|
|
250 |
1/6 |
|
|
|
|
500 |
3/6 |
|
|
|
|
1000 |
5/6 |
|
|
|
|
2000 |
5/6 |
|
|
|
|
4000 |
6/6 |
|
|
|
Guinea pig |
250 |
0/6 |
1283 |
|
|
|
500 |
0/6 |
|
|
|
|
1000 |
0/6 |
|
|
|
|
2000 |
6/6 |
|
|
30 |
Rat |
500 |
5/6 |
Not determined |
|
|
|
1000 |
6/6 |
|
|
|
|
2000 |
5/5 |
|
|
|
Guinea pig |
100 |
0/6 |
364 |
|
|
|
250 |
0/6 |
|
|
|
|
500 |
5/6 |
|
|
|
|
1000 |
6/6 |
|
|
60 |
Rat |
100 |
0/6 |
268 |
|
|
|
250 |
2/6 |
|
|
|
|
500 |
6/6 |
|
|
|
Guinea pig |
25 |
0/12 |
235 |
|
|
|
100 |
1/6 |
|
|
|
|
250 |
2/6 |
|
|
|
|
500 |
6/6 |
|
|
120 |
Rat |
50 |
0/6 |
259 |
|
|
|
100 |
1/6 |
|
|
|
|
250 |
3/6 |
|
|
Exposure Duration (min) |
Species |
Exposure Concentration (ppm) |
Mortality Response |
LC50a(ppm) |
|
|
Guinea pig |
50 |
0/6 |
158 |
|
|
|
100 |
1/6 |
|
|
|
|
250 |
5/6 |
|
|
|
|
500 |
6/6 |
|
|
240 |
Rat |
25 |
0/6 |
58 |
|
|
|
50 |
2/5 |
|
|
|
|
100 |
6/6 |
|
|
|
|
250 |
6/6 |
|
|
|
Guinea pig |
10 |
0/6 |
45 |
|
|
|
25 |
2/5 |
|
|
|
|
50 |
2/6 |
|
|
|
|
100 |
6/6 |
|
|
|
|
250 |
6/6 |
|
|
480 |
Rat |
25 |
1/6 |
35 |
|
|
|
50 |
5/6 |
|
|
|
Guinea pig |
10 |
0/6 |
27 |
|
|
|
25 |
2/6 |
|
|
|
|
50 |
6/6 |
|
|
aLC50 values calculated by probit analysis. Source: Carpenter et al. 1948. Reprinted with permission; copyright 1948, American Medical Association. |
||||
4.4.2.
Susceptible Subpopulations
No data are available to assess the toxicity of propylenimine in potentially susceptible subpopulations.
4.4.3.
Concentration-Exposure Duration Relationship
Data were not available for evaluating the concentration-exposure duration relationship for propylenimine. For ethylenimine, a linear relationship was obtained for the log-log plot of LC50 concentration versus exposure duration for rats and guinea pigs. Because propylenimine and ethylenimine have similar chemical structures and activity; the concentration-exposure duration relationship is expected to be similar.
4.4.4.
Other Data
The oral LD50 for propylenimine is 19 mg/kg body weight (12-30 mg/kg) for rats dosed by gavage (Carpenter et al. 1948). The approximate LD50 is 0.043 mL/kg body weight when applied to the skin of guinea pigs and 0.005 mL when instilled in the eye of rabbits (Trochimowicz et al. 1994).
5.
DATA ANALYSIS AND AEGL-1
5.1.
Human Data Relevant to AEGL-1
No human data were available for deriving AEGL-1 values.
5.2.
Animal Data Relevant to AEGL-1
Animal data were not available for deriving AEGL-1 values.
5.3.
Derivation of AEGL-1
Data are not available for deriving AEGL-1 values for propylenimine; therefore, no values are recommended (Table 8-4).
6.
DATA ANALYSIS AND AEGL-2
6.1.
Human Data Relevant to AEGL-2
Human data were not available for deriving AEGL-2 values.
6.2.
Animal Data Relevant to AEGL-2
No animal data were available for deriving AEGL-2 values.
6.3.
Derivation of AEGL-2
The relative toxicity approach is utilized for deriving AEGL-2 values for propylenimine, because no data on propylenimine are available. Propylenimine is structurally similar to ethylenimine. The toxicity of propylenimine is qualitatively similar to that of ethylenimine; however, propylenimine is four to eight times less toxic than ethylenimine by the inhalation route (Carpenter et al. 1948). The data for relative toxicity are summarized in Table 8-5. In acute inhalation studies reported by Carpenter et al. (1948), propylenimine is four or five times less toxic than ethylenimine to the guinea pig and eight times less toxic to the rat. The relative potency factor of 5 is the geometric mean of the three relative toxicity values calculated from the inhalation studies with rats and guinea
TABLE 8-4 AEGL-1 Values for Propylenimine
|
Chemical |
10 min |
30 min |
1 h |
4 h |
8 h |
|
Propylenimine |
NR |
NR |
NR |
NR |
NR |
|
NR: Not Recommended. The absence of AEGL-1 values does not imply that exposures below the AEGL-2 are without adverse health effects. |
|||||
pigs. Therefore, a relative potency factor value of 5 was used to derive AEGL-2 values for propylenimine. Use of the geometric mean is common practice in toxicology when calculating means of values associated with risk assessments or other expressions of comparative toxicity. It is typically used because it does not give excessive weight to extreme values (“outliers”). It draws outliers toward the center of the distribution, decreasing the sensitivity of the parameter to undue influence of the outlier (Gad 2005). The geometric mean was used for the propylenimine AEGL-2 assessment because of the variability in relative potency values between ethyleneimine and propylenimine (relative potencies were 8-fold in a rat study and 4- and 5-fold in two guinea pig studies). The AEGL-2 values for ethylenimine (see Chapter 4) are presented below along with the currently derived values for propylenimine (Table 8-6). The AEGL-2 values for ethylenimine were derived from the no-observed-effect level for extreme respiratory difficulty in guinea pigs (10 ppm for 240 min); no deaths occurred at this concentration (Carpenter et al. 1948). Uncertainty factors of 3 for both interspecies sensitivity and intraspecies variability (total = 10) were applied to the exposure concentration (see Chapter 4). Scaling across time frames was accomplished using the equation, Cn × t = k, where the value n = 0.91 was calculated from LC50 data for guinea pigs exposed to ethylenimine for durations ranging from 5 to 480 min. A modifying factor of 2 was applied to account for additional uncertainty due to the deficient database for propylenimine; a larger value was not selected because the AEGL-2 values for propylenimine would approximate those of ethylenimine. Propylenimine is carcinogenic to animals exposed by the oral route; IARC (1975) classified propylenimine as Group 2B (possibly carcinogenic to humans). The AEGL-2 values do not take into consideration the potential carcinogenicity of propylenimine to humans.
TABLE 8-5 Relative Potency of Propylenimine and Ethylenimine in Laboratory Animals
|
Route |
Species |
Propylenimine |
Ethylenimine |
Relative Toxicity |
|
Inhalation |
Rata |
500 ppm for 240 min; mortality: 5/6 deaths |
500 ppm for 30 min; mortality: 5/6 deaths |
Ethylenimine 8 times more toxic |
|
|
Guinea piga |
500 ppm for 240 min; mortality: 6/6 deaths |
500 ppm for 60 min; mortality: 6/6 deaths |
Ethylenimine 4 times more toxic |
|
|
Guinea piga |
500 ppm for 60 min; mortality: 1/6 |
100 ppm for 60 min; mortality: 1/6 |
Ethylenimine 5 times more toxic |
|
|
Mouseb |
Lethal concentration >856 ppm for 10 min. |
899 ppm for 10 min; mortality: 3/20 |
Relative potency undetermined |
|
Skin |
0.043 mL/kg bw |
0.014 mL/kg bw |
Ethylenimine 3 times more toxic |
|
|
aCarpenter et al. 1948. bRTECS 2008; Silver and McGrath 1948. cTrochimowicz et al. 1994. |
||||
TABLE 8-6 AEGL-2 Values for Ethylenimine and Derived Values for Propyleniminea [ppm (mg/m3)]
7.
DATA ANALYSIS AND AEGL-3
7.1.
Human Data Relevant to AEGL-3
No human data were available for deriving AEGL-3 values.
7.2.
Animal Data Relevant to AEGL-3
Two acute inhalation studies were available for evaluating the toxicity of propylenimine. One study showed that exposure to propylenimine at a concentration of 500 ppm resulted in 5/6 deaths in rats exposed for 240 min and no deaths after exposure for 120 min (Carpenter et al. 1948). The other study showed that exposure to 500 ppm for 60 min resulted in 1/6 deaths in guinea pigs and 500 ppm for 30 min resulted in no deaths (Carpenter et al. 1948).
7.3.
Derivation of AEGL-3
A 30-min exposure to 500 ppm propylenimine was not lethal to guinea pigs; this concentration was used to derive AEGL-3 values. An uncertainty factor of 3 for interspecies differences and 3 for intraspecies variability (total uncertainty factor = 10) was applied to the no-effect levels for lethality. Effects experienced after exposure to propylenimine are expected to be qualitatively similar but less severe compared with the same concentration of ethylenimine. Therefore, the rationale for selecting uncertainty factors is the same as that used for ethylenimine (see Chapter 4). An interspecies uncertainty factor of 3 was selected because propylenimine is a very reactive direct-acting alkylating agent, and the AEGL-2 effects are expected to be confined to the respiratory tract. Respiratory tract damage appears to be due to a direct effect of an alkylating agent on the respiratory epithelium; this mechanism is expected to be similar among species (NRC 2003). The time of onset of signs and symptoms of exposure on the eyes and respiratory tract is expected to be delayed in both humans and animals. An uncertainty factor of 3 was applied for intraspecies variability because the effects appear to involve direct contact of the eyes or respiratory epithelium with a very reactive alkylating agent and these effects are not expected to differ considerably among members of the population. Studies have
shown that DNA damage is probably the initiating step in a cascade of events leading to cell damage (Papirmeister et al. 1985) after exposure to alkylating agents, and DNA damage can persist in respiratory and systemic organs following inhalation exposure to these agents (Rao et al. 1999). The resulting value was scaled across the relevant exposure durations using the equation: Cn × t = k, where n = 0.91 derived by regression analysis of LC50 data for guinea pigs exposed to ethylenimine. AEGL-3 values are presented in Table 8-7. Propylenimine is carcinogenic to animals exposed by the oral route; IARC (1975, 1999) classified propylenimine as Group 2B (possibly carcinogenic to humans). The AEGL-3 values do not take into consideration the potential carcinogenicity to humans.
8.
SUMMARY OF AEGLs
8.1.
AEGL Values and Toxicity End Points
AEGL values for propylenimine are summarized in Table 8-8. Human data were not available for deriving AEGL values and animal data were available only for AEGL-3 derivation.
Data were not available for deriving AEGL-1 values for propylenimine; therefore, no values are recommended. The absence of AEGL-1 values does not imply that exposure below AEGL-2 is without adverse health effects. Data were not available for deriving AEGL-2 values. Propylenimine is structurally and toxicologically similar to ethylenimine, AEGL-2 values were derived based on the relative potency compared with the AEGL values for ethylenimine. The relative potency factor was 5 was applied to the AEGL-2 values for ethylenimine, and then a modifying factor of 2 was applied to account for a limited database on propylenimine..
The AEGL-3 values were derived from the concentration (500 ppm) of propylenimine and the longest exposure duration that resulted in no deaths to guinea pigs. Uncertainty factors of 3 each for interspecies differences and 3 for interspecies variability (total = 10) were applied to the no-effect level for mortality.
8.2.
Comparison of AEGLs with Other Standards and Criteria
The recommended threshold limit value − B time-weighted-average (TLV-TWA) for propylenimine is 2 ppm (ACGIH 2000). This value was based on the comparison of toxicity to ethylenimine (TLV-TWA = 0.5 ppm). A skin notation was also recommended because of its similarity to ethylenimine (ACGIH 1991). The IDLH recommended by the National Institute for Occupational Safety and Health (NIOSH 1996) is 100 ppm based on the data of Carpenter et al. (1948,
1949). The Occupational Safety and Health (29 CFR 1910.1000 [1997]) permissible exposure level (PEL) for propylenimine is 2 ppm (5 mg/m3) with a skin designation. Table 8-9 compares existing standards and guidelines with the derived AEGL values. No other standards or guidelines have been published for propylenimine.
TABLE 8-7 AEGL-3 Values for Propylenimine [ppm (mg/m3)]
|
Chemical |
10 min |
30 min |
1 h |
4 h |
8 h |
|
Propylenimine |
170 (398) |
50 (120) |
23 (54) |
5.1 (12) |
2.4 (5.6) |
TABLE 8-8 AEGL Values for Propyleniminea,b [ppm (mg/m3)]
|
Classification |
10 min |
30 min |
1 h |
4 h |
8 h |
End Point (Reference) |
|
AEGL-1c |
No data available for deriving AEGL-1 values |
|||||
|
AEGL-2c |
83 (200) |
25 (58 ) |
12 (28 ) |
2.5 (5.8 ) |
1.2 (2.8) |
NOEL for extreme respiratory difficulty (Carpenter et al. 1948) |
|
AEGL-3c |
170 (398) |
50 (120 ) |
23 (58) |
5.1 (12 ) |
2.4 (5.6 ) |
Lethality threshold (Carpenter et al. 1948) |
|
aAEGL-2 and -3 values do not account for the potential cancer risk due to inhalation of propylenimine. bEffects including death, irritation to eyes, and irritation to the respiratory tract may be delayed until after exposure is terminated. cAEGL values for propylenimine = AEGL for ethylenimine × 5 (relative potency factor) ÷ 2 (modifying factor). |
||||||
TABLE 8-9 Extant Standards and Guidelines for Propyleneimine
|
Guideline |
Exposure Duration |
||||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
|
AEGL-1 |
No values were derived |
||||
|
AEGL-2 |
83 ppm |
25 ppm |
12 ppm |
2.5 ppm |
1.2 ppm |
|
AEGL-3 |
170 ppm |
50 ppm |
23 ppm |
5.1 ppm |
2.4 ppm |
|
IDLH (NIOSH)a |
NA |
500 ppm |
NA |
NA |
NA |
|
REL-TWA (NIOSH)b |
2 ppm (8-h TWA), skin designation |
||||
|
PEL-TWA (NIOSH)b |
2 ppm (8-h TWA); skin designation |
||||
|
TLV-TWA (ACGIH)c |
2 ppm (8-h), skin notation |
||||
|
MAK (Germany)d |
No value (carcinogenicity category 2: shown to cause cancer in animals; skin absorption) |
||||
|
MAC (the Netherlands)e |
0.63 mg/m3 (8-h) |
||||
|
aIDLH (Immediately Dangerous to Life and Health, National Institute of Occupational Safety and Health) (NIOSH 1996) represents the maximum concentration from which one could escape within 30 min without any escape-impairing symptoms, or any irreversible health effects. |
|||||
8.3.
Data Quality and Research Needs
Numerous data gaps exist concerning toxicity of propylenimine to humans and animals. Human studies are precluded because propylenimine is carcinogenic in animals and is, therefore, a suspect carcinogen in humans. Because the data set for deriving AEGL values for propylenimine was deficient, a relative toxicity approach was used to derive AEGL-2 values for propylenimine. The available data indicate that propylenimine is structurally and toxicologically similar to ethylenimine. Therefore, the relative potency approach was a reasonable approach for deriving the AEGL-2 values for propylenimine. Concentration-response data were not available for propylenimine; therefore, a no-effect level for lethality from a time-response study (rats and guinea pigs were exposed to the same concentration for different time periods) was used to derive the AEGL-3 values. Standard acute inhalation studies for propylenimine would provide definitive data for deriving AEGL-1, -2 and -3 values. A single-exposure carcinogenicity study would provide data for carcinogen risk assessment.
9.
REFERENCES
ACGIH (American Conference of Governmental Hygienists). 2000. P. 59 in Threshold Limit Values for Chemical Substances and Physical Agents and Biological Exposure Indices. American Conference of Governmental Hygienists, Cincinnati, OH.
Carpenter, C.P., H.F. Smyth, Jr., and C.B. Shaffer. 1948. The acute toxicity of ethylene imine to small animals. J. Ind. Hyg. Toxicol. 30(1):2-6.
Carpenter, C.P., H.F. Smyth, Jr., and U.C. Pozzani. 1949. The assay of acute vapor toxicity, and the grading and interpretation of results on 96 chemical compounds. J. Ind. Hyg. Toxicol. 31(6):343-346.
DFG (Deutsche Forschungsgemeinschaft). 2000. List of MAK and BAT Values 2000. Maximum Concentrations and Biological Tolerance Values at the Workplace Report No. 36. Weinheim, Federal Republic of Germany: Wiley VCH.
Gad, S.C. 2005. Basic principles. Pp. 5-20 in Statistics and Experimental Design for Toxicologists and Pharmacologists, 4th Ed. Boca Raton: CRC Press.
Gresham, G.A., and I.E. West. 1975. Injury and repair of tracheobronchial cartilage following accidental exposure to ethyleneimine. J. Clin. Pathol. 28(7):564-567.
Ham, G.E. 1981. Imines, cyclic. Pp. 142-166 in Kirk-Othmer Encyclopedia of Chemical Technology, Vol 13, 3rd Ed. New York: John Wiley & Son.
HSDB (Hazardous Substance Data Bank). 2006. Propylenimine. TOXNET, Specialized Information Services, U.S. National Library of Medicine, Bethesda, MD [online]. Available: http://toxnet.nlm.nih.gov/cgi-bin/sis/search/f?./temp/~1vBki0:1 [accessed Nov. 18, 2008].
IARC (International Agency for Research on Cancer). 1975. 2-Methylaziridine. Pp. 61-65 in Some Aziridines, N-, S- & O-Mustards and Selenium. IARC Monograph on the Evaluation of the Carcinogenic Risk of Chemicals to Man Vol. 9. Lyon, France: IARC.
IARC (International Agency for Research on Cancer). 1999. 2-Methylazaridine. Pp. 1497-1502 in Re-evaluation of Some Organic Chemicals, Hydrazine and Hydrogen Peroxide (Part 3). IARC Monograph on the Evaluation of Carcinogenic Risk to Humans Vol. 71. Lyon, France: IARC.
Lewis, R.J., Jr. 1993. P. 971 in Hawley’s Condensed Chemical Dictionary, 12th Ed. New York: Van Nostrand Reinhold Co.
MSZW (Ministerie van Sociale Zaken en Werkgelegenheid). 2004. Nationale MAC-lijst 2004:2-Methylaziridine. Den Haag: SDU Uitgevers [online]. Available: http://www.lasrook.net/lasrookNL/maclijst2004.htm [accessed Oct. 24, 2008].
NIOSH (National Institute of Occupational Safety and Health). 1996. Documentation for Immediately Dangerous to Life or Health Concentrations (IDLH): NIOSH Chemical Listing and Documentation of Revised IDLH Values (as of 3/1/95)-Propylene imine. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute of Occupational Safety and Health [online]. Available: http://www.cdc.gov/niosh/idlh/75558.html [accessed Nov. 18, 2008].
NIOSH (National Institute of Occupational Safety and Health). 2005. NIOSH Pocket Guide to Chemical Hazards: Propylene Imine. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute of Occupational Safety and Health, Cincinnati, OH. September 2005 [online]. Available: http://www.cdc.gov/niosh/npg/npgd0537.html [accessed Oct. 16, 2008].
NRC (National Research Council). 2003. Sulfur mustard (Agent HD). Pp. 301-383 in Acute Exposure for Selected Airborne Chemicals Vol. 3. Washington, DC: National Academies Press.
NRC (National Resource Council). 2001. Standing Operating Procedures for Developing Acute Exposure Guideline Levels for Hazardous Chemicals. Washington, DC: National Academy Press.
NRC (National Research Council). 1993. Guidelines for Developing Community Emergency Exposure Levels for Hazardous Substances. Washington, DC: National Academy Press.
Papirmeister, B., C.L. Gross, H.L. Meier, J.P. Petrali, and J.B. Johnson. 1985. Molecular basis for mustard-induced vesication. Fundam. Appl. Toxicol. 5(6 Pt. 2):S134-S149.
Rao, P.V.L., R. Vijayaraghavan, and A.S. Bhaskar. 1999. Sulphur mustard induced DNA damage in mice after dermal and inhalation exposure. Toxicology 139(1-2):39-51.
RTECS (Registry of Toxic Effects of Chemical Substances). 2008. Aziridine, 2-methyl. RTECS No. CM8050000. National Institute for Occupational Safety and Health [online]. Available: http://www.cdc.gov/niosh/rtecs/cm7ad550.html [accessed Nov. 18, 2008].
Silver, S.D., and F.P. McGrath. 1948. A comparison of acute toxicities of ethylene imine and ammonia to mice. J. Ind. Hyg. Toxicol. 30(1):7-9.
Speck, W.T., and H.S. Rosenkranz. 1976. Mutagenicity of azathioprine. Cancer Res. 36(1):108-109.
Trochimowicz, H.J., G.L. Kennedy, Jr., N.D. Krivanek. 1994. Heterocyclic and miscellaneous nitrogen compounds. Pp. 3285-3521 in Patty’s Industrial Hygiene and Toxicology, Vol. IIB Toxicology, 4th Ed., G.D. Clayton, and F.E. Clayton, eds. New York: John Wiley & Sons, Inc.
Ulland, B., M. Finkelstein, E.K. Weisburger, J.M. Rice, and J.H. Weisburger. 1971. Carcinogenicity of industrial chemicals propylene imine and propane sultone. Nature 230(5294):460-461.
Vogel, E.W., and M.J. Nivard. 1997. The response of germ cells to ethylene oxide, propylene oxide, propylene imine and methyl methanesulfonate is a matter of cell stage-related DNA repair. Environ. Mol. Mutagen. 29(2):124-135.
Weightman, J., and J.P. Hoyle. 1964. Accidental exposure to ethylenimine and N-ethylethylenimine vapors. JAMA 189:543-545.
Wright, G.J., and V.K. Rowe. 1967. Ethylenimine: Studies of the distribution and metabolism in the rat using carbon-14. Toxicol. Appl. Pharmacol. 11(3):575-584.
APPENDIX A
Derivation of AEGL Values for Propylenimine
DERIVATION OF AEGL-3
|
Key Study: |
Carpenter et al. 1948 |
|
Toxicity End Point: |
Lethality: NOEL for lethality: 500 ppm for 30 min |
|
Time Scaling: |
Cn × t = k; n = 0.91 based on regression analysis of the guinea pig data. |
|
|
C = 500 ppm/10 (uncertainty factor) = 50 ppm Cn × t = k; C = 50 ppm, t = 30 min, n = 0.91 k = 1054.8336 ppm minutes |
|
Uncertainty Factors: |
Total = 10: 3 for interspecies sensitivity, because propylenimine a reactive direct-acting alkylating agent, and the AEGL-2 effects are expected to be confined to the respiratory tract. Respiratory tract damage appears to be due to direct effect of an alkylating agent on the respiratory epithelium; this mechanism is expected to be similar among species. The time of onset of signs and symptoms of exposure on the eyes and respiratory tract is expected to be delayed in both humans and animals. |
|
|
3 for intraspecies variability, because the effects appear to involve direct contact of the eyes or respiratory epithelium with a very reactive alkylating agent. Studies have shown that DNA damage is probably the initiating step in a cascade of events leading to cell damage after exposure to alkylating agents, and DNA damage is persistent in respiratory and systemic organs following inhalation exposure to these agents. The alkylating activity of propylenimine is not expected to vary appreciably among individuals in the population |
|
Calculations: |
|
|
10-min AEGL-3 |
C = (k/t)1/0.91 = (1054.8 ppm minutes/10 min)1/0.91 = 170 ppm |
|
30-min AEGL-3 |
C = (k/t)1/0.91 = (1054.8 ppm minutes/30 min)1/0.91 = 50 ppm |
|
1-h AEGL-3 |
C = (k/t)1/0.91 = (1054.8 ppm minutes/60 min)1/0.91 = 23 ppm |
|
4-h AEGL-3 |
C = (k/t)1/0.91 = (1054.8 ppm minutes/240 min)1/0.91 = 5.1 ppm |
|
8-h AEGL-3 |
C = (k/t)1/0.91 = (1054.8 ppm minutes/480min)1/0.91 = 2.4 ppm |
APPENDIX B
Carcinogenicity Assessment
QUANTITATIVE CANCER ASSESSMENT FOR PROPYLENIMINE
Only one carcinogenicity study (Ulland et al. 1971) was available for propylenimine. In this study rats were administered propylenimine by gavage at two different doses for different time periods. The study author combined incidences of tumors at different anatomical sites but provided no data indicating that the incidence of each tumor type was related to administration of propylenimine. Therefore, these data are not suitable for a dose-response assessment of propylenimine.
APPENDIX C
Derivation Summary of AEGL Values for Propylenimine
AEGL-1 VALUES
|
30 min |
1 h |
4 h |
8 h |
|
Not recommended. |
|||
|
Reference: Not applicable. |
|||
|
Test Species/Strain/Number: Not applicable. |
|||
|
Exposure Route/Concentration/Durations: Not applicable. |
|||
|
Effects: Not applicable. |
|||
|
End Point/Concentration/Rationale: Not applicable. |
|||
|
Uncertainty Factors/Rationale: Not applicable. Total uncertainty factor: Not applicable. Interspecies: Not applicable. Intraspecies: Not applicable. |
|||
|
Modifying Factor: Not applicable. |
|||
|
Animal to Human Dosimetric Adjustment: Not applicable. |
|||
|
Time Scaling: Not applicable. |
|||
|
Data Quality and Support for AEGL Values: No data are available. |
|||
AEGL-2 VALUES
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
83 (200) |
25 (58) |
12 (28) |
2.5 (5.8) |
1.2 (2.8) |
|
Reference: Carpenter, C.P., H.F. Smyth, Jr., and C.B. Shaffer. 1948. The acute toxicity of ethylene imine to small animals. J. Ind. Hyg. Toxicol. 30(1):2-6 (see Chapter 4). |
||||
|
Test Species/Strain/Number: Ethylenimine: male guinea pigs, 6 per group. |
||||
|
Exposure Route/Concentration/Durations: Ethylenimine: inhalation; 10, 25, 50, 100, or 250 ppm for 240 min. |
||||
|
Effects: Ethylenimine: Guinea pigs were exposed for 240 min. Clinical signs: eye and respiratory irritation, and extreme respiratory difficulty at 25-250 ppm; prostration at 250 ppm; no effects at 10 ppm. Gross pathologic effects: congestion and hemorrhage in the lungs, congestion in all internal organs at 25-250 ppm; no effects at 10 ppm. Microscopic effects: lung congestion leakage of fluid and red blood cells into bronchioles, tubular necrosis and cloudy swelling in the kidneys at 25-250 ppm; no effects at 10 ppm. Mortality: 10 ppm, (0/6), 25 ppm (2/6), 50 ppm (2/6), 100 ppm (6/6), and 250 ppm (6/6). |
||||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
83 (200) |
25 (58) |
12 (28) |
2.5 (5.8) |
1.2 (2.8) |
|
End Point/Concentration/Rationale: Ethylenimine: No-effect-level for lethality in the guinea pig, 10 ppm exposure for 4 h; effects at 25 ppm and higher were above the definition for AEGL 2. The AEGL-2 values for propylenimine were derived based on the relative potency approach. Ethylenimine is five times more toxic than propylenimine. |
||||
|
Uncertainty Factors/Rationale: Ethylenimine Total uncertainty factor: 10 Interspecies: 3 - ethylenimine is a very reactive direct-acting alkylating agent, and the AEGL-2 effects would be confined to the respiratory tract. Respiratory tract damage appears to be due to the direct effect of an alkylating agent on the respiratory epithelium, and this mechanism is not expected to be different among species. Humans and animals exhibit delays between the time of exposure and the onset of symptoms and the eyes and respiratory tract are the most sensitive targets in both species. Intraspecies: 3 - the effects appear to involve direct contact of the eyes or respiratory epithelium with a very reactive alkylating agent. Studies have shown that DNA damage is likely the initiating step in a cascade of events leading to cell damage and DNA damage is persistent in respiratory and systemic organs following inhalation exposure to alkylating agents. |
||||
|
Modifying Factor: 2 because of a deficient database. |
||||
|
Animal to Human Dosimetric Adjustment: 1 |
||||
|
Time Scaling: Ethylenimine: Cn × k = t, where n = 0.91 derived empirically from guinea pig LC50 data with exposure times ranging from 5 min to 480 min. |
||||
|
Data Adequacy: No acute toxicity data for propylenimine were available for deriving AEGL-2 values. Therefore, AEGL-2 values for propylenimine were derived by the relative potency method; a relative potency of 5 was selected for propylenimine (based on lethality data, propylenimine was considered to be 5 times less toxic than ethylenimine). The resulting AEGL values were reduced by a factor of 2 because of a deficient database. The AEGL 2 values for ethylenimine were 33, 9.8, 4.6, 1.0, and 0.47 for 30 min, 1 h, 4 h, and 8 h, respectively. |
||||
AEGL-3 VALUES
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
170 (398) |
50 (120) |
23 (54) |
5.1 (12) |
2.4 (5.6) |
|
Key Reference: Carpenter, C.P., H.F. Smyth, Jr., and C.B. Shaffer. 1948. The acute toxicity of ethylene imine to small animals. J. Ind. Hyg. Toxicol. 30(1):2-6. |
||||
|
Test Species/Strain/Number: guinea pig/6 per group. |
||||
|
Exposure Route/Concentration/Durations: inhalation/500 ppm for 5, 10, 30, 60, 120, or 240 min. |
||||
|
Effects: lethality, 1/6, 3/5, 6/6 at 60, 120, and 240 min, respectively; no deaths after exposures ≤30 min. |
||||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
170 (398) |
50 (120) |
23 (54) |
5.1 (12) |
2.4 (5.6) |
|
End Point/Concentration/Rationale: no effect level for lethality |
||||
|
Uncertainty Factors/Rationale: Total uncertainty factor: 10 Interspecies: 3 - propylenimine is a reactive direct-acting alkylating agent, and the AEGL-3 effects are expected to be confined to the respiratory tract. Respiratory tract damage appears to be due to the direct effect of an alkylating agent on the respiratory epithelium; this mechanism is expected to be similar among species. The time of onset of signs and symptoms of exposure on the eyes and respiratory tract is expected to be delayed in both humans and animals. Intraspecies: 3 - the effects appear to involve direct contact of the eyes or respiratory epithelium with a very reactive alkylating agent. Studies have shown that DNA damage is probably the initiating step in a cascade of events leading to cell damage after exposure to alkylating agents, and DNA damage is persistent in respiratory and systemic organs following inhalation exposure to these agents. |
||||
|
Modifying Factor: 1 |
||||
|
Animal to Human Dosimetric Adjustment: None applied. |
||||
|
Time Scaling: Cn × k = t, where n = 0.91 derived empirically from LC50 data in which guinea pigs were exposed to ethylenimine for times ranging from 5 min to 480 min. |
||||
|
Data Adequacy: Very few data were available for deriving AEGL-3 values for propylenimine. A standard acute lethality study has not been conducted for propylenimine; therefore, the time-response study in which rats and guinea pigs were exposed to 500 ppm for different time periods was used to derive AEGL-3 values. The data for the guinea pig showed that this species is more sensitive to propylenimine exposure than the rats; lethality occurred after exposure of guinea pigs to 500 ppm for 60 min and after exposure of rats to 500 ppm for 240 min. |
||||