Draft Strategic National Vaccine Plan
Table of Contents
Page
Preface: Letter from the Assistant Secretary for Health
Given the importance of vaccines and immunizations in the prevention of an expanding number of infectious diseases, it is important that we – as a Department and as a Nation --clearly articulate our vision for the vaccine and immunization enterprise. In my role as the Assistant Secretary for Health and the Director of the National Vaccine Program, I have directed and monitored the efforts to update the 1994 National Vaccine Plan
The accompanying draft strategic National Vaccine Plan reflects current priorities and potential future directions for the next decade. This draft Plan offers a clear signal about our goals for vaccines and immunizations to our domestic and international partners in the United States vaccine and immunization enterprise and abroad, both public and private in the United States and abroad. It is a strong beginning, but requires extensive consultation with and input from many partners, including the public. This input will also enable us to develop an implementation plan with discrete activities and measurable milestones. With release of this draft Plan, consultations with stakeholders will begin, led by the National Vaccine Program Office and the National Vaccine Advisory Committee. We all look forward to the final products of these efforts in late 2009.
This draft strategic National Vaccine Plan responds to the mandate of Congress contained in P.L. 99-660, in which the vision of the National Vaccine Program was first outlined. The first National Vaccine Plan had fourteen expected outcomes, most of which have been achieved at least in part. However, the world of vaccines has changed dramatically since 1994, with more diseases for which vaccines could be available, multiple new research tools, many available new vaccines, heightened interest in vaccine safety, new communications tools, and many more people for whom vaccines are routinely recommended.
This draft Plan describes proposed strategies for ways in which the United States can promote immunization to protect the health of all people. Subsequent work with our many partners will enhance and improve this draft, so the final plan can implement strategies to assure all people can benefit from the prevention of infectious diseases.
Joxel Garcia, M.D., M.B.A.
Assistant Secretary for Health And Director, National Vaccine Program
Acronyms and Abbreviations
AAFP American Academy of Family Physicians
AAP American Academy of Pediatrics
ACIP Advisory Committee on Immunization Practices
AEFI adverse events following immunization
ASPA Assistant Secretary for Public Affairs (Department of Health and Human Services)
ASPR Assistant Secretary for Preparedness and Response (Department of Health and Human Services)
BARDA Biomedical Advanced Research and Development Authority
BLA Biologics License Application
CDC Centers for Disease Control and Prevention
CBER Center for Biologics Evaluation and Research
CMS Centers for Medicare and Medicaid Services
DARPA Defense Advanced Research Projects Agency
DoD Department of Defense
DHS Department of Homeland Security
DoJ Department of Justice
DTP diphtheria, tetanus toxoids, and pertussis vaccine
DTaP diphtheria, tetanus toxoids, and acellular pertussis vaccine
DTRA Defense Threat Reduction Agency
DVIC Division of Vaccine Injury Compensation
FDA Food and Drug Administration
FY fiscal year
GAVI Global Alliance for Vaccines and Immunization, now formally known as the GAVI Alliance
GMP Good Manufacturing Practices
HBV Hepatitis B virus
HEDIS Healthcare Effectiveness Data and Information Set
HHS U.S. Department of Health and Human Services
Hib Haemophilus influenzae type b
HIV human immunodeficiency virus
HP2010 Healthy People 2010
HRSA Health Resources and Services Administration
IDSA Infectious Diseases Society of America
IHS Indian Health Service
IOM Institute of Medicine
IPV inactivated polio vaccine
MCOs managed care organizations
MMR measles, mumps, and rubella virus vaccine (combined)
NCIRD National Center for Immunization and Respiratory Diseases
NCPDCID National Center for Preparedness, Detection, and Control of Infectious Diseases
NGO Non-governmental organization
NIAID National Institute of Allergy and Infectious Diseases
NIH National Institutes of Health
NVAC National Vaccine Advisory Committee
NVP National Vaccine Program
NVPO National Vaccine Program Office
OD Office of the Director
OMHHD Office of Minority Health and Health Disparities
OPDIV Operating Division of the Department of Health and Human Services (e.g., CDC)
OPV oral polio vaccine
PHEMCE Public Health Emergency Medical Countermeasures Enterprise
PHS Public Health Service
P.L. public law
STAFFDIV Staff Division of the Department of Health and Human Services (e.g., Office of Public Health and Science, of which NVPO is included)
TB tuberculosis
Td tetanus and diphtheria toxoids (adult formulation)
Tdap tetanus and diphtheria toxoids, and acellular pertussis vaccine (adult formulation)
UNICEF United Nations International Children’s Emergency Fund (now United Nation’s Children’s Fund)
USAID U.S. Agency for International Development
USAMRIID U.S. Army Medical Research Institute of Infectious Diseases
VA Department of Veterans Affairs
VAERS Vaccine Adverse Event Reporting System
VFC Vaccines for Children program
VHA Veterans Health Administration
VICP National Vaccine Injury Compensation Program
VPD vaccine-preventable disease
VRC Vaccine Research Center
VSD Vaccine Safety Datalink project
Executive Summary
The National Vaccine Program was established in 1986 to achieve optimal prevention of infectious diseases through immunization and optimal prevention of adverse reactions to vaccines. The purpose of the National Vaccine Plan is to promote achievement of the National Vaccine Program mission by providing strategic direction and promoting coordinated action by vaccine and immunization enterprise stakeholders.
Federal involvement in civilian and military vaccination programs is longstanding, including in research and development, regulation, vaccine delivery and the evaluation of the impacts of immunizations. This draft strategic National Vaccine Plan builds on the many achievements of the vaccine and immunization enterprise prior to and since the establishment of the National Vaccine Program in 1986 and the completion of the first National Vaccine Plan in 1994. New vaccine preventable disease targets have been identified and new vaccines have been developed and licensed; many of these new vaccines are now recommended for children, adolescents and adults. These new vaccines have expanded the number of infections that can be prevented, and more effectively and safely prevent some diseases for which earlier generation vaccines already existed. In addition, federal immunization financing programs have reduced or eliminated many financial barriers to immunizations, particularly for children. The number of infections that are preventable by vaccination has decreased significantly while vaccination coverage in the United States has increased, and coverage for many vaccines has reached record levels. More robust systems have been developed to identify adverse events following immunization and to assess potential associations of those events with vaccination. Globally, the United States has worked with multilateral and bilateral partners and non-governmental organizations in contributing to improvements in child health status and the prevention of hundreds of thousands of child deaths each year through improved vaccine coverage and introduction of new vaccines. Of the fourteen anticipated outcomes included in the 1994 National Vaccine Plan, most were substantially or fully realized (see Appendix 1).
Despite these successes, however, many of the challenges that stimulated establishment of the National Vaccine Program and the development of the 1994 National Vaccine Plan remain relevant today. Vaccine shortages have frequently been experienced for many routinely recommended vaccines. Despite improved vaccination coverage among children, the occurrence of several recent vaccine preventable disease outbreaks serves as a reminder that these diseases still occur. Among older adults both vaccination coverage and the effectiveness of some routinely recommended vaccines remain sub-optimal. As the number of vaccines has increased and vaccine preventable diseases have declined, vaccine safety concerns are expressed more prominently today and may be more widely shared. Enhancing the current vaccine safety system is important to keep pace with several factors influencing it: an increasing number of vaccines and vaccine combinations, expanding target populations, and a better understanding of human biology, especially the human immune system. As the cost of vaccination has increased, financial barriers to vaccination have emerged for health departments, healthcare providers, and the public. Significant scientific challenges remain in the development of
safe and effective vaccines against existing global health threats, such as HIV, TB and malaria. Vaccines that have been developed and are in use in industrialized countries have the potential to make major contributions to health in developing countries, but are being underused. Additionally, emerging and pandemic infections and bioterrorist threats pose new challenges for vaccine development and regulation, manufacturing, vaccine delivery and access in the US and abroad.
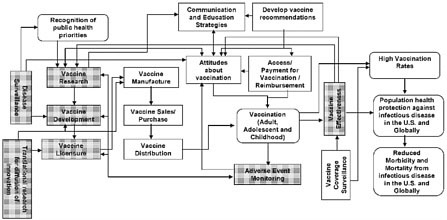
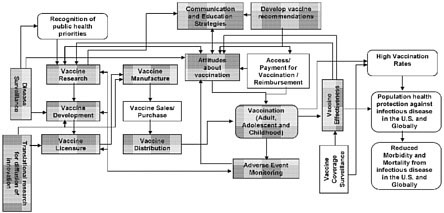
In the context of the many challenges and opportunities that exist, updating the 1994 National Vaccine Plan is an opportunity to provide a strategic focus for the nation’s efforts to improve disease prevention and enhance vaccine safety. This draft strategic National Vaccine Plan is primarily the result of deliberation, analysis, and input from multiple Federal agencies under the coordination of the National Vaccine Program Office (NVPO). A committee empanelled by the National Academy of Sciences’ Institute of Medicine (IOM) reviewed the 1994 National Vaccine Plan and provided guidance on the development of the updated Plan (see Appendix 2). Because successfully preventing infectious diseases and enhancing vaccine safety are outcomes of a complex process, identifying objectives and strategies that lead to and sustain these outcomes is facilitated by understanding the many interconnected determinants of these outcomes. A framework that identifies components of the vaccine and immunization enterprise and illustrates their interrelationships is shown in Figure 1. While a simplification of a complex system, this framework provides an overview of key processes from beginning to end (critical components are shown as rectangular boxes and intermediate and long term outcomes as rounded boxes). The intermediate and long term inputs and outcomes of the vaccine and immunization enterprise include the Recognition of public health priorities, Vaccination (adult, adolescent and childhood), High vaccination rates, Population health protection against infectious disease in the U.S. and globally, and Reduced morbidity and mortality from infectious diseases in the U.S. and globally. The critical components of the vaccine and immunization enterprise that contribute to achieving the desired outcomes include Translational research for diffusion of innovation, Disease surveillance, Vaccine research, Vaccine development, Vaccine licensure, Vaccine manufacture, Vaccine sales/purchase, Vaccine distribution, Communications and education strategies, Attitudes about vaccination, Develop vaccine recommendations, Access/payment for vaccination reimbursement, Adverse event monitoring, Vaccine effectiveness, and Vaccine coverage surveillance.
Figure 1. Overview of the vaccine and immunization enterprise
The goals, objectives, and strategies included in this plan were drafted by Federal departments and agencies for which vaccines and immunization are a significant part of their mission and provide a comprehensive approach to reach these outcomes through coordinated efforts of Federal, State, local, multinational and non-governmental stakeholders. Recognizing that success is facilitated by careful planning that includes defining specific activities, milestones and measurable outcomes, an implementation plan will be developed based on this draft plan. With a ten-year horizon, the framework balances a strategic vision, which requires development and implementation of new initiatives, with the recognition that changing circumstances and new opportunities and challenges will occur over the next decade. The ten-year horizon also allows incorporation of the HealthyPeople 2020 objectives once those are established. Annual monitoring of progress and a mid-course review will promote both accountability and flexibility.
The Plan is built around the achievement of five broad goals:
Goal 1: Develop new and improved vaccines
Goal 2: Enhance the safety of vaccines and vaccination practices
Goal 3: Support informed vaccine decision-making by the public, providers, and policy-makers
Goal 4: Ensure a stable supply of recommended vaccines and achieve better use of existing vaccines to prevent disease, disability and death in the United States
Goal 5: Increase global prevention of death and disease through safe and effective vaccination
These goals will be achieved by pursuing objectives and strategies that address each of the key determinants of those outcomes. Success in achieving these goals will be assessed by tracking progress in achieving measurable outcomes (“indicators”) associated with each goal. Final definition of the indicators and the development of specific numeric targets will occur through further consultation with stakeholders and the IOM Committee.
The indicators for each of the goals are listed below (Table 1):
Table 1. Measurable Indicators by Goal in the draft strategic National Vaccine Plan
|
|
|
|
Goal 1: Develop new and improved vaccines |
• Within one year, create an evidence-based list of new vaccine targets to prevent infectious diseases that are high priorities for development. |
|
|
• Identify X candidate vaccines (e.g., for HIV, malaria, TB, and a cross-protective vaccine for influenza) and advance Y priority vaccine candidates along the research and development pipeline including Z candidates into advanced clinical trials. |
|
|
• Advance X new delivery strategies that will improve effectiveness, feasibility, acceptability, safety, or ease of administration of new or improved vaccines into clinical trials. |
|
|
• In X years, have the capability to test potential vaccine candidates in clinical trials developed in response to an emerging infectious disease health threat within six months of the identification of the need for a vaccine. |
|
Goal 2: Enhance the safety of vaccines and vaccination practices |
• Conduct and disseminate the results of active and passive surveillance-based safety assessments for newly recommended vaccines or for vaccines with expanded recommendations: |
|
|
|
|
|
• Develop and disseminate plans for further investigation, if any, of newly detected AEFI signals |
|
|
and the rationale for those plans within X months of signal detection. |
|
|
• By X year, X % of infants, children, adolescents, adults, and pregnant women will be under active surveillance for AEFls |
|
|
• Conduct research to explore host factors and biological mechanisms associated with serious AEFIs and annually report results to the Assistant Secretary for Health, vaccine advisory committees, vaccine policy makers and other stakeholders |
|
Goal 3: Support informed vaccine decision-making by the public, providers, and policy-makers |
• Enhance communication with stakeholders and the public to more rapidly inform them (within _X_ days) about urgent and high-priority vaccine and vaccine-preventable disease issues (e.g., outbreaks, supply shortages, vaccine safety concerns). |
|
|
• _ X_ % of the public will report that they are satisfied with how their health care provider answers their questions about the benefits and risks of vaccines by Y (year). |
|
|
• _ X_ % of the public will report they have access to information which allows them to make informed vaccination decisions for themselves or their children by Y (year). |
|
|
• _X_% of health care providers will report that they have access to accurate and complete information about vaccine benefits and risks and are able to adequately answer questions of parents and patients by Y (year). |
|
|
• _ X_ % of key decision- and policy-makers will report they have access to vaccine benefits, risks, and costs to make informed decisions about vaccine policy by Y (year). |
|
|
• By Y (year) all health professional schools and training programs will include vaccine and vaccine-preventable disease content in their curricula, and assess students’ and trainees’ knowledge. |
|
|
• By Y (year) all relevant health professional certifying examinations will include vaccine and vaccine-preventable disease questions. |
|
Goal 4: Ensure a stable supply of recommended vaccines and achieve better use of existing vaccines to prevent disease, disability and death in the United States |
• The United States will have 6 months’ supply of all vaccines appropriate to stockpile. |
|
• Reduce financial and nonfinancial access barriers, such as cost, availability, and language, to immunization by 2020 so that: |
|
|
○ _X_% of parents of infants and children report no barriers to immunization; |
|
|
|
|
|
• Reach or exceed HealthyPeople 2020 vaccine coverage levels once established, through incrementally increasing coverage rates for pediatric, adolescent and adult populations using coverage levels in 2010 as a baseline. |
|
|
• X% of electronic health record systems and Y% of immunization information systems will include reminder and recall systems for vaccination by Y (year). |
|
|
• Within Y years after its ACIP recommendation, surveillance for at least one major disease outcome for each routinely recommended vaccine will be implemented in X% of states. |
|
|
• The Vaccine Injury Table is updated as needed (at least every X years). |
|
Goal 5: Increase global prevention of death and disease through safe and effective vaccination |
• Transmission of wild polio virus will be eradicated by Y (year). |
|
|
• Mortality from measles will be reduced by X% by Y (year) compared with an X (year) baseline. |
|
|
• X% of countries will achieve DTP3 vaccination coverage of 90% or greater nationally (and 80% or greater in each country’s district) by Y (year). Support introduction of new vaccines as part of national vaccination programs: |
|
|
|
|
|
• X countries establish immunization advisory committees by Y (year) that make evidence-based decisions on adding new vaccines to the routine program and monitor program quality, vaccination coverage, and vaccine safety. |
|
|
• X countries enhance injection safety by Y (year) through the use of auto-disable syringes or other safe injection devices (e.g., needle free delivery) for all immunizations. |
The current draft strategic National Vaccine Plan is based largely on input received from Federal Departments and agencies. Recognizing that success can best be achieved through a national plan that includes coordinated action by public and private sector stakeholders in pursuit of the Plan’s goals, extensive outreach and consultation will be implemented as the Plan is finalized. An IOM committee is holding a series of national meetings focused around each of the goals in which perspectives from many of the stakeholders will be obtained. Following these meetings, the IOM committee will prepare a report that includes conclusions and recommendations about priority actions within major components of the Plan. The National Vaccine Advisory Committee, a Federal advisory committee that includes representatives from many of the key vaccine and immunization enterprise stakeholders, also will implement a process to obtain input from a wide range of stakeholder groups. This input will include comments on this draft Plan and additional strategies that they can contribute to achieve Plan goals. In addition, input from the public will also be solicited to identify priority areas from their perspective. This draft will serve as the basis for the development of the updated National Vaccine Plan and based on this range of input, indicators of measurable outcomes will be determined and priorities will be presented. In addition, an implementation plan will be drafted that identifies specific actions that will be undertaken by government and other vaccine and immunization enterprise stakeholders to achieve the objectives and strategies in the plan and milestones will be established that will allow progress to be measured.. The updated National Vaccine Plan and an accompanying implementation plan is expected to be completed by fall 2009.
Introduction
Federal involvement in civilian and military vaccination programs is longstanding including research and development, regulation, vaccine delivery and the evaluation of the impacts of immunizations. The National Vaccine Program was established by Congress (Title XXI of the Public Health Service Act [Public Law 99-660]) in 1986 to achieve optimal prevention of infectious diseases through immunization and optimal prevention of adverse reactions to vaccines. The Act called for the development of a National Vaccine Plan to guide activities in pursuit of program goals. This initial plan, completed in 1994, defined goals, objectives and strategies to achieve the Program’s mission through coordinated action by Federal agencies, State and local governments, and private sector partners including manufacturers and healthcare providers. The four goals of the 1994 National Vaccine Plan were: 1) Develop new and improved vaccines; 2) Ensure the optimal safety and effectiveness of vaccines and immunization; 3) Better educate the public and members of the health professions on the benefits and risks of immunizations; and 4) Achieve better use of existing vaccines to prevent disease, disability, and death.
The National Vaccine Program was established in an environment characterized by U.S. outbreaks of several vaccine preventable diseases, increased public concern regarding vaccine safety, and liability concerns among vaccine manufacturers that had led some companies to stop producing vaccines for the U.S. market. Since 1986, there have been substantial changes in the nation’s vaccine program aimed at addressing these issues. The National Vaccine Injury Compensation Program, created through the same legislation as the National Vaccine Program, established a Federal claims process for persons experiencing adverse events following immunization. New vaccine recommendations and increased vaccination coverage among infants and children have reduced the risk of vaccine preventable disease outbreaks and reduced morbidity and mortality from infectious diseases that are now effectively prevented by vaccination. New vaccines have also been licensed by the U.S. Food and Drug Administration (FDA) that result in fewer adverse events compared with previously recommended vaccines (e.g., DTaP), improve effectiveness (e.g., conjugate Hib and pneumococcal vaccines), and expand the number of diseases that can be prevented in children, adolescents, and adults (e.g., rotavirus, varicella, human papillomavirus, zoster). Release of the 1994 National Vaccine Plan1 coincided with passage of the Vaccines for Children (VFC) amendments to Medicaid (Public Law 103-66), which provides for Federal vaccine purchase for children enrolled in Medicaid, uninsured children, American Indians and Alaska Natives, and underinsured children served through Federally Qualified Health Centers. Through these and other achievements, substantial progress has been made in addressing National Vaccine Program objectives and National Vaccine Plan goals (see Appendix 1).
Despite the successes that have been achieved in disease prevention and enhancement of vaccine safety, many of the challenges that stimulated establishment of the National Vaccine Program and led to the goals defined in the 1994 National Vaccine Plan remain relevant today. Although the specific issues are different, many have concerns about
|
1 |
See http://www.hhs.gov/nvpo/vacc_plan/ for the 1994 National Vaccine Plan |
vaccination and vaccine safety, and new communications channels such as the internet have resulted in widespread dissemination of these concerns. Despite an increased number of vaccine manufacturers, several routinely recommended vaccines are produced by single manufacturers and intermittent supply shortages continue to occur. Recent mumps and measles outbreaks and continued circulation of pertussis are reminders that despite changes in vaccine recommendations and higher coverage, vaccine preventable diseases still occur. The pace of development of new vaccines is much slower than advances in our understanding of immunology and several significant infectious diseases remain leading causes of death globally. In the U.S., the success in achieving high vaccination coverage among children has not been replicated for routinely recommended vaccinations among adolescents or adults. And as the cost of vaccination has increased, financial barriers to optimal program implementation have emerged for health departments, healthcare providers, and the public.
Development of an updated National Vaccine Plan to address current challenges and continue progress toward prevention of disease and enhanced safety is informed by an evaluation of the 1994 Plan and its implementation, conducted by a committee empanelled by the Institute of Medicine (IOM). The committee reviewed the goals, objectives, and strategies of the plan; assessed vaccination program progress; heard presentations from Department of Health and Human Services (HHS) agencies; met with government officials and others who were involved or familiar with development of the plan; and reviewed relevant literature. While the committee noted “many remarkable achievements” of Federal agencies working in collaboration with other stakeholders in the U.S. vaccine and immunization enterprise, they also noted that there was little evidence that the 1994 plan guided or motivated activities and it was difficult to attribute the changes that occurred to the plan.2
Based on its evaluation, the IOM committee made several recommendations to consider in developing and implementing an updated National Vaccine Plan (see Appendix 2 for the complete IOM recommendations). The committee emphasized the importance of a national rather than Federal plan, recognizing the important roles of many stakeholders in achieving program goals and objectives (see Appendix 3) for a list of key stakeholders in the vaccine and immunization enterprise). Non-Federal stakeholders should be involved in the development of the plan and approaches should be developed to motivate stakeholders to implement activities that achieve plan objectives. The committee also suggested that an updated plan include specific milestones, assignments of responsibility, objective measures, and evaluation mechanisms to increase the likelihood and success of implementation. Recognizing that a strategic plan will not include every activity that should be undertaken, the committee recommended describing the rationale for decisions on what was included.
With respect to the specific content of an updated plan, the committee highlighted several issues. They recommended that the plan include the following:
|
2 |
See http://www.iom.edu/CMS/3793/55143.aspx for the IOM letter report, Initial Guidance for an Update of the National Vaccine Plan. |
-
Mechanisms to assess the potential (“horizon”) for innovation and new developments in vaccines and promote timely decision-making in response to opportunities and challenges;
-
Consideration of vaccine financing issues;
-
Focus on disparities in access to vaccines;
-
A comprehensive framework for communications with the public and other stakeholders;
-
Creative solutions to vaccine supply problems;
-
Increased attention to global immunization issues.
IOM committee guidance on process and content has contributed to the development of this draft Plan and each of the issues that have been highlighted is addressed in the Plan or the approach to its implementation.
Purpose, Perspective, and Scope
The purpose of the updated National Vaccine Plan is to promote achievement of the National Vaccine Program mission to prevent infectious diseases and reduce adverse reactions to vaccines by providing strategic direction and promoting coordinated implementation by vaccine and immunization enterprise stakeholders.
Consistent with this purpose, this draft Plan is strategic, defining goals, objectives, and strategies. The Plan is national, defining goals and objectives to pursue through coordinated actions by a broad range of governmental and non-governmental stakeholders. The National Vaccine Advisory Committee, a Federal advisory committee that includes representatives from many of the key vaccine and immunization enterprise stakeholders, will implement a process to obtain input from a wide range of stakeholder groups. This input will include comments on this draft Plan and additional strategies that they can contribute to achieve Plan goals. In addition, input from the public will also be solicited to identify priority areas from their perspective. This draft will serve as the basis for the development of the updated National Vaccine Plan and based on this range of input, indicators of measurable outcomes will be determined and priorities will be presented. Recognizing, however, that success is facilitated by defining and monitoring specific activities, milestones and measurable outcomes, an implementation plan will also be developed. A ten-year horizon was chosen, balancing strategic vision which requires development and implementation of new initiatives with the recognition that changing circumstances and new opportunities and challenges are likely and may require adjustments to strategies and measurable outcomes.
The scope of the plan is broad, including vaccines and vaccine-related issues for the U.S. and globally. While several currently recommended vaccines prevent non-infectious outcomes – hepatitis B vaccine preventing hepatocellular carcinoma and HPV vaccine preventing cervical cancer – and future vaccines may prevent other cancers, autoimmune diseases, or other non-infectious conditions, based on the statute that established the National Vaccine Program, the focus for this plan is prevention of infectious diseases and adverse reactions to vaccines. Nevertheless, it is clear that progress made for
vaccines that prevent infections will also lead to progress more broadly. In addition, whereas emergency preparedness and vaccines and vaccination for pandemic or bioterrorist threats are important issues and are included in this draft Plan, they are considered more completely in other HHS strategic plans.3
Vaccine development, regulation, and program implementation are major components of HHS agency activities and those of other federal departments and agencies (see Appendix 4 for a description of relevant federal agency roles and responsibilities with respect to vaccines and the immunization enterprise). Strategic plans developed by these agencies identify vaccine-related objectives that are consistent with those included in this plan. Appendix 5 lists the agency and department strategic plans relevant to this plan. The HealthyPeople 2020 objectives will also be addressed in the Plans once those are established.
Approach to Developing the Draft Plan
This draft strategic National Vaccine Plan is primarily the result of deliberation, analysis, and input from multiple Federal agencies under the coordination of NVPO. Development of the draft Plan proceeded through a series of steps, with additional actions proposed that will lead to its completion (Table 2).
Table 2. Steps in developing and finalizing the draft strategic National Vaccine Plan
|
• Collection of information from Federal agencies regarding priority issues to address in the Plan. |
|
• Consideration of outcomes and lessons learned from the 1994 National Vaccine Plan and its implementation |
|
• Establishment of the Plan’s guiding perspective, structure, and overarching goals |
|
• Development of objectives and strategies that will lead to achievement of the Plan’s goals |
|
Next Steps: |
|
• Review and revision, as needed, of objectives and strategies based on input from vaccine and immunization enterprise stakeholders and an IOM committee [ongoing] |
|
3 |
For example, see BARDA strategic plan http://www.hhs.gov/aspr/barda/phemce/eiiterprise/strategy/mdex.html. |
|
• Public engagement to define values, perspectives, and priorities for the vaccine and immunization enterprise [pending] |
|
• Finalization of the National Vaccine Plan and development of implementation plans that will lead to achievement of Plan goals and strategies [pending] |
Analysis and recommendations from the IOM committee, based on its review of the 1994 National Vaccine Plan, provided useful perspectives that contributed to defining the scope of the plan (e.g., as a national rather than Federal plan), the process of development and implementation, and some of the key issues to include. A framework identifying key activities and pathways to achieve vaccine and immunization enterprise outcomes (see Framework) also was developed4 to facilitate a systematic approach to identifying objectives and strategies whose implementation will lead to achievement of Plan goals.
Objectives, strategies, and indicators by which success could be measured were drafted by lead Federal agencies for each of the five goals. In an iterative process, input was received from other agencies that will contribute to reaching the specific goal and all Federal agencies with a stake in the vaccine and immunization enterprise. Concurrent with this process, the IOM committee held an open meeting at which it reviewed the proposed objectives and strategies for the Plan goal to “Ensure a stable supply of recommended vaccines and achieve better use of existing vaccines to prevent disease, disability and death in the United States.” Comments from this meeting also contributed to revision of the draft for this goal.
Stakeholder review is a critical component of finalizing the draft Plan. Because stakeholder activities in pursuit of its goals and objectives will be essential to success, such input will contribute to buy-in, implementation planning, and coordination as activities are implemented. Stakeholder input will be obtained in four primary ways:
-
By input gathered by the IOM committee at four scheduled public meetings that will focus on different goals in the plan and will take place before June 2009. These meetings will include participation by stakeholders in medicine, public health, industry, and vaccinology and will include review of the draft Plan and other HHS planning documents5;
-
From interviews of Federal vaccine advisory committee members6 conducted by NVPO;
-
Through a process to engage domestic and international stakeholders coordinated by the National Vaccine Advisory Committee; and
-
From input by the public and others in response to a notice in the Federal Register.
|
4 |
RAND Corporation assisted in the development of the framework, under a contract from NVPO . |
|
5 |
More about this committee’s work can be found at http://www.iom.edy/CMS/3793/51325.aspx. |
|
6 |
Committees include the National Vaccine Advisory Committee (NVAC), the Advisory Committee on Immunization Practices (ACIP), the Vaccine and Related Biological Products Advisory Committee (VRBPAC), and the Advisory Commission on Childhood Vaccines (ACCV). |
Public input also will contribute to finalization of the Plan. Public values and perspectives can contribute to identifying the most important priorities for achieving and sustaining an effective and safe vaccination program. NVPO will seek input from the general public to assess their expectations and priorities about the Nation’s vaccine and immunization activities and programs.
Input and comments from stakeholders, the public, and recommendations from the IOM committee all will contribute to the final Plan to be released in the fall 2009. This plan, in turn, will serve as the basis for implementation planning by Federal agencies that will define specific actions and timelines and measurable outcomes to assess progress in accomplishing each of the strategies in the Plan. Non-Federal stakeholders also will be engaged in this process in order to develop specific actions that they will contribute to achieving the vision and goals of the National Vaccine Plan.
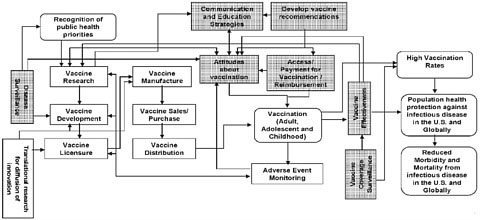
Framework
Disease prevention and enhanced vaccine safety are ultimate outcomes of a successful vaccination program. Identifying objectives and strategies that lead to and sustain these outcomes is facilitated by understanding the many processes or determinants of these outcomes. Figure 2 provides a simplified overview of these complex processes from beginning to end. It shows the key components (the rectangular boxes in Figure 2), the intermediate and long term outcomes (rounded boxes), how they relate to each other, and how they fit together to support the overall mission of the plan.
Figure 2. Overview of the vaccine and immunization enterprise
This complex process of preventing infectious diseases safely and effectively by vaccination begins with the identification of public health priorities informed by disease
surveillance data, which in turn, can guide vaccine research and development priorities. Following the licensure of a vaccine and recommendations for its use, receipt of vaccination requires manufacturing (with ongoing monitoring of product safety and quality) and sales, distribution, storage and handling of vaccines, vaccine payment and reimbursement policy, and communications and education to support decision making about vaccination. Attitudes, vaccination coverage and the effectiveness of disease prevention also are influenced by issues related to vaccine safety and effectiveness. Ultimately, attitudes, safety and effectiveness inform the development of the next generation of new vaccines. The desired outcomes of this process include high vaccination rates which lead to reduced morbidity and mortality from infectious disease in the U.S. and globally, and improved population health. The complex pattern of connections between these components suggests that to achieve the goals of the Plan, the objectives and strategies need to be comprehensive, addressing the vaccine and immunization enterprise as a whole rather than focusing on specific activities in isolation.
Toward that end, the components relevant to each goal were identified. A graphical representation and description of those components is provided for each goal. Then, in an effort to identify the factors critical to the achievement of each goal, a more detailed schematic was developed and analyzed. Objectives were developed to comprehensively address the critical factors for each goal along with the strategies needed to accomplish them. Finally, for each objective, relevant stakeholders were identified.
This approach to the development of the Plan provides a systematic process for identifying objectives and strategies and a rationale for their inclusion in the Plan. Moreover, it helps to ensure that the Plan is comprehensive in its approach to achieving the stated goals. The graphical representation of each component of the Plan, and the identification of key stakeholders involved, form the foundation for accountability in achieving each goal.
As is evident from a review of the figure, some vaccine-related activities are crucial to achieving more than one goal. To simplify the presentation of the plan, objectives and strategies were assigned to a single primary goal area. However, overlap between the goals is identified in the introduction to each goal area and are reflected in the figures showing the key components related to each goal.
A similar approach is being used to develop the corresponding Implementation Plan; that is, mapping the key activities associated with each objective and strategy. Identifying the sequence of steps in this manner facilitates the identification of milestones, for both Federal and nonfederal stakeholders, which can be monitored and reported to ensure that the plan is being implemented.
National Vaccine Plan Structure
The Plan is built around the achievement of five broad goals:
Goal 1: Develop new and improved vaccines
Goal 2: Enhance the safety of vaccines and vaccination practices
Goal 3: Support informed vaccine decision-making by the public, providers, and policy-makers
Goal 4: Ensure a stable supply of recommended vaccines and achieve better use of existing vaccines to prevent disease, disability and death in the United States
Goal 5: Increase global prevention of death and disease through safe and effective vaccination
Vaccine development, safety, and improved vaccine use goals also were included in the 1994 plan. Supporting informed decision-making expands the 1994 plan goal to better educate the public and healthcare professionals on the benefits and risks of immunization. Inclusion of a global immunization goal in this plan emphasizes the key role that U.S. governmental and non-governmental stakeholders can play in enhancing prevention of disease and death throughout the world
Each goal is realized by achieving several objectives that are pursued through defined strategies. Strategies can be mapped back to the key components of the vaccine and immunization enterprise diagram and each of the key determinants of an outcome is addressed by one or more strategies. Reaching goals and objectives generally requires action by many stakeholders in the vaccine and immunization enterprise, while individual strategies may require action by one or a small number of stakeholders.
Success in reaching the goals defined in the final Plan will be monitored by assessing progress in achieving measurable outcomes for each goal. The proposed indicators are measurable outcomes related to this draft strategic Plan’s objectives and are directly in a path toward, and critical to, realizing the Plan’s goals. Revision and definition of specific numeric targets (milestones) will occur through further consultation with stakeholders and the IOM. Approaches currently exist or can feasibly be established to provide dependable information on progress in reaching these quantitative outcomes. In contrast to milestones or measurable outcomes of individual action steps, indicators are broader and quantify outcomes rather than processes. Indicators in this plan also differ from the fourteen “predicted outcomes” in the 1994 plan which generally were not quantitative and often defined aspirations rather than specific targets.
Monitoring and Evaluation
Monitoring progress toward achievement of the indicators included in this Plan and the milestones in the ensuing implementation plan will contribute to success: both because monitoring stimulates action (“what gets monitored gets done”) and because measuring
progress can help identify problems and barriers that require the development and implementation of new approaches.
The National Vaccine Program Office (NVPO) is the principal coordinating office for the National Vaccine Program and reports to the Assistant Secretary for Health, who is the Director of that program. NVPO is responsible for providing leadership, facilitating coordination, and monitoring progress as the National Vaccine Plan is implemented. NVPO interacts directly with each of the HHS agencies and other federal Departments with vaccine and immunization portfolios.
The National Vaccine Advisory Committee (NVAC) is a federally chartered advisory committee which reports to the Assistant Secretary for Health (http://www.hhs.gov/nvpo/nvac). The Committee includes non-governmental members with expertise in vaccine development, public health and healthcare, and representatives from the vaccine industry and consumers. Liaison members represent Federal agencies with interests in vaccines and vaccination, including the Veterans Administration, Department of Defense and U.S. Agency for International Development; representatives from other vaccine-related advisory committees; and representatives from major stakeholder organizations. NVPO supports NVAC and the NVPO Director is the Executive Secretary for the committee. Given the broad participation in NVAC – either as members or liaisons – of public and private sector vaccine and immunization enterprise stakeholders, the Committee also will have an important role in monitoring progress in achieving National Vaccine Plan goals.
NVPO will be responsible for assuring coordination and for monitoring federal actions and accomplishments on an ongoing basis and NVPO and NVAC will report their findings to the Assistant Secretary for Health annually. This report will include a summary of progress, identify areas where progress is lagging, and propose corrective action where needed. The report also will be presented at an NVAC meeting, which is open to the public and is attended by many stakeholders not represented directly on the Committee.
Key federal stakeholders in global immunization include CDC and USAID. Many of the global immunization targets included in the Plan were established by international organizations (e.g., the World Health Organization) in consultation with U.S. stakeholders. However, the role of those stakeholders in achieving these targets most often involves providing technical assistance and support rather than direct implementation
Many factors may affect the ability to achieve National Vaccine Plan indicators and implementation plan milestones. Existing challenges and barriers may be more difficult to overcome than anticipated and new challenges may emerge. For example, a range of scientific and technical issues may delay development and licensure of new vaccines; safety concerns may affect vaccine uptake; financial constraints may affect vaccination delivery. One of the biggest challenges to success is the availability of resources to implement activities pursuing Plan strategies and objectives. Conversely, opportunities
may emerge that facilitate more rapid progress such that strategies and objectives are reached sooner than anticipated. Recognizing these uncertainties, NVPO will coordinate a mid-course review of the Plan after five years allowing changes to be made which respond to the reality of the environment. Modified indicators, strategies, actions, and milestones will guide subsequent annual evaluation through the overall ten-year horizon of the Plan.
Goal 1:
Develop new and improved vaccines
Introduction
Vaccines have changed humanity. In the United States and around the world vaccines (along with clean water and sanitation) are responsible for the most significant impacts on the health of the public. Not only has the investment in vaccine research and development and the implementation of effective vaccine delivery programs led to the eradication and elimination of several serious infectious diseases that were once common, but by preventing diseases from occurring, vaccines can reduce the emergence of antimicrobial resistance, reduce health care costs and pressure on health care systems.
At the core of this progress is the vaccine research and development enterprise that brings scientific ideas forward and, if successful, into the clinic. Vaccine development in the US is comprised of networks of public and private stakeholders that have been successful at bringing many candidates to licensure for commercial use in the US and globally. These stakeholders support various segments of the vaccine research and development process through collaborative and cooperative relationships. Today hundreds of vaccine candidates are in public and private sector vaccine development pipelines. These candidates address a myriad of diseases and utilize an arsenal of new and existing tools to design safe and effective vaccines that can aim at disease targets of public health and biodefense importance. Target populations for these candidate vaccines include routine use in healthy pediatric and adult populations, travelers, the military, and potentially among many sectors in the event of an emerging biological threat.
Understanding the priorities for development and encouraging collaboration between stakeholders (including researchers, manufacturers, funders, and policy makers) is essential to addressing the challenges of making new and improved vaccines for the future. Vaccine development is time and resource intensive. In addition to the technical and scientific challenges that face vaccine researchers and developers, gaps in funding also exist particularly between early phases of pre-clinical and clinical development and the later stages of advanced development (Phase II or III). Fostering continued investment from all sectors ill be increasingly important to harness the full potential of emerging scientific leads as the potential technological approaches and disease targets has expanded and as the costs of developing, licensing, and delivering vaccines has increased. The focus of Goal 1 is to address research and development aspects of vaccines. The research needs of other aspects of vaccines and immunization (e.g., operations research, translational research, behavior and communications research) are included within their respective Goals.
Goal 1 Indicators
-
Within one year, create an evidence-based list of new vaccine targets to prevent infectious diseases that are high priorities for development.
-
Identify X candidate vaccines (e.g., for HIV, malaria, TB, and a cross protective vaccine for influenza) and advance y priority vaccine candidates along the
-
research and development pipeline including Z candidates into advanced clinical trials.
-
Advance X new delivery strategies that will improve effectiveness, feasibility, acceptability, safety, or ease of administration of new or improved vaccines into clinical trials.
-
In X years, have the capability to test potential vaccine candidates in clinical trials developed in response to an emerging infectious disease health threat within six months of the identification of the need for a vaccine.
Identifying Goal 1. Objectives and Strategies
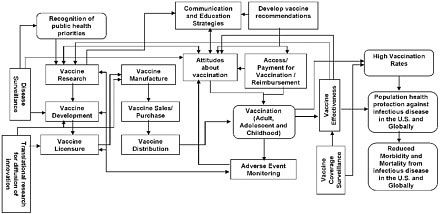
Figure 3 is the same as Figures 1 and 2, but it shows the components of the vaccine and immunization enterprise (i.e., the shaded rectangular boxes) that play a role in achieving Goal 1 – developing new and improved vaccines. While these components, including disease surveillance, vaccine research, translational research, development and licensure, occur mainly in the early part of the overall process, they are influenced by others such as surveillance related to vaccine effectiveness and adverse event monitoring, which occur later in the process. Disease surveillance continues throughout the process and provides information and feedback for multiple activities. The information from these components feeds back into and informs new vaccine research and development priorities.
Figure 3. Components of the Vaccine and Immunization Enterprise Relevant to Goal 1
Proposed Objectives
Objective 1.1: Prioritize the needs for developing new vaccines.
Federal Departments and Agencies7: HHS (BARDA, CDC, FDA, IHS, NIH, NVPO), DoD, USAID, VA
Non-federal Stakeholders8: Academia, health care providers, philanthropic organizations, the public, state, local, and tribal governments and public health agencies, vaccine industry, WHO
Strategies:
1.1.1 With stakeholder input, develop, implement, and evaluate a process for prioritizing the needs for new vaccines that considers the leading causes of morbidity and mortality from infectious diseases in populations for which vaccines could be a component of an effective prevention strategy.
1.1.2 Conduct surveillance to continuously inform the priorities for potential new vaccines.
Objective 1.2: Support research to develop new vaccine candidates and improve current vaccines to prevent infectious diseases, particularly those determined to be priorities.
Federal Departments and Agencies: HHS (BARDA, CDC, FDA, NIH), DoD, USAID, VA
Non-federal Stakeholders: Academia, philanthropic organizations, vaccine investors, vaccine industry
Strategies:
1.2.1 Advance research and development toward new and/or improved vaccines that prevent diseases, including those that protect against emerging, re-emerging, and important biodefense related pathogens.
1.2.2 Conduct and support expanded vaccine research to meet medical and public health needs of specific populations including neonates, infants, the elderly, pregnant women, and immunocompromised individuals.
|
7 |
Federal Departments and Agencies having a significant role in each objective are listed after the objective, with HHS as the first Department, followed by its agencies in parentheses, then by other Federal Departments and agencies in alphabetical order. These Departments and agencies are listed in Appendix 3. |
|
8 |
Non-federal stakeholder sectors considered to have a significant role in an objective are listed after the objective in alphabetical order. The sectors are defined in Appendix 3. |
1.2.3 Advance the science of neonatal and maternal immunization including the development of immunological models with which to study maternal immunization and effects on offspring.
1.2.4 Develop a process that identifies current vaccines that would benefit from improved performance characteristics (effectiveness, safety, number of doses, and/or delivery characteristics) and conduct and support studies to bring them to licensure.
Objective 1.3: Support research on novel vaccine delivery methods.
Federal Departments and Agencies: HHS (BARDA, CDC, NIH), DoD (DARPA), USAID, VA
Non-federal Stakeholders: Academia, philanthropic organizations, vaccine investors, vaccine industry,
Strategies:
1.3.1 Develop and evaluate alternate delivery methods to improve the protective immune response, safety, effectiveness, and/or efficiency (e.g. number of doses) of immunization.
1.3.2 Expand knowledge of the mechanisms by which induction of protective immunity can be stimulated by immunization through mucosal surfaces and other routes of administration. Include studies to identify and mitigate host factors that may have an impact on the effectiveness of immunizing by these routes.
Objective 1.4: Support development of vaccine candidates and the scientific tools needed to evaluate these candidates for licensure.
Federal Departments and Agencies: HHS (BARDA, CDC, FDA, NIH), DoD (DTRA), DHS, USAID
Non-federal Stakeholders: Academia, vaccine industry, philanthropic foundations, vaccine investors
Strategies:
1.4.1 Support applied research to develop rapid and cost efficient production, and optimize formulations and stability profiles of currently available vaccines.
1.4.2 Support research on and development of platform technologies that are applicable to vaccine design and production.
1.4.3 Improve access to appropriately designed pilot lot manufacturing facilities that produce clinical grade material for promising vaccine candidates.
1.4.4 Improve identification of useful biomarkers and immune correlates of protection.
1.4.5 Support translational research that accelerates the development of information that can be used in the product evaluation and licensure process.
1.4.6 Enhance methods and timeliness for conducting risk assessments of emerging variants or strains of vaccine-preventable disease agents, such as emerging strains of human and animal influenza virus.
1.4.7 Establish and strengthen partnerships to address urgent needs in vaccine research and development.
1.4.8. Establish alternative development and manufacturing approaches to support licensure for those vaccines which have a limited market.
Objective 1.5: Increase understanding of how the host immune system influences vaccine response.
Federal Departments and Agencies: HHS (CDC, FDA, NIH) DoD (DARPA), VA
Non-federal Stakeholders: Academia, philanthropic organizations, vaccine industry
Strategies:
1.5.1 Expand basic and applied research on innate and adaptive immune responses to infections at different stages of life (e.g., neonate, infant, pregnancy, elderly) in order to advance the understanding of immune protection.
1.5.2 Gain a better understanding of how induction and recall of immune memory may inform the development of vaccines that provide life-long protection.
1.5.3 Enhance research on vaccine effectiveness by continuing to support development of immunomodulators such as new adjuvants and use insights from such research to create novel vaccines and novel formulations of existing vaccines.
1.5.4 Expand knowledge of host related factors that impact severity of disease, and use this information to inform vaccine development.
Objective 1.6: Strengthen the science base for the development and licensure of safe and effective vaccines.
Federal Departments and Agencies: HHS (CDC, FDA, NIH), DoD
Non-federal Stakeholders: Academia, vaccine industry
Strategies:
1.6.1 Better characterize product safety and efficacy through research in areas including assay development and characterization of novel cell substrates.
1.6.2 Develop better animal models to study potential correlates of immune response to predict safety and efficacy in humans.
1.6.3 Conduct research to inform feasible ways to provide data to support evaluation and licensure of new vaccines for biodefense related pathogens and rare diseases.
1.6.4 Develop better methods for ensuring control and quality for laboratory, clinical and manufacturing practices related to developing a vaccine.
Goal 2:
Enhance the safety of vaccines and vaccination practices
Introduction
The United States has a robust vaccine safety system that has the goal of minimizing the occurrence of serious adverse events from routinely administered vaccines and detecting them in a timely manner when they do occur. Enhancing the current vaccine safety system is important in order to keep pace with a number of factors influencing the system, including: an increasing number of vaccines and vaccine combinations, expanding target populations, and a better understanding of human biology, especially the human immune system. Learning from historic successes and failures (e.g. the Cutter incident) in vaccine safety also offers opportunities for enhancements to the vaccine safety system. In addition to advances in vaccine research and development, prelicensure evaluation, and post-licensure monitoring advances in medical and information technologies are creating opportunities to further enhance immunization safety. For example, advances in genomics may lead to new ways to identify those at increased risk of adverse events following immunization (AEFI). Expansion of healthcare databases and new approaches to statistical analysis will also provide opportunities to more rapidly identify and evaluate AEFI.
Vaccine safety is a focus of virtually all stakeholders in the vaccine and immunization enterprise including government, research scientists, manufacturers, healthcare providers, professional medical societies, philanthropic foundations, and vaccine recipients. Goal 2 specifically addresses safety-related issues of vaccines and their biological effects as well as the system that monitors the safety of vaccines from production to vaccine administration and use in vaccination programs. Several important vaccine safety issues are also considered elsewhere in this plan. For example, research and development of safer vaccines, including pre-clinical and clinical safety evaluation of candidate vaccines and licensure, are addressed in Goal 1 (Develop new and improved vaccines). Issues related to behavioral science research, education, risk communications and public engagement on vaccine safety are included in Goal 3 (Support informed vaccine decision-making by the public, providers, and policy-makers). Vaccine safety is an important component of every immunization program. These other factors are included in Goal 4 (Ensure a stable supply of recommended vaccines and achieve better use of existing vaccines to prevent disease, disability, and death in the United States) and Goal 5 (Increased global prevention of death and disease through safe and effective vaccination).
Throughout Goal 2, two terms are used that deserve clarification: ‘adverse event following immunization’ (AEFI), and ‘signal.’ Respectively, these are defined:
-
Adverse event following immunization (AEFI) is an adverse event temporally associated with an immunization that may or may not be causally related to the immunization. The term “vaccine adverse event” is also commonly used to convey the same meaning.
-
Signal: While there are multiple definitions of signals, in this document a signal refers to a concern that an AEFI could be temporally occurring more often than anticipated based on chance alone (i.e., that the event could be related to the
-
receipt of the vaccine). Signals may arise from a variety of sources, including from pre-licensure clinical trials, case series, surveillance, clinical experience, the literature, expert committee reviews, the media, and/or the public. A signal may also arise from a single individual with a convincing clinical pattern such as a challenge/rechallenge case or instances where the vaccine strain organism (e.g., attenuated virus) is isolated and associated with a pathologic process.
Goal 2 Indicators
-
Conduct and disseminate the results of active and passive surveillance-based safety assessments for newly recommended vaccines or for vaccines with expanded recommendations:
-
Within 1 year of publication in CDC’s Morbidity and Mortality Weekly Report of new or revised ACIP recommendations.
-
Within 1 year after X million doses have been distributed
-
-
Develop and disseminate plans for further investigation, if any, of newly detected AEFI signals and the rationale for those plans within X months of signal detection.
-
By X year, X % of infants, children, adolescents, adults, and pregnant women will be under active surveillance for AEFIs.
-
Conduct research to explore host factors and biological mechanisms associated with serious AEFIs and annually report results to the Assistant Secretary for Health, vaccine advisory committees, vaccine policy makers and other stakeholders.
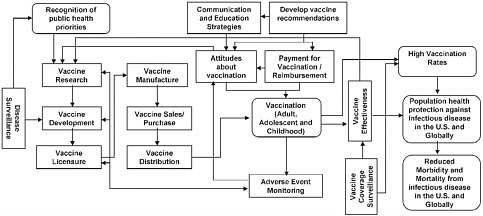
Identifying Goal 2 Objectives and Strategies
Figure 4 is the same as Figures 1 and 2, but it shows the components of the vaccine and immunization enterprise (i.e., the shaded rectangular boxes) that play a role in achieving Goal 2 – enhancing the safety of vaccines and vaccination programs. The vaccine safety-related components shown here occur both pre- and post-licensure. However, as noted before, the focus of Goal 2 is on those components which contribute to enhancing the safety of vaccines and vaccination programs post licensure. These components include disease surveillance, vaccine research and development, translational research, manufacturing, developing vaccine recommendations, communications and education strategies, distribution, administration, vaccination of target populations, and monitoring vaccine effectivenes s and AEFIs through both passive and active surveillance. The pre-licensure safety-related components, such as research and development of safer vaccines, and the associated objectives and strategies are captured in Goal 1.
Figure 4. Components of the Vaccine Process Relevant to Goal 2
Objective 2.1: Facilitate the continuous modernization of manufacturing sciences and regulatory approaches relevant to manufacturing, inspection and oversight to enhance product quality and patient safety.
Federal Departments and Agencies: HHS (FDA)
Primary Non-HHS Stakeholders: Academia, vaccine industry
Strategies:
Strategy 2.1.1: Facilitate the enhancement of vaccine manufacturing sciences and quality systems, including production technologies, in process controls and testing, and identification of best practices in preventive quality systems and oversight.
Strategy 2.1.2: Develop, implement and periodically reassess risk-based scientific approaches to identify inspectional priorities and best practices.
Strategy 2.1.3: Support new technologies and modernization of both industry and FDA testing of product quality to better prevent and more rapidly detect potential quality or safety issues.
Strategy 2.1.4: Evaluate current regulations, guidance documents, policies and procedures that are relevant to manufacturing to determine enhancements that could be made to promote and enhance product safety.
Objective 2.2: Enhance timely detection and evaluation of vaccine safety signals.
Federal Departments and Agencies: HHS (CDC, CMS, FDA, HRSA, IHS), DoD, VA
Non-federal Stakeholders: Academia, health care system, the public, public and private payers and plans, state, local, and tribal governments and public health agencies, vaccine industry
Strategies:
2.2.1: Improve the effectiveness and timeliness of AEFI signal identification and assessment through coordinated use of national passive and active surveillance systems.
2.2.2: Enhance collection of medical histories and biological specimens from selected persons experiencing serious AEFI reported to the Vaccine Adverse Event Reporting System (VAERS), petitioning the National Vaccine Injury Compensation Program (VICP), and available through active surveillance to enhance study of biological mechanisms and individual risk factors.
2.2.3: Assess lay public and professional questions and concerns about vaccine safety.
2.2.4: Improve the process for assessing AEFI signals to determine which signals should be evaluated further in epidemiological and clinical studies.
Objective 2.3. Improve timeliness of the evaluation of vaccine safety signals when a high priority new vaccine safety concern emerges, a new vaccine is recommended or vaccination recommendations are expanded, and during public health emergencies such as in an influenza pandemic or other mass vaccination campaign.
Federal Departments and Agencies: HHS (BARDA, CDC, FDA, HRSA, IHS), DoD, VA
Non-federal Stakeholders: Academia, health care system, public and private payers and plans, state, local and tribal governments/public health agencies, vaccine industry
Strategies:
2.3.1: Increase the size of the population under active surveillance for serious AEFIs that can be included in high quality, rigorously conducted epidemiological studies to test vaccine safety hypotheses.
2.3.2: Expand collaboration with clinical, laboratory, genetic and statistical experts to conduct clinical research studies to investigate the role of host genetics in AEFI.
2.3.3: Enhance capacity to monitor immunization safety in the event of a mass vaccination campaign.
2.3.4: Provide safety data necessary to conduct informed risk-benefit assessments for utilization of vaccines in mass vaccinations for public health emergencies.
Objective 2.4: Improve causality assessments of vaccines and related AEFIs.
Federal Departments and Agencies: HHS (CDC, FDA, HRSA, IHS), DoD, VA
Non-federal Stakeholders: Academia, health care system, public and private payers and plans, state, local and tribal governments/public health agencies, vaccine industry
Strategies:
2.4.1 As appropriate, develop algorithms and assess the evidence on an individual-level for a causal relationship between certain vaccines and specific serious AEFI.
2.4.2 Assess the evidence on a population level for a causal relationship between certain vaccines and specific serious AEFI.
2.4.3 Regularly update the Vaccine Injury Table based upon individual and population level causality assessments.
Objective 2.5: Improve scientific knowledge about the risk of vaccine adverse events and their mechanisms.
Federal Departments and Agencies: HHS (CDC, FDA, HRSA, NIH), DoD, VA
Non-federal Stakeholders: Academia, Vaccine industry
Strategies:
2.5.1 Identify host risk factors, such as previous or concurrent illness or genetic characteristics that may be associated with increased risk for specific AEFI through basic, clinical, or epidemiological research.
2.5.2 Identify the biological mechanism(s) for specific AEFI that, based upon causality assessments (Strategy 2.4.2), are likely to be causally associated with vaccination.
2.5.3 Assess whether the risk of specific AEFI is increased in specific populations such as pregnant women, premature infants, elderly persons, those with immunocompromising or other medical conditions, or based on gender or race/ethnicity.
Objective 2.6: Improve clinical practice to prevent, identify and manage AEFIs.
Federal Departments and Agencies: HHS (CDC, HRSA, IHS), DoD, VA
Non-federal Stakeholders: Academia, health care system, MCOs, public and private payers and plans, state, local, tribal governments and public health agencies, vaccine industry
Strategies:
2.6.1 Improve training, availability of and access to vaccine safety clinical experts to provide consultation to healthcare providers and public health practitioners.
2.6.2 Develop additional evidence-based guidelines for vaccination or revaccination, as appropriate, for persons at increased risk of AEFI. Identify additional contraindications and precautions to vaccination, as needed.
2.6.3 Reduce errors in vaccine administration (e.g., wrong vaccine, dose, injection site, or timing) and associated adverse patient outcomes.
Objective 2.7: Improve cross-cutting scientific capabilities to enhance vaccine safety and the vaccination safety system.
Federal Departments and Agencies: HHS (CDC, FDA, IHS, NIH), DoD, VA
Non-federal Stakeholders: Academia, the Brighton Collaboration, vaccine industry
Strategies:
2.7.1 Enhance the immunization safety science workforce to recruit and retain additional highly trained scientists and clinicians.
2.7.2 Develop additional standard case definitions for AEFI for use in immunization safety surveillance and research, vaccine safety standards such as concept definitions, standardized abbreviations, and standardized study designs.
2.7.3 Improve laboratory, epidemiological and statistical methods used in vaccine safety research.
Objective 2.8: Enhance integration and collaboration of vaccine safety activities
Federal Departments and Agencies: HHS (AHRQ, CDC, CMS, FDA, HRSA, NVPO), DoD, VA, USAID
Non-federal Stakeholders: Academia, MCOs, public and private payers and plans, vaccine industry, World Health Organization
Strategies:
2.8.1 Improve collaboration, such as data sharing arrangements, across agencies and departments.
Goal 3:
Support informed vaccine decision-making by the public, providers, and policy-makers
Introduction
HHS is committed to providing accurate, timely, transparent, complete, and audience-appropriate information about immunizations and vaccines to parents making vaccination decisions for their children (birth through age 18), adults considering vaccines for themselves, public health partners, providers, and others. Communication tools and channels used to disseminate immunization and vaccine information span the spectrum: publication of evidence-based recommendations, use of mass media and new media in campaigns throughout the year, provider education and training in a variety of formats, and support to partner organizations and state immunization programs through provision of resources, trainings, updates, and announcements. HHS also provides immunization and vaccine information to decision-makers in a variety of settings (e.g., health system, executive and legislative government entities).
Current communication efforts are informed by adequate research as well as the principles of effective risk communication. Communication research should be enhanced to facilitate the development of the best messages and methods for transparently, clearly, and effectively communicating about the benefits and risks of vaccines, and addressing information needs and concerns unique to various audiences. The importance of timely audience and message testing research to inform effective communications must not be underestimated. Uninformed strategies could be detrimental to public confidence and ultimately to vaccine coverage and control of vaccine-preventable diseases. By working together, communication scientists, health services researchers and others can develop and implement comprehensive, long-term, sustainable plans for gathering real-time, reliable, and representative data about facilitators of, and barriers to, vaccine acceptance and translate those data into practical solutions.
While the focus of Goal 3 is on communication and education issues relevant to informed decision-making, they are also relevant to each of the other goals of the National Vaccine Plan. Topic-specific communications and education activities are described in Goals 2, 4, and 5.
Goal 3 Indicators:
-
By Y (year), enhance communication with stakeholders and the public to more rapidly inform them (within _X_ days) about urgent and high-priority vaccine and vaccine-preventable disease issues (e.g., outbreaks, supply shortages, vaccine safety concerns).
-
_X__ % of the public will report that they are satisfied with how their health care provider answers their questions about the benefits and risks of vaccines by Y (year).
-
_X__ % of the public will report they have access to information which allows them to make informed vaccination decisions for themselves or their children by Y (year).
-
_X_ % of health care providers will report that they have access to accurate and complete information about vaccine benefits and risks and are able to adequately answer questions of parents and patients by Y (year).
-
_X_ % of key decision- and policy-makers will report they have access to vaccine benefits, risks, and costs to make informed decisions about vaccine policy by Y (year).
-
By Y year, all health professional schools and training programs will include vaccine and vaccine-preventable disease content in their curricula, and assess students’ and trainees’ knowledge.
-
By Y year, all relevant health professional certifying examinations will include vaccine and vaccine-preventable disease questions.
Identifying Goal 3 Objectives and Strategies
Figure 5 is the same as Figures 1 and 2, but it shows the components of the vaccine and immunization enterprise (i.e., the shaded rectangular boxes) that play a role in achieving Goal 3 - supporting informed vaccine decision-making by the public, providers, and policy-makers. The key components include communication and education strategies, development of vaccine recommendations, attitudes about vaccination, access to, and payment for, vaccination, vaccine effectiveness, disease surveillance, and assessment of vaccine coverage.
Figure 5. Components of the Vaccine and Immunization Enterprise Relevant to Goal 3
Proposed Objectives
Objective 3.1: Conduct research and utilize findings in an ongoing fashion to identify communication and education needs and inform communication and education efforts.
Federal Departments and Agencies: HHS (CDC, CMS, FDA, HIS), DoD, VA
Non-federal Stakeholders: Healthcare payers and plans (public and private), healthcare Systems, the public, state, local and Tribal Governments and public health agencies, vaccine industry
Strategies:
3.1.1 Conduct ongoing research to regularly take a “pulse of the public” to identify knowledge, beliefs and concerns about vaccines and vaccine-preventable diseases.
3.1.2 Conduct research on factors (positive influences and barriers) that go into decision-making about vaccination for individuals and families, providers, and policymakers.
3.1.3 Develop and test educational strategies that better enable public audiences and policymakers to read, understand, and use information about vaccine benefits and risks when making immunization decisions.
3.1.4 Continue to assess the effectiveness of specific messages and materials in addressing information needs and concerns based on public and provider attitudes toward the benefits and risks of vaccines.
3.1.5 Evaluate the effectiveness of vaccine benefit and risk communication, overall and for populations known to be at risk of under immunization, and, as needed, update communications.
3.1.6 Gather data to inform communications about the accessibility of vaccines (i.e., where and when to get vaccinated).
3.1.7 Gather data to inform communications activities and vaccine program managers on the direct and indirect costs of vaccination. This includes, but is not limited to, information on federal and state programs that offer low cost vaccines.
Objective 3.2: Utilize collaborations and partnerships to leverage communication efforts.
Federal Departments and Agencies: HHS (CDC, CMS, FDA, HRSA, IHS), USAID, VA
Non-federal Stakeholders: Healthcare payers and plans (public and private), healthcare systems, the public, state, local and tribal governments and public health agencies, vaccine industry
Strategies:
3.2.1 Emphasize cross-agency and intra-agency collaboration to inform development of communication research agendas, protocols, campaigns and messages.
3.2.2 Strengthen partnerships and coalitions supporting immunization of children, adolescents, and adults.
3.2.3 Collaborate with partners and stakeholders to communicate vaccine benefits and risks in appropriate languages, methods, and literacy levels.
Objective 3.3: Enhance delivery of timely, accurate, and transparent information to public audiences and key intermediaries (such as media) about what is known and unknown about the benefits and risks of vaccines and the vaccination program.
Federal Departments and Agencies: HHS (CDC, CMS, FDA, HRSA, IHS), USAID
Non-federal Stakeholders: Healthcare payers and plans (public and private), healthcare Systems, the public, state, local and Tribal Governments and public health agencies, vaccine industry
Strategies:
3.3.1 Enhance communication of scientific findings about vaccine safety and effectiveness studies to the public, partners, and providers in a clear, transparent and timely manner.
3.3.2 Consistently and effectively respond in a rapid and coordinated manner to emerging vaccine issues and concerns (e.g. supply, safety or public health emergencies).
3.3.3 More rapidly and completely disseminate research findings through peer-reviewed journals, conferences, and partner communications to facilitate implementation of evidence-based strategies.
Objective 3.4: Increase public awareness of vaccine preventable diseases, and benefits and risks of vaccines and immunization, especially among populations at risk of under immunization.
Federal Departments and Agencies: HHS (CDC, CMS, FDA, HRSA, IHS)
Non-federal Stakeholders: Healthcare payers and plans (public and private), healthcare Systems, the public, state, local and Tribal Governments and public health agencies, travel industry, vaccine industry
Strategies:
3.4.1 Develop, implement, and evaluate a long-term strategic communications plan and program aimed at educating parents of children and adolescents about vaccine preventable diseases and the benefits and risks of vaccines.
3.4.2 Maintain up-to-date, easily accessible, evidence based web-based information on vaccine preventable diseases and vaccines, including benefits and risks and the basis of immunization recommendations, for all audience groups.
3.4.3 Use and evaluate new media (such as mobile technologies and social networking), as appropriate, to reach target audiences with accurate and timely information about vaccines and to respond to emerging concerns and issues.
3.4.4 Develop, use, and evaluate evidence-based communication tools to educate parents, adolescents, and adults about vaccine-preventable diseases, recommended vaccines, and preventive health care visits.
3.4.5 Develop, implement, and evaluate interventions to increase knowledge among all travelers about benefits and risks of immunizations before travel.
Objective 3.5: Assure that key decision and policy-makers (e.g., third-party payers, employers, legislators, community leaders, hospital administrators, health departments) receive accurate and timely information on vaccine benefits, risks, and economics, and on public and stakeholder knowledge, attitudes, and beliefs.
Federal Departments and Agencies: HHS (CDC,CMS, FDA, HRSA, IHS), VA
Non-federal Stakeholders: Healthcare payers and plans (public and private), healthcare systems, the public, state, local and tribal governments and public health agencies, vaccine industry
Strategies:
3.5.1 Develop, disseminate, and evaluate business case evidence and guidance for purchasers of health care and for health plans that address the coverage of vaccines in routine health care.
3.5.2 Develop, disseminate, and evaluate broad-based education of key groups (e.g., legislators, community leaders, hospital administrators, health departments) on the benefits, risks, and economics of vaccines, the basis of immunization recommendations, vaccine policy development, and on the standards of immunization practice.
3.5.3 Improve capacity for public engagement initiatives at the national, state and local levels.
Objective 3.6: Improve the knowledge of vaccines and vaccine-preventable diseases, understanding of basis for immunization recommendations, and immunization practices of all healthcare providers.
Federal Departments and Agencies: HHS (CDC, CMS, FDA, HRSA, IHS), DoD, USAID, VA
Non-federal Stakeholders: Academia, healthcare payers and plans (public and private), healthcare system, the public, state, local and tribal governments and public health agencies, vaccine industry
Strategies:
3.6.1 Expand and implement training and education of immunization providers at all levels of their education on the proper use of vaccines, the proper storage and handling of vaccines, the basis of immunization recommendations, vaccine safety, and on the standards of immunization practice.
3.6.2 Develop and implement educational strategies for providers on vaccine-preventable diseases, including diagnosis, modes of transmission, prevention and control, and reporting requirements.
3.6.3 Widely disseminate information about vaccines and vaccine use that will assist clinicians assess, report, and manage vaccine adverse events.
3.6.4 Determine the most effective and efficient mechanisms to communicate to health care providers about reporting to VAERS.
Objective 3.7: Develop and disseminate communication materials that help facilitate active and involved immunization decision-making.
Federal Departments and Agencies: HHS (AHRQ, CDC, CMS, FDA, HRSA, IHS), DoD, USAID, VA
Non-federal Stakeholders: Healthcare payers and plans (public and private), healthcare system, the public, state, local and tribal governments and public health agencies, vaccine industry
Strategies:
3.7.1 Conduct research on factors that go into decision-making about vaccination.
3.7.2 Conduct research to identify the kinds of information that would support decision-making about vaccination for individuals and families, providers, and policymakers.
Goal 4:
Ensure a stable supply of recommended vaccines, and achieve better use of existing vaccines to prevent disease, disability, and death in the United States
Introduction
Vaccine-preventable disease incidence is at or near record-low levels for most diseases against which children are routinely vaccinated and vaccination rates are at or near record high levels for infants and children. However, coverage levels are below Healthy People 2010 targets for many vaccines targeted to adolescents and adults, and substantial disparities exist among racial and ethnic groups in vaccination levels for adults. Ongoing efforts through partnerships among national, state, local, private, and public entities are needed to sustain and improve the use of vaccines in the United States.
Challenges remain for further improving vaccination rates, and for incorporating new vaccines into child and adolescent vaccination schedules. Three new vaccines have been recommended for adolescents by the Advisory Committee for Immunization Practices (ACIP) since 2005: meningococcal conjugate vaccine (1 dose), tetanus, diphtheria, acellular pertussis vaccine (1 dose), and quadrivalent human papillomavirus vaccine (3 doses). ACIP also recommends that adolescents should receive recommended vaccinations that were missed during childhood.
Vaccination rates for adults remain below the HealthyPeople 2010 targets and indicate where substantial challenges lie. These challenges include overcoming health access and financial barriers as well as barriers related to knowledge and attitudes of the public, health care professionals, and health policy and decision-makers. Translational research to assess how best to implement strategies to overcome such barriers is also important.
An additional barrier is assuring a reliable and steady supply of all vaccines in the United States, where shortages of several commonly used vaccines have occurred since 2000 (including Haemophilus influenzae type b, hepatitis A, influenza, meningococcal conjugate, pneumococcal conjugate, and rabies). New 21st century vaccine supply concerns are vaccines for pandemic influenza, emerging diseases, and biothreats that present different models for sustainability and surge manufacturing capacity as compared to traditional vaccine models.
Strong public health surveillance that monitors and evaluates vaccine-preventable diseases and the effectiveness of licensed vaccines provides the link between vaccination policy and outcomes. Such public health surveillance is a key component of strategies to overcoming barriers and achieving better use of existing vaccines.
Goal 4 identifies eight objectives and related strategies to keep our nation’s vaccination program strong and overcome barriers to continue to improve it. Enhancing communication and education activities to allow improved decision-making about vaccination, are also a key approach to overcome many of the current challenges. Those
concepts are discussed in Goal 3, and have significant relationships to the objectives and strategies presented in Goal 4.
Goal 4 Indicators
-
The United States will have 6 months’ supply of all vaccines appropriate to stockpile.
-
Reduce financial and non-financial barriers to access immunization services, such as cost, availability, and language, by Y (year) so that:
-
_X_% of parents of infants and children report no barriers to immunization;
-
_X_% of parents of adolescents report no barriers to immunization; and
-
_X_% of adults report no barriers to immunization.
-
-
Reach or exceed HealthyPeople 2020 vaccine coverage levels, once established, for pediatric, adolescent and adult populations using coverage levels in 2010 as a baseline.
-
X% of electronic health record systems and Y% of immunization information systems will include reminder and recall systems for vaccination by Y (year).
-
Implement surveillance for at least one major disease outcome for each routinely recommended vaccine in X% of states within Y years after its ACIP recommendation.
-
Through the evaluation of evolving science, ensure that the Vaccine Injury Table is updated on an as needed basis (at least every X years).
Identifying Goal 4 Objectives and Strategies
Figure 6 is the same as Figures 1 and 2, but it shows components of the vaccine and immunization process (i.e., the shaded rectangular boxes) that play a role in achieving Goal 4 - ensuring a stable supply of recommended vaccines and achieving better use of existing vaccines. These focus on what happens post licensure, and include processes related to vaccine manufacturing, purchasing/sales of vaccines, and distribution channels and process for getting vaccines to people, and processes related to vaccine administration. Importantly, they also include processes related to determining which vaccines are recommended for use, and their indications, communication and education about vaccines — to and from the public, providers, and policymakers, as well as assessing attitudes about vaccination. Finally, surveillance related to disease and to coverage levels informs efforts to achieve high vaccination rates through communication and education strategies, and addresses financial and non financial barriers to vaccines.
Figure 6. Components of the Vaccine and Immunization Enterprise Relevant to Goal 4
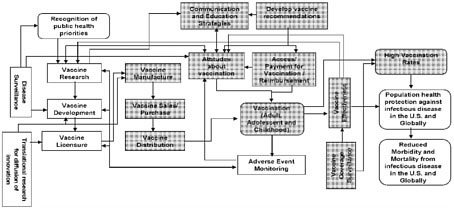
Proposed Objectives
Objective 4.1: Ensure consistent and adequate availability of vaccines for the United States.
Federal Departments and Agencies: HHS (BARDA, CDC, FDA, IHS, NVPO)
Non-Federal Stakeholders: Health care system, international organizations, national regulatory agencies, state, local, and tribal governments/public health, vaccine industry
Strategies:
4.1.1 Increase US licensed vaccine suppliers to have at least two suppliers of each vaccine antigen recommended for routine use by infants, children, adolescents and adults.
4.1.2 Promote development of high quality harmonized vaccine standards internationally.
4.1.3 Improve product quality and availability through advancing manufacturing sciences, through communication and training in best practices and through better manufacturing and production oversight.
4.1.4 Improve vaccine ordering, distribution and tracking systems for routine use, for public health emergencies, and for management of supply disruptions.
4.1.5 Optimize use, and content, and distribution of vaccine stockpiles.
4.1.6 Improve the development, communication, and tracking of adherence to recommended changes in vaccine use during national vaccine shortages.
4.1.7 Enhance support for international regulatory information sharing and collaboration.
Objective 4.2: Reduce financial and non-financial barriers to vaccination.
Federal Departments and Agencies: HHS (BARDA, CDC, CMS, HRSA, IHS), USAID, VA
Non-Federal Stakeholders: Health care payers and plans (public and private), health care system, state, local, and tribal governments and public health agencies, vaccine industry
Strategies:
4.2.1 Ensure that out of pocket costs for purchase and administration of all ACIP recommended vaccines for children, adolescents, and adults by publicly funded health insurance plans do not represent a significant financial barrier (i.e., Medicare, Medicaid, TRICARE, VA, FEHBP, DoD).
4.2.2 Reduce financial barriers to immunization by increasing the proportion of people with private healthcare insurance who have only minimal cost sharing for purchase, counseling, and administration of all ACIP recommended vaccines for children, adolescents, and adults (regardless of where the vaccines are administered).
4.2.3 Identify and regularly monitor financial and non-financial (e.g., vaccine availability and language) barriers to receipt of ACIP recommended vaccines for children, adolescents, and adults, and regularly publicize the findings.
4.2.4 Strengthen the ability of States to purchase and expand access to ACIP recommended vaccines for people who qualify for publicly supported vaccinations.
4.2.5 Develop, implement, and evaluate strategies to reduce the financial burden on vaccination providers for purchase of initial and ongoing vaccine inventories.
4.2.6 Enhance public sector infrastructure to support and sustain adult immunization activities.
4.2.7 Expand access to vaccination at medical care sites for children, adolescents, and adults.
4.2.8 Expand access to vaccination at sites outside of traditional medical settings.
Objective 4.3: Maintain and enhance the capacity to monitor immunization coverage for vaccines routinely administered to infants, children, adolescents, and adults.
Federal Departments and Agencies: HHS (CDC, IHS), DoD, USAID, VA
Non-federal Stakeholders: Health care payers and plans (public and private), health care system (e.g., electronic health record vendors), state, local, and tribal governments and public health agencies
Strategies:
4.3.1 Identify, implement, and evaluate cost-effective and rapid methods for assessing vaccination coverage:
-
among children, adolescents, adults overall and by State, immunization grantee, and within states and grantees;
-
among persons in key population subgroups (e.g., racial/ethnic groups, pregnant women, healthcare workers); and
-
by type of vaccination financing (e.g., VFC, other public sector program, private sector).
4.3.2 Improve the completeness and use of Immunization Information Systems (IIS) and electronic medical records (EMR) to monitor vaccination coverage.
Objective 4.4: Enhance tracking of vaccine preventable diseases and monitoring of the effectiveness of licensed vaccines.
Federal Departments and Agencies: HHS (CDC, IHS), DoD, USAID, VA
Non-federal Stakeholders: Academia, health care system, public and private payers, philanthropic organizations, state, local, and tribal governments/public health, vaccine industry
Strategies:
4.4.1 Strengthen epidemiologic and laboratory methods and tools to diagnose vaccine-preventable diseases and characterize the impact of vaccination coverage on relevant clinical outcomes.
4.4.2 Monitor circulating strains of relevant vaccine-preventable pathogens.
4.4.3 Monitor ongoing disease burden and determine epidemiologic and clinical characteristics of cases of relevant vaccine-preventable diseases.
4.4.4 Conduct studies to assess vaccine effectiveness and indirect (community or herd) protection.
4.4.5 Monitor long term protection from vaccines administered to infants, children, adolescents, and adults.
4.4.6 Assure rapid and comprehensive identification, investigation, and control of vaccine preventable disease outbreaks.
Objective 4.5: Educate about, and support, healthcare and other vaccination providers in vaccination counseling and delivery.
Federal Departments and Agencies: HHS (CDC, CMS, FDA, HRSA, IHS), DoD, USAID, VA
Non-federal Stakeholders: Health care payers and plans (public and private), health care system (including health care provider organizations), philanthropic organizations, state, local, and tribal governments/public health, vaccine industry
Strategies:
4.5.1 Expand knowledge regarding the value of vaccination, the vaccination program, and vaccine administration by traditional healthcare providers, medical and nursing trainees, and other vaccinators (e.g., pharmacists, community vaccinators).
4.5.2 Improve counseling and referral of patients for immunization by healthcare providers who do not offer immunization services.
4.5.3 Promote and support educational and technical assistance to improve business practices associated with providing immunizations.
4.5.4 Incentivize direct health care providers, health systems, and health insurers to provide vaccines by incorporating vaccination in quality assessment programs (e.g., HEDIS, Quality Measures and Pay for Performance programs).
4.5.5 Ensure appropriate reimbursement for vaccine counseling and administration by providers under public sector and private health plans.
4.5.6 Support research to evaluate the capacity (accommodating the increased number of patient visits required to receive recommended vaccines) of health care providers to implement childhood, adolescent, and adult vaccination recommendations.
4.5.7 Develop, implement, and evaluate communication tools as part of comprehensive programs to ensure health care professionals are appropriately immunized with recommended vaccines.
4.5.8 Promote the development, implementation, and evaluation of employer-based immunization programs (including free vaccines, convenient access, education, and compliance monitoring) to increase the coverage of health-care personnel with recommended vaccines.
4.5.9 Assess whether changes in health care facility and professional licensure and regulation can improve the safety of the health care environment by increasing vaccination rates of health care professionals.
4.5.10 Develop and monitor policies promoting vaccination for patients and health care personnel in long-term care facilities and hospitals.
Objective 4.6: Maintain a strong, science-based, transparent process for developing and evaluating immunization recommendations.
Federal Departments and Agencies: HHS, (BARDA, CDC, FDA, HRSA, NVPO, IHS), DoD, USAID, VA
Non-federal Stakeholders: Academia, health care system (including health care provider organizations), media, the public and citizen advocacy groups, state, local, and tribal governments and public health agencies, vaccine industry,
Strategies:
4.6.1 Obtain broad-based input from the public and stakeholders contributing to new immunization policies and to assessment of existing policies.
4.6.2 Support and strengthen immunization advisory committees at the state and national levels.
4.6.3 Assess the impact of new vaccines and vaccine recommendations on the overall immunization schedule, including programmatic implementation, safety, and efficacy.
4.6.4 Evaluate the cost-effectiveness of proposed and existing immunization recommendations.
Objective 4.7: Strengthen the Vaccine Injury Compensation Program (VICP) and Public Readiness and Emergency Preparedness (PREP) Act compensation fund
Federal Departments and Agencies: HHS (BARDA, CDC, HRSA, IHS, NVPO), DoJ
Non-federal Stakeholders: Academia, health care system, media, state, local, and tribal governments/public health, the public, media, vaccine industry, citizen advocacy groups
Strategies:
4.7.1 Increase knowledge about the VICP and PREP act among all stakeholders.
4.7.2 Assure the program is responsive to evolving science, including regularly updating the Vaccine Injury Table.
4.7.3 Continue to ensure fair and efficient compensation.
4.7.4 Examine alternative approaches for adjudication of claims for illnesses not included in the Vaccine Injury Table (and seek Federal legislation as necessary).
Objective 4.8: Enhance the effectiveness of state and federal immunization programs
Federal Departments and Agencies: HHS (ASPR, CDC, HRSA, IHS), DoD, DHS, VA
Non-federal Stakeholders: Health care system, health care payers and plans (public and private) state, local, and tribal governments and public health agencies, vaccine industry
Strategies:
4.8.1 Implement, monitor, and evaluate evidence-based interventions, and translational research, designed to raise and sustain high vaccination coverage in children, adolescents, and adults.
4.8.2 Monitor and evaluate the impact of state immunization laws including childcare, school, and college prematriculation requirements, the role of exemptions, insurance mandates, and immunization information systems requirements.
4.8.3 Prepare, practice, and evaluate mass vaccination activities for containment of an outbreak of a vaccine-preventable disease, for a biological attack, for the critical workforce in advance of an influenza pandemic, and for the entire population, prior to and during, an influenza pandemic.
Objective 4.9: Enhance Immunization Coverage of International Travelers Who Are at Risk of Acquiring Vaccine-Preventable Diseases.
Federal Departments and Agencies: HHS (CDC), USAID
Non-federal Stakeholders: Health care system, health care payers and plans (public and private) state, local, and tribal governments and public health agencies, travel industry, vaccine industry
Strategies:
4.9.1 Define the populations at risk for acquiring international travel-related vaccine-preventable diseases, and identify and address barriers to their receiving immunizations.
4.9.2 Implement and evaluate activities to enhance immunization coverage among travelers.
Goal 5:
Increase global prevention of death and disease through safe and effective vaccination
Introduction
Infectious diseases are the leading cause of death among children globally and contribute substantially to disease and disability among persons of all ages. Immunization programs have been remarkably successful preventing millions of childhood deaths, eradicating smallpox, and eliminating circulation of polio and measles from many countries around the world. However, the continued morbidity and mortality burden from diseases for which vaccination already is routinely recommended, for which vaccines are available but not used in most countries (e.g., pneumococcus and rotavirus), and from diseases for which vaccines are being developed (e.g., HIV, tuberculosis, and malaria) highlights the substantial challenges that remain. Achieving the United Nations Millennium Development Goals of reducing the under-five mortality rate and the proportion of 1 year-old children immunized against measles by two-thirds will require addressing these challenges.
The goals of global vaccination are to eradicate, eliminate or control infectious diseases in a way that supports and strengthens health systems and can be sustained and expanded as new vaccines are developed and introduced. Success in global immunization requires action by the full range of stakeholders involved in the vaccine and immunization enterprise, from research and development, through regulation and manufacturing, to program implementation and monitoring. New partnerships such as the Global Alliance for Vaccines and Immunization (the GAVI Alliance) have led to increased focus and support for immunization worldwide and have catalyzed introduction of new vaccines in most countries and expanded vaccination coverage. U.S. governmental and non-governmental organizations have contributed to progress through vaccine research and development, participation in multilateral and bilateral partnerships, and technical assistance and program support.
Given the breadth of global immunization activities, some of the Objectives and Strategies relevant to this topic are included elsewhere in this Plan. All vaccine research and development are included under Goal 1 as the approach to achieving these objectives and the key stakeholders are not different for the U.S. and the rest of the world. By contrast, issues related to vaccine safety, communications, and program implementation are included under the Global Immunization goal as well as under other goals of the Plan. Whereas many of the objectives in these areas are similar for the U.S. and abroad, the strategies differ as internationally, U.S. stakeholders focus on partnerships and providing assistance rather than on direct implementation as described elsewhere in the Plan. Because the role of U.S. stakeholders in reaching global immunization targets, many of which were established by international organizations such as the World Health Organization, is largely indirect, achievement of these indicators will require actions by the involved countries and international stakeholders and, thus, may be largely outside the control of the United States.
Goal 5 Indicators
-
Transmission of wild polio virus will be eradicated by Y (year).
-
Mortality from measles will be reduced by X% by Y (year) compared with a X (year) baseline.
-
X% of countries will achieve DTP3 vaccination coverage of 90% or greater nationally (and 80% or greater in each country’s district) by Y (year).
-
Support introduction of new vaccines as part of national vaccination programs:
-
Meningococcal vaccine in all African countries in the “meningitis belt” by Y (year);
-
Rotavirus vaccine in X countries by Y (year); and
-
Pneumococcal conjugate vaccine in Z countries by Y (year).
-
-
X countries establish immunization advisory committees by Y (year) that make evidence-based decisions on adding new vaccines to the routine program and monitor program quality, vaccination coverage, and vaccine safety.
-
X countries enhance injection safety by Y (year) through the use of auto-disable syringes or other safe injection devices (e.g., needle free delivery) for all immunizations.
Identifying Goal 5 Objectives and Strategies
Figure 7 is the same as Figures 1 and 2, but it shows components of the vaccine and immunization enterprise (i.e., the shaded rectangular boxes) which contribute to achieving Goal 5 – increasing global prevention of death and disease through safe and effective vaccination. In essence, Goal 5 incorporates elements of each of the other domestically-oriented goals and considers them from a global perspective. The process begins with global disease surveillance informing the recognition of global public health priorities and guiding vaccine research and development activities. The steps from vaccine development to vaccination proceed through a set of activities related to vaccine licensure, developing vaccine recommendations, manufacturing, distribution, communication and education about vaccines, and monitoring adverse events and vaccine effectiveness. Vaccine coverage is influenced by financial and non-financial factors, including attitudes toward vaccination and payment policies. Attitudes and coverage are, in turn, influenced by issues related to vaccine safety and vaccine effectiveness. Issues related to safety and effectiveness inform the next generation of vaccine research and development. The desired outcomes of this process include high vaccination rates which lead to reduced morbidity and mortality from infectious disease, and improved population health around the world. All of these components take place across a wide range of countries with different cultures, environments, and levels of infrastructure. Developing objectives and strategies that recognize and account for these differences is critical for achieving Goal 5.
Figure 7. Components of the Vaccine Process Relevant to Goal 5
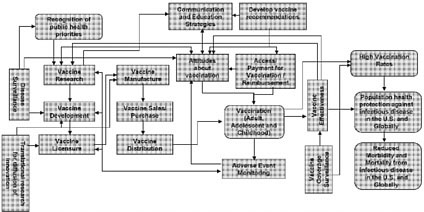
Proposed Objectives
Objective 5.1: Improve global surveillance for VPDs and strengthen health information systems to monitor vaccine coverage, effectiveness, and safety.
Federal Departments and Agencies: HHS (CDC, NIH), USAID, DoD
Non-federal stakeholders: Academia, international organizations, NGOs, public and private philanthropic organizations, UNICEF, World Health Organization
Strategies:
5.1.1 Achieve sustainable WHO certification quality surveillance for eradication targeted VPDs.
5.1.2 Expand and improve sustainable surveillance systems for all current VPDs and for diseases for which vaccine introduction is being considered.
5.1.3 Strengthen all levels of global laboratory networks (including national, regional, and global reference laboratories) to sustain and improve VPD diagnosis in order to establish baseline disease burden, detect outbreaks, detect newly emerging variants of vaccine-preventable diseases, and monitor the impact of new vaccines.
5.1.4 Enhance assessments of emerging variants or strains of vaccine-preventable disease agents.
5.1.5 Develop new diagnostic tests, tools and procedures to improve both field-based and laboratory confirmation of diagnoses.
5.1.6 Improve coverage monitoring of vaccines and other health services linked with the vaccination program and the use of information at district and local levels.
5.1.7 Introduce and improve programs that monitor the occurrence of AEFI.
Objective 5.2: Improve and sustain immunization programs that deliver vaccines safely and effectively as a component of healthcare delivery systems and promote opportunities to link immunization delivery with other priority health interventions, where appropriate.
Federal Departments and Agencies: HHS (CDC), USAID
Non-federal stakeholders: Academia, international organizations, NGOs, public and private philanthropic organizations, UNICEF, World Health Organization
Strategies:
5.2.1 Provide support to countries and partners to strengthen key components of immunization program management and implementation, including epidemiological analysis, comprehensive planning, vaccine distribution and administration, monitoring, and program evaluation.
5.2.2 Provide technical support to countries to introduce, sustain, and monitor recommended safe injection practices for all vaccinations, including the use of auto disable syringes or needle-free devices.
5.2.3 Support linking delivery of immunization and other health services in ways that do not jeopardize immunization coverage, and develop standardized methods for monitoring and evaluating the efficiency, effectiveness and impact of combined interventions to improve coverage and public health.
5.2.4 Encourage establishment of programs, as appropriate, for vaccination beyond the traditional infant target age groups (e.g., among older children, adolescents and adults).
Objective 5.3: Support introduction and availability of new and under-utilized vaccines to prevent diseases of public health importance.
Federal Departments and Agencies: HHS (CDC, FDA, NIH), USAID
Non-federal stakeholders: Academia, international organizations, NGOs, public and private philanthropic organizations, UNICEF, World Health Organization
Strategies:
5.3.1 Collaborate with global organizations and partners to accelerate the clinical testing and licensure, where appropriate, in developing countries of vaccines already licensed in developed countries.
5.3.2 Strengthen country capacity to make informed decisions on introduction of new vaccines based on evaluation of epidemiology, financial sustainability, safety, and programmatic considerations.
5.3.3 Support the integration of new and under-utilized vaccine into each GAVI-eligible country’s multi-year national plan of action and provide training and logistical support necessary to successfully incorporate new vaccines into routine programs.
5.3.4 Conduct post-licensure evaluations of the impact of new vaccines on immunization programs, disease patterns, and the occurrence of AEFI.
Objective 5.4: Improve communication of research-based and culturally and linguistically appropriate information about the benefits and risks of vaccines to the public, providers, and policy-makers.
Federal Departments and Agencies: HHS (CDC), USAID
Non-federal stakeholders: Academia, international organizations, NGOs, public and private philanthropic organizations, UNICEF, World Health Organization
Strategies:
5.4.1 Support appropriate economic studies to inform the understanding of the costs and benefits of immunization among key decision and policy-makers.
5.4.2 Develop and support capabilities to communicate vaccine risks and to respond to emerging vaccine safety issues and concerns to the public, providers, and other stakeholders in a clear, transparent and timely manner.
5.4.3 Provide assistance in determining the most effective and efficient mechanisms to communicate with health care providers about reporting on AEFI; evaluate providers’ knowledge and adherence to recommendations to prevent AEFI; and improve and assess adherence to these recommendations.
5.4.4 Assist countries to develop, implement and assess comprehensive evidence-based communication plans to increase provider and public awareness of vaccine preventable
diseases and promote immunization recommendations, especially among populations at risk of under-immunization.
5.4.5 Assist countries to develop and implement sustainable communication research to gather timely and reliable data from the public and providers on knowledge, attitudes and beliefs about the benefits and risks of vaccines.
5.4.6 Provide technical assistance and training to behavioral and communications scientists and promote their participation on Technical Advisory Groups.
Objective 5.5: Support the development of regulatory environments and manufacturing capabilities that facilitate access to safe and effective vaccines in all countries.
Federal Departments and Agencies: HHS (BARDA, CDC, FDA), USAID
Non-federal stakeholders: Academia, international organizations, NGOs, public and private philanthropic organizations, vaccine industry, World Health Organization
Strategies:
5.5.1 Promote and support the efforts of the World Health Organization to develop and harmonize international standards and norms to assure the quality, safety and efficacy of vaccines and to provide a predictable environment for vaccine development.
5.5.2 Promote and support the efforts of the World Health Organization to improve regulatory capacity in countries with limited infrastructures to assure vaccine quality, evaluate new vaccines when appropriate and assure that clinical trials are conducted in accordance with Good Clinical Practices.
5.5.3 Support efforts to harmonize international vaccine licensing regulations.
5.5.4 Provide technical assistance to developing country vaccine manufacturers to support development and production of safe and effective vaccines and related technologies.
Objective 5.6: Build and strengthen multilateral and bilateral partnerships and other collaborative efforts to support global immunization and eradication programs.
Federal Departments and Agencies: HHS (CDC, FDA, NIH), USAID
Non-federal stakeholders: Academia, international organizations, public and private philanthropic organizations, UNICEF, World Health Organization
Strategies:
5.6.1 Participate in establishing global immunization priorities, goals and objectives and provide technical assistance at global, regional, and national forums.
5.6.2 Strengthen international collaborations for basic and applied research, especially onsite research in disease endemic areas or those with the greatest burden of disease.
5.6.3 Work with global partners to establish an international system that facilitates rapid response to emerging infections through the development of vaccine reference strains and candidate vaccines.
5.6.4 Contribute to development and implementation of a research agenda establishing the scientific basis for VPD eradication/elimination; identifying optimal vaccination approaches; and developing strategies to minimize risks in the post-eradication period.
5.6.5 Build and strengthen bilateral and multilateral partnerships and other collaborative efforts to support availability, access, sustainable financing, and use of current, under-utilized, and new vaccines.
5.6.6 Work with global partners to secure and maintain adequate stockpiles/strategic reserves of vaccines to maintain uninterrupted supply, for emergency response to outbreaks, and for special purposes.
5.6.7 Work with global partners to develop a global advocacy agenda and create a positive environment for vaccine use.
Appendix 1.
Anticipated outcomes from the 1994 National Vaccine Plan and the extent to which each has been achieved at the time the release of draft strategic National Vaccine Plan (November 2008)
|
Anticipated outcome |
Status |
|
Age-appropriate immunization with all recommended vaccines will be extended to at least 90% of infants and children, and access to affordable vaccination services will be made available for every person in the United States. |
|
|
Diphtheria, tetanus, poliomyelitis, measles, rubella mumps, some forms of hepatitis, pertussis (whooping cough), and bacterial meningitis (from Haemophilus influenzae type b) will be essentially eliminated as significant causes of death, disease and disability in the United States. |
|
|
Educational communication networks will be in place that will inform all healthcare providers, communities, and families of the benefits and risks of |
|
|
vaccination. |
providers, families, and communities about vaccine benefits and risks, internet-based information is significantly more available than it was in 1994. For example, for providers, the AAP and AAFP newsletters and email blasts constitute a network, as does IDSA’s similar capacity with members. CDC/NCIRD’s Immunization Works is a regular effort to update programs and partners, and DoD’s Milvax internet site has abundant information about their vaccination programs. |
|
In a global context, polio will be drastically reduced, if not eliminated, and neonatal tetanus and measles will be better controlled. |
|
|
Pneumococcal pneumonia and influenza in American adults over the age of 65 will be significantly reduced. |
|
|
A nationwide system will monitor the vaccines that children receive, and will remind parents when |
|
|
individual infants and children should be vaccinated. |
vaccination varies (see http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5 711a4.htm) |
|
A nationwide surveillance system will report and investigate cases of vaccine-preventable-diseases. |
|
|
Vaccine safety and efficacy will be continuously monitored, and adverse events following immunization will be reported and carefully analyzed. |
|
|
Improved vaccines will replace some of the vaccines in current use. |
|
|
Some vaccines requiring multiple doses and multiple contacts with the health care system will be replaced by more cost-effective ones that will improve people’s access to immunization. |
|
|
Many new vaccines will be developed or be much closer to licensure, for diseases for which effective |
|
|
vaccines do not now exist. |
|
|
New mechanisms for the more rapid assessment of vaccines proposed for licensure will be in place. |
|
|
A reliable supply of all recommended vaccines and a capability to respond to emergencies and emergent threats to public health will be achieved and sustained. |
|
|
Information on the costs and benefits of the National Vaccine Plan will be made available on an ongoing basis to the American people. |
|
Appendix 2:
IOM committee recommendations from the June 11, 2008 letter report “Initial Guidance for an Update of the National Vaccine Plan: A Letter Report to the National Vaccine Program Office” and National Vaccine Program Office responses9
The National Vaccine Program Office (NVPO) and other offices and agencies in the Department of Health and Human Services (HHS) have reviewed the recommendations in the June 11, 2008 letter report of the Institute of Medicine’s Committee on the Review of Priorities in the National Vaccine Plan. We appreciate the Committee’s work and thoughtful recommendations. Our response to each recommendation follows its citation below.
Recommendation 1: The committee recommends that NVPO and its partners include for each strategic initiative listed under the four plan goals the following details:
-
The primary responsible party (government agency or other stakeholder)
-
Secondary participant(s) (government agency or other stakeholder)
-
Measurable short, mid, and longer term outcomes to assess success of the initiative
-
Identification of costs and potential funding sources (e.g., professional judgment budgets) to support pursuit of the initiative
-
The plan also should include a timetable and process for regular updates that reflect the dynamic nature of the field.
The draft strategic National Vaccine Plan has listed HHS agencies and other federal Departments responsible for each objective in it, as well as possible non-federal stakeholder sectors. . Input and comments from non-federal stakeholders (including the public), and recommendations from the IOM committee all will contribute to final Plan to be released in the fall, 2009. This Plan, in turn, will serve as the basis for implementation planning by Federal agencies and non-Federal stakeholders which will define specific actions that will be undertaken to accomplish each of the strategies in the Plan and timelines and measurable outcomes to assess progress.
Identification of resource needs will follow identification of specific actions in the implementation plan, in consultation with stakeholders.
NVPO will be responsible for assuring coordination and for monitoring federal actions and accomplishments on an ongoing basis. NVAC will establish a working group to promote coordination and monitor progress by the broader group of stakeholders. Each year, NVPO and NVAC will report their findings to the Assistant Secretary for Health.
|
9 |
Recommendations from the Institute of Medicine Committee on the Review of Priorities in the National Vaccine Plan letter report “Initial Guidance for an Update of the National Vaccine Plan: A Letter Report to the National Vaccine Program Office,” June 11, 2008 available at http://www.iom.edu/CMS/3793/55143.aspx. |
This report will include a summary of progress, identify areas where progress is lagging, and propose corrective action where needed. The report also will be summarized annually at an NVAC meeting, which is open to the public and is attended by many stakeholders not represented directly on the Committee.
Recommendation 2: The committee recommends that NVPO and its partners identify specific and creative strategies (not limited to funding) that federal agencies and programs could use to motivate stakeholders to implement objectives in the national vaccine plan.
NVPO believes the three following steps will be essential strategies motivating stakeholders to implement the National Vaccine Plan: 1) reviewing and commenting on the Goals, Objectives and Strategies in the draft Plan; 2) participating in developing, and assuming responsibility for implementing, measurable milestones for the implementation plan; and 3) working with NVPO and NVAC to monitor progress in implementing these milestones.
Recommendation 3: The committee recommends that NVPO and its partners explain in the draft update to the National Vaccine Plan what was important to include and why, and the process by which items were selected for inclusion or discarded.
The draft strategic National Vaccine Plan represents the aggregate input from HHS federal agencies and Departments most involved in vaccine issues about how to improve the current United States vaccine system. The component maps of the vaccine and immunization enterprise address these issues and offer a framework for the Goals, Objectives, and Strategies. Some elements have not been included, post-exposure prophylaxis, and therapeutic vaccines. The National Vaccine Program was established by congressional legislation (Title XXI of the Public Health Service Act [Public Law 99-660]) in 1986 to achieve optimal prevention of infectious diseases through immunization and optimal prevention of adverse reactions to vaccines. NVPO recognizes the need for strategic planning for therapeutic vaccines and will seek to identify mechanisms to address this need separately from this Plan.
Recommendation 4: The committee recommends that NVPO and its partners include in the update to the National Vaccine Plan mechanisms to assess the “horizon” of innovation and new developments in vaccines, and explore strategic objectives or initiatives that enable timely consideration of and decision making to address emerging opportunities and challenges.
NVPO agrees that a flexible approach is important to assure timely response to emerging opportunities and challenges. In developing this draft Plan, NVPO analyzed the strengths, weaknesses, opportunities, and threats to the U.S. vaccine system. The draft Plan has incorporated the results of that analysis, and believes the regular monitoring of the progress in the Plan, as well as its objectives and strategies, will permit rapid assessment and response to those emerging opportunities and challenges.
Recommendation 5: The committee recommends that the update to the National Vaccine Plan include a comprehensive framework for communicating with the public and other key stakeholders such as healthcare providers about the benefits (both individual and community) and risks of vaccination. Communication strategies that are implemented should be evaluated for their effect on knowledge and behavior.
The draft Plan has maintained the focus of Goal 3 from the 1994 Plan on communications and education issues, including benefits and risks of vaccines, and including vaccine issues in health professional schools’ curricula and training programs. Evaluation and monitoring of proposed communication and education strategies is outlined in the draft, and are essential to improving this area, as they are for all aspects of the Plan.
Recommendation 6: The committee recommends that NVPO and its partners consider ways the update to the National Vaccine Plan could spur research for creative solutions to vaccine supply problems.
Goal 4 is now entitled Ensure a stable supply of recommended vaccines, and increase uptake of existing vaccines to prevent disease, disability, and death in the United States, demonstrating the importance of vaccine supply issues in the draft Plan. The Plan discusses improving vaccine stockpiles, centralized vaccine distribution, and Good Manufacturing Practices. The Committee also emphasized the importance of addressing health disparities in vaccination levels, including access and financial issues. Concepts from the NVAC Vaccine Financing Working Group’s recommendations have been incorporated into the draft Plan.
Appendix 3:
Stakeholders In the United States National Vaccine System*
Academic research
-
Researchers
-
Universities
-
Vaccine manufacturers
-
Research Organizations
-
-
Research funders
-
National Institutes of Health
-
Philanthropic organizations
-
-
Institute of Medicine
-
The Brighton Collaboration
Adocacy groups
Federal government Departments and Agencies
-
Department of Health and Human Services
-
Office of the Assistant Secretary for Health
-
National Vaccine Program Office
-
Office of the Surgeon General
-
-
Office of the Assistant Secretary for Legislation
-
Office of the Assistant Secretary for Planning and Evaluation
-
Office of the Assistant Secretary for Public Affairs
-
Office of the Assistant Secretary for Preparedness and Response
-
Biomedical Advanced Research and Development Authority
-
-
Office of the Assistant Secretary for Public Affairs
-
Office of the General Counsel
-
Office of Global Health Affairs
-
Office of Intergovernmental Affairs
-
Office of the National Coordinator for Health Information Technology
-
Administration on Aging
-
Agency for Healthcare Research and Quality
-
Centers for Disease Control and Prevention
-
Centers for Medicare and Medicaid Services
-
Food and Drug Administration
-
Health Resources and Services Administration
-
Indian Health Service
-
National Institutes of Health
-
Substance Abuse and Mental Health Services Administration
-
-
Department of Agriculture
-
Department of Defense
-
Assistant Secretary for Health Affairs
-
US Army Medical Research Institute of Infectious Diseases (USAMRIID)
-
The Military Vaccine (MILVAX) Agency
-
Defense Advanced Research Projects Agency
-
-
-
Defense Threat Reduction Agency
-
-
Department of Homeland Security
-
Department of Justice
-
Department of Labor
-
Occupational Safety and Health Administration
-
-
Department of State
-
U.S. Agency for International Development
-
-
Department of Veterans Affairs
The general public
-
Individuals
-
Families
-
Schools
-
Employers
-
Churches and other faith-based institutions
HHS Federal advisory committees
-
Advisory Commission on Childhood Vaccines (ACCV)
-
Advisory Committee on Immunization Practices (ACIP)
-
National Advisory Allergy and Infectious Disease Council (NAAIDC)
-
National Biodefense Science Board (NBSB)
-
National Vaccine Advisory Committee (NVAC)
-
Vaccines and Related Biologicals Products Advisory Committee (VRBPAC)
Health care system
-
Health care providers, hospitals, clinics, and federally qualified health centers, long-term care facilities, managed care organizations (MCOs)
-
Medical professional societies
-
Health care professional schools
-
Health care training programs
-
Pharmacies
-
Community vaccinators
-
Electronic health record vendors
Health care payers and plans (public and private)
-
Benefit managers
-
Coverage policy, reimbursement
-
Quality monitoring systems
International organizations
-
WHO, UNICEF
-
Other countries
-
National regulatory agencies
Media
Non-governmental organizations
Philanthropic organizations
State, local, and tribal governments and public health agencies
Travel industry
Vaccine industry (manufacturers [large companies, biotechnology companies, both foreign and U.S.-based]
Vaccine distributors
Vaccine investors
|
* |
Adapted from the Initial Guidance for an Update of the National Vaccine Plan, A Letter Report to the National Vaccine Program Office, June 11, 2008, available at http://www.iom.edu/CMS/3793/55143.aspx, The sectors, and groups listed under each sector, are examples and may not be all-inclusive. |
Appendix 4:
Roles and Responsibilities of Department of Health and Human Services Agencies and Offices, and other federal Departments in the draft strategic National Vaccine Plan
|
Secretary of Health and Human Services |
|
|
Office of the Assistant Secretary for Health |
|
|
Office of the Assistant Secretary, Administration on Aging |
|
|
|
|
Office of the Assistant Secretary, Administration for Children and Families |
|
|
|
Office of the Assistant Secretary for Legislation |
|
|
Office of the Assistant Secretary for Planning and Evaluation |
|
|
|
|
|
Office of Assistant Secretary for Preparedness and Response (ASPR) |
|
|
|
|
|
Office of the Assistant Secretary for Public Affairs |
|
|
Office of Intergovernmental Affairs |
|
|
Office of the Surgeon General |
|
|
Office of the General Counsel |
|
|
Office of Global Health Affairs |
|
|
|
coordination with the Department of State |
|
Office of the National Coordinator for Health Information Technology |
|
|
|
|
National Vaccine Program Office (NVPO) |
|
|
|
|
Agency for Healthcare Research and Quality |
|
|
|
|
Centers for Disease Control and Prevention (CDC) |
|
|
|
|
|
|
|
efforts and accelerating use of new and underutilized vaccines |
|
Centers for Medicare and Medicaid (CMS) |
|
|
Food and Drug Administration (FDA) |
|
|
|
|
|
National Institutes of Health (NIH) |
|
|
Health Resources and Services Administration (HRSA) |
|
|
|
|
|
Indian Health Service |
|
|
Substance Abuse & Mental Health Services Administration |
|
|
|
|
|
Department of Agriculture |
|
|
|
|
|
Department of Defense |
|
|
Assistant Secretary of |
|
|
Defense for Health Affairs |
Secretary of Defense for health service support for pandemic influenza preparedness and response
|
|
Defense Advanced Research Projects Agency |
|
|
Defense Threat Reduction Agency |
|
|
The Military Vaccine (Milvax) Agency |
|
|
|
|
|
|
|
|
United States Army Medical Institute for Infectious Diseases |
|
|
|
|
|
|
|
|
United States Agency for International Development |
|
|
vaccine preventable diseases such as measles and neonatal tetanus.
|
|
|
|
|
Department of Veterans Affairs (VA) |
Appendix 5:
HHS Agency and Other Federal Department Strategic Plans relevant to the draft strategic National Vaccine Plan
|
HHS: |
U.S. Department of Health and Human Services Strategic Plan - FY 2007-2012 |
|
|
|
||
|
ASPR: |
HHS PHEMCE Strategy and HHS PHEMCE Implementation Plan |
|
|
|
http://www.hhs.gov/aspr/barda/phemce/enterprise/strategy/index.html |
|
|
|
Draft BARDA Strategic Plan for Medical Countermeasure Research, Development, and Procurement |
|
|
|
http://www.hhs.gov/aspr/barda/phemce/enterprise/strategy/bardaplan.html |
|
|
CDC: |
National Immunization Program Strategic Plan 2000-2005 |
|
|
|
Immunization Information Systems Strategic Plan 2002-2007 http://www.cdc.gov/vaccines/programs/iis/activities/strategic-plan.htm |
|
|
|
CDC Global Immunization Strategic Framework 2006-2010 http://www.cdc.gov/vaccines/programs/global/downloads/gisf-2006-2010.pdf |
|
|
FDA: |
FDA Strategic Action Plan, Fall, 2007 |
|
|
ODPHP: |
|
HealthyPeople 2010 |
|
|
|
HealthyPeople 2020 |
|
HRSA: |
HRSA Strategic Plan FY 2005-2010 |
|
|
NIH: |
NIAID Strategic Plan for Biodefense Research, 2007 http://www3.niaid.nih.gov/topics/BiodefenseRelated/Biodefense/about/strategicplan.htm |
|
|
|
NIAID: Planning for the 21st Century http://www3.niaid.nih.gov/about/overview/planningPriorities/strategicplan/ |
|
|
Department of Agriculture |
||
|
|
USDA Strategic Plan 2005-2010 |
|
|
DoD: |
U.S. Department of Defense Military Health System Strategic Plan |
|
|
DARPA |
Defense Advanced Research Projects Agency Strategic Plan 2007 http://www.darpa.mil/body/pdf/DARPA2007StrategicPlanfinalMarchl4.pdf |
|
|
DoJ |
U.S. Department of Justice Strategic Plan 2007-2012 |
|
|
VA: |
VA Strategic Plan (2006-2011) |
|
|
USAID: |
|
Strategic Plan: Fiscal Years 2007-2012 http://www.usaid.gov/policy/coordination/stratplan_fy07-12.html |