1
Introduction
GENESIS AND SCOPE OF THE STUDY
This report, which was prepared by a committee of the National Research Council's Energy Engineering Board, assesses possible applications of concentrated solar energy to various processes other than the production of electricity.
The committee was asked to perform the following tasks:
-
Assess potential applications for the ''near term" and "long term" of concentrated solar energy in nonelectric areas;
-
Assess the knowledge base for those applications; and
-
Recommend research paths and priorities to enhance the knowledge base and increase the potential for development of successful applications.
The committee was asked to restrict its scope to terrestrial applications of concentrated solar energy. (See Appendix A for a complete statement of task.)
Technical Background
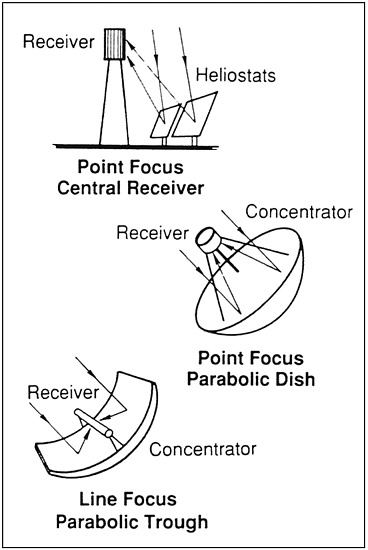
The Department of Energy's Solar Thermal Technology Program was started in 1975 and was concerned mainly with the use of concentrated solar energy to produce electricity. In solar thermal systems, solar radiation is concentrated with mirrors onto a receiver, where it is absorbed as heat. The resulting heat is then converted into electricity by a thermal converter, such as a turbine. There are several different types of solar concentrators: parabolic troughs, parabolic dishes, and heliostat/central receiver systems (Figure 1-1) [1]. Parabolic troughs are line focus reflectors that concentrate sunlight onto receiver tubes along their focal lines. Parabolic dishes track the sun and use mirrors and lenses to focus the sunlight onto a receiver. Central receiver systems employ fields of heliostats (tracking mirrors) to focus the solar energy on a tower-mounted receiver. These devices differ in the degree of concentration of solar flux, available temperature, and cost (Table 1-1). Solar concentration with holographic elements and Fresnel lenses is also possible. The energy flux of unconcentrated solar energy (1 sun) depends on
TABLE 1-1 Typical Characteristics of Systems Concentrating Solar Photons
|
Nominal Concentration Ration (suns) |
Concentrating System |
Typical Focal Length (m) |
Nominal Flux Densitya |
Spectral Flux Densityb |
Typical Temperatures (°C) |
Heating Ratec (°C/s) |
Working Area |
Installed Cost ($/kWt) |
||
|
(W/cm2) |
(ph/s/cm2) |
300–400 nm (ph/s/cm2× 10-16) |
300–700 nm (ph/s/cm2× 10-16) |
|||||||
|
1 |
Flat plate collector |
N/A |
0.1 |
3.5 × 1017 |
0.5 |
9.9 |
<100 |
— |
30–40f |
|
|
100 |
Parabolic trough |
1 |
10 |
3.5 × 1019 |
50 |
990 |
<400 |
— |
300–400h |
|
|
1000 |
Parabolic dish; heliostats |
5–10 (dish) 500 (central receiver) |
100 |
3.5 × 1020 |
500 |
9900 |
1200c |
4 × 109 |
0.1 M2 (dish) 1000 m2 (central) |
400–500g |
|
10,000e |
Compound parabolic secondary concentrator |
7e (primary) |
1000 |
3.5 × 1021 |
5 × 103 |
99 × 103 |
2300c |
4 × 1010 |
15 mm diametere |
50,000i |
|
>50,000e |
High refractive index secondary concentrator |
7e (primary) |
>5000 |
17.5 × 1021 |
>25 × 103 |
>500 × 103 |
3600c |
2 × 1011 |
7 mm |
|
|
a Based on a nominal insolation of 1000 W/m2. Reflection losses, etc., are not shown. Losses for a one-reflection system (troughs, dishes, heliostats) are less then 10%; for a two-reflection system (heliostat/concentrator), losses are less then 20%; with a secondary concentrator, losses are on the order of 30–40%. Aluminum reflectors will preserve higher frequencies better than silver reflectors, but they have lower overall reflectance in the solar spectrum. b Based on air mass 1.5. Does not include absorption losses, which depend on the spectral properties of elements in the optical train. c Temperature based on the equilibrium, uniform temperature of a small particle, α = ε = 1. Heating rate assumes 1-µm particle; density, 2000 kg/m3; specific heat, 0.2 J/kg K. Actual temperature determined by receiver/reactor design, including ability to contain high temperatures. For surfaces, heating rate and surface temperature depend on thermal diffusivity and emissivity of material exposed. d Not limited by physical considerations (subject to plant size requirements and economic optimization). e Based on the Solar Energy Research Institute's (SERI) 10-kW high-flux solar furnace and research at the University of Chicago. An average concentration on the target area of 21,000 suns has been measured at SERI. f Based on flat plate collector system technology. g Based on a number of dish and central receiver cost estimates. h Bascd on actual installed costs in existing systems (IST). i Based on SERI's high-flux solar furnace. Costs for the furnace at Sandia National Laboratories (with a maximum concentration of 5000 suns) are approximately $13,000 kWt. Both are one-of-a-kind research facilities. Commercial furnaces dedicated to specific processes and applications will undoubtedly cost considerably less. SOURCE: SERI, private communication, 1990. |
||||||||||
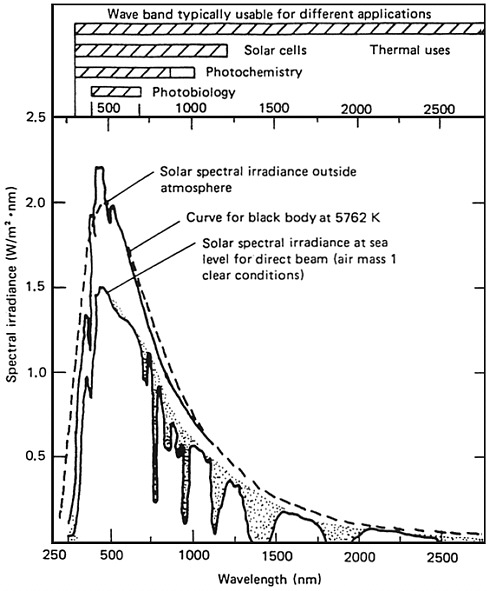
geographical location, time of day, and season. For reference purposes the solar spectrum at AMI (air mass 1) is shown in Figure 1-2; the maximum flux in the southwestern part of the United States is about 1000 W/m2, and the average flux is about 250 W/m2 [2]. Solar thermal electricity systems are now developed to the point where they are entering the commercial electricity market. For example, parabolic trough-based systems with a capacity of 200 MW are being installed in southern California, and central receiver systems have operated in Barstow, California (10 MW), and Almeria, Spain (1 MW).
The oil crisis in the 1970s spawned rather extensive efforts in the use of light energy to drive useful chemical reactions (solar photochemistry and photoelectrochemistry). Reactions of interest included those that produce possible fuels (mainly hydrogen), interesting synthetic reactions, and reactions for decomposition of waste materials in water. There was also substantial work on the use of aqueous and nonaqueous systems involving semiconductor materials as electrodes for the production of electricity [3].
The use of semiconductor particles, such as TiO2, for the decomposition of low concentrations of waste materials in aqueous solutions was reported as early as 1977 [4].
Photochemistry is an old and developed field [5,6]. Thousands of photochemical reactions have been studied, and the nature of the interaction of radiant energy with molecules is considered a well-understood field, having been investigated for many years. Most studies in these areas, however, have involved light intensities on the order of 1 sun or less. Photochemical and photophysical processes are widely encountered in industry, offices, and the environment. These applications include laser cutting and welding, photoresist technology in microelectronics, radiative modification of fibers, and photocopying methods. The environmental conversions of pollutants in the atmosphere and water, the formation of photochemical smog, the dynamics of the ozone layer, and biological photosynthesis represent important, large-scale processes involving photochemistry. Thus, photochemistry and photophysics are important in many applications and natural processes.
The goal of the present work was to determine whether special advantages might result from using concentrated solar photons for photochemical, photoelectrochemical, and thermal processes and to assess the state of the art of potential applications, such as water and waste treatment. Other possible applications of solar photons, such as materials processing and solar pumping of lasers, were also considered.
In assessing potential applications of solar energy, its particular advantages and limitations must be considered. Solar energy is an environmentally friendly and clean energy source. It does not produce pollutants that lead to acid rain or lead to production of greenhouse gases that could contribute to global warming. Solar energy provides a virtually unlimited fuel supply, and solar energy systems can be configured on both large and small scales. Thus, small decentralized collection installations are possible, with potential decreased transmission or distribution losses and costs. However, sunlight is a diffuse energy source, and its collection requires large areas. In other words, solar energy installations have a large "footprint" on the order of square meters per kilowatts, compared with other energy technologies in which areas on the order of square meters per megawatts are typical. Large arrays of collectors may be unsightly and result in rather high capital equipment costs. Solar energy is an intermittent and unreliable energy source. It can be used for only a fraction of a day and may not be available on demand. Moreover, solar radiation itself is not directly storable. These considerations become important in comparison of solar-energy-based technologies with competitive ones. Finally, the energy distribution of solar radiation is largely in the visible and near-infrared regions, regions generally not suitable for most photochemical or semiconductor photoelectrochemical reactions of interest. For example, terres-
trial sunlight only has about 4 percent of its spectrum in the ultraviolet region (energies greater than about 3.0 eV). Solar energy is most useful in locations remote from other available power sources (e.g., space applications). This report, however, deals mainly with potential terrestrial applications.
ORGANIZATION OF THE STUDY AND REPORT
Beyond reliance on its members' expertise and examination of an extensive literature on solar chemistry and applications, the committee invited a number of experts to provide briefings on pertinent issues in a two-day meeting and workshop (see Appendix B). (The proceedings of the workshop are published separately.)
Furthermore, as part of the committee's mandate to review relevant international research and development activities in solar chemistry and technology, it undertook two efforts. The committee chairman, Allen Bard, attended the International Energy Agency Symposium on Solar High-Temperature Technologies, in Davos, Switzerland, on August 27–31, 1990, and exchanged perspectives with various European experts at the symposium. In a subsequent effort, committee member Nick Serpone and study director, Kamal J. Araj visited a number of European research establishments and facilities to exchange views and perspectives and gather information (see Appendix C).
This report consists of four chapters and is intended to be short and succinct as it draws heavily, for background information and details, on the workshop proceedings. Chapter 2 provides a critical review and evaluation of six potential nonelectric applications of concentrated solar energy, and identifies new opportunities for long-term basic research. Chapter 3 addresses organizational and structural aspects of the research and development process. Chapter 4 presents the committee's conclusions and recommendations.
REFERENCES
1. Becker, M., ed. "Solar Thermal Central Receiver System." Proceedings of the 3rd International Workshop (1986).
2. Kreith, F. and Kreider, J.F. Principles of Solar Engineering. Hemisphere Publishing Corp., Washington, DC. (1978).
3. Cardon, F., Gomes, W.P., Dekeyser, W., eds., Photovoltaic and Photoelectrochemical Solar Energy Conversion. Plenum Press. New York (1981).
4. Frank, S.N., Bard. A.J. J. Amer. Chem. Soc., 99, 303 (1977).
5. Calvert, J.G., and Pitts, Jr., J.N. Photochemistry. Wiley, New York (1966).
6. Rabek, J.R., ed. Photochemistry and Photophysics. CRC Press, Boca Raton, Fla. (1990).