1
Ballast Water and Nonindigenous Species
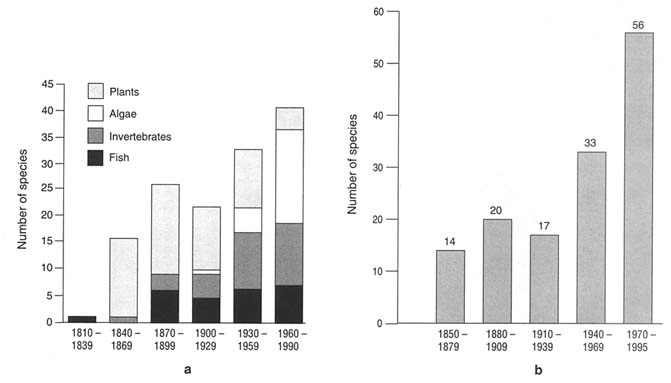
The discovery in the 1980s of the fouling European zebra mussel in the Great Lakes, of a toxic Japanese dinoflagellate in Australia, and a carnivorous North American comb jellyfish in the Black Sea—all of whose successful establishments led to critical economic, human health, and ecological concerns—focused scientific, governmental, and public attention on an age-old phenomenon: the introduction of nonindigenous organisms transported to new locations by ships. There are many dispersal vectors for the transport or organisms on board ships. The most important of these appears to be water in ships' ballast1 tanks and in cargo/ballast holds (Carlton, 1985; Williams et al., 1988; Carlton and Geller, 1993). It has been estimated that in the 1990s ballast water may transport over 3,000 species of animals and plants a day around the world (NRC, 1995), and there is evidence that the number of ballast-mediated introductions is steadily growing. More than 40 species have appeared in the Great Lakes since 1960; more than 50 have appeared in San Francisco Bay since 1970 (see Figure 1-1).
Ballast water and sediment frequently contain abundant living organisms reflecting in large part whatever is in the water around and under the ship at the time of ballasting. In turn, ballast water released from ships acts as an inoculation mechanism for nonindigenous species. Nonindigenous species—also known as exotic species, alien species, and biological invasions—are defined in this report as any species or other viable biological material that enters an ecosystem beyond
|
1 |
Ballast is defined as any solid or liquid placed in a ship to increase the draft, to change the trim, to regulate the stability, or to maintain stress loads within acceptable limits. For the purposes of this study, the term ballast includes the sediment that accumulates in ballast tanks, which may be discharged with ballast water (see Chapter 2). |
TABLE 1-1 Examples of Shipborne Introductions Worldwide since the 1980s
|
Species |
Origin |
Location |
|
Dinoflagellates |
|
|
|
Gymnodinium catenatum |
Japan |
Australia |
|
Comb Jellyfish (Ctenophora) |
|
|
|
Mnemiopsis leidyi |
North America |
Black and Azov Seas |
|
American Comb Jellyfish |
|
|
|
Polychaete Worms (Annelida) |
|
|
|
Marenzelleria viridis |
North America |
Western and Northern Europe |
|
Spionid Tubeworm |
|
|
|
Mussels and Clams (Bivalvia) |
|
|
|
Ensis americanus |
North America |
Western and Northern Europe |
|
American Razor Clam |
|
|
|
Musculista senhousia |
Japan |
New Zealand |
|
Japanese Mussel |
|
|
|
Crabs (Decapoda) |
|
|
|
Charybdis helleri |
Mediterranean |
Colombia, Venezuela, Cuba, and United States |
|
Indo-Pacific Swimming Crab |
|
|
|
Seastars (Asteroidea) |
|
|
|
Asterias amurensis |
Japan |
Australia |
|
North Pacific Seastar |
|
|
|
Sources: Carlton and Geller, 1993; Carlton et al., 1995; LeMaitre, 1995. |
||
its historic range, including any such organism transferred from one country into another (P.L. 101-646, 1990). Tables 1-1 and 1-2 provide examples of shipborne introductions.2
Humans and ships have moved across the oceans for centuries; however, trade routes and the vessels that sail on them have changed and continue to change, as have the environments that donate and receive nonindigenous species. Because the mechanisms that transport organisms from one region to another and to the ocean are dynamic systems and are everchanging, nonindigenous species continue to be introduced (Carlton, 1996). Such introductions may have critical economic, industrial, human health, and ecological consequences. Thus, there are compelling arguments for reducing the role of ships as a vector of nonindigenous species, particularly through ballast water. For example, the three introductions noted above (i.e., zebra mussel, dinoflagellates, and comb jellyfish) have cost tens of millions of dollars and have had deep and broad ecological repercussions (Hallegraeff, 1993; Nalepa and Schloesser, 1993; Harbison and Volovik, 1994).
TABLE 1-2 Examples of Shipborne Introductions in the United States since the 1970s
|
Species |
Origin |
Location |
|
Jellyfish (Hydromedusae) |
|
|
|
Maeotias inexspectata |
Black Sea |
Chesapeake Bay |
|
Black Sea Jellyfish |
|
San Francisco Bay |
|
Blackfordia virginica |
Black Sea |
Chesapeake Bay |
|
Black Sea Jellyfish |
|
San Francisco Bay |
|
Water Fleas (Cladocera) |
|
|
|
Bythotrephes cederstroemi |
Europe |
Northeastern North America |
|
Spiny water flea |
|
|
|
Copepods (Copepoda) |
|
|
|
Limnoithona sinensis |
China |
San Francisco Bay |
|
Oithona davisae |
Japan |
San Francisco Bay |
|
Sinocalanus doerrii |
China |
San Francisco Bay |
|
Pseudodiaptomus marinus |
Japan |
San Francisco Bay |
|
Pseudodiaptomus inopinus |
Asia |
Columbia River |
|
Pseudodiaptomus forbesi |
China |
San Francisco Bay |
|
Crabs (Decapoda) |
|
|
|
Hemigrapsus sanguineus |
Japan |
Massachusetts to Virginia |
|
Japanese shore crab |
|
|
|
Mussels, Clams, and Snails (Mollusca) |
|
|
|
Dreissena polymorpha |
Eurasa |
Eastern North America |
|
Zebra Mussel |
|
|
|
Dreissena bugensis |
Eurasia |
Eastern North America |
|
Quagga Mussel |
|
|
|
Perna perna |
South America |
Gulf of Mexico |
|
South American Mussel |
|
|
|
Potamocorbula amurensis |
China, Japan |
San Francisco Bay |
|
Asian clam |
|
|
|
Philine auriformis |
New Zealand |
California |
|
New Zealand Seaslug |
|
|
|
Moss Animals (Bryozoa) |
|
|
|
Membranipora membranacea |
Europe |
Gulf of Maine to New York |
|
Kelp bryozoan |
|
|
|
Fish (Osteichtyes) |
|
|
|
Neogobius melanostamus |
Eurasia |
Great Lakes |
|
Round goby |
|
|
|
Preteorhinus Marmoratus |
Eurasia |
Great Lakes |
|
Tubenose goby |
|
|
|
Gymnocephalus cernuus |
Europe |
Great Lakes |
|
Ruffe |
|
|
|
Mugiligobius parvus |
Philippines |
Hawaii |
|
Philippine Goby |
|
|
|
Source: Carlton, 1985; Carlton et al., 1990, 1995; Gosliner, 1995; Mills et al., 1993; Mills and Sommer, 1995; Randall et al., 1993. |
||
DIVERSITY OF BALLAST WATER BIOTA
Virtually all ballast water samples taken to date in Canadian, Australian, German, and U.S. studies contained living macro-organisms, indicating that ballast water, including sediment, is a viable habitat for a wide variety of freshwater, brackish water, and saltwater organisms (Bio-Environmental Services, Inc., 1981; Jones, 1991; Hallegraeff and Bolch, 1992; Carlton and Geller, 1993). The potential diversity of biota in ballast is vast; virtually all organisms less than 1 cm in size that are adjacent to the vessel—naturally swimming in the water, stirred up from bottom sediment, or rubbed off harbor pilings—could be ballasted into a vessel. These include viruses, bacteria, protists (including protozoans), fungi and molds, and plants and animals. It is important to note that the parasites and symbionts of all of these organisms will, of course, also be transported.
The maximum size range of organisms that can be taken into a ship depends upon the method of ballasting and the size of the intake screens. Pumped water passes organisms through pump impellers, which may kill some organisms. If gravity-loading is used, organisms are not passed through an operating pump, thus eluding possible mechanical destruction, although there are still external and internal screens through which larger organisms generally cannot pass. However, exceptions may occur in poorly maintained vessels, allowing these larger organisms to be transported.3
Among the plants transported, phytoplankton, especially diatoms and dinoflagellates, have been found to be particularly common in ballast water (Carlton and Geller, 1993; Hallegraeff, 1993). The potential is high for the transport of floating, detached plants, including seaweeds (algae) and seagrasses (such as the eelgrass, Zostera, and the turtle grass, Thalassia). It has been suggested that the recent invasion of the Asian brown kelp, Undaria pinnatifida, in Australia may be related to ballast water release (Sanderson, 1990).
Among the animals, zooplankton can be both diverse and dense in ballast water. These include those planktonic organisms that spend most or all of their life cycle in the water, such as many types of protozoans, rotifers, copepods, opossum shrimp (mysids), arrow worms (chaetognaths), and fish. Zooplankton also include those planktonic animals that spend only a portion of their lives in the water column (meroplankton), in particular the larvae of many benthic invertebrates, including sea anemones, corals, hydroids, mollusks (snails, mussels, clams, oysters, and scallops), crustaceans (barnacles, shrimp, lobsters, crabs, hermit crabs), polychaete worms, echinoderms (seastars, brittle stars, sea urchins, sea cucumbers), tunicates (sea squirts), and the larvae of fish.
The biota of the sediment in ballasted cargo holds of arriving ships are not well known. Williams et al. (1988) reported that vessels arriving in Australia contained diverse and abundant invertebrates that could be released if and when the sediment is discharged. Hallegraeff and Bolch (1992) further reported that ballast sediment may contain numerous dinoflagellate cysts. The biota of the sediment in ballast tanks are also not well known. The committee directly observed benthic communities established on the bottom of a dedicated ballast tank on a coastal trade vessel that, when deballasted, held unpumpable ballast to a depth of 15 to 30 cm (see Appendix B). A permanently submerged assemblage of polychaete worms, amphipods, shrimp, hydroids, nematodes, and other species had become established. These organisms could release planktonic larvae into the overlying ballast water, which could subsequently be discharged, and the resident adult community would remain on the bottom of the tank.
FATE OF DISCHARGED ORGANISMS
The release of nonindigenous species into a novel environment constitutes their inoculation but not necessarily their successful introduction. Inoculation is followed by differential survival; a long-standing observation is that most individuals disappear after release and do not form established populations (Carlton et al., 1995). It is not known how long most inoculated individuals simply survive. Older, isolated individuals of nonindigenous species that do not form reproducing populations are occasionally found, which indicates that a certain number grow to adulthood (Carlton, 1995).
As is true of all transport vectors (natural and synanthropic) and of all quarantine concerns, the greater the difference in the physical and chemical states of the donor (source) and receiver (target) regions, the lesser the probability of survival. Thus many organisms from tropical ports will not survive or reproduce in cooler, temperate or boreal ports, and vice-versa.4 However, some species of marine invertebrates and algae occur from subpolar to tropical waters. Thus, the transport of organisms from a warm-water port to a cold-water port, or vice versa, cannot be classified as a "zero risk" scenario. Further, it is impossible to make a complete list of all potential unwanted introductions from a foreign source because many species do not manifest nuisance characteristics within their native ranges. Biological science cannot predict whether a species that is harmful at its source will present a risk when introduced to a new location. Thus, it is not possible to identify areas of zero risk where ballast controls are unnecessary.
Regardless of the match or mismatch in any one physical or chemical variable between a source environment and a release site for a species, numerous
other considerations come into play when determining whether a given species will become established (see also Box 3-3). Thus, even when temperatures of discharged ballast water and receiving waters are identical, factors such as salinity, oxygen, light, food resources, competitors, predators, substrate availability, and many others may be inhospitable or limiting.5 These biological and ecological factors frequently vary dramatically over seasons and years, with the result that a species that is not successful at one time may later become established (Carlton, 1996).
COMPLEXITY OF BALLASTING PATTERNS
A critical concept in managing ballast water is that the source regions and release sites of ballast water frequently occur in a complex fashion along the vessel's route. The following hypothetical scenario is an example of how a vessel may have ballast water from multiple sources, with different water in different tanks or mixed in the same tank.
A vessel off-loads its cargo in a discharge port, having arrived with some ballast on board from a prior harbor (site 1). After completing cargo discharge, the vessel ballasts (site 2) in preparation for its transoceanic crossing to pick up cargo for another port. During passage additional water is loaded into storm ballast and cargo/ballast holds because of rough sea conditions (site 3). Some days later, due to fuel and freshwater consumption, more ballast is added mid-ocean for additional stability (site 4). Finally, additional ballast is taken aboard for clearance under a harbor bridge in the arrival port (site 5).
The result of this hypothetical scenario translates biologically into the vessel accumulating organisms from multiple ballastings at several sites. Thus, organisms arriving in ballast water are not necessarily strictly estuarine or coastal in origin, nor do all of them necessarily represent the last port of call. Ballast water may be hours to months old and may contain living organisms for an extended period of time. As noted above, sediment in ballast tanks may reflect an even longer history of ballasting and may include an accumulation of life forms from many ports around the world that have survived in the shipboard environment.
The movement and release patterns of ballast water are such that no coastal site, whether it receives direct shipping or not, is immune to ballast-mediated introductions. It is frequently assumed that only major port systems are at risk from introductions of organisms. However, ships may release ballast water as they pass along coastlines, often sufficiently close to shore that natural onshore advection may carry ballast-discharged organisms into small lagoons, bays, or any other coastal location. Coastal vessel traffic and coastal currents may also disperse introduced species from larger port systems to remote sites all along a coast.
IMPLICATIONS FOR MANAGING BALLAST WATER
The overall diversity of biota found in ballast and the complexity of ballasting patterns and operations mean that predicting the presence of a particular unwanted species in any one vessel is a scientific challenge. Similarly, the great diversity of the larval or juvenile stages of marine invertebrates and fish in ballast water, many of which are not identifiable, means that certifying a vessel as free or safe from all unwanted species is not possible. A further complication is that many of the most prominent introductions of recent years were not recognized as problem species in their donor regions. Thus the Asian clam, Potamocorbula amurensis, which has led to fundamental changes in the energy flow and dynamics of the San Francisco Bay estuary since its appearance in 1986 (Carlton et al., 1990; Alpine and Cloern, 1992), was not signalled as a species of concern in Western Pacific estuaries, nor would its larvae have been identifiable if ballast sampling programs had been in place in California prior to 1986.
In terms of managing ballast water, the multiplicity of factors influencing the establishment of nonindigenous organisms means that species (particularly those from comparable environments) found in ballast water samples that have not previously or subsequently been found in the receiving port cannot be assumed to be safe species that could be put on a ''clean" list. Possible explanations have been postulated for the observation that a species may eventually become extraordinarily successful in waters where it may have been inoculated for many decades. The zebra mussel, Dreissena, in the North American Great Lakes (Carlton, 1996) is one example. Important factors include changes in the donor region, new donor regions, changes in the recipient region—including improved water quality—the opening of introduction windows (the proper combination of environmental conditions), stochastic inoculation events, and changes in the dispersal vector. As long as there are transport mechanisms, such as ballast water, there may be new introductions.
The objective of developing strategies for managing ballast water is to reduce the likelihood of new invasions to an acceptable level without in any way compromising ship safety. This requirement is complicate by the diversity of potential invaders and by the many environmental factors—often overlapping and synergistic—determining the fate of organisms discharged with ballast water.
OTHER DISPERSAL VECTORS
A ship is a biological island (see Box 1-1). Besides the potential complement of synanthropic hitchhikers (e.g., mice, rats, flies, and cockroaches) and the rich bouillabaisse of ballast water and sediment, living marine and estuarine organisms can occur elsewhere in and on the vessel. In this regard, it is necessary to distinguish the unique role of ballast water an sediment in transporting non-indigenous species from other ways in which a vessel may transport organisms.
|
BOX 1-1 Life beyond Ballast The modern ship transports living organisms on the hull, in sea chests, and elsewhere on and in the vessel, such as in seawater piping systems, on the rudder, entangled in the anchor or in the anchor chain, in chain lockers or caught up in fish nets. Ballast water has a clearly identified role in directly and consistently releasing large numbers of organisms in every major port of the world every day. This report focuses on ballast water as the major vector for the dispersal of nonindigenous aquatic organisms. |
Of particular concern are organisms carried on the ship's hull and in sea chests (also known as sea boxes or suction bays). Despite numerous modern studies on the diversity of organisms transported in ballast water, relatively little is known about the diversity of marine life transported on hulls or in sea chests. In a number of cases there is obvious taxonomic overlap. For example, barnacles occur commonly both as adults on ships' hulls and as larvae in ships' ballast water. Should a new barnacle invasion occur, it would be difficult to determine which of these two vectors was responsible.
In the case of sea chests, the extent of potential overlap is much less clear. Thus, although starfish (seastars) are not likely to survive long sea voyages on the exposed hull of a vessel and seastar larvae occur in ballast water, sea chests may also conceivably support seastar populations. Sea chests, because of their protected nature, may harbor relatively larger accumulations of organisms—including mobile species such as snails that would normally be washed away from the ship's hull—than the outside of the ship. Carlton et al. (1995) suggested that the modern sea chest may be an analogue of the deep and protected shipworm galleries off wooden sailing ships. However, the committee is not aware of any modern scientific studies of sea chest fouling.
The general perception is that, because of anti-fouling paints, modern vessels typically do not develop the massive fouling communities that were the bane of mariners a century and more ago. However, the role of ships' fouling in transporting nonindigenous species remains unclear. Increased modern vessel speeds, decreased port time, and increased vessel performance (to offset vessel operating costs) suggest that the number of marine organisms transported in fouling communities may have decreased over time. Conversely, the role of ship fouling may have increased for some species and for some trade routes for a number of reasons, including the evolution of strains of certain seaweeds resistant to anti-fouling paint; the greater sea-going speeds of modern vessels, which lead to decrease in
transport times and a resultant increase in survival rates of oligohaline organisms; and the decreased use globally of certain types of anti-fouling paints, such as those containing tributyltins (Hall, 1981; Weis and Cole, 1989; Carlton et al., 1995). The net result of these negative and positive influences on the diversity of fouling communities is not known.
Temporal considerations further hinder predictions of the relative roles of ballast versus other ship-mediated vectors. Fouling communities are transported to a "new" port with every visit of every ship; significant amounts of ballast water are released only by those vessels loading cargo, and different ports receive significantly different amounts of ballast water. On the other hand, for fouling communities to act as effective vectors of non-native species, the organisms involved must either reproduce in the short time a vessel is in port or must be dislodged and fall of the ship. The latter situation is less likely for organisms residing in the protected sea chest.
References
Alpine, A.E., and J.E. Cloern. 1992. Trophic interactions and direct physical effects control phytoplankton biomass and production in an estuary. Limnology and Oceanography 37:946–955.
Bio-Environmental Services, Inc. 1981. The Presence and Implication of Foreign Organisms in Ship Ballast Waters Discharged into the Great Lakes, vols. 1 and 2. Ottawa, Canada: Environment Canada for the Water Pollution Control Directorate, Environmental Protection Service.
Carlton, J.T. 1985. Transoceanic and interoceanic dispersal of coastal marine organisms: The biology of ballast water. Oceanography and Marine Biology: An Annual Review23:313–371.
Carlton, J.T. 1995. Personal communication to the Committee on Ships' Ballast Operations, October 3, 1995.
Carlton, J.T. 1996. Pattern, process, and prediction in marine invasion ecology. Biological Conservation, Special Issue (University of California Davis Invasions Symposium), in press.
Carlton, J.T., and J.B. Geller. 1993. Ecological roulette: Biological invasions and the global transport of nonindigenous marine organisms . Science 261:78–82.
Carlton, J.T., J.K. Thompson, L.E. Schemel, and F.H. Nichols. 1990. Remarkable invasion of San Francisco Bay (California, USA) by the Asian clam Potamocorbula amurensis: I. Introduction and dispersal. Marine Ecology Progress Series 66:81–94.
Carlton, J.T., D.M. Reid, and H. van Leeuwen. 1995. The Role of Shipping in the Introduction of Nonindigenous Aquatic Organisms to the Coastal Waters of the United States (other than the Great Lakes) and an Analysis of Control Options. Washington, D.C.: U.S. Coast Guard and U.S. Department of Transportation, National Sea Grant College Program/Connecticut Sea Grant. USCG Report Number CG-D-11-95. NTIS Report Number AD-A294809.
Cohen, A.N., and J.T. Carlton. 1995. Biological Study. Nonindigenous Aquatic Nuisance Species in a United States Estuary: A Case of the Biological Invasions of the San Francisco Bay and Delta. Washington, D.C.: National Oceanic and Atmospheric Administration for the U.S. Fish and Wildlife Service and the National Sea Grant College Program, Connecticut Sea Grant. NTIS Report Number PB96-166525.
Gosliner, T. 1995. The introduction and spread of Philine auriformis (Gastropoda: Opisthobranchia) from New Zealand to San Francisco Bay and Bodega Harbor. Marine Biology122:249–255.
Hall, A. 1981. Copper accumulation in copper-tolerant and non-tolerant populations of the marine fouling alga, Ectocarpus siliculosus (Dillwyn) Lyngbye. Botanica Marina 24:223–228.
Hallegraeff, G.M. 1993. A review of harmful algal blooms and their apparent global increase. Phycologica 32:79–99.
Hallegraeff, G.M., and C.J. Bolch. 1992. Transport of diatom an dinoflagellate resting spores in ships' ballast water: Implications for plankton biogeography and aquaculture. Journal of Plankton Research 14:1067–1084.
Harbison, G.R., and S.P. Volovik. 1994. The ctenophore, Mnemiopsis leidyi, in the Black Sea: A holoplanktonic organism transported in the ballast water of ships. Pp. 25–36. Proceedings of the National Oceanic and Atmospheric Administration Conference and Workshop on Nonindigenous Estuarine and Marine Organisms. Washington, D.C.: U.S. Government Printing Office.
Jones, M.M. 1991. Marine Organisms Transported in Ballast Water: A Review of the Australian Scientific Position. Bureau of Rural Resources Bulletin No. 11, Department of Primary Industries and Energy. Canberra, Australia: Australian Government Publishing Service.
LeMaitre, R. 1995. On the introduction of the Indo-Pacific portunid crab Charybdis helleri, in Florida. Proceedings, Biological Society of Washington 108:643–648.
Mills, C.E., and F. Sommer. 1995. Invertebrate introductions in marine habitats: Two species of hydromedusae (Cnidaria) native to the Black Sea, Maeotias inexspectata and Blackfordia virginica, invade San Francisco Bay. Marine Biology 122:279–288.
Mills, E.L., J.H. Leach, J.T. Carlton, and C.L. Secor. 1993. Exotic species in the Great Lakes: A history of biotic crises and anthropogenic introductions. Journal of Great Lakes Research 19:1–54.
Nalepa, T.F., and D.W. Schloesser (eds). 1993. Zebra Mussels: Biology, Impacts, and Control. Boca Raton, Florida: Lewis Publishers, Inc. (CRC Press).
NRC. 1995. Understanding Marine Biodiversity: A Research Agenda for the Nation. Ocean Studies Board, National Research Council. Washington, D.C.: National Academy Press.
Public Law 101-646. 1990. The Nonindigenous Aquatic Nuisance Prevention and Control Act. Washington, D.C.: U.S. Congress.
Randall, J.E., J.L. Earle, T. Hayes, C. Pittman, M. Severns, and R.J.F. Smith. 1993. Eleven new records and validations of shore fishes from the Hawaiian Islands. Pacific Science 47:222–239.
Ruiz, G., and J.T. Carlton. 1995. Personal communication from J.T. Carlton to the Committee on Ships' Ballast Operations, August 22, 1995.
Sanderson, J.C. 1990. A preliminary survey of the distribution of the introduced macroalga, Undaria pinnatifidia (Harvey) Suringar, on the east coast of Tasmania, Australia. Botanica Marina 33:153–157.
Weis, J.S., and L.A. Cole. 1989. Tributyltin and public policy. Environmental Impact Assessment Review 9:33–47.
Williams, R.J., F.B. Griffiths, E.J. Van der Wal, and J. Kelly. 1988. Cargo vessel ballast water as a vector for the transport of nonindigenous marine species. Estuarine and Coastal Shelf Science 26:409–420.