A
Origin and Framework of the Development of Dietary Reference Intakes
This report is the first in a series of publications resulting from the comprehensive effort being undertaken by the Food and Nutrition Board's (FNB) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes (DRI Committee) and its panels and subcommittees.
ORIGIN
This initiative began in June 1993, when the FNB organized a symposium and public hearing entitled “Should the Recommended Dietary Allowances Be Revised?” Shortly thereafter, to continue its collaboration with the larger nutrition community on the future of the Recommended Dietary Allowances (RDAs), the FNB took two major steps: (1) it prepared, published, and disseminated the concept paper “How Should the Recommended Dietary Allowances Be Revised?” (IOM, 1994), which invited comments regarding the proposed concept, and (2) it held several symposia at nutrition-focused professional meetings to discuss the FNB's tentative plans and to receive responses to this initial concept paper.
The five general conclusions presented in the 1994 concept paper are as follows:
-
Sufficient new information has accumulated to support a reassessment of RDAs.
-
Where sufficient data for efficacy and safety exist, reduction in the risk of chronic degenerative disease is a concept that should be included in the formulation of future recommendations.
-
Upper levels of intake should be established where data exist regarding risk of toxicity.
-
Components of food of possible benefit to health, although not meeting the traditional concept of a nutrient, should be reviewed, and if adequate data exist, reference intakes should be established.
-
Serious consideration must be given to developing a new format for presenting future recommendations.
Subsequent to the symposium and release of the concept paper, the FNB held workshops at which invited experts discussed many issues related to the development of nutrient-based reference values, and FNB members have continued to provide updates and engage in discussions at professional meetings. In addition, the FNB gave attention to the international uses of the earlier RDAs and the expectation that the scientific review of nutrient requirements should be similar for comparable populations.
Concurrently, Health Canada and Canadian scientists were reviewing the need for revision of the Recommended Nutrient Intakes (RNIs) (Health Canada, 1990). Consensus following a symposium for Canadian scientists cosponsored by the Canadian National Institute of Nutrition and Health Canada in April 1995 was that the Canadian government should pursue the extent to which involvement with the developing FNB process would be of benefit to both Canada and the United States in terms of leading toward harmonization.
Based on extensive input and deliberations, the FNB initiated action to provide a framework for the development and possible international harmonization of nutrient-based recommendations that would serve, where warranted, for all of North America. To this end, in December 1995, the FNB began a close collaboration with the government of Canada and took action to establish the DRI Committee. It is hoped that representatives from Mexico will join in future deliberations.
RATIONALE FOR THE FRAMEWORK
The 1993 symposium and subsequent activities have provided substantial evidence that a comprehensive, coordinated approach to developing DRIs is needed for diet planning, nutritional assessment, and nutrition policy development. The current framework is based on the following four assumptions:
-
Since the publication of the tenth edition of Recommended Dietary Allowances (NRC, 1989a) in the United States and Canadian Recommended Nutrient Intakes (Health Canada, 1990), there has been a significant expansion and evolution of the research base in relation to a move toward defining functional endpoints that are relevant to the understanding of nutrient requirements and food constituents and their relationship to a number of aspects of human health.
-
These advances allow the refinement of the conceptual framework for defining nutrient requirements quantitatively and a clearer determination of the legitimate uses of nutrient requirement estimates and their derivatives in the interpretation and use of dietary intake data. Such uses might broadly be categorized according to whether they are (a) prescriptive or planning applications, where suitable levels of nutrient intake by individuals and/or population groups are established, and (b) diagnostic or assessment applications, where determinations are made about the likely nutritional adequacy of the observed intake when considered in relation to appropriate nutrient requirement data. Major differences in the types of information required about nutrient needs and relevant nutrient intake data are fundamental to appropriately focusing on the individual or on a defined population group (Beaton, 1994).
-
Neither the RDAs nor the Canadian RNIs have been applied appropriately in many settings. The availability of only a single type of reference value in the face of various needs has led to inappropriate applications. Moreover, inconsistent methods and criteria for deriving certain RDAs and RNIs and insufficient documentation of approaches and criteria have also contributed to inappropriate applications.
-
In these times of extensive international collaboration, agricultural and food exchange, and global nutrition-related health problems, harmonization of nutrient-based dietary standards between Canada and the United States is viewed as a first step, with the expectation that Mexico will be able to join in the near future. Such harmonization within the North American continent would further global development of similar efforts. Although the same general approaches have been used by most countries in developing recommended nutrient intakes (e.g., RDAs in the United States, RNIs in Canada, and Dietary Reference Values [DRVs] in Great Britain), and physiological requirements for nutrients are expected to be similar across healthy population groups, many of the quantitative values that have emerged from the different national expert groups are quite divergent, largely reflecting differences in the interpretation and use of scientific data and often based on different food
-
habits and indigenous diets. A mechanism is needed to determine the commonality of the bases on which recommendations are made and to use scientific data to indicate differences in requirements among apparently similar population groups in different geographic locations.
DESCRIPTION OF THE FRAMEWORK
In 1995, the DRI Committee was appointed to oversee and conduct this project. To accomplish this task over a period of 5 years, the DRI Committee devised a plan that involves the work of seven or more expert related nutrient group panels and two overarching subcommittees (Figure A-1). The process described below is expected to be used for subsequent panels, which will interact with an additional Subcommittee on the Uses and Interpretation of the DRIs.
The related nutrient group panels, composed of experts on those nutrients, are responsible for (1) reviewing the scientific literature concerning specific nutrients under study for each stage of the lifespan, (2) considering the roles of nutrients in decreasing risk of chronic and other diseases and conditions, and (3) interpreting the current data on intakes in North American population groups. The panels are charged with analyzing the literature, evaluating possible criteria or indicators of adequacy, and providing substantive rationales for their choices of each criterion. Using the criterion or criteria chosen for each stage of the lifespan, the panels estimate the average requirement for each nutrient or food component reviewed, assuming that adequate data are available. As the panel members review data on requirements, they also interact with two subcommittees regarding their group of nutrients. The Subcommittee on Upper Reference Levels is charged with reviewing possible risk assessment models for estimating levels of nutrients that may increase risk of toxicity or adverse effects and then assisting the panel to apply the model to each nutrient or food component reviewed. Similarly, a Subcommittee on the Uses and Interpretation of the DRIs is proposed to assist the panels and DRI Committee in the development of practical information and guidance using the many DRIs appropriately. Based on interaction with and from information provided by the panels and subcommittees, the DRI Committee determined the DRI values to be included in the report.
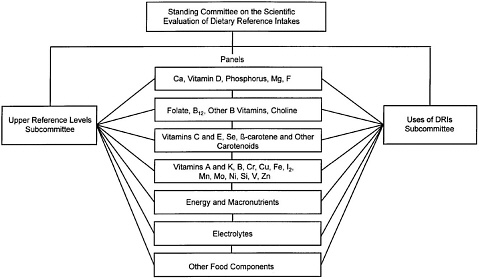
FIGURE A-1 Dietary Reference Intakes.
CHARGE TO THE PANEL ON CALCIUM AND RELATED NUTRIENTS AND SUBCOMMITTEE ON UPPER REFERENCE LEVELS
The National Institutes of Health's National Heart, Lung and Blood Institute, the U.S. Food and Drug Administration, and the U.S. Department of Agriculture's Agricultural Research Service requested that the Institute of Medicine review and develop dietary reference intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. In part, this nutrient group was given priority because of the high prevalence of osteoporosis among the growing population of people over 50 years of age, in addition to possible links of these nutrients to the development of risk factors for stroke and cardiovascular disease. Additionally, the need to establish upper levels of intake, which although not evaluated as having any benefit to the individual, would also not result in increased risk, was an important component of the task.
In April 1996, the DRI Committee of the FNB established the Panel on Calcium and Related Nutrients and the Subcommittee on Upper Reference Levels. The panel was charged with the following:
-
Review the literature and interpret the depth of current knowledge of calcium, phosphorus, magnesium, vitamin D, and fluoride requirements for each stage of the lifespan.
-
Analyze the literature coupled with information available from large epidemiological studies to determine dietary intakes of calcium, phosphorus, magnesium, vitamin D, and fluoride in the U.S. and Canadian populations and the potential for such intakes to be associated with decreased risk of chronic disease.
-
Review the other components in foods that affect the utilization of calcium and related nutrients in human diets, including bioavailability from nonfood sources.
-
Evaluate comparable standards from other countries and multinational groups for whom recommended nutrient ranges have been developed and consider the applicability of the recommended nutrient range for use in North America.
-
Develop dietary reference intake values for calcium, phosphorus, magnesium, vitamin D, and fluoride, where adequate data are available, for each stage of the lifespan including people older than 50 years.
-
Identify research needed to improve knowledge of calcium, phosphorus, magnesium, vitamin D, and fluoride requirements.
The Subcommittee on Upper Reference Levels was charged with the following:
-
Develop a model to estimate the maximum level of nutrient intake that would pose a low risk of adverse effects.
-
Apply the model to calcium, phosphorus, magnesium, vitamin D, and fluoride to develop Tolerable Upper Intake Levels.






